Abstract
Abnormality in Src PSD-95 NR2B signaling complex assemble occurs in levodopa-induced dyskinesia (LID).
Introduction
Levodopa-induced dyskinesia (LID) is one of the thorniest troubles during the treatment of Parkinson’s disease (PD). Dyskinesia commonly presents as chorea or choreoathetosis. This major motor complication of long-term levodopa treatment has restricted levodopa use for PD treatment in the hemi-parkinsonian rat model by 6-hydroxydopamine (6-OHDA) lesion; levodopa results in equal side effects named abnormal involuntary movements (AIMs) affecting cranial, trunk, and limb muscles on the side of the body contralateral to the lesion. 1 –3 Dyskinesia is difficult to treat and, therefore, efforts should be made to prevent them.
The underlying mechanisms for dyskinesia are unclear. Recent studies indicate the importance of pulsatile stimulation of striatal postsynaptic receptors in their pathogenesis. It is suggested that with the gradual reduction in the level of striatal dopamine with the progression of PD, the fluctuation of exogenous levodopa level brings about the striatal dopamine receptor aberrant stimulation, which can further overactivate ionotropic glutamatergic receptors and signal cascade reactions.
1
–5
On this side, we had found evidences that chronic levodopa alternative treatment induced the overactivation of striatal
In addition, another very important gaseous messenger molecule, nitric oxide (NO), derived from neuronal NO synthase (nNOS) has also been involved in the pathogenesis of LID. NO plays an important role in motor control within the central nervous system. nNOS inhibition can reduce the development of dyskinesia in PD rats. 9 –11 As an important downstream effector of NMDAR, nNOS functionally colocalized in the synaptic site with NMDAR. 12,13 The nNOS signal recently was proved to participate the activation of Src function by regulating S-nitrosylation of signal molecular. 13 Thus, nNOS signal might in turn regulate the NR2B activation. Yet, the detailed signal mechanism about the interactions of NR2B, Src, and nNOS is still unknown.
Based on the abovementioned evidences, in order to uncover the relationship between NR2B, Src, and nNOS, we investigated the effects of 7-NI (nNOS inhibitor) and CP-101,606 (NR2B/NMDAR antagonist) on the S-nitrosylation and activation of Src and the further activation of NR2B by Src.
Materials and methods
Animals
Adult female Sprague–Dawley rats (
Parkinsonian rat model
Rat head was placed onto stereotaxic (Narishige, Japan) after pentobarbital anesthesia (50 mg/kg body weight, intraperitoneally (i.p.)) and injected 6-OHDA (8 µg in 4 µl physiological saline) into the right medial forebrain bundle (bregma 4.5 mm; lateral 0.9 mm and dura 7.5 mm). 14 Three weeks after 6-OHDA microinjections, the lesioned rats were injected with apomorphine (APO; 0.25 mg/kg in physiological saline water, i.p.) and were placed in a stainless steel bowl. The rats that showed APO-induced rotation of over seven turns per minute away from the lesioned side were chosen for the next study. 15 Forty successfully parkinsonian rats were selected for the next research.
Drug treatment scheme and behavior assessments
We established five groups (10 rats per group), including control group, PD group (hemi-6-OHDA lesion), dyskinesia group (PD rats with chronic levodopa treatment), 7-NI group (PD rats with chronic levodopa treatment plus 7-NI injection), and CP-101,606 group (PD rats with chronic levodopa treatment plus CP-101,606 injection). The control rats and PD rats were treated twice daily i.p. with normal saline for 23 days. Other PD rats were treated twice daily i.p. with levodopa methyl ester for 22 days (25 mg/kg with benserazide 6.25 mg/kg). 16 On day 23, these rats received i.p. levodopa or levodopa plus 7-NI (25 mg/kg) or levodopa plus CP-101,606 (0.5 mg/kg) 30 min before each levodopa dose treatment. 7,13 During the drug treatment, AIMs were assessed in the light of the previous approach. 17 Briefly, we assessed locomotor, axial, limb, and orolingual movement for each rat after L-DOPA administration on the testing day of 1, 8, 15, 22, and 23. Within every 20-min intervals over 3 h, AIMs were graded for 60 s. Severity scoring from 0 to 4 was designated to each AIMs. In the end, the four categories of AIM scores were added up at each time point (Figure 1). After the last levodopa treatment, the rats were killed, and the striatum was dissected and frozen at −80°C for next immunoblotting (IB).

