Abstract
Objective:
To investigate the therapeutic effect and mechanism of sodium tanshinone IIA sulfate (STS) on paraquat (PQ)-induced myocardial injuries in a rat model.
Methods:
Healthy adult Sprague Dawley rats were randomly divided into normal control, PQ, and PQ + STS groups. PQ group was given a single intragastric administration of PQ (80 mg/kg). PQ + STS group was intraperitoneally injected with STS (1 ml/kg) at 30 min following PQ exposure. Rats in control and PQ groups were injected with equal amount of saline. After 12, 24, 48, and 72 h, rats were killed, and the apoptosis of myocardial cells was detected. Myocardial expression of Bax and Bcl-2 was measured. The activity of the nuclear erythroid 2-related factor 2 (Nrf2) pathway was assessed by Western blot.
Results:
The apoptotic cells in PQ group were significantly increased in a time-dependent manner compared with the control group (p < 0.01). The rats in PQ group exhibited significantly lower Bcl-2 expression, but notably higher Bax expression at 12, 24, 48, and 72 h after PQ exposure (p < 0.05 or 0.01). STS intervention markedly reduced the proportion of apoptotic myocardial cells, increased Bcl-2 expression, and decreased Bax expression at 24, 48, and 72 h after treatment (p < 0.05 or 0.01). The expression of phosphorylated Nrf2 and heme oxygenase 1 in PQ + STS group was significantly increased compared with PQ and control groups (p < 0.05 or 0.01).
Conclusion:
STS effectively inhibits PQ-induced myocardial cell apoptosis in rats via modulating the Nrf2 pathway, suggesting its potential as a promising therapeutic agent for PQ-induced myocardium damage.
Introduction
Paraquat (PQ; 1,1′-dimethyl-4,4′-bipyridinium) is an important fast-acting broad-spectrum contact herbicide that has been widely used since 1960s. 1 The chemical is toxic through ingestion, inhalation, or dermal exposure. 2 PQ, when ingested orally, is highly toxic to mammals including humans, leading to lung, heart, liver, and kidney failure and even death. 3 The reported oral lethal dose (LD50) value of PQ ranges from 30 mg/kg to 150 mg/kg in mammals. 4 PQ is easily available and inexpensive, and there are currently no specific medical treatments available, which makes poisoning management extremely difficult. 5 The incident of PQ poisoning due to accidental or intentional ingestion has been reported each year, leading to a fatality rate between 60% and 80%. 6,7
Although the mechanism of PQ toxicity has not been fully elucidated, it has been suggested that the chemical initiates a sustained redox-cycling reaction to generate a large amount of reactive oxygen species (ROS), leading to oxidative stress and extensive inflammation. 8 Excess ROS and the resulting oxidative stress induce oxidative modification of cellular macromolecules, inhibit protein function, and ultimately promote apoptosis; increasing evidences support that oxidative stress and apoptosis are closely associated with physiological phenomena; Bcl-2, a member of the Bcl family, has been shown to prevent cell apoptosis via an anti-oxidative mechanism. 9,10 Recent studies have also shown that PQ impairs myocardial contractile function through the generation of ROS, which ultimately results in cardiopulmonary failure. 11,12 Numerous researches have attempted to develop efficient treatments for PQ poisoning, but so far have achieved disappointing clinical therapeutics. 13 –16 Therefore, alternative treatment agents for acute PQ poisoning are urgently required for clinical practice.
Medicinal plants have provided a rich source for the screening of novel therapeutic agents. 17 –19 Salvia miltiorrhiza, also known as Danshen, is a traditional Chinese medicine that has been commonly used for centuries for improving microcirculation. 20,21 Tanshinone IIA (TIIA), one of the active components of Salvia miltiorrhiza, might exert a wide range of strong biochemical effects, including anti-oxidant and anti-inflammatory effects. 22 –24 Sodium tanshinone IIA sulfate (STS), a clinical formulation of TIIA, is suggested to protect myocardium and blood vessels against ischemia/reperfusion injury during ischemic heart diseases and ischemic stroke. 25,26 However, whether STS produces protective effects on myocardium against PQ-induced toxicity is not yet clear. In this study, we aimed to evaluate the therapeutic effect of STS on PQ-induced myocardial dysfunction in a rat model after PQ ingestion. We further investigated the underlying mechanism of the effects of STS in this model by analyzing the key apoptosis-related markers (Bax and Bcl-2) and the proteins in the nuclear erythroid 2-related factor 2 (Nrf2) pathway.
