Abstract
Inhalant abuse is a significant public health issue, particularly for adolescents, the predominant group of inhalant users. Adolescence is a critical growth period, and inhalant abuse has been associated with growth impairments, including reduced body weight and height. However, the extent to which inhalant abuse affects growth remains unquantified, and potential moderators remain unknown. To address this knowledge gap, a systematic review and meta-analysis of clinical human and preclinical animal studies utilizing toluene exposure (the primary solvent in abused products) was conducted. Five-hundred and sixty-nine studies were screened; 31 met inclusion criteria, yielding 64 toluene-control comparisons for body weight and 6 comparisons for height. Toluene exposure was negatively associated with body weight (d = −0.73) and height (d = −0.69). Concentration of inhaled toluene, but not duration, moderated the effect of toluene exposure on body weight, with more severe impairments at higher concentrations. Differences in effect size for body weight were observed for study characteristic subgroups including sex, age at first exposure, administration route and species. However, these findings should be interpreted cautiously due to low study numbers. Growth impairments, particularly during adolescence, can cause long-term health consequences. These effects on growth are therefore an important clinical outcome for individuals with a history of inhalant abuse.
Introduction
Inhalant abuse is the deliberate inhalation of products containing volatile solvents, in order to achieve an altered mental state. 1 Demographic studies into inhalant abuse show that usage patterns are similar across sexes 2 and overwhelmingly indicate that abuse is most commonly associated with adolescence (particularly young adolescence) patterns that are consistent worldwide. 3 –5 For example, population-based surveys indicate that 19.1% of Australian 12- to 13-year olds self-report experimenting with inhalants. 3 While large population-based surveys do not collect data from children under the age of 12, and smaller cohort studies have identified that children under the age of 12 may also misuse inhalants. 6
Preadolescence and adolescence is a critical period of growth, with the peak growth spurt for females occurring between 10 years and 14 years of age, and a slightly later spurt for males occurring at 12–15 years of age. 7 Studies indicate that for many inhalant users, the duration of abuse occurs over periods greater than 10 years, with users reporting abuse patterns equivalent to intermittent high usage, 8 thus overlapping with the period when rapid growth is occurring in many young inhalant users. 3 It is, therefore, unsurprising that inhalant abuse has an effect on growth, which has been observed in both males 6,9 and females 10 ; however, this effect is poorly understood.
Case studies from the 1960s report that inhalant users present as emaciated, 9 and a reduction in weight is well-accepted as a consequence of inhalant abuse, to the extent that it is often listed as a warning sign of this type of drug abuse. 1 Indeed weight impairments can be so severe in humans that inhalant abuse is associated with failure to thrive (FTT). 6 Weight impairments (either a loss of weight or reduction in weight gain) is also a common outcome in animal experiments that utilize toluene exposure as a model of inhalant abuse, occurring immediately after the exposure period, 11 –13 and body weight is often included as a descriptive statistic, even if it was not the key outcome under consideration. However, the effects of toluene exposure on body weight are not consistent in effect size, suggesting that unknown moderators may play a role in the effect of toluene exposure on weight.
Given that the adolescent growth spurt is reflected in a change to individuals’ weight and height, it is surprising that the effects of inhalant abuse on height have not been studied to the same degree as weight. Height impairments arising from inhalant abuse have been reported in both humans 6,14 and animal models utilizing toluene exposure, 15 and in humans this effect has been shown to persist into sustained abstinence. 16 Again the size of this effect has never been quantified, and the potential moderators remain unknown. Despite our lack of understanding of the factors moderating inhalant-induced changes to growth parameters, the effects of inhalant abuse on weight and height are important outcomes. This is especially due to the potential for long-term health impacts arising from alterations to growth, 17 –19 even if an individual with a history of abusing inhalants remains abstinent.
The majority of the research into the physiological consequences of inhalant abuse is derived from animal models, which incorporate five main study characteristics. Firstly, the range of products inhaled by inhalant users is varied and opportunistic and includes common household items such as glue, thinners and aerosols. However, a common solvent to all these products is toluene, which is a colourless, water-insoluble, aromatic hydrocarbon. Inhalant users will preferably inhale pure toluene if it is available, 20 and thus toluene exposure is utilized as an exemplar for inhalant abuse in animal models in the majority of studies. 21 Study design must consider toluene dosage which when the administration route is inhalation will be reflected as a concentration in parts per million (ppm). Human inhalant users will often inhale via a solvent-soaked cloth or directly from a bag, and the concentration of inhaled toluene from this method is estimated to range from 3000 ppm to 15,000 ppm. 21 It is important to note that exposure to toluene may also occur in the industrial setting; however, this occurs in a different pattern to abuse with exposure at lower concentrations for extended durations. Secondly, many humans abuse inhalants in a chronic yet intermittent nature. 8 Thus, a study design must include a pattern of exposure in minutes/day and days/week for a specified period of time. Despite the diverse range of exposure patterns used experimentally, this can be quantified as a variable called ‘exposure hours’, which can then be multiplied by the dosage to give ‘total toluene exposure’. The third issue is that of administration route. Inhalation is the most reflective of human usage but requires specialized equipment and thus some models utilize intraperitoneal (IP) injections or gavage to administer toluene. How the amount of toluene administered is reported will depend on the administration route, with inhalation reported as a concentration in ppm, IP injections as milligrams/kilogram (mg/kg) and gavage as millilitres/kilogram (mL/kg). The final issue is that of experimental animal. Rats and mice are the predominant species used in inhalant abuse models, with rats having equivalent sensitivity with humans to toluene, 22 but choices must still be made about sex and the age at which exposure will begin, bearing in mind the association between inhalant abuse and young adolescence.