Time course of the experiments described in the text. 6-OHDA: 6-hydroxydopamine; APO: apomorphine; AIM: abnormal involuntary movement; 7-NI: one nNOS inhibitor; CP-101,606: one NR2B/NMDAR antagonist; nNOS: neuronal nitric oxide synthase.
Immunoblotting
Striatal tissues (five rats per group) underwent lysis in the containing phenylmethanesulfonyl fluoride tissue lysis buffer. The supernatant was collected after centrifugation for 10 min at 12,000 ×
IP for determining protein S-nitrosylation
Striatal samples (containing 400 µg of protein) were diluted four-fold with immunoprecipitation (IP) buffer and preincubated with 25 µl of protein A sepharose CL-4B (Amersham) for 1 h at 4°C. After centrifugation, the supernatant was incubated with 2 µg antibody (mouse monoclonal anti-Src antibody) overnight at 4°C; 25 µl of protein A sepharose CL-4B was added and the incubation continued for 2 h at 4°C. Immune complex was isolated by centrifugation and the pellet was washed three times with IP buffer. Bound proteins were eluted by boiling for 5 min in Laemmli sample buffer. Then, sample was centrifuged and supernatant was used for protein analysis by IB with anti-SNO-Cys antibody as described above.
Data analysis
Data were demonstrated as mean ± standard deviation. Statistical analysis of the biochemical results was accomplished by analysis of variance (ANOVA) followed by Dunnett’s
Power analysis for the two-group independent sample
Results
Effect of 7-NI and CP-101,606 on dyskinetic behavior
