Abstract
Occupational exposure to the waste anaesthetic gases (WAGs) is a crucial problem for healthcare personnel. Cancer is among the potential long-term adverse effects of WAGs. The present occupational molecular epidemiology study was conducted in healthcare personnel (anaesthetists, nurses and technicians; n = 46), working in operating rooms (ORs; n = 34) and recovery units (RUs; n = 12) of the same hospital, to assess the genotoxicity risk of WAGs exposure. Twenty-one healthy available hospital staff allocated to other wards, without the history of working in ORs and RUs were the control group. A micronucleus test was carried out for peripheral blood lymphocytes (PBLs) and buccal epithelial cells (BECs). Exposure to the anaesthetics was assessed with sevoflurane concentrations and inorganic fluoride levels in post-shift urine samples of the healthcare staff. As an exposure marker, sevoflurane concentrations in ORs and RUs were measured using passive samplers. The micronuclei frequencies were increased in both PBLs (approximately two times) and BECs (approximately three times) of the healthcare personnel. Urinary sevoflurane concentrations exceeded the biological equivalent level in 23 personnel. Air sevoflurane levels in the breathing zone in three ORs and one RU did not exceed the established occupational exposure limits. Both in surrogate tissue (PBLs) and in target tissue (BECs) of the personnel of RUs and ORs of the same hospital, the genotoxicity risk was evident and similar. Originality of this study, in addition to the WAGs exposure confirmation of the healthcare personnel, was the involvement of the RU personnel for the genotoxicity assessment, which was the first time in the scientific literature.
Introduction
Despite the improvements in anaesthesiological methods, modern climate control and implementation of gas scavenging systems, occupational exposure to waste anaesthetic gases (WAGs) is still possible for the personnel working in the operating rooms (ORs) and in the recovery units (RUs). 1 Long-term adverse effects of anaesthetic gases may be associated with spontaneous abortion, premature births, infertility, renal and hepatic disease and cancer. 2
Of the cytogenetic biomarkers, high levels of micronucleus (MN) frequencies in peripheral blood lymphocytes (PBLs) may predict an increased cancer risk in humans. 3 The buccal epithelial cells (BECs) supply a complementary method for measuring MN in an easily accessible tissue without tissue culture requirement for monitoring human exposure to occupational and environmental genotoxins. 4,5 BEC is a target site for airborne pollutants and may metabolize proximate carcinogens and demonstrate early genotoxic events. 4,6
Genotoxic effects of the volatile anaesthetic agents have been studied in experimental animals, in patients and in healthcare personnel using several cytogenetic tests. 7 Genotoxicity risks associated with exposure to anaesthetics are controversial due to a number of conditions such as the type of the anaesthetics, exposure conditions and the combination of different anaesthetics agents used. 2
Even though healthcare personnel are exposed to the anaesthetics in lower concentrations compared with the patients, the exposure is continuous and long term to the different or/and combination of anaesthetic agents, and there is no adjacent regulation of WAG especially in RU. As a result, they may have the risk of genotoxicity and other toxic effects. The goal of this occupational molecular epidemiology study is to assess the genotoxicity risk using MN test in two different tissues, namely PBL and BEC of OR and RU personnel potentially exposed to WAG. The present study includes RU personnel for the first time in the literature for genotoxicity risk, making this study unique.
Methods
The study group consisted of 34 healthcare staff (13 anaesthetists, 13 nurses and 8 technicians) employed in three ORs located in gastrointestinal and urologic surgery units of Yüksek Ihtisas Hospital, Ankara, Turkey, with contact to anaesthetics such as N2O and halogenated hydrocarbons such as sevoflurane and desflurane. Study group also comprised 12 nurses working in one RU in the same hospital who had no history of working in OR. The subjects spent about 8 h per day and 5 days per week in the OR and RU. RU personnel did not wear any personal protective equipment and clothing; 21 healthy available hospital staff allocated to other wards, without the history of working in ORs and RUs and comparable to the study group with demographic characteristics were taken as a control group. The sample size of study and control group in this study enabled us to detect a type I error rate of 5% for all outcome variables in the study.