To date, the extent to which inhalant abuse affects growth patterns have not been quantified, and the effects of potential moderators are not well-understood. To address this knowledge gap, we conducted a systematic review of human and animal studies and applied meta-analytic techniques to calculate an effect size d. The primary aim of the study was to examine the effects of inhalant abuse (in humans) and toluene exposure (in animals) on weight and height, hypothesizing that ‘as amount of toluene administered (in ppm, mg/kg or mL/kg) increases, height and body weight will decrease’. A secondary aim was to explore the effect on height and body weight of a range of study characteristics that are commonly encountered in inhalant abuse studies, pattern of exposure (including exposure duration and total toluene exposure), administration route and model (including species, sex and age at first exposure).
Methods
This study was registered with the open access database PROSPERO (international prospective register of systematic reviews) at inception (registration number CRD42017069002; https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=69002) and conducted in accordance with PRISMA guidelines. 23 The completed PRISMA flow chart is provided as Online Supplemental material.
Systematic literature review
Peer-reviewed studies of inhalant abuse and toluene exposure were collated via extensive literature searches, searched for in PubMed. To be included, studies must have provided a measure of height or weight that was taken immediately or shortly after exposure, sample sizes in each group along with a measure of variance such as standard deviation (SD) or standard error of the mean (SEM), in order for an effect size to be calculated. Studies that focused on height or weight effects occurring in sustained abstinence (i.e. after exposure to toluene had ceased) were not the focus of this study and were not included. Only published, peer-reviewed studies were considered for inclusion. For the animal studies, only those that utilized toluene as the inhalant were included in this meta-analysis (excluding animal models of inhaled benzene or xylene), as this directly links the experimental model to the products typically abused by humans (e.g. glue, paint and solvents). Given that growth data are often reported as a descriptive statistic in studies, without being a primary outcome, it was not possible to limit the search to keywords such as inhalant + weight. Therefore, a range of strategies were utilized including keyword searches, searches using synonyms for describing growth patterns (Table 1) and keyword searches of physiological outcomes relative to toluene exposure where it might be plausible that body weight would be reported (e.g. reproduction, motor syndrome and food intake).
Key words (in greyed boxes) and synonyms (below) used as alternative terms for the search strategy.

Each combination of keyword and synonym was used as search strings; this strategy meant that 84 unique searches (provided as search strings in Online Supplemental Table 1) were performed in PubMed, which yielded a total of 622 potential studies; 53 of those were duplicates, leaving 569 studies that were checked to determine whether they included a description of height or weight. Of the 569 studies, only 52 reported on at least one of these outcome measures. Studies were then excluded if they did not provide the mean of height or weight along with a measure of variance, for both the treatment and the control groups. This criterion reduced the number of eligible studies from 52 to 31, with 21 studies excluded. It was deemed that this number of studies was sufficient to proceed from systematic review to meta-analysis, and these 31 studies were then used for the meta-analysis. Figure 1 shows the PRISMA flow chart; the 21 excluded studies with reasons box refer to the 21 studies discussed above. Risk of bias within the included studies was assessed using the SYRCLE risk-of-bias tool for animal studies, 24 acknowledging that four of the included studies were clinical observational studies, however, it was not considered appropriate to utilize multiple risk of bias tools. Results of the assessment are provided as Online Supplemental Table 2.

/prismastatement/flowdiagram (PRISMA) flow chart showing study numbers. Source: Moher et al. 23 For more information, see www.prisma-statement.org.
Meta-analysis techniques
The software program Review Manager (RevMan) version 5.3, Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014, was used to analyse the data and conduct the meta-analysis. An effect size statistic was computed for each toluene-to-control comparison; the effect size was estimated by pooling individual effects using a random-effects model. The statistics computed was Cohen’s d, by comparing the mean body weight and/or height from the toluene group to the mean body weight and/or height from the control group. Then, 95% confidence intervals were computed for each d. Heterogeneity is reported using the I 2 statistic, which describes the percentage of variation across studies that is due to heterogeneity rather than chance. Following Higgins et al., 50 we consider I 2 values of 25%, 50% and 75% as low, moderate and high, respectively. Note that these categories do not refer to the absolute amount of observed heterogeneity, but rather to the proportion of the observed effect variance that would remain if the sampling error were to be eliminated, that is, if we were to be able to observe the true effect size for all studies in the analysis.