Material and methods
Main reagents
STS injection (Nuoxinkang) was purchased from Shanghai No. 1 Biochemical & Pharmaceutical Co., Ltd (Shanghai, China). PQ solution (20%) was purchased from Biobest Biotech (Chongqing, China). Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay kit was purchased from Beyotime Institute of Biotechnology (Shanghai, China). RNA extraction kit, PrimeScript RT-PCR kit, and polymerase chain reaction (PCR) amplification kit were purchased from Takara (Japan). The primers used for PCR amplification were synthesized by Promega (Shanghai, China): β-actin-forward: 5′-GGCTGTATTCCCCTCCATCG-3′, β-actin-reverse: 5′-CCAGTTGGTAACAATGCCATGT-3′; Bax-F: 5′-CCAGGATGCGTCCACCAA-3′, Bax-R: 5′-AAAGTAGAAGAGGGCAACCAC-3′; and Bcl-2-F: 5′-GTGGCCTTCTTTGAGTTCG-3′, Bcl-2-R: 5′-ACCCAGCCTCCGTTATCC-3′. Protein extraction kit and BCA kit were purchased from Beyotime Institute of Biotechnology. Mouse anti-rat Bcl-2 and Bax monoclonal antibody, mouse anti-rat β-actin polyclonal antibody, and horseradish peroxidase (HRP)-labeled rabbit anti-mouse immunoglobulin G (IgG) were purchased from Abcam (Cambridge, Massachusetts, USA).
Animals and grouping
This animal study was approved by the Research Ethics Committee at our hospital. Healthy adult Sprague Dawley male rats weighting 220 ± 40 g were purchased from the Department of Animal Science of the School of Medicine, Nanchang University and housed in standard laboratory conditions. These rats were randomly divided into three groups: control (n = 24), PQ (n = 40), and PQ + STS group (n = 40). The rats in PQ group were given a single intragastric administration of PQ (80 mg/kg). The control group was intragastrically administered with equal amount of saline. The PQ + STS group was given a single intraperitoneal injection of STS (1 ml/kg) at 30 min following PQ exposure. Rats in control and PQ groups were intraperitoneally injected with equal amount of saline. Based on our preliminary experiments and previous studies, 27,28 rats exhibit severe cardiac oxidative damage at approximately 72 h after intragastric exposure to 80 mg/kg PQ, and death rate begins to increase thereafter. We, therefore, monitored the condition of rats within the first 72 h after PQ toxicity. At 12, 24, 48, and 72 h after PQ exposure, six randomly selected rats from each group were anesthetized and killed by abdominal aorta bloodletting. The heart was obtained from each rat and stored at −70°C until use.
TUNEL assay
The heart tissue in each group was fixed in 10% formalin, treated with xylene, and dehydrated with serial ethanol. The middle part of the left ventricle coronal was cleared in chloroform, embedded in paraffin, and cut into 4-µm sections. The sections were stained using TUNEL assay kit following the manufacturer’s instructions. The number of positive cells in randomly selected five visual fields was counted and the apoptotic index was calculated as the mean percentage of positive cells.
Western blot analysis
The expression of Bax, Bcl-2, and proteins in the Nrf2 pathway including phosphorylated Nrf2 (p-Nrf2) and heme oxygenase 1 (HO-1) in the heart tissues was detected by Western blot. Briefly, the heart tissue was homogenized. Total protein was extracted using protein extraction kit and quantified using a BCA kit following the manufacture’s instruction. Equal amounts of total protein (10 μg) were separated by 10% sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) electrophoresis and transferred to polyvinylidene difluoride membranes. The membrane was blocked with 5% skim milk overnight at 4°C and incubated with the appropriate primary antibody (Abcam) at 37°C for 4 h. The membranes were washed with tris-buffered saline plus Tween 20 and incubated with HRP-labeled secondary antibody (1:100) at 37°C for 1 h. The membrane was subjected to enhanced chemiluminescence detection. The intensity of bands was detected by a Molecular Imager® ChemiDocTM XRS System (Bio-Rad Laboratories) and analyzed by Image Lab 2.0 software (Bio-Rad Laboratories, Hercules, CA, USA). The experiment was performed in triplicates. The relative expression of protein was calculated using β-actin as the internal control.