Detailed questionnaires (age, gender, cigarette smoking, body mass index (BMI), recent diagnostic X-ray examination including dental examination (yes/no) prior to the sampling and sports activity information) and informed consents were completed by the study and control groups. This study was approved by the local ethical committee of Ankara, Turkey (17.04.2007 – B.10.4.ISM.4.06.00.15-PER.SB.722.0).
Anaesthesia ventilators were used in semi-closed circuit during maintenance. Operating rooms had an active scavenging system. The volume of OR was 50 m3, air exchange rate was 20 per day, and the exhaust outlets of the anaesthetic machines were connected to the hospital’s central scavenging system with suction flow of 876.6 L/min. Periodic WAG measurements were not available for OR. Air exchange in RU was provided with air conditioning and/or opening the windows.
Biological sampling
BECs and venous blood samples for each subject were simultaneously collected for BEC-MN assay and cytokinesis-blocked micronucleus assay (CBMN), respectively, delivered to the laboratory within the same day and were processed within 5 h of collection. BECs were obtained by scraping both sides of the interior cheeks using pre-moistened wooden tongue depressors after subjects rinsed their mouths. The collected cells were smeared directly onto pre-cleaned and pre-moistened microscope slides and then left to dry. At the same day of BEC and venous blood sample collection, post-shift urine samples in an area far from the working area to avoid sample contamination were collected in a polyvinyl chloride container without preservatives and kept at −80°C until analysis. The sampling of all in the study group completed in 1 month and in the same day of the week. The control group was recruited after the sampling period of the study group.
Exposure assessment
Five passive samplers (SKC 575-002, Eighty Four, Pennsylvania, USA) were placed in ORs (n = 3), RUs (n = 1) and control clinical area (n = 1) on the level of healthcare personnel’s breathing zone in one of the biological sampling days. Sampling duration time was 8 h. Passive samplers were transferred into laboratory with cold chain and kept in −20°C. Sevoflurane was determined with headspace gas chromatography–mass spectrometry (HP Agilent 6890 GC-MS, Santa Clara, California, USA) equipped with Agilent J&W Ultra 2 GC Column, 19091B-105 (50 m × 0.20 mm i.d. × 0.33 mm thickness), Santa Clara, California, USA.
Urine samples (4 ml) and 1 ml 10% (v/v) Triton-X-100 (Sigma, St Louis, MO, USA) were transferred to the headspace vials (AGT-5182-0838 flat-bottom) in an ice bath; then, the vials were sealed with Agilent Hdspc Al crmp cap, PTFE/Si sep, Santa Clara, California, USA. The solutions were vortexed for 1 min and heated to 55°C for 30 min on a hot plate. 8 The gas-phase sevoflurane in the top of the headspace vials was injected with gas-tight injector (HAMILTON 80956 Model 1705 SL, Reno, Nevada, USA) to the GC-MS system (limit of detection (LOD): 0.04 µg l−1).
Sevoflurane in the passive samplers (SKC 575-002) was desorbed with 2.0 ml H2O/MeOH, (60:40; v/v), by vortexing for 1 h; 9 1 ml of desorbed solution was transferred to the headspace vial containing 3 ml of 10% (v/v) Triton-X-100, and then, the vial was sealed, vortexed for 1 min and heated to 55°C for 30 min on a hot plate. 8 After that, the gas-phase sevoflurane in the top of the vial was injected with gas-tight injector to the chromatographic system (LOD: 0.005 ppm).
The GC injector split ratio was 20:1 at 200°C, and the carrier gas was helium (1 ml min−1). The GC oven was programmed to 39°C (4.0 min) and then ramped to 120°C at 40°C min−1 (1 min). MS interface temperature was set to 280°C. 10 The data were acquired in selected-ion monitoring modes with 51, 131 and 181 m/z ions. Air concentration values of sevoflurane were expressed in ppm (1 ppm = 8.34 mg/m3). 11
The amount of fluoride in the urine samples was determined by direct potentiometric analyses with the use of a combination fluoride ion selective electrode (Consort C863 multiparameter analyser, Turnhout, Belgium). Urine samples and fluoride standards were diluted with TISAB(II) to compensate the ionic strength. 12 Five-point external calibration graph was plotted between 0.1 and 100.0 mg l−1 fluoride concentrations daily. Urinary (U) concentrations of fluoride were expressed in milligrams per litre using linear calibration equation (y = −54.12x + 88.82, R2 = 0.9940; y in mV, x is log F−1[F-] in mg l−1).
