Abstract
Little is known on the toxicity of nanomaterials in the user phase. Inclusion of nanomaterials in paints is a common nanotechnology application. This study focuses on the toxicity of dusts from sanding of paints containing nanomaterials. We compared the toxicity of titanium dioxide nanomaterials (TiO2NMs) and dusts generated by sanding boards coated with paints with different amounts of two different types of uncoated TiO2NMs (diameters:10.5 nm and 38 nm). Mice were intratracheally instilled with a single dose of 18, 54 and 162 µg of TiO2NMs or 54, 162 and 486 µg of sanding dusts. At 1, 3 and 28 days post-instillation, we evaluated pulmonary inflammation, liver histology and DNA damage in lung and liver. Pulmonary exposure to both pristine TiO2NMs and sanding dusts with different types of TiO2NMs resulted in dose-dependently increased influx of neutrophils into the lung lumen. There was no difference between the sanding dusts from the two paints. For all exposures but not in vehicle controls, mild histological lesions were observed in the liver. Pulmonary exposure to pristine TiO2NMs and paint dusts with TiO2NMs caused similar type of histological lesions in the liver.
Keywords
Introduction
Nanomaterials (NMs) are added to products to improve their properties or function. Since the Danish Nanodatabase was launched in 2012, an increasing number of such nanocomposite products has been registered. 1 Examples of nanocomposite products include paints, cosmetics and electronics with titanium dioxide nanomaterials (TiO2NMs) or carbon black (CB), and epoxy with carbon nanotube (CNT). In a life cycle of a product, NM may be released either as pristine particles or encapsulated in the matrix.
While the scientific literature on the toxicity of pristine NM is extensive, the knowledge of toxicity of nanocomposites is still limited. 2 –5 In this study, we focused on the toxicity of dust generated by sanding of paint containing TiO2NM. While larger sized TiO2 has been used for decades as white pigment, TiO2NM are, for example, added to paints to provide self-cleaning or odour removing properties or to increase scratch resistance. 6
Some TiO2NM have been shown to be inflammogenic and/or DNA damaging after pulmonary exposure. 7 –10 In 2006, International Agency for Research on Cancer (IARC) evaluated TiO2 as carcinogenic to experimental animals, while the evidence for carcinogenicity to humans was evaluated as insufficient. On this background, IARC classified TiO2 as possibly carcinogenic to humans (Group 2B) 11 and the European Chemicals Agency has opened a proposal for classification as a carcinogen (Carc 1B) for commenting. 12
We have previously published a dose–response study on the toxicity in mice following pulmonary deposition of sanding dust from acryl paint with and without a 17-nm coated TiO2NM. No increased toxicity of dust from the nano-doped paint (containing 10% TiO2NM) was observed compared to the reference paint without TiO2NM. Since the characteristics of the NM (e.g. coating and size) and the content of NM in the paint matrix may affect the toxicity of the sanding dust, the aim of the present study was to investigate the toxic effects of sanding dusts from paints with a similar design as the previous study but with a higher content of two differently sized and uncoated TiO2NM in the paints. In addition, histological changes in the liver were investigated.
Methods
Study design
The present study was designed to detect possible effects of large amounts of TiO2NM in paint dusts. We wanted to compare toxicity of sanding dusts containing either 100 nm TiO2NM or 10 nm TiO2NM or a mix of these two. However, our physicochemical characterization of the TiO2NMs revealed that the diameter of the largest TiO2NM was only 38 nm. Thus, the study design is suboptimal for detecting NM-specific effects. Mice were intratracheally instilled with a single dose of either pristine TiO2NM in two different sizes: TiO2 10.5 nm, TiO2 38 nm or two different dusts obtained by sanding paints with different amounts of the two TiO2NM samples: Sanding dust from a paint which contained 36% of the largest TiO2NM is denoted SD-TiO2 38 nm. Sanding dust from the other paint (denoted SD-TiO2 10.5nm+38 nm) which contained a mix of the two TiO2NM (12% TiO2 10.5 nm and 24% TiO2 38 nm, in total 36% TiO2NM). Animal exposures, sample collection and toxicological analyses were described previously. Therefore, these techniques are only described in brief in this section with references given for further information. 4,13,14 The data on DNA damage, cell count in broncheoalveolar lavage (BAL) fluid and histology from mice instilled with TiO2 10.5 nm, TiO2 38 nm, SD-TiO2 38 nm or SD-TiO2 10.5nm+38 nm have not been published previously.