The data suggested that the individual and total AIM scores showed progressively rise after chronic-repeated levodopa treatment in PD rats, as shown in Figure 2. On day 23, coadministration of 7-NI or CP-101,606 significantly reduced the AIM scores with statistically differences in comparison with dyskinesia group (

Effects of 7-NI or CP-101,606 on total and individual AIMs in dyskinetic rats (
Effects of 7-NI on the augmented SNO-Src, p-Src, and NR2B tyrosine phosphorylation in the lesioned striatum
As demonstrated in Figure 3, the expression of SNO-Src, p-Src, and pNR2B-Tyr1472 were upregulated obviously in dyskinesia group. On the day of 23 during levodopa treatment, the abundance of SNO-Src, p-Src, and pNR2B-Tyr1472 increased to 130.2 ± 11.2%, 145.5 ± 7.3%, and 142.3 ± 8.1%, respectively, of control group (
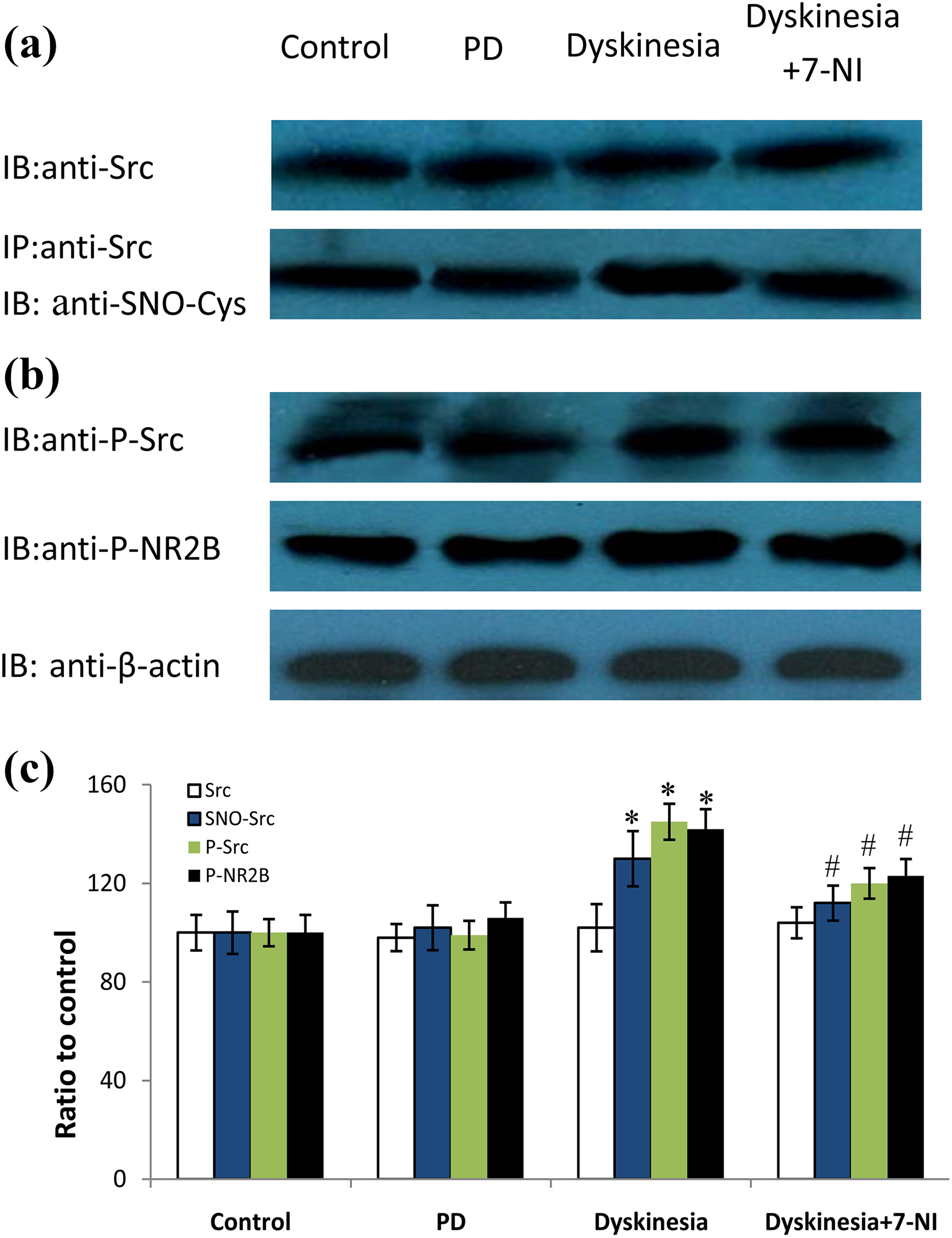
Effects of 7-NI on Src S-nitrosylation, Src autophosphorylation, and phosphorylated NR2B levels in dyskinetic rats. (a) The band represents typical immunoblot images detected by antibody against Src and co-IP analysis of Src with SNO-Cys from control, PD, dyskinesia, and 7-NI-treated rats. (b) The band represents typical immunoblot images detected by antibodies against p-Src and pNR2B-Tyr1472 from control, PD, dyskinesia, and 7-NI-treated rats. (c) Bands corresponding to Src, SNO-Src, p-Src, and p-NR2B on immunoblots in the lesioned side striatum shown as in (a) and (b) were scanned and their optical density quantified by densitometry. *
Effects of CP-101,606 on the p-nNOS-S847, p-Src, SNO-Src, and NR2B tyrosine phosphorylation in the lesioned striatum
The expression of p-nNOS-S847 was downregulated obviously in dyskinesia group. However, coadministration of CP-101,606 reversed the p-nNOS-S847 level in dyskinetic rats to 110.1 ± 5.6%, respectively, of control group (