Genotoxicity assessment
CBMN in PBL
Whole blood (0.5 ml) samples were added to 4.5 ml culture medium (Roswell Park Memorial Institute supplemented with 20% foetal calf serum, 2% phytohaemagglutinin and 0.4%
MN test in BECs
Buccal epithelial slides were stained by the Feulgen reactive and then counterstained with Fast Green (Merck) according to the criteria of Stich and Rosin. 14 A total of 1000 differentiated and basal cells were identified and scored for the presence of MN per subject according the criteria of Tolbert et al. 15 using light microscopy at 400× magnification and confirmed at 1000× magnification (Zeiss Axioscope 2 Microscope). The frequency of MN per thousand BECs (MN frequency in BECs) was determined via blind evaluation of coded slides by a single scorer.
Statistical analysis
Data analysis was performed using IBM SPSS version 17.0 software (IBM Corporation, Armonk, NY, USA). Whether the continuous variables were distributed normally or not was determined by Kolmogorov–Smirnov test. Levene test was used for the evaluation of homogeneity of variances. Continuous variables were expressed as mean ± standard deviation or median (min–max), where applicable. Number of cases and percentages were used for categorical data. While the mean differences between control and anaesthesia groups were compared by Student’s t-test, Mann–Whitney U-test was applied for comparisons of not normally distributed data. When the number of independent groups was more than two, data were evaluated by one-way analysis of variance or Kruskal–Wallis test, where appropriate. When the p-values from Kruskal–Wallis test statistics were statistically significant, Conover’s multiple comparison test was used to know which group(s) differ from which others. Degrees of association between continuous variables were evaluated by Spearman’s rank correlation analyses. Categorical variables were analysed by Fisher’s exact test when more than 20% of cells have expected frequencies less than 5; otherwise, χ2 test was performed. A p-value less than 0.05 was considered statistically significant.
Results
Our study group comprising OR personnel (n = 34) and RU personnel (n = 12) as a total exposed group (n = 46) was consistent with the control group (n = 21) according to the age, gender distribution, smoking status, frequency of the smoking cigarettes per day, alcohol intake and BMI (Table 1; p > 0.05). The X-ray exposure was found to be significantly lower in total exposed group (p = 0.017) and in RU personnel (p = 0.030) when compared with the control group.
Demographic characteristics of study subjects.a