Nanomaterials
The two TiO2NM samples, TiO2 10.5 nm and TiO2 38 nm, were obtained from NanoAmor (Nanostructured & Amorphous Materials, Houston, Texas, USA) and NaBond (NaBond Technologies, Limited, Shenzhen, China), respectively. The toxicity of other batches of the same TiO2-materials has been studied extensively in the ENPRA (Risk Assessment of Engineered NanoParticles) project under the designations; TiO2 10.5 nm as NRCWE-001 and TiO2 38 nm as NRCWE-004 (see Wallin et al. 9 and Kermanizadeh et al. 15 and references therein). TiO2 38 nm was included under the presumption that it was 100 nm but our transmission electron microscopy analysis showed that the diameter was 38 nm. The carbon black NM Printex 90 (CB) was a gift from Degûssa AG (Germany) and was included as an internal reference particle. This batch of Printex 90 has been studied extensively in our and other laboratories in Europe. The physicochemical characteristics of the NMs and the sanding dusts have been published previously. 8,16 –19 Key characteristics are summarized in Table 1.
Overview of samples and average data on key physical chemical characteristics.a

BET: Brunauer–Emmett–Teller; CB: carbon black; SD-TiO2 38 nm: sanding dust with TiO2 38 nm, SD-TiO2 10.5 nm+38 nm: sanding dust with TiO2 38 nm and TiO2 10.5 nm; Wt%: weight %; NRCWE: National Research Centre for the Working Environment.
aData has been published previously. 20
Painted boards and sanding dusts
The two tested alkyd paints were prepared and applied on wooden boards by Flügger A/S (Denmark): SD-TiO2 38 nm and SD-TiO2 10.5nm+38 nm contained 36% w/dry weight paint 38 nm of TiO2 (TiO2 38 nm), and 12% w/dry weight paint 10.5 nm of TiO2 (TiO2 10.5 nm) plus 24% w/dry weight paint TiO2 38 nm, respectively. Sanding dusts were collected during sanding of painted boards by an electrostatic sampler as previously described. 13 The physicochemical characteristics of the sanding dusts have been described before. 18,19 Briefly, scanning electron microscopy of the sanding dusts SD-TiO2 10.5nm+38 nm and SD-TiO2 38 nm showed that TiO2NM particles mostly remained embedded in the matrix. The sanding dusts seemed to consist of aggregates of sub-µm-size particles. The key characteristics have been summarized in Table 1.
Animals
Housing and acclimatization of mice before exposure has been described previously. 13,19 In brief, female C57BL/6 mice 5–7 weeks old (Taconic, Ry, Denmark) were acclimatized for 1–3 weeks before onset of the experiment. Mice were given food (Altromin no. 1324, Christian Petersen, Denmark) and water ad libitum during the whole experiment. The cages were stored at controlled temperature 21 ± 1°C and humidity 50 ± 10% with a 12-h light:12-h dark cycle. Mice were studied at 8 weeks of age.
Characterization of dispersion of TiO2NMs and sanding dusts in the exposure media
The characterization of exposure media has been described in detail previously. 19 In brief, size distributions measured by dynamic light scattering (DLS) show that the Z-average varied between 126 nm (162 µg) and 133 nm (18 µg) for TiO2 10.5 nm, between 178 nm (54 µg) and 219 µg (18 µg) for the TiO2 38 nm, between 274 nm (486 µg) and 280 nm (162 µg) for the SD-TiO2 10.5nm+38 nm and between 372 nm (54 µg) and 517 nm (162 µg) for the SD-TiO2 38 nm, respectively. The TiO2NM samples were very well dispersed. The sanding dust samples appeared to be relatively well-dispersed and were dominated by sub-µm particles. However, for all exposure media, minor amounts of approximately 3–6-µm-sized particles were detected in the intensity size distribution.
Exposure
The exposure of the mice has been described previously. 13,19 In brief, female C57BL/6 mice (Taconic, Ry, Denmark; N = 6 per exposure group) were exposed to TiO2 10 nm, TiO2 38 nm, SD-TiO2 38 nm or SD-TiO2 10.5nm+38 nm by a single intratracheal instillation at 8 weeks of age. Particle (3.24 mg/ml) and paint dust (9.72 mg/ml) were dispersed in 2% serum in water by sonication as described previously. 13 The doses were 18, 54 and 162 µg for the TiO2NM and 54, 162 and 486 µg for the paint dust. The dose levels, 18, 54 and 162 µg/mouse, were chosen to reflect occupational exposure levels. The Danish occupational exposure limit for TiO2 is 10 mg/m3. Based on previously measured particle size distribution of aerosolized TiO2 nanoparticles (NPs), 20,21 the deposited dose of TiO2 NPs 20,21 was estimated to be 12.4 µg/mouse during an 8-h working day. Thus, the chosen dose levels for TiO2 NPs reflect exposure during 1.5, 5 and 13 working days, respectively. The dose levels of paint dust were chosen to allow comparison of the effect of embedded TiO2 NP to the same dose of pristine TiO2 NPs as previously described. The animal exposure experiments were performed on different calendar days for different particles. In each experiment, vehicle instilled mice were included. The vehicle controls were combined for each post-exposure day (i.e. for post-exposure days 1, 3 and 28). Therefore, the control groups consisted of 24, 22 and 24 mice, respectively, for post-exposure days 1, 3 and 28, respectively. Each treatment group consisted of six mice. All animal procedures followed the guidelines for the care and handling of laboratory animals according to the EC Directive 86/609/EEC and the Danish law. The experiments were approved by the Danish ‘Animal Experiment Inspectorate’ under the Danish Ministry of Justice (2012-15-2934-00223).
