Abstract
Objective:
Nitrogen oxide (NOx) pollution and human immunodeficiency virus (HIV)/AIDS intensify inflammation during pregnancy and linked with adverse birth outcomes (ABOs). MicroRNA (miRNA)-146a plays a crucial role in regulating inflammation in the NF-κB pathway. The G/C rs2910164 dampens miRNA-146a activity and linked with inflammatory diseases. The present study investigated whether HIV/AIDS and NOx exposure throughout pregnancy further intensifies ABO in Black South African women genotyped for the rs2910164.
Methods:
Pregnant women (n = 300) were subdivided into low, medium and high NOx exposure groups, genotyped for the miRNA-146a G/C rs2910164 using polymerase chain reaction-restriction fragment length polymorphism, and further stratified based on HIV status.
Results:
Unstratified data (HIV+ and HIV− mothers combined): Mothers from the high NOx group with the variant C-allele had low blood iron levels (p = 0.0238), and had babies with reduced birthweights (p = 0.0283). As NOx increased, the prevalence of preterm birth and low birth weight also increased in mothers with the variant C-allele versus wildtype G-allele. HIV-infected mothers: In all NOx exposure groups, mothers with the variant C-allele had higher systolic blood pressure (low: p = 0.0386, medium: p = 0.0367 and high: p = 0.0109) and had babies with lower Appearance, Pulse, Grimace, Activity and Respiration scores at 1 min (low: p = 0.0190, medium: p = 0.0301 and high: p = 0.0361).
Conclusion:
Maternal rs2910164 variant C-allele, NOx pollution and HIV/AIDS might collectively play a role in intensifying gestational hypertension and ABO.
Introduction
Inflammation is a key point during pregnancy due to imbalances in humoral and cell-mediated immune responses and poor maternal and foetal communication. 1 Maternal and foetal stress is further triggered by a broad spectrum of communicable (human immunodeficiency virus (HIV)/AIDS) and non-communicable (hypertension and obesity) diseases, environmental insults (pollution and teratogens) and genetic factors (genetic variations) which further intensifies inflammation and oxidative stress. 2 As a consequence, adverse birth outcomes (ABOs) could arise, namely, intrauterine growth retardation, preterm births (PTBs), low birth weights (LBWs), small for gestational age births (SGAB) and foetal mortality. 3,4
Air pollution is considered to be the leading force behind the manifestation of ABO. 5 In particular, nitrogen oxides (NOx), comprising nitrogen dioxide (NO2) and nitric oxide (NO), are potent prooxidants which are formed in combustion engines and are considered to be an important marker for traffic pollution. 6 Several studies linked NOx with defective foetal development and ABO. 7 –11 Apart from NOx, maternal obesity and hypertension 12,13 and HIV/AIDS 14 are also associated with ABO. Genetic variations, including random single base mutations in DNA sequences, often referred to as single nucleotide polymorphisms (SNPs), can diminish the overall functionality of protein coding genes crucial for healthy pregnancies 15 and linked with ABO. 16
MicroRNAs (miRNAs) are a class of small (19–22 nucleotides long) non-coding RNAs that negatively regulate gene expression at the post-transcriptional level. 17 The 2–7 nucleotides long conserved ‘seed’ region of miRNAs, positioned at the 5′-untranslated region (5′-UTR), binds to the 3′-UTR of mRNAs with near-perfect complementarity. As a result, translational repression and/or mRNA deadenylation/decay can occur. 18 MiRNAs are implicated in several physiological pathways that control cell cycle, organogenesis, inflammation, apoptosis and haematopoiesis. 19 They are also associated with embryogenesis and placental functionality and found in several human tissues and biofluids. 19,20 Pollution can dysregulate miRNA expression and linked with widespread gene expression changes and several pathophysiological processes associated with inflammatory disorders. 21
MiRNA-146a is situated on human chromosome 5q34 and plays a significant role in regulating inflammatory and immune response pathways. 22 Toll-like receptors (TLRs), interleukin (IL)-1β and tumour necrosis factor (TNF)-α stimulate the induction of miRNA-146a. In order to modulate and prevent overstimulation of inflammatory responses in the TLR/NF-κB pathways, miRNA-146a primarily targets TNF receptor associated factor 6 (TRAF6) and IL receptor associated kinase 1 (IRAK1) to elicit its regulatory functions. 23 Pollution negatively affects miRNA-146a expression and triggers oxidative stress and inflammation. 24 Pollution, HIV/AIDS and pregnancy-based studies involving miRNA-146a are limited. Nonetheless, miRNA-146a was found to be a therapeutic target and biomarker for peripartum cardiomyopathy, 25 upregulated in HIV-1-infected individuals 26 and downregulated in placentas taken from active smokers. 27
The miRNA-146a G/C rs2910164 (due to a C:U miss-pairing taking place instead of a normal G:U pairing) is associated with the pathogenesis of several inflammatory diseases, including sepsis, 28 diabetes, 29 autoimmune disorders 30 and cardiovascular disease. 31 This SNP is located within the crucial stem region of pre-miRNA-146a and disrupts the expression of mature miRNA-146a. 32,33 The rs2910164 was associated with severe preeclampsia in HIV+ Black South African women on highly active antiretroviral therapy (HAART). 34 To date, studies investigating the role of rs2910164 in ABO are scarce.