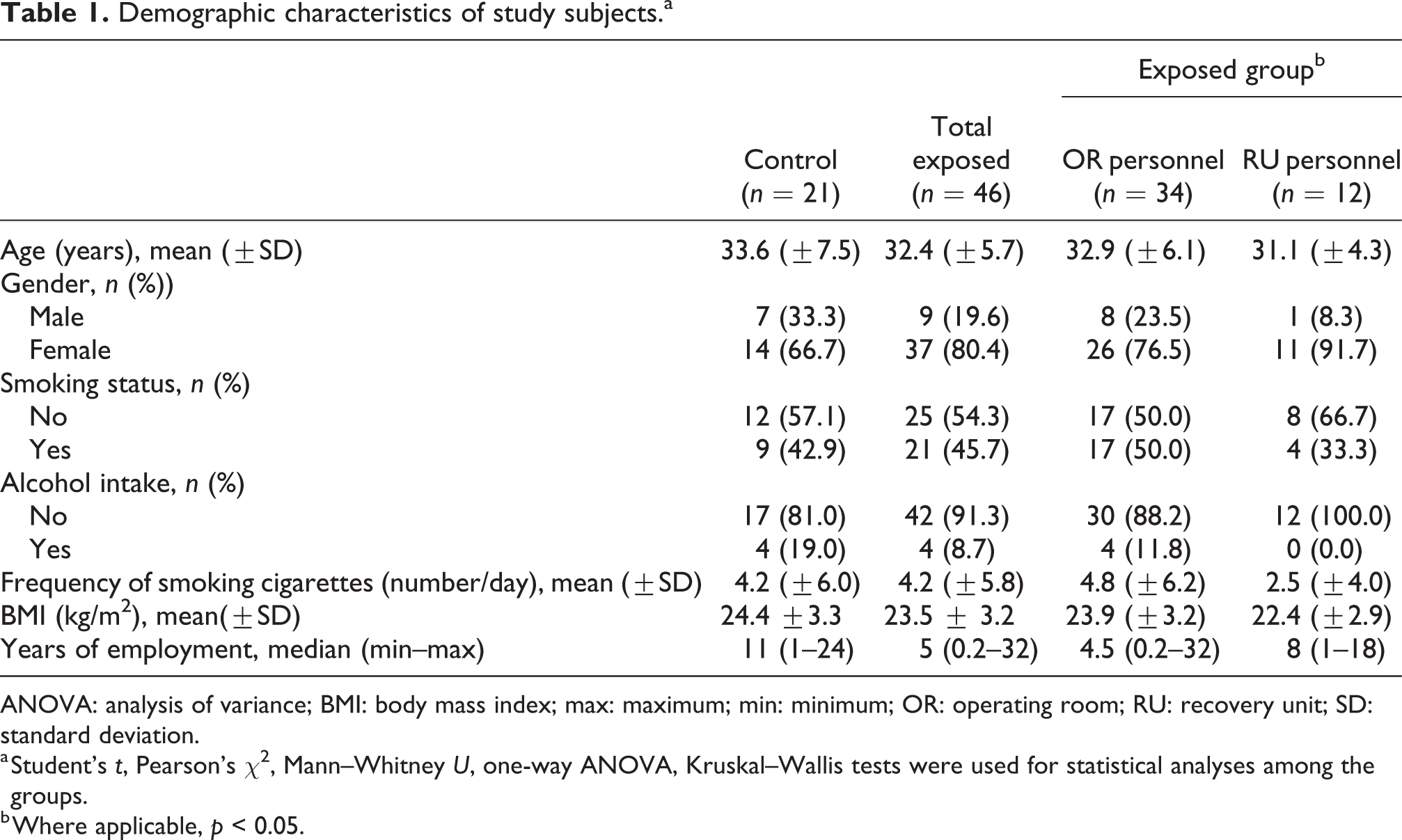
ANOVA: analysis of variance; BMI: body mass index; max: maximum; min: minimum; OR: operating room; RU: recovery unit; SD: standard deviation.
a Student’s t, Pearson’s χ2, Mann–Whitney U, one-way ANOVA, Kruskal–Wallis tests were used for statistical analyses among the groups.
b Where applicable, p < 0.05.
Air sevoflurane levels in three ORs were measured as 0.32, 0.38 and 0.58 ppm. The level was 0.43 ppm in an RU. Finally, in the control area from the same hospital, the air sevoflurane level was lower than LOD value.
U-sevoflurane could not be detected in the samples of the control group (Table 2). The OR and RU groups were not significantly different from each other for the U-sevoflurane levels (Table 2; p > 0.05).
Descriptive characteristics for exposure parameters in urine samples of study subjects.a

LOD: limit of detection; max: maximum; min: minimum; OR: operating room; RU: recovery unit.
a Mann–Whitney U and Kruskal–Wallis tests were used for statistical analyses among the groups.
b Where applicable, *p < 0.05; The urinary samples were not obtained from one subject from each OR and RU personnel.
c Urinary sevoflurane was lower than LOD for control group.
d Urinary sevoflurane was lower than LOD for two of the OR personnel.
U-fluoride levels were not different between control group versus total exposed, OR and RU groups, while the OR personnel had the highest U-fluoride levels (Table 2; p > 0.05). U-fluoride levels were significantly higher for males versus females (p = 0.011), smokers versus non-smokers (p = 0.001) and age of 35 and older subjects versus younger ones (p = 0.004). U-fluoride levels were found to be significantly correlated with age, BMI, number of daily smoked cigarettes and smoking years (respectively, r = 0.367, 0.349, 0.373 and 0.433; p < 0.01). U-fluoride levels were positively and significantly correlated with working years for the OR personnel (r = 0.488 vs. p = 0.003) and negatively and significantly correlated for the RU personnel (r = −0.773 vs. p = 0.005).
The MN frequency in PBL and the frequency of PBL with MN were significantly higher in total exposed group, in OR and RU personnel, than that of control group (Table 3; p < 0.05). Grouping the subjects according to the smoking (yes/no; 1–10 cig./≤1–10 cig.; non-smokers/<10 years smoking/≤10 years smoking), age (<35/≤35) and sex (male/female) did not show differences in the MN frequency parameters in PBL (p > 0.05). The MN frequency and frequency of lymphocytes with MN were found to be significantly correlated (r = 0.984; p < 0.001).