Yet, as demonstrated in Figure 4, the expression of SNO-Src, p-Src, and pNR2B-Tyr1472 was upregulated obviously in dyskinesia group. Coadministration of CP-101,606 reduced the SNO-Src, p-Src, and pNR2B-Tyr1472 levels in dyskinetic rats to 108.4 ± 6.9%, 115.1 ± 4.1%, and 113.4 ± 5.3%, respectively, of control group (
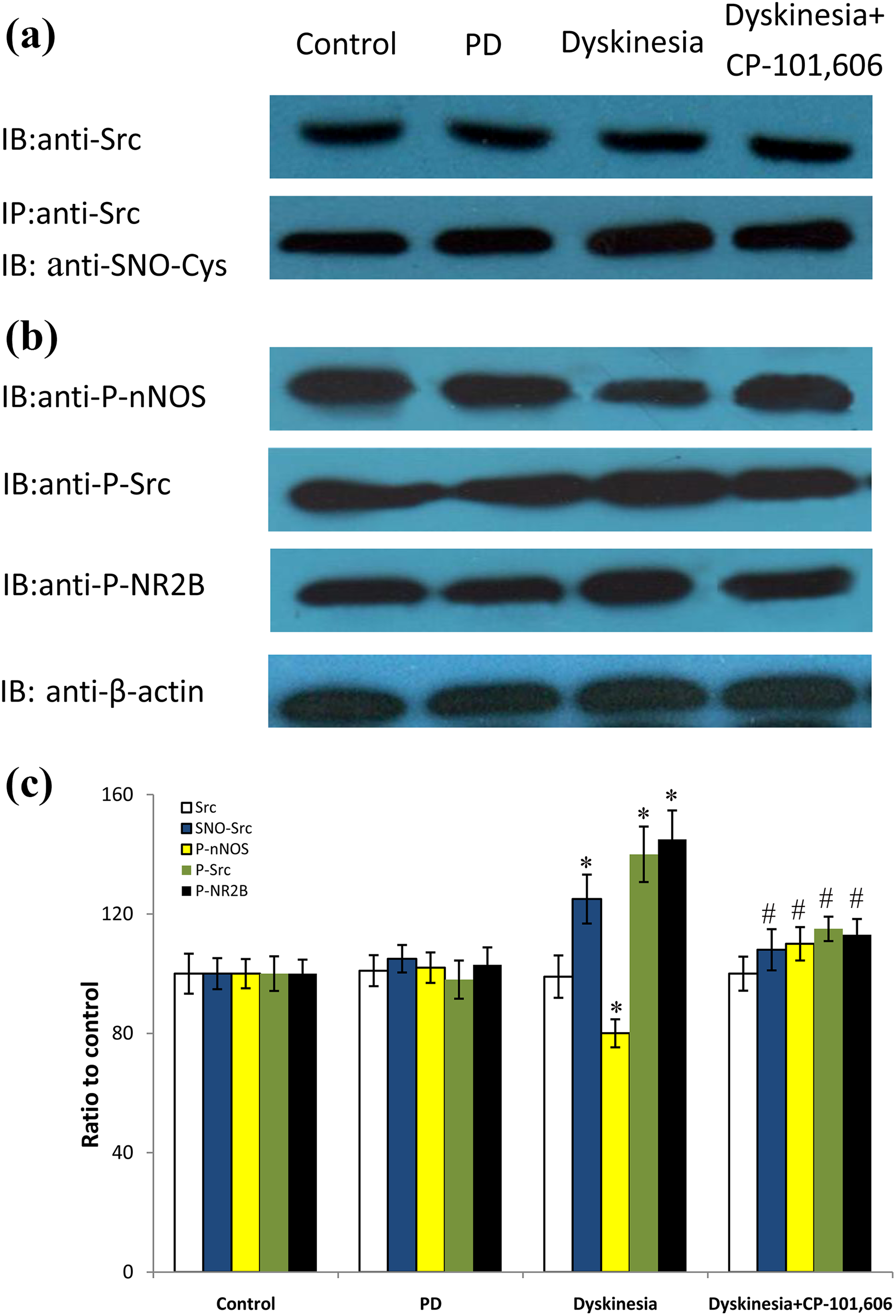
Effects of CP-101,606 on Src S-nitrosylation, nNOS phosphorylation, Src phosphorylation, and NR2B phosphorylation levels in dyskinetic rats. (a) The band represents typical immunoblot images detected by antibody against Src and co-IP analysis of Src with SNO-Cys from control, PD, dyskinesia, and CP-101,606-treated rats. (b) The band represents typical immunoblot images detected by antibodies against p-nNOS-S847, p-Src, and pNR2B-Tyr1472 from control, PD, dyskinesia, and CP-101,606-treated rats. (c) Bands corresponding to Src, SNO-Src, p-nNOS, p-Src, and p-NR2B on immunoblots in the lesioned side striatum shown as in (a) and (b) were scanned and their optical density quantified by densitometry. *
Discussion
In the present study, we found that chronic levodopa treatment promoted Src kinase activation by Src S-nitrosylation and tyrosine-416 autophosphorylation. The activated Src enhanced NMDAR activation via exacerbating NR2B tyrosine phosphorylation, which closely correlated with the expression of dyskinesia. These findings demonstrated that inhibition of SNO-Src could attenuate NMDAR overactivation in dyskinesia rat model via suppressing the postsynaptic “NR2B/NMDAR–nNOS/NO–SNO-Src–p-Src–NR2B/NMDAR” signal cycle.