Preparation of tissues
One, 3 or 28 days post-exposure mice were anesthetized and killed and the following was sampled: (1) BAL cells were separated from the BAL fluid by centrifugation before determination of the cellular composition by May–Grünwald–Giemsa stain and the total number of cells by using the NucleoCounter (Chemometec, Allerød, Denmark) as previously described, (2) small pieces of lung and liver tissue were snap frozen in liquid nitrogen and stored at −80°C until comet analysis 13 and (3) the specimens of liver tissue were fixed in 4% neutral buffered formaldehyde, paraffin-embedded.
Comet assay
The level of DNA strand breaks in frozen lung and liver tissue was determined by the alkaline comet assay using IMSTAR (France) as described previously. 14 In brief, deep frozen lung or liver tissues were pressed through a stainless steel sieve into ice-cold Merchant’s medium. We have previously shown that this procedure results in the lowest background levels of DNA strand breaks. 14 Aliquots of cell suspensions were embedded in agarose. Slides were lysed in 4°C cold lysing solution and kept overnight at 4°C. Slides were rinsed in cold electrophoresis buffer, alkaline treated in ice-cold electrophoresis buffer directly in electrophoresis chamber (0.3 M of sodium hydroxide, 1 mM of Na2EDTA, pH 13.2) placed on ice and subjected to electrophoresis (pH > 13) with 4°C cold circulating buffer. Slides were neutralized, the gels/slides were fixed in 95% ethanol, stained with SYBR® Gold and comets were scored. DNA damage was quantified as %DNA in tail (%T DNA). The samples were scored by fully automated PathFinder™ system from IMSTAR. As a positive assay control and to estimate the electrophoresis-to-electrophoresis variation, 0 and 30 μM of hydrogen peroxide (H2O2) exposed A549 cells from a single-batch exposure were included on each GelBond film in all electrophoresis runs. DNA damage in lung tissue from the control group (vehicle instilled mice) belonging to the TiO2 10.5 nm and TiO2 38 nm instilled samples varied greatly. However, the H2O2 treated A549 control cells included as plate control were within our historically expected ranges, suggesting that the large variation was caused by non-optimal sample treatment. Therefore, we disregarded the lung comet results from the TiO2NM instilled mice.
Histology
Four to six micrometre sections of formaldehyde, paraffin-embedded liver tissue were stained with haematoxylin and eosin for histological examination as previously described. Necrotic changes were represented by microfoci of necrosis, eosinophilic necrosis of single hepatocytes and by hepatocytes with pyknotic nuclei.
Statistics
The data on inflammation and DNA damage were assessed by non-parametric three-way analysis of variance (ANOVA) with post hoc Tukey-type multiple comparison test for effects showing statistical significance in the overall ANOVA test. The statistical analyses were performed in SAS version 9.4 (SAS Institute Inc., Cary, North Carolina, USA). The data on the incidence of histological changes in the liver were analysed by Fisher’s exact test (Graph Pad Prism 7.02, Graph Pad Software Inc., La Jolla, California, USA).
Results
Cell composition in bronchoalveolar lavage fluid
We determined the total number of BAL fluid cells and the number of macrophages, neutrophils, eosinophils and lymphocytes in the BAL cells. The number of inflammatory cells recruited into the lung lumen in mice instilled with TiO2NM and sanding dusts is shown in Tables 2 and 3, respectively. Neutrophil influx normalized to mass or specific surface area, respectively, is also shown in Figures 1 and 2 (only day 1), respectively. Dose-dependent pulmonary neutrophil influx was observed for all test materials and the response decreased over time. When normalized to mass, the neutrophil influx was larger for TiO2 10.5 nm compared to TiO2 38 nm (the difference was statistically significant (p < 0.05) for the 54-µg dose on day 1 post-exposure and 54- and 162-µg dose on day 3 post-exposure). There were no differences between mice intratracheally instilled with SD-TiO2 38 nm and SD-TiO2 10.5nm+38 nm at any time point for any cell type.
BAL fluid counts in mice, 1, 3 and 28 days post-exposure to 18, 54 and 162 µg TiO2NM and control mice.a

BAL: Broncheoalveolar lavage; CB: Carbon black; TiO2NM: titanium dioxide nanomaterial.
aValues are mean ± standard error of the mean.
bp < 0.01 compared to controls.
cp < 0.05 compared to TiO2 38 nm,
dp < 0.05 compared to controls.
ep < 0.01 compared to TiO2 38 nm.