When entering the data into RevMan, the reported SEM values were converted to SD (
The studies identified in the systematic review utilized a wide range of concentrations of toluene (minimum 80 ppm, maximum 10,000 ppm). To analyse concentration effectively, these were collapsed into the following groups: 0–500 ppm, 501–2000 ppm, 2001–5000 ppm and >5000 ppm. These groupings were chosen for consistency with previous meta-analysis on toluene exposure 52 and provided an approximately even pool of comparisons across the subgroups. Additional analysis was performed post hoc comparing occupational exposure concentrations (≤1000 ppm) with inhalant abuse concentrations (>1000 ppm). Studies using a non-inhalation route such as IP injection or gavage were not assigned a concentration. Exposure hours reflected the total duration of exposure to toluene and were calculated by multiplying the exposure duration in hours and the total number of exposures. For example, 1 h/day and 3 days/week for 4 weeks would be reported as 12 exposure hours. Total toluene exposure (in ppm h) was calculated by multiplying exposure hours and the concentration in ppm, and then collapsed into subgroups of low (0–50,000 ppm h), medium (50,001–200,000 ppm h), high (>200,000 ppm h), which were chosen to provide approximately even distribution of studies across the subgroups. Fourteen comparisons could not be allocated a concentration (due to either utilizing a non-inhalation route or because it was a human study of inhalation and concentration was not measured) and did not have exposure hours and, therefore, could not have total toluene exposure computed.
Each study characteristic was assigned subgroups (e.g. sex: male and female), and χ 2 tests were used to quantify the differences between subgroups for each study characteristic. Characterization of age at first exposure was into either adolescent or adult and was based on data extracted from studies; some studies directly stated adult/adolescent, whereas others stated postnatal day and/or body weight, from which the category was calculated. 53 Pearson’s correlation was used to explore the relationship between concentration and exposure for the identified experimental models. There are limited published data available for toluene exposure, and owing to incomplete data in all categories, a meta-regression could not be undertaken, which would allow the relationship between study characteristics to be explored more fully. The small number of studies also did not provide sufficient power for the inclusion of funnel plots to assess publication bias. The approach taken was therefore exploratory in nature, estimating the effects of the various study characteristics. All p values for individual comparisons (seven in total) were therefore reported, without multiplicity correction, and all Cohen’s d results are reported with the 95% confidence interval in brackets.
Results
Overall effect sizes and study characteristics
A summary of the study characteristics and effect sizes is provided in Table 2. The overall effect size on body weight was −0.73 [95% CI −0.99, −0.47], with I 2 of 76% categorized as high heterogeneity (Figure 2), and the overall effect size on height was −0.69 [95% CI −1.16, −0.21], with I 2 of 65% categorized as moderate–high heterogeneity (Figure 3), indicating that exposure to toluene resulted in significantly decreased body weight and height, though the relatively high heterogeneity suggests that subgroups may exist that affect the outcome. 50
Summary of study characteristics and effect sizes used in the meta-analysis.a

IP: intraperitoneal.
aThirty-one studies were included in the meta-analysis, yielding 64 unique toluene-to-control comparisons for body weight and 6 unique comparisons for height. ID is the study number followed by the comparison number. The characteristics of each toluene-to-control comparison are outlined in Table 2, along with the mean effect size and 95% confidence interval in brackets. Boxes are greyed out when there are no data available. Total toluene exposure was first calculated by multiplying concentration and exposure hours (ppm h). These were then collapsed into categories based on low (0–50,000 ppm h), medium (50,001–200,000 ppm h), high (>200,000 ppm h).

Exposure to toluene impaired body weight. Bars represent the effect size for each study with a 95% confidence interval. Multiple toluene-to-control comparisons (c) existed within a single study, and these are noted as c1, c2, and so on.

Exposure to toluene impaired height. Bars represent the effect size for each study with a 95% confidence interval. Multiple toluene-to-control comparisons (c) existed within a single study, and these are noted as c1, c2, and so on.
The effect of toluene concentration on body weight and height
As hypothesized, toluene concentration was a clinically meaningful moderator of body weight (χ 2 = 25.50, df = 3, p < 0.00001), as shown in Figure 4, with a full forest plot in Online Supplemental Figure 1. Clear separation between the 95% confidence intervals was observed between the upper and lower concentration subgroups, suggestive of a concentration–response relationship, though there was overlap between the confidence intervals of the two middle subgroups. The lowest concentration subgroup (0–500 ppm) had no observable effect on body weight, with decreases in body weight observed in the three upper concentration subgroups. Furthermore, when concentration was differentiated post hoc by occupational-level exposure (≤1000 ppm) or inhalant abuse exposure (>1000 ppm), this was also a significant moderator of body weight (χ 2 = 25.81, df = 1, p < 0.00001). Only inhalant abuse exposure had an effect on body weight (Online Supplemental Figure 2), with no observable effect on body weight for occupational-level exposure. The 14 comparisons that could not be ascribed a concentration in ppm were not able to be included in these subgroup analyses. In addition, due to the unavailability of data in all categories for height, these analyses could not be completed for height as an outcome.