Reverse transcription PCR
The expression of Bax and Bcl-2 mRNA in the heart tissues was measured by reverse transcription PCR (RT-PCR). Briefly, total RNA was extracted using RNA extraction kit and reverse transcribed into cDNA using reverse transcription kit according to the manufacture’s instructions. PCR amplification was performed using cDNA as a template and PCR Master Mix (TaKaRa, Japan) following the manual. The reaction condition is 95°C for 5 min, followed by 35 cycles of 95°C 10 s, 49°C 30 s, and 72°C 30 s, and 72°C for 10 min. PCR products were analyzed by 1% agarose gel electrophoresis. The relative expression of mRNA was expressed as its intensity to that of β-actin. The experiment was performed in triplicates.
Statistical analysis
All data were represented as mean ± standard deviation and analyzed using SPSS 13.0 (SPSS Inc, Chicago, Illinois, USA). Difference among groups was analyzed by one-way analysis of variance followed by Student-Newman-Keuls tests (SNK) in case of significant difference. The value of p < 0.05 is considered statistically significant.
Results
STS reduced the apoptosis of myocardial cells induced by PQ exposure
The apoptosis index of myocardial cells in different groups was compared by TUNEL assay. As shown in Figure 1, the apoptotic cells in PQ group was significantly increased in a time-dependent manner compared with the control group (p < 0.01). STS intervention markedly reduced the proportion of apoptotic cells induced by PQ toxicity at 24, 48, and 72 h after treatment (p < 0.05 or 0.01). The apoptosis index in PQ + STS group was similar to that in control group at 48 and 72 h (p > 0.05).

Comparison of apoptosis index of myocardial cells in different groups by TUNEL assay at 12, 24, 48, and 72 h after PQ exposure (n = 6). *p < 0.05 and **p < 0.01 compared with control group; # p < 0.05 and ## p < 0.01 compared with PQ group. TUNEL: terminal deoxynucleotidyl transferase dUTP nick-end labeling; PQ: paraquat.
STS modulated the expression of Bax and Bcl-2 protein
The expression of apoptosis-related proteins (Bax and Bcl-2) was compared by Western blot. As shown in Figure 2, the rats in PQ group exhibited significantly lower Bcl-2 expression but notably higher Bax expression at 12, 24, 48, and 72 h after PQ exposure (p < 0.05 or 0.01). Bcl-2 expression in PQ + STS group was significantly higher, but Bax expression was substantially lower compared with PQ group at the same time point (p < 0.05 or 0.01), suggesting that STS treatment time-dependently increased Bcl-2 expression and decreased Bax expression.

Comparison of the expression of apoptosis-related proteins in different groups by Western blot at 12, 24, 48, and 72 h after PQ exposure (n = 6). (a) A representative image of Western blot, (b) Bcl-2 protein, and (c) Bax protein. *p < 0.05 and **p < 0.01 compared with control group; # p < 0.05 and ## p < 0.01 compared with PQ group. PQ: paraquat.
STS regulated the expression of Bax and Bcl-2 mRNA
The expression of Bax and Bcl-2 mRNA was further determined by RT-PCR. As shown in Figure 3, the change trend of Bax and Bcl-2 mRNA expression in PQ and PQ + STS groups was similar to that of their corresponding proteins during the experiment, indicating that STS intervention significantly increased Bcl-2 expression and decreased Bax expression in a time-dependent manner.