BAL fluid counts in mice, 1, 3 and 28 days post-exposure to 54, 162 and 486 µg sanding dust from paint and control mice.a

BAL: broncheoalveolar lavage; TiO2: titanium dioxide; SD-TiO2 38 nm: sanding dust with TiO2 38 nm; SD-TiO2 10.5 nm+38 nm: sanding dust with TiO2 38 nm and TiO2 10.5 nm; CB: carbon black.
aValues are mean ± standard error of the mean.
bp < 0.01 compared to controls.
cp < 0.05 compared to controls.

Neutrophil influx in the lungs of mice instilled with 0, 18, 54 or 162 µg of TiO2 10.5 nm (a) or TiO2 38 nm (b) or 0, 54, 162 or 486 µg of SD-TiO2 38 nm (c) or SD-TiO2 10.5 nm+38 nm (d). TiO2: titanium dioxide; SD-TiO2 38 nm: sanding dust with TiO2 38 nm, SD-TiO2 10.5 nm+38 nm: sanding dust with TiO2 38 nm and TiO2 10.5 nm.
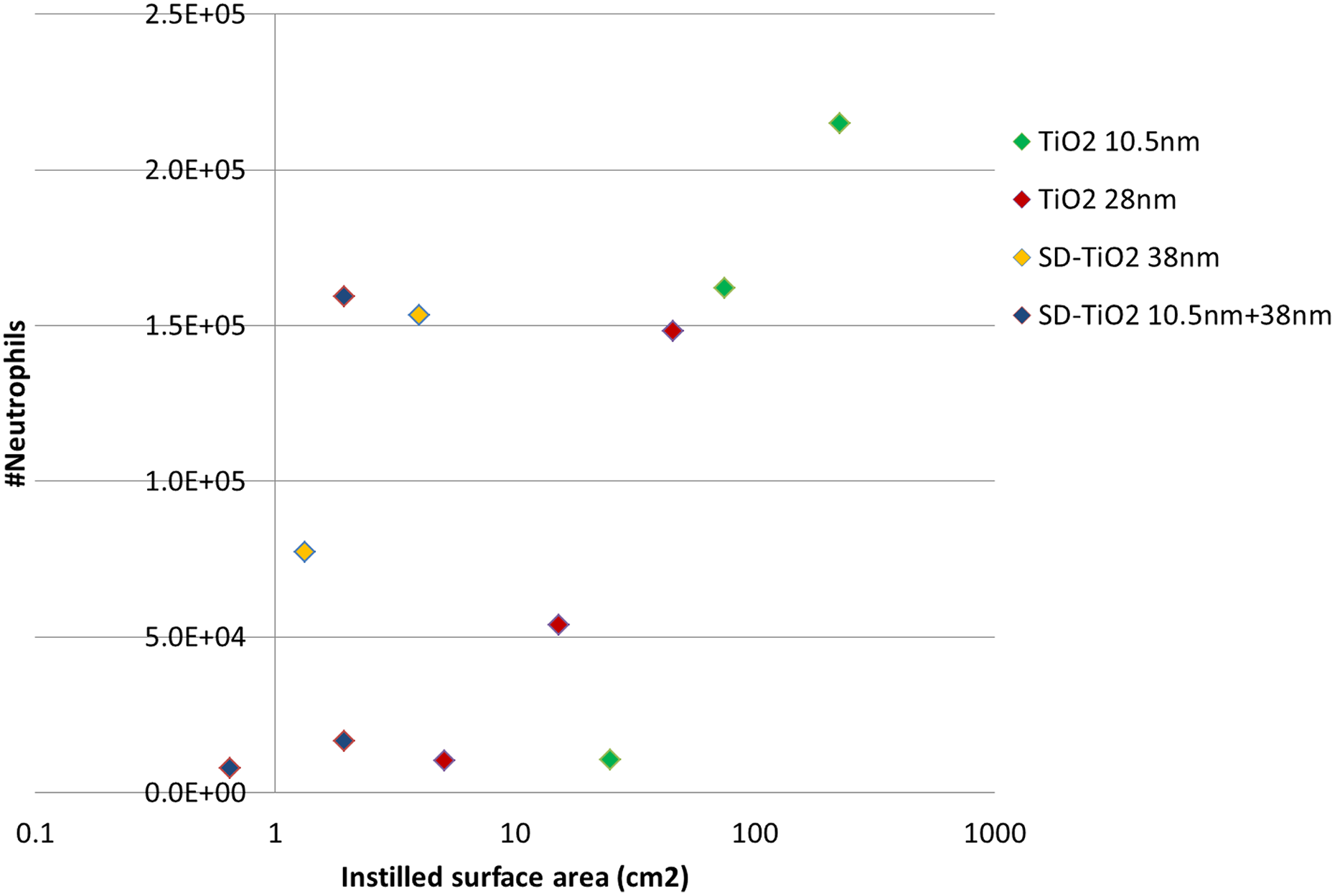
Correlation between surface area and neutrophil influx 1-day post-instillation.