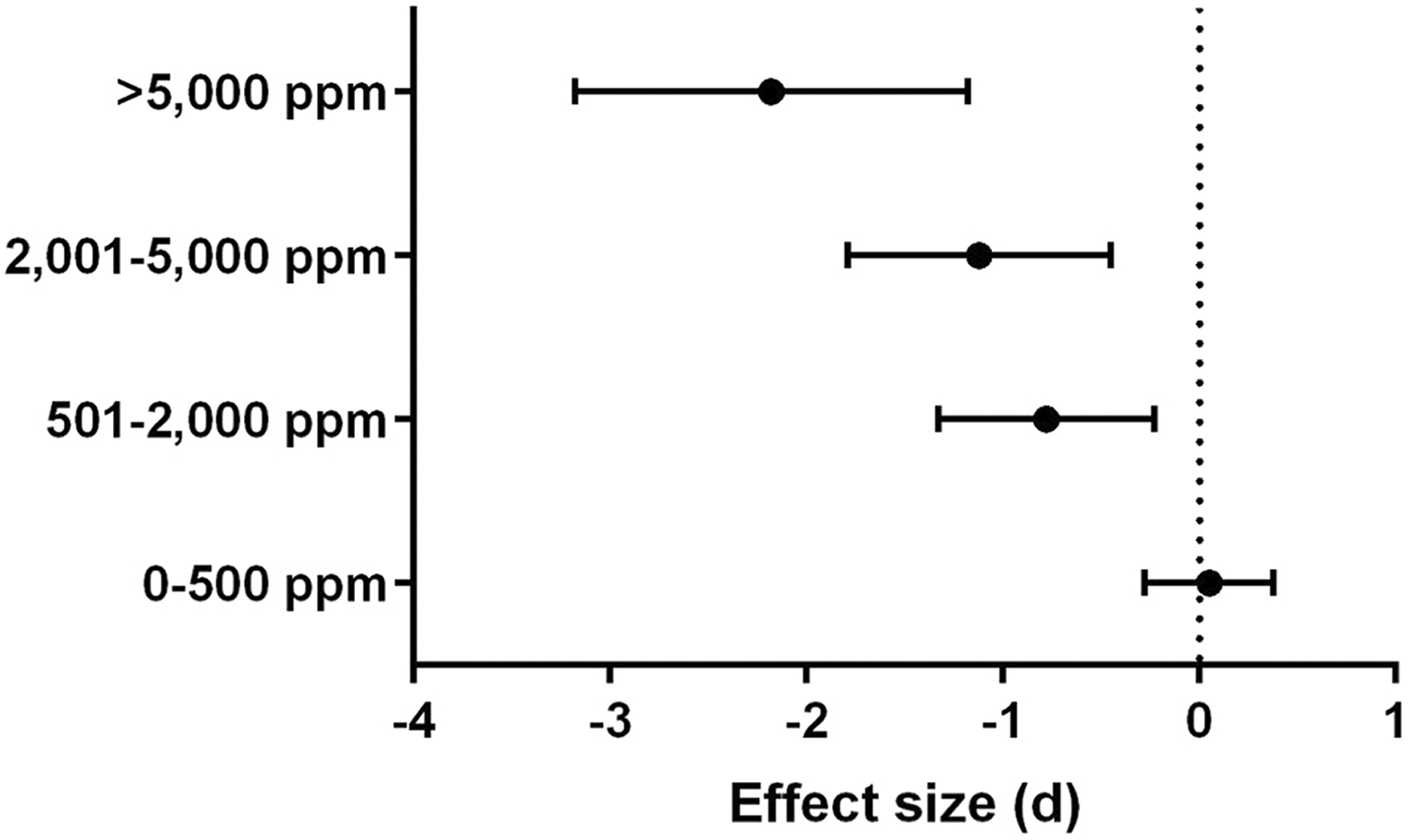
As toluene concentration increased, body weight was more impaired. The mean effect size for each subgroup within toluene concentration is shown with a 95% confidence interval. Only studies that cited a specific concentration of toluene (in ppm) were included, which excluded 14 comparisons. Inhaled concentration was a significant moderator of body weight, with the highest negative effect size observed at the highest concentration.
Estimating the effect of study characteristics on body weight
The effect of all study characteristics on body weight was investigated, and the findings are summarized in Table 3 and Figure 5 (full forest plots in Online Supplemental Figures 3 to 8).
The effects of toluene exposure on body weight, by subgroup.

IP: intraperitoneal; k = the number of effect sizes (i.e. the number of toluene-to-control comparisons); I 2 = heterogeneity.