The striatal pulsatile dopamine receptor stimulation can lead to aberrant signal cascade reactions in dyskinesia rat model. 1,4,19 Previous research have suggested that Src tyrosine kinase was one important protein kinases for intracellular signal transduction related to dopamine receptor reaction. 20,21 Calcium influx dependent on the NMDAR can modulate many downstream signaling pathways after chronic L-DOPA treatment, including Src or CaMKII activation. 22 –24 Our previous research had shown that NR2B/NMDAR antagonist could inhibit Src activation in dyskinesia rat model by attenuating interactions among NR2B and Src. 7 Yet, the underlying mechanisms of Src activation in dyskinesia rat model have not been clarified.
In the light of intramolecular modulation, Src phosphorylation sites comprise the inhibitory phosphorylation site at tyrosine-527 (p-Src527) and the stimulatory phosphorylation site at tyrosine-416 (p-Src416) which is attributed to the Src autophosphorylation. In vivo, most of Src is phosphorylated under normal circumstance, which is prone to be activated by several mechanisms, for example, Src tyrosine-527 dephosphorylation and Src tyrosine-416 autophosphorylation. 25,26 The NO toxicant metabolic substance, peroxynitrite (ONOO–), can also provoke the Src activity and phosphorylation by enhancing Src nitration. 27 Similar to the phosphorylation, S-nitrosylation is one reversible targeted protein function modification in the process of molecular signaling transduction. As reflected in the results, levodopa treatment leads to significant upregulation of SNO-Src and p-Src. Thus, in the present research, we found two forms of Src activation, which are the p-Src416 by Src autophosphorylation and the SNO-Src at cysteine by NO signal.
nNOS is one of the sources of NO signal. 9 –11 Our further research demonstrated that calcium influx via the NMDAR modulated the downstream nNOS signal pathways. Chronic levodopa treatment could result in downregulation of p-nNOS-S847 which reflected nNOS overactivation in dyskinesia rat model. 28,29 And inhibition of nNOS with 7-NI could lead to lower phosphorylation and S-nitrosylation of Src and NR2B tyrosine phosphorylation. NR2B/NMDAR antagonist CP-101,606 could upregulate p-nNOS-S847 levels and reduce the nNOS activation and thus produce the similar effects like nNOS inhibitor 7-NI, which were consistent with the change of the behavioral profile of this drug. Taken together, the S-nitrosylation of Src is caused by nNOS/NO signal, which is overactivated via Ca2+ influx dependent on NR2B/NMDAR and subsequently facilitates Src tyrosine autophosphorylation and further phosphorylates NR2B. The “NR2B/NMDAR–nNOS/NO–SNO-Src–p-Src–NR2B/NMDAR” signaling cycle may be the molecular basis of NR2B tyrosine phosphorylation upward positive feedback.
Conclusion
Now, it is well considered that Src tyrosine kinase plays an important role in modulation of NMDAR function in dyskinesia. Our findings indicate that the agents, which suppress Src S-nitrosylation and autophosphorylation, can indirectly reduce overactivation of NMDAR and obviously improve behavioral responses in dyskinesia. As we known, direct NMDAR antagonists for treating dyskinesia can bring about undesirable side effects in preclinical and clinical trials. 30,31 That is the reason that we explore the relation between Src activation and NR2B tyrosine phosphorylation. These can be hopeful as agents for treating dyskinesia in PD.
Footnotes
Authors’ contribution
Maowen Ba and Weifang Ding contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chinese National Natural Science Foundation (no 81571234), Key Research and Development Plan of Shandong Province (2018GSF118235), and Yantai Science and Technology Development Project (2016WS037).