DNA damage
We determined DNA damage in lung and liver tissue by measuring DNA strand breaks by the comet assay. As described in the ‘Method’ section, for technical reasons we had to disregard the lung comet results from the TiO2NM instilled mice. Neither TiO2NM38 nm and TiO2NM10.5nm+38 nm nor CB resulted in significant increases in DNA strand break levels in liver tissue at any of the tested doses and time points (data not shown).
The comet results from the sanding dust instilled mice are shown in Table 4. DNA strand break levels in lung and liver were unaffected by pulmonary exposure to both types of sanding dust.
DNA damage (%T DNA) in mice, 1, 3 and 28 days post-exposure to 54-, 162- and 486-µg sanding dust from paint, 162 µg of CB and control mice.a
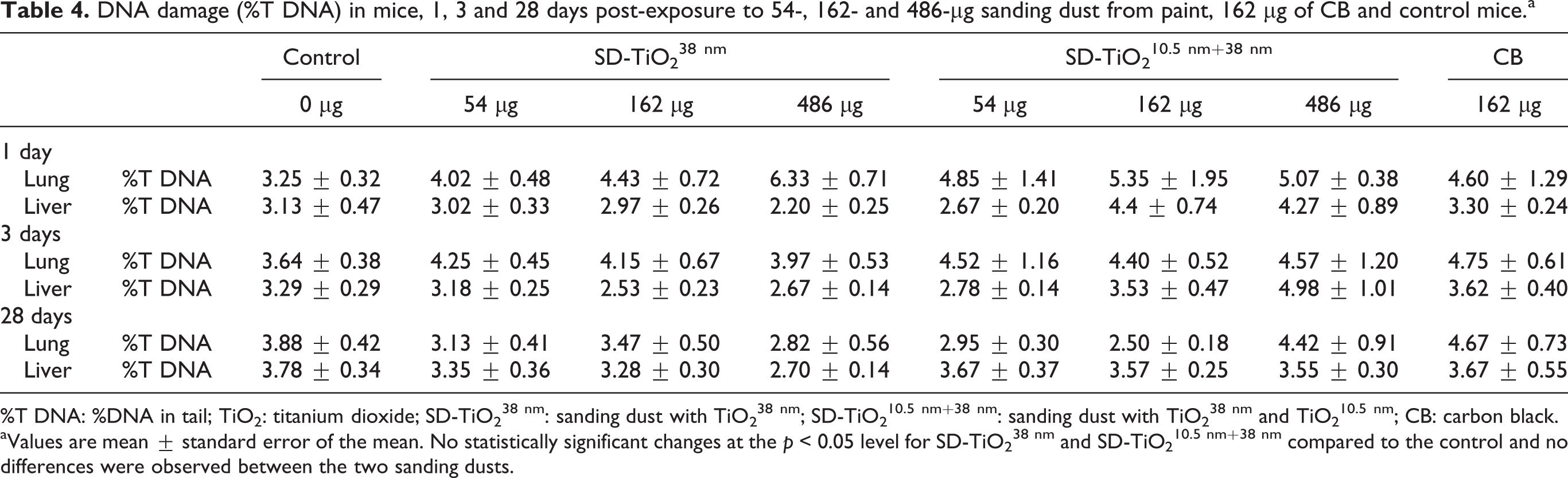
%T DNA: %DNA in tail; TiO2: titanium dioxide; SD-TiO2 38 nm: sanding dust with TiO2 38 nm; SD-TiO2 10.5 nm+38 nm: sanding dust with TiO2 38 nm and TiO2 10.5 nm; CB: carbon black.
aValues are mean ± standard error of the mean. No statistically significant changes at the p < 0.05 level for SD-TiO2 38 nm and SD-TiO2 10.5 nm+38 nm compared to the control and no differences were observed between the two sanding dusts.
Liver histology
Several histological lesions of similar type were observed in the livers from the exposed mice when compared to the controls (Table 5 and Figure 3). The most frequently recorded inflammatory lesions were polymorphonuclear cell foci. The observed eosinophilic necrosis of single hepatocytes was mostly located at central venules and surrounding granulomas and sporadically close to polymorphonuclear cell foci. Hyperplasia of Kupffer cells was not observed by following exposure to the SD-TiO2 10nm+38 nm, and hypertrophy of Kupffer cells was recorded only in one liver at day 3 after instillation to this test material. In the other exposure groups, the hypertrophy of Kupffer cells was often seen along with hyperplasia of Kupffer cells (Figure 3(m)).
Type and incidence of histological changes in the liver on days 1, 3 and 28 following intratracheal instillation with TiO2 38 nm, TiO2 10.5 nm, SD-TiO2 38 nm, SD-TiO2 10.5 nm+38 nm or CB.