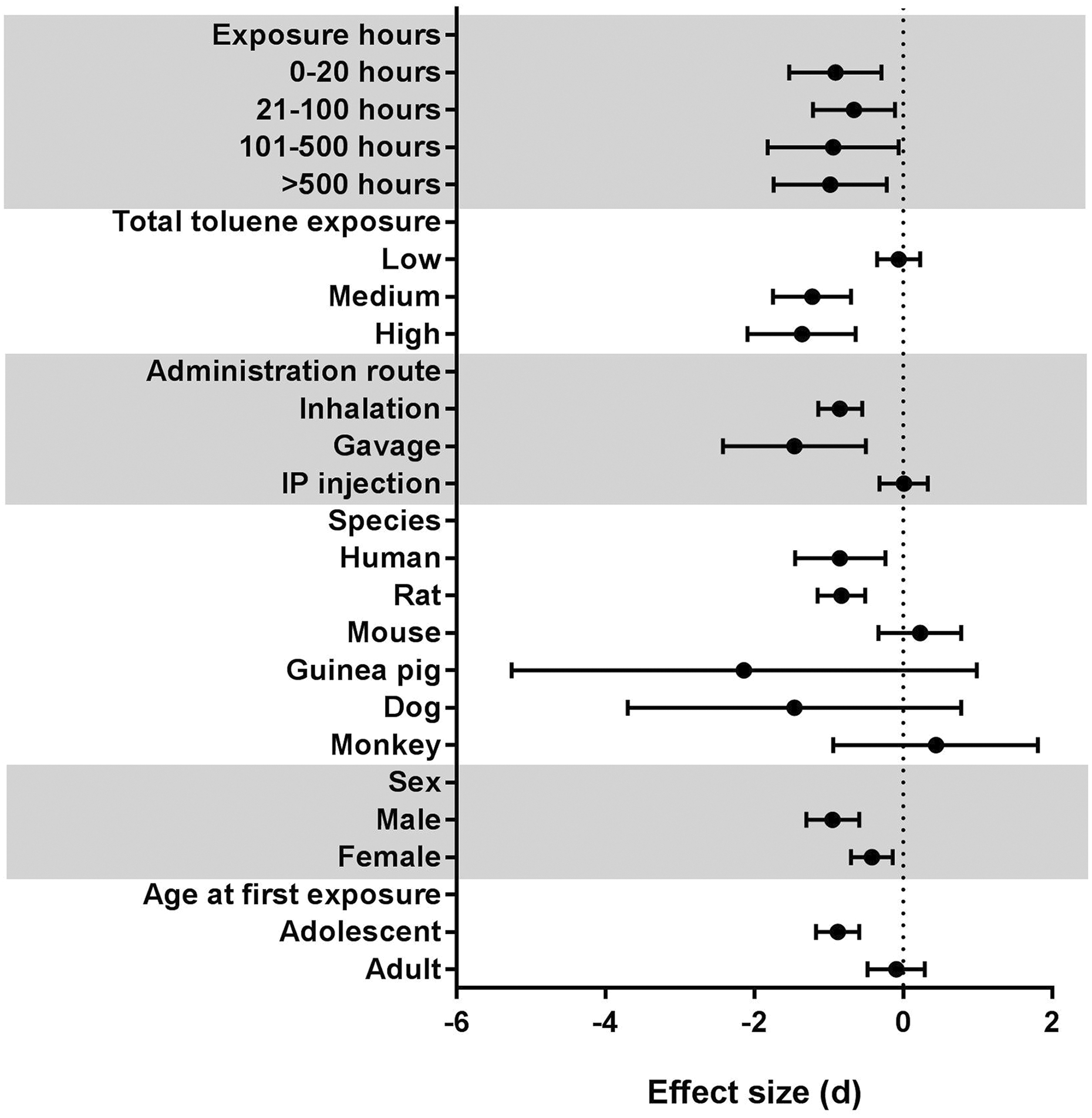
The effect size of toluene exposure on body weight differed by study characteristics, such as exposure method, species and age at first exposure. The effect of each study characteristic on body weight was investigated and the mean effect size for each subgroup within the study characteristic is shown with a 95% confidence interval. For exposure hours and total toluene exposure, only studies that cited a specific concentration of toluene (in ppm) were included, which excluded 14 comparisons.
There was not a clear relationship between exposure hours and body weight (χ 2 = 0.65, df = 3, p = 0.89, Table 3, Figure 5 and Online Supplemental Figure 3), and overlaps between the confidence intervals were observed. However, there was a significant negative relationship (Pearson’s correlation coefficient = −0.366, p = 0.009) between concentration and exposure hours in the experimental models within the studies, and the highest concentrations only occurred in the lowest exposure subgroup (Figure 6). Total toluene exposure was computed to account for this relationship, and increased total toluene exposure was associated with decreased body weight (χ 2 = 21.55, df = 2, p < 0.00001, Table 3, Figure 5 and Online Supplemental Figure 4), with the low exposure subgroup having no very small effect on body weight; however, there was overlap between the confidence intervals of the medium and high subgroups, both of which showed large effect sizes.