As mentioned, ambient air pollution is associated with ABO in non-HIV-infected pregnant women, 5 but it is hypothesized to be more pronounced in HIV-infected women. Additionally, ABO in HIV-infected pregnant women is associated with the rs2910164, 34 and it is hypothesized that both the rs2910164 and ambient air pollution exposure can collectively further intensify the prevalence of ABO. Hence, based on the inflammatory milieu in pregnancy and HIV, the link between NOx pollution-induced inflammation and ABO, the role of miRNA-146a in regulating inflammatory responses and the association between rs2910164 and inflammatory disease, the present study investigated the role of rs2910164 in adverse maternal health and ABO in HIV-infected Black South African women residing in Durban, a city with heavy traffic congestion and high NOx levels. 35 –37 In this study, mothers were subdivided into low, medium and high NOx exposure groups, genotyped for the rs2910164, and further stratified based on HIV status. We then investigated any possible association between the rs2910164, HIV/AIDS and NOx with maternal body mass index (BMI), blood pressure, haemoglobin and iron levels, and with neonates gestational age, birthweight, birth length, head circumference and Appearance, Pulse, Grimace, Activity and Respiration (APGAR) scores.
Methods
Patient recruitment and data collection
A total of 300 pregnant women (HIV+ and HIV− combined) were selected from public sector antenatal clinics in participating communities and enrolled in the Mother and Child in the Environment (MACE) Birth Cohort Study situated in Durban, Kwa-Zulu Natal, South Africa. Thereafter, they were subdivided into low (n = 100), medium (n = 100) and high (n = 100) NOx exposure groups. Durban was considered to be an ideal location for this study due to the presence of several heavy polluting industries and freeway roads that are situated in close proximity to residential areas. In 2007, Naidoo and colleagues determined the exposure levels of all conventional air pollutants, volatile organic compounds, total reduced sulphur and airborne metals within Durban 35 ; these findings lead to the formation of the MACE study. The study was approved by the Biomedical Research Ethics Committee at the University of KwaZulu-Natal (BF263/12). Each participant was given a comprehensive explanation of this study and a written consent form was obtained. Participation in this study was purely voluntary and withdrawal at any time was permitted.
The study inclusion criteria involved all study participants, irrespective of their race, having: (1) resided in the same geographical area for the entire duration of pregnancy; (2) been in their third trimester of pregnancy; (3) not experienced any pregnancy related complications such as placenta previa, genital tract infections, preeclampsia, hypertension and diabetes; and (4) not had multiple pregnancies. Participants who tested positive for HIV and experienced multiparity were not excluded from this study. The list of antiretroviral therapy (ART)/HAART drugs administered to HIV-infected mothers during pregnancy includes nevirapine, lamivudine, stavudine, odimune, tenofovir disoproxil fumarate, efavirenz, tribuss and atroiza.
Clinical, demographic, lifestyle and ambient and indoor air pollution exposure data were collected. Prenatal and postnatal data were obtained from participating clinics and hospitals where the neonates were born, respectively.
Characterization of NOx exposure
The land use regression model was used to determine the exposure of pregnant women to ambient NOx levels, that is, low, medium and high. The model was developed using the ESCAPE approach 38 which involved monitoring NOx levels at selected locations within the study area and regressing these measurements against a predefined set of geographic predictor variables in a multivariate regression model. The model parameters were then used to derive estimates of NOx exposure at the participant’s home. Data from this study have been published elsewhere. 36
DNA extraction
The FlexiGene® DNA Isolation Kit (Qiagen, Hilden, Germany) was used to isolate genomic DNA from whole blood obtained during the third trimester of pregnancy. Briefly, 750-µl cell lysis buffer was added to 300-µl whole blood and centrifuged (10,000 × g, 20 s). The supernatants were removed and 150 µl denaturation buffer was added to the pellet and incubated (65°C, 5 min). Thereafter, 150-µl 100% isopropanol was added to precipitate the DNA which was then recovered by centrifugation (10,000 × g, 3 min). The DNA was washed in 150-µl 100% ethanol, dried at room temperature, resuspended in 15-µl hydration buffer (10 mM Tris-Cl, pH 8.5) and incubated (65°C, 1 h). The samples were then stored at −20°C until further use. The Nanodrop2000 spectrophotometer (Biolab, Scoresby, VIC, Australia) was used to quantify the DNA and the samples were standardized to a concentration of 10 ng/µl.