TiO2: titanium dioxide; SD-TiO2 38 nm: Sanding dust with TiO2 38 nm; SD-TiO2 10.5 nm+38 nm: Sanding dust with TiO2 38 nm and TiO2 10.5 nm; CB: carbon black.
ap < 0.05 compared to controls.
bp < 0.001 compared to controls.
cp < 0.01 compared to controls.

Morphological structure of the mouse liver on day 1 (a, d, g, j and m), day 3 (b, e, h, k and n) and day 28 (c, f, i, l and o) after intratracheal instillation. (a) to (c): exposure to TiO2 38 nm (NRCWE-25): (a) to (c) – vacuolar degeneration (short arrows), (a) – hyperplasia of the bile ducts epithelium (long arrows), (a) and (b) – eosinophilic necrotic hepatocytes (asterisks), (b) – polymorphonuclear cell focus (head of arrows), (b) and (c) – numerous binucleate hepatocytes (long arrows), (c) – hypertrophy hepatocytes with pyknotic nucleus (head of arrows); (d) to (f): exposure to TiO2 10.5 nm (NRCWE-030): (d) to (f) – vacuolar degeneration (short arrows), numerous binucleate hepatocytes (long arrows), (d) – sinusoidal dilatation, (d) and (e) – eosinophilic necrotic hepatocytes (asterisks), (e) and (f) – polymorphonuclear cell foci (head of arrows), hyperplasia of Kupffer cells (numerous black ‘points’); (g) to (i): exposure to SD-TiO2 38 nm (NRCWE-032): (g) – numerous binucleate hepatocytes (short arrows), hypertrophy of Kupffer cells (long arrows), sinusoidal dilatation, (g) and (h) – eosinophilic necrotic hepatocytes (asterisks), (g) and (i) – polymorphonuclear cell foci (head of arrows), (h) – vacuolar degeneration (short arrows), granuloma (head of arrows); (j) to (l): exposure to SD-TiO2 10.5+38 nm (NRCWE-033): (j) – sinusoidal dilatation, (j) and (k) – polymorphonuclear cell foci (head of arrows), (j) to (l) – eosinophilic necrotic hepatocytes (asterisks), (l) – granuloma, hypertrophy of Kupffer cells (arrows); (m) to (o): liver of mice exposed to carbon black (Printex 90): (m) – parenchymatous degeneration, numerous binucleate hepatocytes (thick arrows), hyperplasia of Kupffer cells (short arrow), (n) – sinusoidal dilatation, (m) and (n) – hypertrophy of Kupffer cells (long thin arrows), (m) to (o) – polymorphonuclear cell foci (head of arrows), (m) and (o) – eosinophilic necrotic hepatocytes (asterisks), (o) – vacuolar degeneration (short arrows), hyperplasia of Kupffer cells (numerous black ‘points’); (p): The control group – typical microscopic structure of the mouse liver. Staining HE, magnification on the figures (a) to (p) as scale on (p) (bar = 50 µm). TiO2: titanium dioxide.
Hyperplasia of bile ducts epithelium and sinusoidal dilatation were observed sporadically in mice exposed to CB as compared to the livers from the mice exposed to either TiO2NM or to sanding dusts with TiO2NM. Hyperplasia of connective tissue near bile ductules or venules and oedemateus endothelial cells and portal venules or closed to blood vessels were solely observed in some of the mice exposed to CB.
Discussion
In the present study, we investigated the dose–response relations of inflammation, DNA damage and histological changes of (1) two different sized uncoated rutile TiO2NMs, NanoAmor (10.5 nm, code: TiO2
10.5 nm) and NaBond (38 nm, code: TiO2
38 nm), and (2) sanding dusts from paints with either a mix of TiO2
10.5 nm and TiO2
38 nm (SD-TiO2
10.5nm+38 nm) or only TiO2
38 nm (SD-TiO2
38 nm) in mice. Toxicity was evaluated 1, 3 and 28 days after a single intratracheal instillation. We observed very similar dose-dependent inflammatory response for the two paint dusts, SD-TiO2
10.5nm+38 nm and SD-TiO2
38 nm. For the pristine TiO2NPs, the inflammatory response measured as neutrophil influx per mass unit was larger for TiO2
10.5 nm compared to TiO2
38 nm. The difference in response was statistically significant for the 54
Study design
The present study was designed to detect possible effects of large amounts of two differently sized, but otherwise similar TiO2NM in paint dusts. Both TiO2NMs were rutile and uncoated to exclude a possible toxicological effects of crystal phase and coating, respectively. To maximize the ability to detect a possible effect of TiO2NM in paint matrix and the effect of differently sized TiO2NM, we had paints with the largest possible amount (36% (w/w dry paint)) of two TiO2NM (10.5 nm and 38 nm) prepared. The two tested paints, SD-TiO2 38 nm and SD-TiO2 10.5nm+38 nm contained 36% 38 nm of TiO2 (TiO2 38 nm), and 12% 10.5 nm of TiO2 (TiO2 10.5 nm) plus 24% TiO2 38 nm, respectively. A third paint containing 36% TiO2 10.5 nm was also prepared. However, we had to exclude this from the toxicological testing because the paint cracked into large flakes making sanding impossible. The dose levels in the current study were chosen to reflect occupationally relevant exposure levels and to allow comparison with a previous paint dust study using the same dose levels of TiO2NM (18, 54 and 162 µg) and paint dust (54, 162 and 486 µg), and the same post-exposure time points (1, 3 and 28 days). The doses of 18, 54 and 162 µg of TiO2NM correspond to the estimated pulmonary deposition during 1.5, 5 and 13 eight-hour working days at the Danish occupational exposure limit for TiO2 (6 mg/m3 Ti equivalent to 10 mg/m3 TiO2). 19 As described in detail previously, the amount of TiO2NM in the sanding dust doses of 54, 162 and 486 µg corresponds to 19, 58 and 175 µg of TiO2NM, respectively, 19 thus allowing comparison of the toxicity of pristine and embedded TiO2NM.