There is an inverse relationship between exposure hours and concentration (r = −0.3657 p = 0.009, Pearson’s correlation test) within the experimental models used for the identified studies, demonstrating that the highest concentrations of toluene have the shortest exposure hours, and vice versa. This analysis excluded the 14 comparisons that did not report a toluene concentration (in ppm).
Administration route affected body weight (χ 2 = 18.42, df = 2, p = 0.0001, Table 3, Figure 5 and Online Supplemental Figure 5), with toluene administered via IP injection having no effect on body weight, but decreases being observed for both inhalation and gavage, with large effect sizes. Differences in the effect of toluene exposure on body weight were also observed for both species (χ 2 = 14.95, df = 5, p = 0.01, Table 3, Figure 5 and Online Supplemental Figure 6) and sex (χ 2 = 5.32, df = 1, p = 0.02, Table 3, Figure 5 and Online Supplemental Figure 7). Body weight decreases, with large effect sizes, were observed for humans and rats following toluene exposure, but results were inconclusive for dogs and guinea pigs exacerbated by the low number of studies for comparison, and results for monkeys and mice trended towards an increased body weight following toluene exposure, despite all these species undergoing exposure via inhalation. While both males and females showed a decrease in body weight following toluene exposure, the effect size was greater for males than females. Age at first exposure also had an effect on body weight (χ 2 = 10.28, df = 1, p = 0.001, Table 3, Figure 5 and Online Supplemental Figure 8), with adolescents showing decreased body weight following toluene exposure, whereas there was no clear effect on body weight for adults, and the confidence interval crossed zero.
Estimating the effect of study characteristics on height
Because of the small number of comparisons for height, full analysis of all study characteristics could not be undertaken. No characteristic had all subgroups represented, and the only characteristics for which there was more than one subgroup represented for height were species (human and rat), concentration (2001–5000 ppm and >5000 ppm) and exposure hours (101–500 h and >500 h). These results are not shown, due to the incompleteness of the data set.
Discussion
We conducted a systematic review and meta-analysis of the inhalant abuse literature, incorporating the use of toluene exposure as an experimental model of inhalant abuse and including both human and animal studies. We demonstrate that inhalant abuse in humans and toluene exposure in animal models are associated with significant impairments to both body weight and height, with effect sizes that would be categorized as medium/large according to Cohen. 54 Furthermore, the results indicate a concentration–response relationship between inhaled toluene concentration and body weight impairment, with low concentrations (<500 ppm) having no significant effect on body weight. These changes to growth have the potential to affect an individual’s long-term health outcomes, through both physiological and psychological consequences. 17 –19
The finding that toluene concentration is a clinically meaningful moderator of the effect of toluene on body weight is juxtaposed by the non-significant effect of exposure hours, with similar medium–large effect sizes observed across the four subgroups. This may be due to the experimental design, where exposure to the highest concentrations have shorter exposure hours, thus this result may be confounded by the uneven distribution of concentrations within those experimental models. We attempted to overcome this issue by computing total toluene exposure, as a way of combining both toluene concentration and the duration of exposure. Total toluene exposure affected body weight more significantly than exposure hours (time) alone, a finding consistent with Bowen et al. 55 ; however, there was no expected distinction between the medium and the high exposure subgroups. This suggests that while both toluene concentration and time in combination (i.e. total toluene exposure) affect body weight, the relative importance of these two factors requires further exploration.
The additional study characteristics of administration route, species, sex and age at first exposure meet the threshold for significance with p values less than 0.05; however, these results should be interpreted with caution. Some findings are consistent with the literature; for example, toluene exposure has been previously associated with weight gain in mice. 52 It would also be expected that toluene would have a larger effect during adolescence, when growth is accelerating, as opposed to adulthood when growth (particularly height) is more stable. However, data are not consistently available across all subgroups. For example, in the systematic review, the studies with mice utilized only adult animals, whereas the guinea pig, dog and monkey data all came from adolescent animals. Therefore, we cannot identify whether it is species, age at first exposure or both that is affecting the body weight. Furthermore, having identified a concentration–response relationship, we found differences in the concentrations of toluene that different subgroups within each study characteristic were exposed to. For example, in the highest inhaled concentration subgroup of >5000 ppm, there are only males and no females, only adolescents and no adults and only rats and no other animal species. Given that higher effect sizes were observed in males, adolescents and rats, it is possible that this is simply reflecting the response to the high concentration of toluene, rather than sex, age and species moderating the relationship between toluene exposure and growth changes.