MN frequencies (‰) in lymphocytes of study subjects.a

max: maximum; min: minimum; MN: micronucleus; OR: operating room; RU: recovery unit.
a Mann–Whitney U and Kruskal–Wallis tests were used for statistical analyses.
b Two subjects from exposed group and two subjects from control group were excluded because of the technical problems.
cp < 0.01 versus control group.
dp < 0.001 versus control group.
The frequency of BEC-MN was significantly higher in total exposed group than that of the control group (Table 4; p < 0.05). The MN frequency in BEC was not significantly different in the subgroups, namely OR and RU personnel, than that of the control group (Table 4; p > 0.05). Grouping the subjects according to the smoking (yes/no; 1–10 cig./≤1–10 cig.; non-smokers/<10 years smoking/≤10 years smoking), age(<35/≤35 years) and sex (male/female) did not show differences in the MN frequency in BEC (p > 0.05).
MN frequencies (‰) in buccal epithelial cells of study subjects.

max: maximum; min: minimum; MN: micronucleus; OR: operating room; RU: recovery unit.
a Total exposed group versus control group (p = 0.037); Mann–Whitney U and Kruskal–Wallis tests were used for statistical analyses.
No significant correlations were found between PBL and BEC MN frequencies, between U-sevoflurane levels and MN frequencies and between working years and MN frequencies obtained from two tissues (p > 0.05).
Discussion
The present occupational molecular epidemiology study of OR and RU personnel of the same hospital assessed the exposure (sevoflurane in air and sevoflurane and inorganic fluoride levels in post-shift urine samples) and genotoxicity risk (MN test in two different tissues; PBLs and BECs) of WAG. Passive sampling of air sevoflurane levels, representative for the WAG exposure, in the breathing zone in three ORs and one RU in the duration of the research period were found to be not exceeding the established occupational exposure limits. Median levels of U-sevoflurane in OR and RU personnel were around 4 μg/lurine. U-sevoflurane levels by job categories (e.g. anaesthetists, nurses and technicians) of OR personnel were not significantly different. The inorganic fluoride levels were not different between the healthcare personnel and control group and also between OR and RU personnel. The genotoxicity increased in both PBL and BEC tissues of the healthcare personnel exposed to WAG.
In the present study, two different tissues were collected in which BECs were assumed to be the target tissue. The use of exposure biomarker as urine and exposure marker as passive samplers was confirmation of the exposure to WAG in both OR and RU. Another originality of the study is to include RU personnel for their likely exposure to the WAG and genotoxic effect.
There is a wide range of recommended limits in different parts of the world. 16 The workplace recommended exposure limit for halogenated inhaled anaesthetics is 2 ppm (1-h ceiling) when used alone or 0.5 ppm for halogenated anaesthetics combined with 25 ppm N2O (time-weighted average) during use. 17 In a few European countries, the occupational exposure limit (8 h) has been settled as 10 ppm for sevoflurane and desflurane. 18
In the present study, the air sevoflurane data of RU show the exposure potential to the WAG. There have been only a few studies on the exposure to WAG in the RU. 19 RU personnel may come across occupational exposure to WAG from the patients instead of anaesthesia delivery system. WAG in the breathing zone of RU personnel may contain higher levels compared with that in random room samples. 1
U-sevoflurane level may be used as an internal dose biomarker reflecting very recent exposure, and biologically equivalent limit (BEL) of 3.9 μg/lurine is recommended for sevoflurane corresponding to the National Institute for Occupational Safety and Health exposure limit, 2 ppm. 20 Biological exposure indices (BEIs) for U-fluoride are published as 2 mg l−1 (prior to shift) and 3 mg l−1 (end of shift). 21 In a research end of shift, U-sevoflurane levels in paediatric OR personnel (n = 36) ranged between ND and 18.5μg/lurine with a median level of 0.60 μg/lurine. 20 In another study, levels in OR personnel (n = 100) ranged between <0.1 and 17.28 μg/lurine with a median level of 0.62 μg/lurine. 22 Gentili et al. 23 demonstrated that U-sevoflurane levels of anaesthetists were significantly greater when compared with other OR personnel, with a median value of 2.1 μg/lurine. In this study, U-sevoflurane levels exceeded the BEL in 23 instances (nine anaesthetists, five nurses, three technicians of OR personnel and six RU personnel), indicating OR and RU personnel are occupationally exposed to WAG. There is no biological exposure information for RU personnel in the literature.