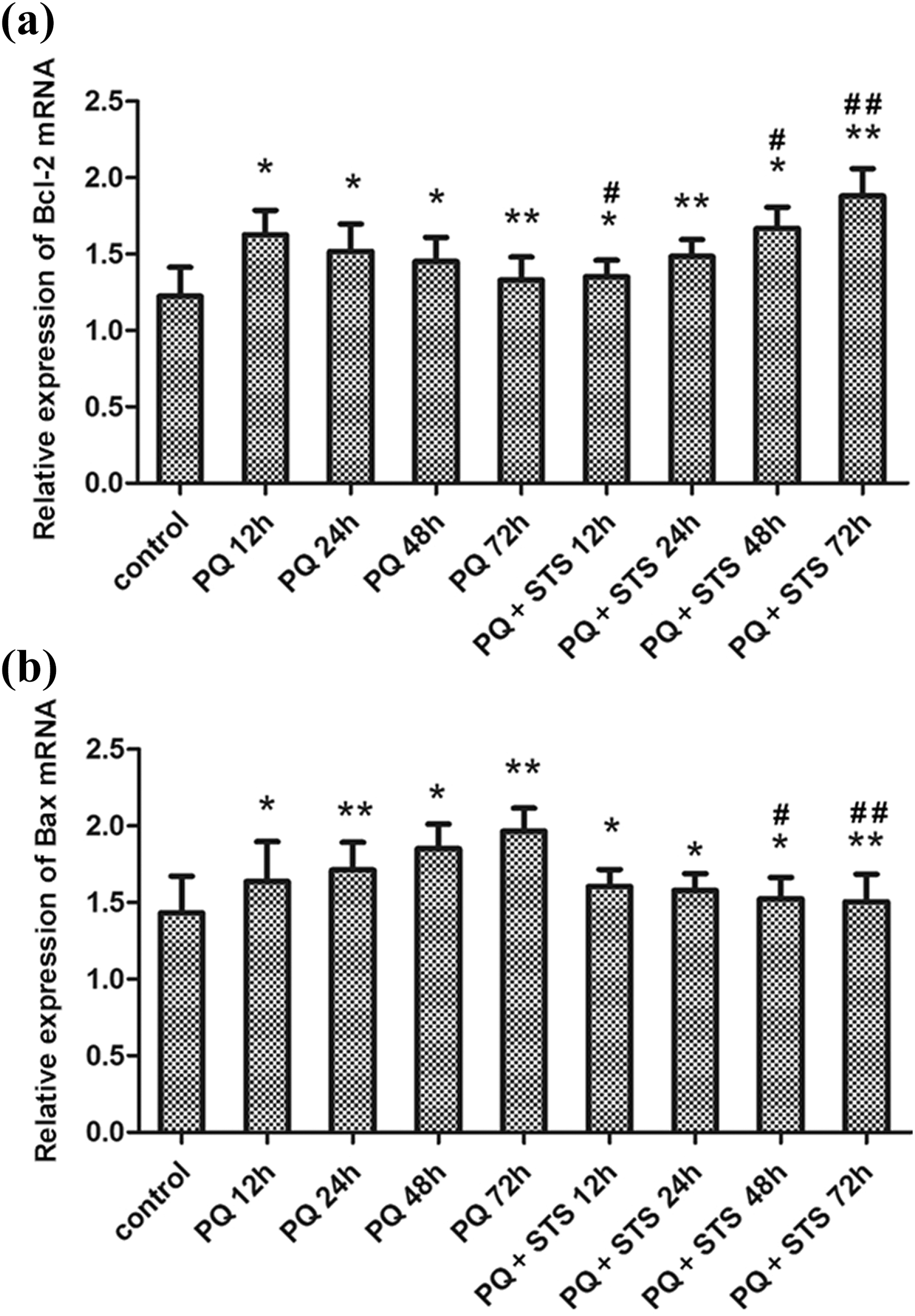
Comparison of the expression of (a) Bcl-2 mRNA and (b) Bax mRNA in different groups by RT-PCR at 12, 24, 48, and 72 h after PQ exposure (n = 6). *p < 0.05 and **p < 0.01 compared with control group; # p < 0.05 and ## p < 0.01 compared with PQ group. RT-PCR: reverse transcription PCR; PQ: paraquat.
STS activated the Nrf2 signaling pathway
The expression of key proteins in the Nrf2 signaling pathway (p-Nrf2 and HO-1) in different groups was measured by Western blot. As shown in Figure 4, the expression of p-Nrf2 and HO-1 in PQ + STS group was significantly increased compared with PQ and control groups at 24, 48, and 72 h (p < 0.05 or 0.01), whereas the expression of both proteins in PQ group was only slightly higher than that in control group at all time points (all p > 0.05). The results indicated that the STS treatment had activated the Nrf2 signaling pathway to protect the mice against PQ-induced toxicity.