It is important that valid animal models of toluene exposure exist, which reflect human patterns of inhalant abuse and which yield comparable outcomes. The differences between effect sizes for administration route are of concern, particularly as toluene given via IP injection resulted in no significant effect on body weight. The data for gavage is difficult to interpret due to the low number of studies, but exposure via inhalation appears to yield results more consistent with clinical outcomes. Rats are the most common animal model used for inhalant abuse, and it is positive to note the large overlap in the confidence intervals between rats and humans, suggesting that they appear to be a valid experimental model. However, as noted, at the highest concentrations, only male rats are represented. Given that inhalant abuse has similar prevalence across sexes, 56 future animal experiments need to consider whether the concentration–response relationship between male and female rats is equivalent. This is particularly relevant as only one human inhalant abuse study could be found that directly compared the growth effects of inhalant abuse between males and females and showed that weight impairments in females were more severe than in males. 47 The lack of studies using females is particularly concerning because a weight impairment and any associated loss of adiposity 11 during adolescence can cause disruptions to pubertal and metabolic processes due to the endocrinological activity of adipose tissue. 57 Furthermore, as shown by Callan et al., exposure to toluene during pregnancy can impair developmental body weight, 52 and inhalant abuse is most prevalent in females during childbearing years. 2,3 Thus, inhalant abuse in females has the potential to affect not only the individual but also her offspring.
The effects of inhalant abuse in females have been explored before in a meta-analysis that considered whether toluene exposure during the prenatal period affected birth weight of the offspring. 52 This meta-analysis only included animal studies, and the only growth outcome was birth weight; however, many of the study characteristics explored were the same. The average effect size for weight in the individual from this meta-analysis was −0.73, compared to −0.39 for the effect on birth weight following prenatal exposure. 52 This suggests that the growth impacts of inhalant abuse may be more severe in the exposed individual than to a developing foetus. In contrast to our findings, Callan et al. did not identify a linear concentration–response relationship and observed a reversal of the weight impairment trend at the highest concentration of inhaled toluene but did find a linear relationship between total toluene exposure and weight impairments. 52 This may reflect the fact that, due to the distribution of the available data, the same subgroups for total toluene exposure could not be used between the two meta-analyses. It is also possible that for the exposed individual, toluene concentration is a more meaningful variable, whereas for a developing foetus, it is the total toluene exposure that causes the greatest effect on birth weight. Consistent with our findings, Callan et al. found that toluene exposure resulted in increased birth weight for mice, compared to rabbits and rats, suggesting that mice are not a good experimental model for exploring the growth effects of inhalant abuse. 52
To understand the relationship between inhalant abuse and growth more fully, there are a range of experiments that would assist in filling the identified knowledge gaps, and while studies may measure weight, weight data are not always included in the publication but is a valuable output to report on. Firstly, experiments should be conducted to understand the relative importance of concentration versus exposure time, within total toluene exposure. Secondly, experiments should be done to understand whether sex, age at first exposure and species do moderate the effect of toluene exposure on body weight, by conducting experiments in which all subgroups have equivalent total toluene exposure. Thirdly, it would be beneficial for future experiments to also consider height as a growth outcome, which can be easily measured in experimental animals (crown-to-rump length) but is often missed as a descriptive outcome. This would allow better understanding of how toluene exposure affects height and what moderates this effect.
Weight impairment and emaciation can result in a diverse range of harmful outcomes including impaired cognition, renal failure and osteoporosis. 17 Weight impairment may normalize to re-establish homeostasis if the factor suppressing weight is removed. 58 However, this process is associated with rapid weight regain (predominantly through increased fat deposition), which then increases the risk of subsequent health issues such as visceral adiposity and insulin resistance. 18 Thus, weight impairments can cause long-term health impacts to individuals and are therefore an important clinical outcome for individuals with a history of inhalant abuse. Future studies should focus on adolescence as the predominant time for inhalant use, 2,3 overlapping with a critical growth period, and seek to incorporate subsequent outcomes such as adiposity and insulin resistance, as well as the persistence of weight and height effects in abstinence, to enable a better understanding of the long-term health consequences of inhalant abuse.