Pulmonary exposure by inhalation is the gold standard for assessing the toxicity of particles. However, intratracheal instillation is a time- and cost-effective alternative to inhalation. 22,23 Instillation was the only possibility due to a limited amount of sanding dust. We have previously shown that the global transcriptional response to TiO2NM inhalation was very similar to the global pulmonary transcriptomic pattern following intratracheal instillation, 10 indicating that the biological response to inhaled and instilled TiO2NM is very similar.
Pulmonary effects of TiO2NM
Many studies have demonstrated that the total surface area of deposited low-toxicity low-solubility particles is a better predictor for inflammation than mass. 8,17,24,25 The data on neutrophil influx following exposure to the pristine TiO2NM are consistent with this observation (Figure 2, day 1). Both TiO2NMs induced inflammation 28 days post-exposure, but only at the highest dose, 162 µg/mouse. The observed inflammatory response is in agreement with previously published data on global transcription patterns in lung tissue from the same mice as in the present study, where we detected that the number of differentially expressed genes were much higher in mice instilled with TiO2 10.5 nm compared to mice instilled with the larger TiO2 38 nm. Both TiO2NMs induced biological pathways related to inflammation and immune response. 19
For technical reasons, we had to disregard the lung Comet results from the TiO2NM instilled mice. However, we recently reported that another batch of TiO2 10.5 nm increased the DNA damage in lung tissue (measured as %T DNA) on day 28 after instillation of 18, 54 or 162 µg/mouse, while this endpoint was unaffected for all doses on day 1 and day 3. 9 This suggests that TiO2 10.5 nm is able to induce pulmonary DNA damage.
Pulmonary toxicity of sanding dust from nano alkyd paints
Both sanding dusts induced less influx of neutrophils compared to the pristine NMs when considering mass. Neutrophil influx was only observed 28 post-exposure at the very high dose of 486-µg SD-TiO2 38 nm. The inflammatory response was overall similar for the two paint dusts, and thus neutrophil influx was unaffected by the addition of the small TiO2NM to the paint. The Brunauer–Emmett–Teller (BET) surface areas of the two paint dusts were quite similar. Thus, addition of 12% of the small TiO2 10.5 nm did not affect the BET surface area of the sanding dust, suggesting that the TiO2NMs are embedded in the paint matrix. When normalized to the specific surface area, the paint dusts were more inflammogenic compared to TiO2NM (Figure 2). This may reflect that the alkyd paint matrix contributes to the inflammatory influx. Overall, the observed pulmonary toxic responses are in line with our two previous mouse studies on sanding dust from (1) a single-dose study of nine different NM-containing paints and lacquer and six corresponding conventional products without NMs 3 and (2) a dose–response study of acryl-based paint with and without 10% of a coated 17 nm of TiO2NM: None of the studies showed increased toxicity of the NM-containing product in terms of inflammation and genotoxicity compared to the reference products. To our knowledge, only one additional in vivo study has been published on the inflammatory effects mimicking potential exposures to downstream products of TiO2NM, in this case the mechanical disintegration fragments of paints. 5 In that study, little to no adverse toxicological effects was observed when mice were exposed by oropharyngeal aspiration to powdered particles from paint NM-containing paint. Based on the above studies we conclude 3,5 that the toxicity of sanding dust from paint in terms of inflammation and DNA damage was unaffected by the presence of NM.
Hepatic toxicity of sanding dust from nano alkyd paints
No effect on DNA damage levels was detected in the livers of mice instilled with sanding dusts compared to the controls. However, we observed several mild histological changes in the livers from mice instilled with both types of TiO2NM and both types of sanding dusts from alkyd paint.