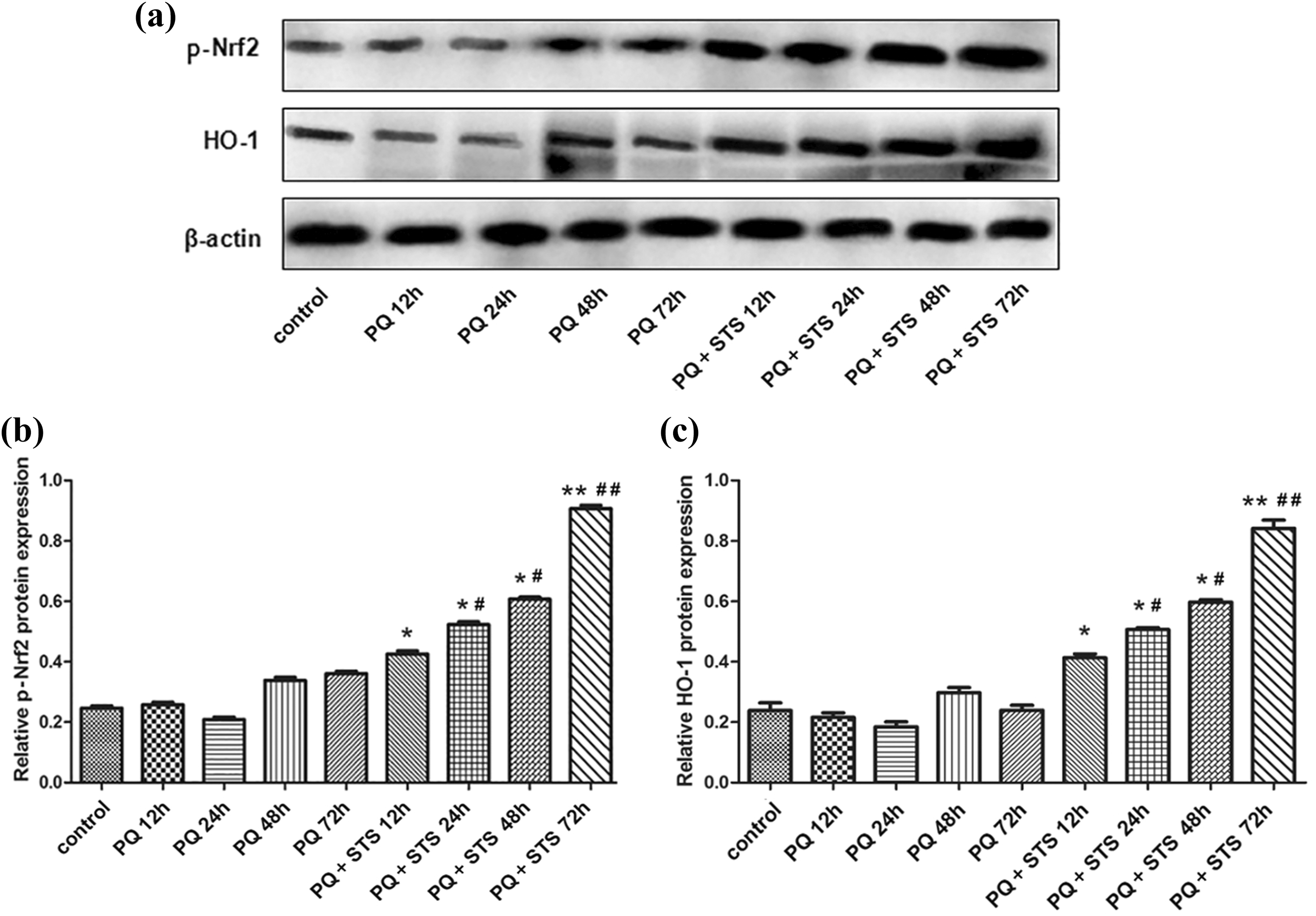
Comparison of the expression of apoptosis-related proteins in different groups by Western blot at 12, 24, 48, and 72 h after PQ exposure (n = 6). (a) A representative image of Western blot, (b) Nrf2 protein, and (c) HO-1 protein. *p < 0.05 and **p < 0.01 compared with control group; # p < 0.05 and ## p < 0.01 compared with PQ group. PQ: paraquat; Nrf2: nuclear erythroid 2-related factor 2; HO-1: heme oxygenase 1.