Genotyping
The miRNA-146a G/C rs2910164 SNP was genotyped using polymerase chain reaction (PCR)-restriction fragment length polymorphism. The GoTaq® G2 Flexi DNA Polymerase PCR kit (Promega, Madison, Wisconsin, USA) was used together with the CFX96 TouchTM Real-Time PCR Detection System (Bio-Rad, Hercules, California, USA) for this analysis. Briefly, the 147 bp gene product was amplified in a 30-µl reaction consisting of 1X Green GoTaq Flexi buffer, 2.5 mM magnesium chloride, 200 µM of each deoxyribonucleotide triphosphate (dNTP), 0.2 Units GoTaq Flexi DNA polymerase, 20 pmol of each primer and 30-ng genomic DNA template. A no-template DNA sample was used as a quality control measure against PCR contamination. The forward and reverse primer sequences used were 5′-CATGGGTTGTGTCAGTGTCAGAGCT-3′ and 5′-TGCCTTCTGTCTCCAGTCTTCCAA-3′, respectively. PCR was performed using the following cycling conditions: 94°C for 10 min (initial denaturation), followed by 30 cycles at 94°C for 30 s (denaturation), 65°C for 30 s (annealing) and 72°C for 30 s (extension). This was followed by a final extension at 72°C for 7 min. The PCR products were electrophoresed on 1.8% agarose gel containing 2-µl GelRed and visualized using the ChemiDoc™ XRS+ Molecular Imaging System (Bio-Rad, Hercules, California, USA). The Sac I restriction enzyme (5 Units; New England BioLabs, Massachusetts, USA) was used to digest the PCR products at 37°C for 16 h. Thereafter, the restriction products were electrophoresed on 3% agarose gels containing 2-µl GelRed and visualized as mentioned earlier. The homozygous wild-type G-allele (GG genotype) resulted in no cleavage of the 147 bp product. The homozygous variant C-allele (CC genotype) yielded two fragments of 122 and 25 bp. The heterozygous GC genotype yielded three fragments of 147, 122 and 25 bp. A DNA ladder was run together with the restriction products to accurately determine the different genotypes.
Statistical analysis
All statistical analysis was performed using the IBM SPSS statistical software (version 24) and GraphPad Prism software (version 5.0) packages. The Kolmogorov–Smirnov and Shapiro–Wilk tests for normality, followed by the Tukey’s honest significant difference multiple range post hoc test and unpaired t-test was used to analyse all data. Pre and postnatal data were expressed as mean ± standard deviation (SD). Results were interpreted as the mean difference (MD) ± SD (95% confidence interval (CI)). A p < 0.05 was considered as being significant.
Results
NOx exposure levels for all study subjects
Mothers subdivided into low, medium and high exposure groups had a median NOx exposure of 26.46 (12.78 to 31.44) µg/m3, 36.64 (31.72 to 39.74) µg/m3 and 46.85 (39.94 to 56.89) µg/m3 during pregnancy, respectively (p < 0.0001; Figure 1 and Table 1).
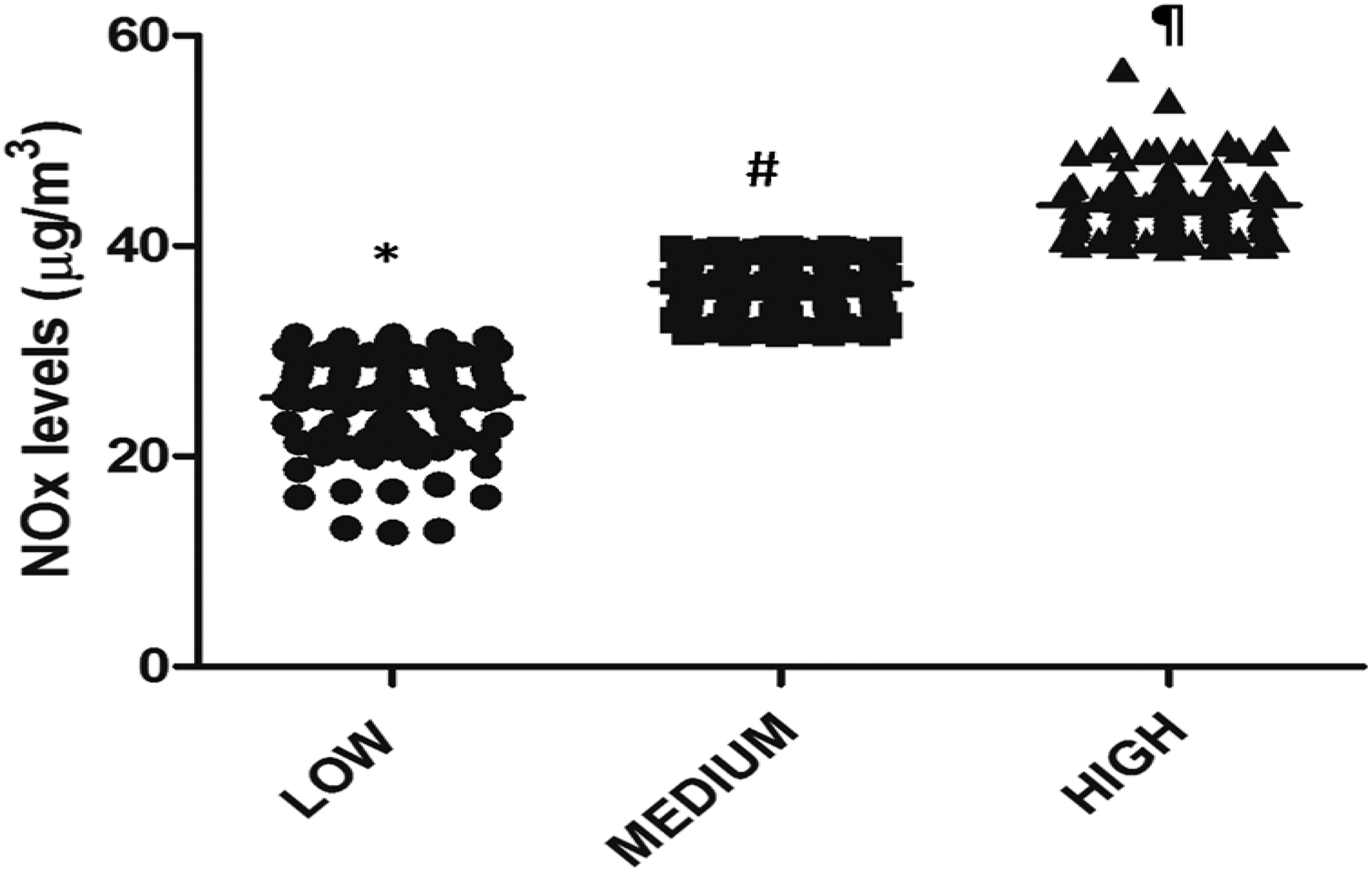