The hepatic lesions were primarily of the inflammatory and necrotic type (Table 5), and the different particles and sanding dusts generally induced the same type of lesions with a few exceptions. Pyknotic nuclei were not present in the SD-TiO2 38 nm group, hyperplasia of Kupffer cells was not observed in SD-TiO2 10.5nm+38 nm and in this exposure group, hypertrophy of Kupffer cells was seen only in one mouse. In contrast, no histological changes were found previously in livers from mice instilled with sanding dusts from an acryl-based paint with and without a coated 17 nm of TiO2NM (TiO2 17 nm).
The observed differences in liver histopathological responses could be caused by translocation of NM from the lungs to the blood circulation followed by accumulation in Kupffer cells in the liver (previously discussed in the study by Saber et al. 13 ). TiO2 17 nm and TiO2 10.5 nm have been detected in the livers of mice at 1, 28 and 180 days post-intratracheal instillation using hyperspectral imaging, 26,27 and it is therefore likely that a minute fraction of the deposited TiO2NM in mice in the present study also translocated from the lungs and accumulated in the liver. However, translocation is highly size-dependent and much less translocation would therefore be expected for the paint dust particles. The paint in the previous study contained 10% of TiO2NM 4 while the paint in the present study contained 36% TiO2NM. We have no experimental evidence suggesting that there is a larger fraction of nanosized particles in the alkyd sanding dust, as no significant differences in size distribution were observed when comparing dust generated during sanding of acryl paint 28 with alkyd paint. 18 In the study by Smulders et al., biodistribution of milled-down aged paint with either TiO2NM or bulk-size TiO2 and the corresponding pristine TiO2NM was tested using oropharyngeal aspiration in mice 5 : Thirty days after the first of five consecutive aspiration – doses on days 0, 7, 14, 21 and 28 in mice (total dose 100 µg equivalent to 4 mg/kg) the hepatic content of TiO2 was increased (approximately 0.3 ng/mg tissue) in mice exposed to the paint containing TiO2NM. However, the results are difficult to interpret because exposure to TiO2NM alone did not increase TiO2 content in the liver.
The observed hepatic histological lesions in the present study could also be caused by differences in signalling from the pulmonary region: The toxicity of the paint matrix itself may differ (alkyd in the present study vs. acryl in the previous study) leading to differences in systemic signalling. Previously published transcriptional profiling of lung tissue revealed 1 and 12 differentially regulated genes in lung tissue from mice exposed to conventional paint acryl dust and TiO2NM-containing acryl paint dust, respectively, as compared to 4 and 74 differentially regulated genes following exposure to the alkyd SD-TiO2 38 nm and SD-TiO2 10.5nm+38 nm, respectively, perhaps suggesting matrix-depending toxicity. 19
In a recent study on effects following pulmonary deposition of sanding dust from CNT-epoxy composites, we also observed histological lesions in the liver. 13 These changes seemed to be NM-mediated because the lesions only occurred in mice instilled with pristine CNT or sanding dust from epoxy containing CNT, while no lesions were observed in the liver from mice instilled with sanding dust from reference epoxy (without CNT).
Conclusions
Pulmonary exposure to pristine TiO2NM and sanding dusts from paint with TiO2NM resulted in dose-dependent increase of influx of neutrophils into the lung lumen. The level of pulmonary inflammation in terms of neutrophil influx in mice instilled with sanding dust was not affected by the different types of TiO2NM in the paints. Similarly, the histological lesions in the liver were overall of similar type in mice instilled with the two types of dusts. The results are in agreement with our previous paint dust study regarding the pulmonary inflammation, but in contrast to a previous study reporting no histological in mice instilled with sanding dust from acryl-based paint with or without coated TiO2NM. The results of the study underline that it is still difficult to predict the toxicity of different nanocomposites. The effects of nanocomposites may differ due to a combination of several parameters such as NM (type, amount) and matrix. Further studies are needed to clarify physicochemical predictors of the toxicity of nanocomposites.
Footnotes
Acknowledgements
The technical assistance from Lourdes Pedersen, Elzbieta Christiansen, Michael Guldbrandsen, Eva Terrida, Lisbeth Pedersen, Yahia Kembouche, Anne-Karin Asp and Aleksander Penkowski is gratefully acknowledged. The Danish paint company Flügger A/S kindly supplied the painted boards. Niels Hadrup is thanked for performing the statistical analysis of the histological data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from the European Union Seventh Framework Programme (FP7/2007-2013) under grant agreement no. 247989 (Nanosustain), The Danish Working Environment Research Fund (Danish Centre for Nanosafety, grant 20110092173/3 and Danish Centre for Nanosafety II) and the European Union Seventh Framework Programme (FP7/2007-2013) under grant agreement no. 310584 (NanoREG).