Discussion
PQ is a widely used herbicide, but may lead to multiple organ damage and failure in humans and animals through ingestion, inhalation, or dermal exposure. 2 Once entering the body, PQ acts as a redox cycling agent, producing a large amount of ROS including superoxide anion radical and hydrogen peroxide and hydroxyl radical. 29 The PQ-induced oxidative stress interrupts several essential biochemical processes in the body and thus impairs the cellular functions in target organs. 30 Multiple studies have shown that PQ can severely damage myocardial functions. 11,12 Consistently, we found that the apoptosis index of myocardial cells in rats was significantly increased at 12, 24, 48, and 72 h after PQ exposure, confirming that PQ had induced myocardial damages.
STS has been suggested as an anti-inflammatory, anti-oxidative, and antiapoptotic agent. 22 –24 Although the protective effect of STS on myocardium against ischemia/reperfusion injury has been frequently studied, 31 –33 whether STS protects myocardium against PQ-induced toxicity remains largely unclear. In this study, we assessed the therapeutic effects of STS on myocardium in a rat model with PQ toxicity. It was found that STS intervention significantly reduced the proportion of PQ-induced apoptotic cells at 24, 48, and 72 h after treatment (p < 0.05 or 0.01). The apoptosis index in PQ + STS group was close to that in control group at 48 and 72 h, suggesting that STS exerted protective effects on myocardial cells.
The Bcl-2 protein family has been well known for its regulation on apoptosis. 34 The mitochondrial apoptosis pathway is controlled by the Bcl-2 family. Bcl-2 (an antiapoptotic member) and Bax (a proapoptotic member) are two major important players in modulating the mitochondrial apoptosis pathway. 35 We, therefore, further investigated the mechanism behind the inhibitory effect of STS on the apoptosis of myocardial cells. We measured the expression of Bcl-2 and Bax and found that STS reduced myocardial cell apoptosis through the enhancement of Bcl-2 and the inhibition of Bax (Figures 2 and 3). These results suggested that the therapeutic effect of STS against PQ-induced myocardial damage was achieved through the inhibition of apoptosis.
Nrf2 is an NF-E2-like basic leucine zipper transcription activator that plays an important role in the defense against oxidative stress. 36 Under normal physiological conditions, the cytoplasmic adapter protein Keap1 binds to Nrf2 and prevents its translocation to the nucleus, whereas oxidative stresses trigger the phosphorylation of Nrf2, leading to its dissociation from Keap1 and translocation into the nucleus, where it activates the expression of downstream cytoprotective genes such as HO-1, NQO1 (NAD(P)H: quinone oxidoreductase 1), and so on. 37 The protective effect of the Nrf2 pathway in the defense against oxidative stress-induced injury including PQ toxicity has been frequently reported. 38,39 We, therefore, examined the activity of Nrf2 pathway by Western blot and found that the expression of p-Nrf2 and HO-1 in PQ + STS group was markedly increased compared with PQ and control groups, suggesting that STS had activated the Nrf2 signaling pathway to protect the mice myocardium against PQ-induced toxicity. Our study herein provided a preliminary mechanistic study on the therapeutic effects of STS against PQ toxicity, despite that further in-depth studies are needed.
In summary, our study has shown that STS therapeutically inhibits PQ-induced myocardial cell apoptosis in rats. The associated mechanism was achieved via the enhancement of Bcl-2 and the inhibition of Bax. Our results provide a theoretic basis for the application of STS in the treatment of acute PQ toxicity, despite that further in-depth studies are needed. STS might be a promising therapeutic agent for PQ-induced myocardial damage and failure.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation in Science and Technology Department of Jiangxi Province (no: 20142BBG70092).