Mothers subdivided into low (n = 100), medium (n = 100) and high (n = 100) NOx exposure groups. *,#,¶ Symbols over the plots for a given parameter are significantly different from each other (p < 0.0001). NOx: nitrogen oxides.
Maternal NOx exposure levels in the low (n = 100), medium (n = 100) and high (n = 100) exposure groups (p < 0.0001).

NOx: nitrogen oxides.
Demographics and clinical characteristics for all study participants
Over one-third of mothers in this study were HIV+ (low: 32%, medium: 36% and high: 38%). In all NOx exposure groups, the mean maternal BMI was greater than 30 kg/m2 which indicated maternal obesity. BMI in the high exposure group was significantly increased versus low (MD = 4.93 (1.16 to 8.70) kg/m2) and medium (MD = 4.71 (0.94 to 8.48) kg/m2) exposure groups (p = 0.0024). Mothers in the high exposure group also displayed significantly higher diastolic blood pressure versus low exposure (MD = 3.37 (0.21 to 6.53) mmHg; p = 0.0227) and had neonates with lower birthweights versus low exposure (MD = −239 (−423 to −56) g; p = 0.0088), birth length versus low (MD = −1.68 (−3.12 to − 0.24) cm) and medium (MD = −1.48 (−2.92 to −0.04) cm) exposures (p = 0.0113) and APGAR scores at 5 min (MD = −0.32 (−0.63 to −0.01); p = 0.0408). The prevalence of PTB and LBW were 6% and 15% (low), 8% and 16% (medium) and 15% and 19% (high), respectively (Table 2).
Demographical and clinical characteristics of study participants (n = 300) grouped according to NOx exposure levels.

HIV: human immunodeficiency virus; NOx: nitrogen oxide; BMI: body mass index; APGAR: Appearance, Pulse, Grimace, Activity and Respiration.
a,bValues with different superscript variables within a column are statistically different from each other (p < 0.05).
Genotype and allele frequencies: MiRNA-146a G/C rs2910164
Overall, 34%, 51% and 14% of all mothers in this cohort had the GG, GC and CC genotypes, respectively, and had a G versus C allele frequency of 60% versus 40%, respectively. The G versus C allele frequency in all exposure groups were 62% versus 38% (low), 60.5% versus 39.5% (medium) and 57.5% versus 42.5% (high), respectively (Table 3).
Genotype and allele frequencies for mothers genotyped for the miRNA-146a G/C rs2910164.

miRNA: MicroRNA; NOx: nitrogen oxides; G: guanine; C: cytosine; GG genotype: homozygous wild-type G-allele; GC genotype: heterozygous variant C-allele; CC genotype: homozygous variant C-allele.
Demographics and clinical characteristics: HIV-infected mothers
Mothers that were HIV+ had significantly higher BMI (MD = 4.70 ± 1.36 (2.05 to 7.36) kg/m2; p = 0.0006) and had neonates with reduced gestational age (MD = −0.58 ± 0.20 (−0.97 to −0.18) weeks; p = 0.0045) and APGAR scores at 5 min (MD = −0.25 ± 0.11 (−0.46 to −0.04); p = 0.0225). The prevalence of PTB and LBW was 9% and 16% (HIV−) and 10% and 17% (HIV+), respectively. Although not significant, HIV+ mothers had babies with lower birthweights (Table 4).
Demographical and clinical characteristics of study participants (n = 300) stratified based on HIV status.