It should be noted that one limitation of this meta-analysis is the low number of comparisons, particularly for the height outcome, and the uneven distribution of comparisons across the various subgroups, which makes interpreting the effects of study characteristics difficult. As we were unaware at the time of PROSPERO registration whether there would be sufficient studies to conduct a meta-analysis and/or meta-regression, the methodology was not prospectively registered, which is a further limitation of this study. Overall, the risk of bias was difficult to assess, with unclear results for the majority of studies within the performance domain, predominantly attributable to lack of detailed methods on allocation procedures, housing protocols and blinding. Due to lack of preregistered protocols for any of the included studies, there is a risk of bias due to selective outcome reporting; however, for the majority of the studies, body weight and height were not the primary outcomes of investigation; therefore, this may not be a major limitation for this study. We also acknowledge the difficulties inherent in comparing animal studies to human studies, particularly due to the range of substances (e.g. glue, paint, petrol, thinners, etc.) that humans may inhale. Although all these products contain toluene, there may be unknown substance-specific effects. Defining the level of exposure is difficult, given the multiple combinations between the amount of toluene exposure, the duration of that exposure and how long it occurred for. Although we attempted to address these issues through computation of exposure duration and total toluene exposure, this approach does not take into account peak blood concentrations of toluene (data that would be valuable but is not cited in these studies), and thus a detailed analysis of the pharmacokinetics of toluene under the variety of exposure models could not be undertaken. A further limitation of this study is tlack of long-term health impact data arising from the changes to growth, which means that while we can quantify the effect size on weight and height, we are unable to relate that to any long-term health outcome such as adiposity or insulin resistance. This study does not provide mechanistic explanation for the effect of toluene on body weight and height, which means that we are unable to comment on whether the effect on height and weight are common correlated outcomes of the same mechanism or distinct outcomes arising from different mechanisms. However, understanding the mechanisms behind these effects on body weight and height, including the impact of changes to food intake following toluene exposure 11,13 is necessary and will provide information that is relevant to the treatment of those with a history of inhalant abuse.
In conclusion, this systematic review and meta-analysis forms a comprehensive summary of the human and animal research literature on the growth effects of inhalant abuse and toluene exposure (used as a model of inhalant abuse). We have shown a medium–large negative effect of toluene exposure on body weight, which confirms weight impairment as a warning sign of inhalant abuse. Indeed the effect on body weight is such that it has been previously associated with FTT in humans, and furthermore, identification of FTT can be a strong diagnostic marker for inhalant abuse in relevant clinical populations. 6 We also show that toluene exposure has a medium–large negative impact on height, and despite limited studies, the effect size is similar to that of body weight. It is therefore recommended that height impairment be added to the warning signs of inhalant abuse and as a potential deterrent to adolescents, for whom height impairment compared to their peers may be an undesirable outcome. We show that rats and exposure by inhalation show results consistent with human findings, but other species and administration routes should be utilized with caution, until future experimental research has tested whether these study characteristics do moderate the effect of toluene on growth. The effect of sex is also under-studied, making it difficult to extrapolate these findings to females in the clinical setting. Additional outcomes that arise from growth changes, for example, insulin resistance and adiposity should be included in future studies. Thus, knowledge gaps need to be filled in order to understand potential long-term health risks, stemming from growth impairments, for individuals with a history of inhalant abuse.
Supplemental material
Supplemental Material, Crossin_R_supplementary_figures - Growth changes after inhalant abuse and toluene exposure: A systematic review and meta-analysis of human and animal studies
Supplemental Material, Crossin_R_supplementary_figures for Growth changes after inhalant abuse and toluene exposure: A systematic review and meta-analysis of human and animal studies by R Crossin, AJ Lawrence, ZB Andrews, L Churilov, and JR Duncan in Human & Experimental Toxicology
Supplemental material
Supplemental Material, Crossin_R_supplementary_figure_9 - Growth changes after inhalant abuse and toluene exposure: A systematic review and meta-analysis of human and animal studies
Supplemental Material, Crossin_R_supplementary_figure_9 for Growth changes after inhalant abuse and toluene exposure: A systematic review and meta-analysis of human and animal studies by R Crossin, AJ Lawrence, ZB Andrews, L Churilov, and JR Duncan in Human & Experimental Toxicology
Supplemental material
Supplemental Material, Crossin_R_supplementary_table_1_revised - Growth changes after inhalant abuse and toluene exposure: A systematic review and meta-analysis of human and animal studies
Supplemental Material, Crossin_R_supplementary_table_1_revised for Growth changes after inhalant abuse and toluene exposure: A systematic review and meta-analysis of human and animal studies by R Crossin, AJ Lawrence, ZB Andrews, L Churilov, and JR Duncan in Human & Experimental Toxicology
Supplemental material
Supplemental Material, Crossin_R_supplementary_table_2_revised_(1) - Growth changes after inhalant abuse and toluene exposure: A systematic review and meta-analysis of human and animal studies
Supplemental Material, Crossin_R_supplementary_table_2_revised_(1) for Growth changes after inhalant abuse and toluene exposure: A systematic review and meta-analysis of human and animal studies by R Crossin, AJ Lawrence, ZB Andrews, L Churilov, and JR Duncan in Human & Experimental Toxicology
Footnotes
Author contributions
The study was conceived by RC, AJL, ZBA and JRD. The systematic review was undertaken by RC. RC and LC designed the meta-analysis, which was conducted by RC and checked by LC. All authors were involved in reviewing the manuscript drafts and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of following financial support for the research, authorship and/or publication of this article: This research was supported by the Australian National Health and Medical Research Council (NHMRC) (940835), of which AJL is a Principal Research Fellow (1116930) and ZBA is a Career Development Fellow (1084344) and the Victorian Government’s Operational Infrastructure Support Scheme. RC is funded by a federal RTP scholarship. Funding bodies had no involvement in the design, analysis and decision to publish.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