The inorganic fluoride is a metabolite common to the biotransformation of sevoflurane as well as other all fluorinated volatile anaesthetics. 24 In agreement with this study, U-fluoride is found to be not suitable for the biomonitoring of WAG because of its lack of specificity since fluoride in urine could be from other environmental sources. 20
Chromosomal changes and genetic instability are the main factors in the carcinogenic process. Cytogenetic biomarkers have been used for the early effects of environmental and occupational exposures to the likely genotoxic carcinogens. In two recent reviews, 7,25 occupational exposure of OR personnel to WAG was found to be related with genotoxicity for even though some conflicting results.
Originality of our study is the assessment of MN frequencies in two different tissues of OR and RU personnel with the exposure evidence evaluated in both working environment and biological samples. Additionally, there is no cytogenetic finding in RU personnel exposed to the anaesthetics.
In this study, both OR and RU personnel showed approximately two times higher MN frequencies in PBL as compared to the control subjects, thus reflecting high chromosomal instability. Age, sex, smoking habits, vitamin B12 and folate status and also technical differences may have an effect on MN frequencies in PL. 26 Herewith, age, sex and smoking habits did not influence the MN frequencies. Two measures of MN frequency were used in this study, that is, MN frequency in lymphocytes and frequency of lymphocytes with MN, following a similar tendency in this study. MN frequency in lymphocytes is potentially more sensitive; however, frequency of lymphocytes with MN is more stable. 27 Vodicka et al. 28 have recently reviewed 11 published studies on PBL MN frequencies of OR personnel. The exposure assessment was only available in five studies with airborne anaesthetic levels, and none of them had any biological exposure data. N2O was the most used anaesthetic, followed by sevoflurane, isoflurane, halothane and enflurane. Their meta-analysis showed that there were statistically significant different MN frequencies in OR personnel when compared to those in control subjects. Our results demonstrate that occupational exposure to anaesthetics is associated with cytogenetic damage in PBL after controlling possible confounding factors, in line with the review. 28
Similar to the PBL, genotoxic effect in the most easily accessible target tissue BEC was significantly higher (approximately three times) in the WAG-exposed healthcare personnel. Higher BEC-MN frequencies in both OR and RU personnel were observed than that of the control group.
Age, gender and smoking habits were not found to be confounding factors on BEC-MN frequencies in this study. There can be variations in BEC-MN frequencies according to the study population (i.e. age and sex), region, laboratory and method (i.e. cell numbers counted, staining and reader). 29
Chandrasekhar et al. 30 demonstrated significantly increased BEC-MN frequency in 46 OR personnel exposed to the mixture of WAG (N2O, halothane, sevoflurane, enflurane and desflurane), which is similar to the present study. Environmental concentrations of anaesthetics were not available in that study.
It could be better to have higher number of subjects and the number of the scored BECs. Also, the MN assay in both of the tissues could be carried out as cytome assay with evaluation of nuclear buds; nucleoplasmic bridges in lymphocytes and basal, differentiated, binucleated, condensed chromatin; karyorrhectic, pyknotic and karyolytic cells; and nuclear buds in buccal epithelial cells. 5
In this study, MN frequencies in BEC and PBL seemed to increase chromosomal instability and genotoxicity events although the extent of damage varied between two tissues.
It is suggested that RU and OR personnel may have similar occupational genotoxicity risk. Genotoxic effect evaluation of WAG for RU personnel needs further attention to clarify the results. It is recommended that monitoring and improvement of environmental conditions in OR and RU areas and also the implementation of health survey programs should be considered to avoid health hazards.
Footnotes
Acknowledgements
The authors are grateful to the healthcare personnel for their participation in this study. The authors would like to thank Mr. Salih Ergocen for performing the statistical analysis. The authors also thank Gazi University Academic Writing Center for proofreading this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Gazi University Research Fund, Project No. 02/2010-03.