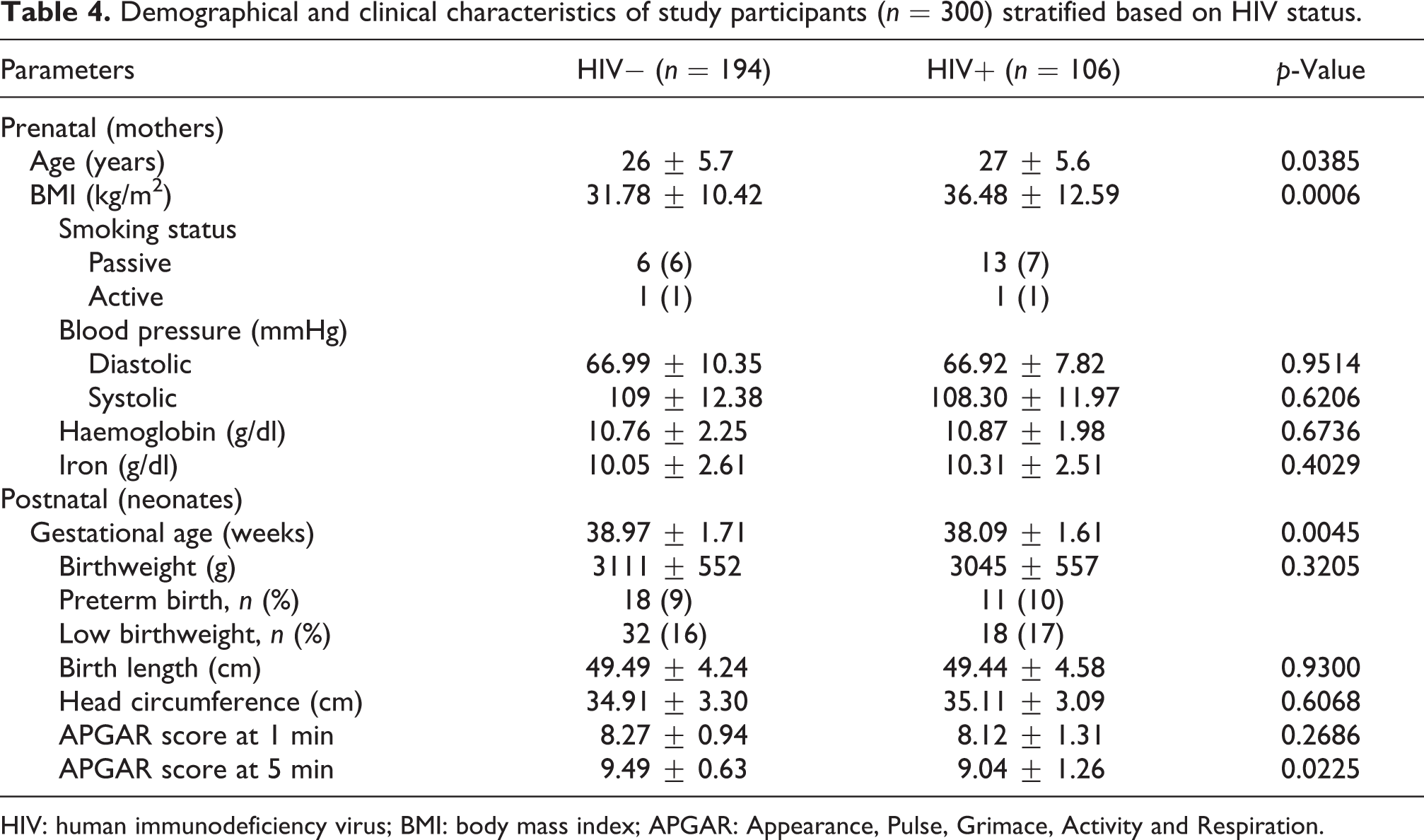
HIV: human immunodeficiency virus; BMI: body mass index; APGAR: Appearance, Pulse, Grimace, Activity and Respiration.
Effects of NOx exposure on HIV-infected mothers
No significant changes in pre and postnatal data were observed in mothers exposed to low levels of NOx pollutants. Conversely, HIV+ mothers in the medium exposure groups had significantly higher BMI (MD = 6.38 ± 2.02 (2.37 to 10.39) kg/m2; p = 0.0021) and diastolic blood pressure (MD = 4.86 ± 2.20 (0.49 to 9.24) mmHg; p = 0.0296). HIV+ mothers in the high exposure groups had higher BMI (MD = 5.25 ± 2.67 (2.01 to 10.54) kg/m2; p = 0.0480), diastolic blood pressure (MD = 3.63 ± 1.74 (0.18 to 7.08) mmHg; p = 0.0392) and systolic blood pressure (MD = 5.36 ± 2.34 (0.72 to 10.00) mmHg; p = 0.0237) and had neonates with reduced gestational age (MD = −0.60 ± 0.28 (−1.17 to −0.04) weeks; p = 0.0373). The prevalence of PTB and LBW in HIV− and HIV+ mothers was 4% and 12% versus 9% and 22% (low), 6% and 17% versus 11% and 14% (medium) and 18% and 21% versus 11% and 16% (high), respectively. Although not significant, HIV+ mothers in all NOx exposure groups had babies with lower birthweights (Table 5).
Clinical characteristics of study participants grouped according to NOx exposure and stratified based on HIV status.

HIV: human immunodeficiency virus; NOx: nitrogen oxide; BMI: body mass index; DBP: diastolic blood pressure; SBP: systolic blood pressure; HGB: haemoglobin; GA: gestational age; BW: birthweight; PTB: preterm birth; LBW: low birth weight; BL: birth length; HC: head circumference; APGAR: Appearance, Pulse, Grimace, Activity and Respiration.
a,bValues with different superscript variables between HIV− versus HIV+ mothers subdivided into low, medium and high NOx exposure groups are statistically different from each other (p < 0.05).
All study subjects exposed to NOx pollution and genotyped for the miRNA-146a rs2910164
Apart from BMI (medium exposure: p = 0.0097), no significant changes in pre and postnatal data were observed in the low and medium exposure groups. For the high exposure group, mothers homozygous or heterozygous for the variant C-allele in comparison with homozygotes with the wild-type G-allele (GC + CC vs. GG) had significantly lower blood iron levels (MD = −1.24 ± 0.54 (−2.32 to −0.17) g/dl; p = 0.0238) and had neonates with reduced birthweights (MD = −242 ± 108 (−458 to −26) g; p = 0.0283) and birth length (MD = −1.92 ± 0.88 (−3.67 to −0.17) cm; p = 0.0315). For low and medium exposure groups, although not significant, mothers with the GC + CC genotypes had neonates with relatively lower birthweights versus GG genotype. The prevalence of PTB and LBW in mothers with the GG versus GC + CC genotypes was 3% and 2% versus 3% and 13% (low), 3% and 3% versus 5% and 13% (medium) and 6% and 4% versus 9% and 15% (high), respectively (Table 6).
Study participants grouped according to NOx exposure and genotyped for the miRNA-146a G/C rs2910164 before stratification for HIV.

HIV: human immunodeficiency virus; NOx: nitrogen oxide; miRNA: MicroRNA; GG genotype: homozygous wild-type G-allele; GC genotype: heterozygous variant C-allele; CC genotype: homozygous variant C-allele; BMI: body mass index; DBP: diastolic blood pressure; SBP: systolic blood pressure; HGB: haemoglobin; GA: gestational age; BW: birthweight; PTB: preterm birth; LBW: low birthweight; BL: birth length; HC: head circumference; APGAR: Appearance, Pulse, Grimace, Activity and Respiration; G: guanine; C: cytosine.
a,bValues with different superscript variables between mothers with the homozygous wild-type G-allele (GG genotype) versus those with the heterozygous variant C-allele (GC genotype) and homozygous variant C-allele (CC genotype), and subdivided into low, medium and high NOx exposure groups are statistically different from each other (p < 0.05).
HIV-infected mothers exposed to NOx pollution and genotyped for the miRNA-146a rs2910164
In all NOx exposure groups, apart from BMI (medium exposure: p = 0.0352), mothers homozygous or heterozygous for the variant C-allele in comparison with homozygotes with the wild-type G-allele (GC + CC vs. GG) had significantly higher systolic blood pressure (low: MD = 9.17 ± 4.24 (0.52 to 17.83) mmHg, p = 0.0386; medium: MD = 7.91 ± 3.64 (0.51 to 15.31) mmHg, p = 0.0367; high: MD = 11.18 ± 4.16 (2.73 to 19.63) mmHg, p = 0.0109) and had neonates with reduced APGAR scores at 1 min (low: MD = −0.77 ± 0.31 (−1.41 to −0.14), p = 0.0190; medium: MD = −0.57 ± 0.25 (−1.09 to −0.06), p = 0.0301; high: MD = −0.98 ± 0.45 (−1.88 to −0.07), p = 0.0361). The prevalence of PTB and LBW in HIV+ mothers with the GG versus GC + CC genotypes was 18% and 18% versus 10% and 14% (low), 8% and 0% versus 13% and 26% (medium) and 13% and 13% versus 7% and 20% (high), respectively (Table 7).
HIV+ study participants grouped according to NOx exposure and genotyped for the miRNA-146a G/C rs2910164.

HIV: human immunodeficiency virus; NOx: nitrogen oxide; miRNA: MicroRNA; GG genotype: homozygous wild-type G-allele; GC genotype: heterozygous variant C-allele; CC genotype: homozygous variant C-allele; BMI: body mass index; DBP: diastolic blood pressure; SBP: systolic blood pressure; HGB: haemoglobin; GA: gestational age; BW: birthweight; PTB: preterm birth; LBW: low birthweight; BL: birth length; HC: head circumference; APGAR: Appearance, Pulse, Grimace, Activity and Respiration; G: guanine; C: cytosine.
a,bValues with different superscript variables between HIV-infected mothers with the homozygous wild-type G-allele (GG genotype) versus those with the heterozygous variant C-allele (GC genotype) and homozygous variant C-allele (CC genotype), and subdivided into low, medium and high NOx exposure groups are statistically different from each other (p < 0.05).
Discussion
Birth cohort studies have shown that NOx pollution affects foetal development which could ultimately lead to ABO. 7 –11 Most studies, to date, were conducted on Caucasian and Asian patients with no studies on an urban African population. To the best of our knowledge, this is the first study in Africa to investigate how NOx pollution, HIV/AIDS and miRNA-146a G/C rs2910164 collectively affect both maternal health and foetal development.
In the present study, neonates from the high NOx exposure group (46.85 (39.94 to 56.89) μg/m3) had the lowest birthweights, birth length and APGAR scores at 5 min compared to low (26.46 (12.78 to 31.44) μg/m3) and medium (36.64 (31.72 to 39.74) μg/m3) groups (Table 2). NOx pollution consists of NO and NO2. NO is produced together with ozone when oxygen reacts with NO2 under the influence of sunlight. When NO emissions are high, ozone is rapidly consumed in order to oxidize NO to NO2. 39 A Swedish cohort study measuring foetal growth from ultrasound scans during the second trimester of pregnancy reported that for every 10 µg/m3 increment of NOx, there was a decrease in femur length, abdominal diameter and foetal weight. During the third trimester of pregnancy, there was a decrease in head circumference, foetal growth and birthweight. 11 A Spanish cohort linked NO2 exposure >40 μg/m3 with reductions in birthweight, birth length and head circumference. Every 10 μg/m3 increment of NO2 during the second trimester increased the risk of SGAB-weight and SGAB-lengths. 8 Similar findings were found in our study. NO2 exposure between 3 months and 5 months of pregnancy also increases the prevalence of LBW. 40
Mothers in the high NOx exposure group also had the highest diastolic blood pressure and BMI (Table 2). Women with BMI > 30 kg/m2 and waist-to-hip ratio > 0.90 compared to those with BMI < 25 kg/m2 were reported to have significantly high serum NO metabolite (nitrate and nitrite) concentrations. 41 Abnormally high NO production and its associated metabolites are linked with the pathogenesis of several disorders, including metabolic syndrome, hyperlipidemia, cardiovascular disease and diabetes. 42,43 Every 10 µg/m3 increment of NOx pollution during pregnancy was associated with intensified pregnancy-induced hypertensive disorders, 6 and long-term NO2 exposure was linked with high blood pressure. 44
Taking into account that inflammation is common during pregnancy and NOx pollution is a potent oxidant associated with adverse maternal health and ABO, this could explain the observed trends seen in Table 2. Evidently, as NOx pollution increased, the prevalence of PTB and LBW also increased (Table 2).
HIV+ mothers in this study displayed symptoms of aggravated obesity and although not significant, were more likely to have LBW and PTB, and lower mean birthweights (Table 4). In addition to NOx, HIV/AIDS patients on HAART were reported to be more overweight and obese, and displayed symptoms of hypertension and dyslipidaemia. 45 Similar findings were reported in African patients from Sub-Saharan Africa. In this study, obesity was strongly correlated with the Black ethnic race groups. 46
HIV+ mothers in the medium and high NOx exposure groups had significantly increased BMI and higher diastolic pressure. Additionally, the high exposure group also had HIV+ mother much older in age and with higher systolic blood pressure (Table 5). Khalid (2006) associated age and BMI with elevated diastolic and systolic blood pressure in women. 47 Malaza et al. (2012) also reported that HIV+ South African women on ART and HAART were morbidly obese and had higher blood pressure. 48 In a South African birth cohort, HAART exposure during pregnancy was not linked with LBW 49 and maternal BMI had no effects on birthweights. 50 Similar findings were found in our study (Tables 4 and 5). In the high NOx group, HIV+ mothers had babies with significantly decreased gestational age (Table 5). Both air pollution 51 and HIV/AIDS 52 are linked with reductions in gestational age and PTB.
Apart from being an air pollutant, NO is also produced within the human body. The role of endogenous NO in HIV infection remains confounding, 53 –55 and studies reported HIV-infected patients had increased 56,57 and decreased 58,59 endogenous NO levels. In 2016, Soccal et al. found untreated HIV-infected patients had the highest endogenous NO levels. Remarkably, the healthy non-HIV-infected control group had a higher endogenous NO level versus HIV patients on HAART. 53 In our study, all HIV-infected mothers were on ART/HAART and it could be deduced that endogenous NO was not a confounding factor.
In all NOx exposure groups, we found that HIV+ mothers with the variant genotypes (GC + CC) versus wild type genotype (GG) had significantly elevated systolic blood pressure, and gave birth to babies with lower APGAR scores at 1 min (Table 7). In the unstratified group (HIV+ and HIV− mothers combined), both HIV/AIDS and high NOx exposure levels might have further intensified the prevalence of reduced birthweights and birth lengths observed in mothers with the variant genotypes (Table 6). In addition, with respect to the variant genotypes, as NOx exposure levels increased the prevalence of LBW and PTB also increased, and significantly low maternal iron levels were observed in the high NOx group (Table 6).
As mentioned, when miRNA-146a expression is high, it inhibits IRAK1 and TRAF6, leading to concomitant reductions in pro-inflammatory cytokine levels (IL-1, IL-6, IL-8, IL-1β and TNF-α). 31 Pro-inflammatory cytokines are potent stimulators for miRNA-146a activation, are found in tracheal, blood and amniotic fluid aspirates and are associated with adverse foetal respiratory outcomes. 60 ABO can also arise if inflammatory cytokines are aberrantly overexpressed. 61 miRNA-146a expression is rapidly increased in response to TNF-α and IL-1β in several cell types, including human rheumatoid arthritis synovial fibroblasts cells 62 and primary human airway epithelial cells. 63
Studies investigating miRNA-146a expression in response to NOx pollution, HIV/AIDS and adverse maternal and foetal health are limited and confounding. Nonetheless, NO2 pollution exposure during the second and third trimester of pregnancy 64 and maternal cigarette smoking 27 downregulated placental miRNA-146a expression. Downregulated miRNA-146a expression was also reported in placentas taken from women who experienced PTB 65 and preeclampsia. 66 HIV-infected patients have significantly elevated miRNA-146a expression versus healthy controls. 26 Quaranta et al. (2015) reported that resting CD4+ T lymphocytes have high miRNA-146a expression, which inhibits co-receptor CXCR4 expression and prevents HIV-1 infection of CD4+ T cells. 67
The miRNA-146a G/C rs2910164 aberrantly dampens the functionality and expression of miRNA-146a and is associated with the pathogenesis of several inflammatory diseases. 28 –31 The variant C-allele lowers miRNA-146a expression and increases the risk of developing severe sepsis. This inflammatory response syndrome was accompanied by an upregulation in IRAK1 and TRAF6 expression levels. 28 Women of childbearing age with the variant C-allele was also more susceptible to metabolic syndrome. The heterozygous GC and homozygous variant CC genotypes had significantly increased BMI, diastolic and systolic blood pressure, total cholesterol, low density lipoprotein (LDL) cholesterol and triglycerides. The GC genotype was associated with high blood glucose and serum insulin levels, and insulin resistance, while the CC genotype was associated with low high density lipoprotein (HDL) cholesterol levels. 68
Studies investigating the collective role of miRNA-146a G/C rs2910164, NOx pollution and HIV/AIDS in ABO are also scarce. In the only known birth cohort study involving the rs2910164 in an African population, Maharaj et al. (2017) found an association between the rs2910164 and severe preeclampsia in HIV+ Black South African women on HAART. In this study, heterozygotes and homozygotes with the variant genotypes (GC + CC) were less susceptible to severe preeclampsia, which was further influenced by co-morbid HIV/AIDS infection and HAART. 34
NOx also disrupts iron homeostasis by converting oxyhaemoglobin to metheamoglobin, an insidious prooxidant. 69 Iron deficiency is also associated with LBW and PTB. 70,71 and elevated systolic blood pressure and hypertension. 72
Conclusion
In conclusion, the present study demonstrates the relationship between the rs2910164 variant C-allele, HIV/AIDS and NOx pollution with adverse maternal health and ABO in a predominantly Black South African population, which has not been previously reported. As NOx pollution increased, maternal diastolic blood pressure increased and neonates birthweights, birth length and APGAR score at 5 min decreased. Mothers with the rs2910164 variant C-allele and exposed to high NOx pollution had low blood iron levels and gave birth to neonates with decreased birthweights and birth length. In the high NOx exposure group, HIV+ mothers displayed higher diastolic and systolic blood pressure levels, and gave birth to neonates with reduced gestational age versus HIV− mothers. In all NOx exposure groups, HIV+ mothers with the variant C-allele had higher systolic blood pressure and had neonates with reduced APGAR scores at 1 min.
Data from this study shed light on the harmful effects of maternal HIV infection and NOx pollution exposure towards foetal growth, which is further intensified in mothers with the rs2910164 variant C-allele. ABO, that is, reductions in gestational age, birthweight, birth length and APGAR scores were associated with maternal rs2910164 variant C-allele, HIV infection and NOx pollution exposure.
Identification of maternal genetic susceptibility loci that are associated with HIV/AIDS and air pollution-induced ABO could allow for early therapeutic interventions in affected mothers in order to prevent and/or minimize the risk of ABO from occurring. This study is the foundation for future work in investigating the influence of maternal rs2910164, HIV status and NOx pollution exposure during pregnancy on adverse health outcomes among children of the cohort.
Footnotes
Acknowledgements
The authors would like to thank Dr Alisa Phulukdaree for her technical expertise, and also thank the fieldworkers and study participants in the Mother and Child in the Environment (MACE) birth cohort study.
Author contributions
PN, RNN, PR and AAC were involved in designing and conceptualizing this project and analysing and interpreting the data. PN performed the experiments, analysed the data and wrote the manuscript. SM developed a land use regression model to estimate NOx exposure. KA is the project manager for the MACE study and was involved in collecting clinical and demographical data for all study participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Research Foundation (grant UID: 90550) and the College of Health Science (grant no.: 533648), UKZN.
