Abstract
Chlorhexidine (CHX) is a cationic biguanide compound that has been widely used for disinfection of skin, mucous membranes, and medical instruments. Poisoning has been occurred occasionally due to its easy accessibility. Some fatal cases developed acute respiratory distress syndrome (ARDS) from aspiration of CHX directly into the lung. There is no preclinical information about the pulmonary toxicity of CHX available since the products of CHX are usually developed for disinfection by topical use. In this study, the acute pulmonary toxic effects of CHX following an intratracheal instillation in rats were investigated. Rats were exposed either to CHX at concentrations of 0.02% and 0.2% or to distilled water at a volume of 500 μl/kg b.w. CHX at concentration of 0.2% caused changes in hematological and biochemical values including white blood cell count (WBC), total protein (TP), albumin (ALB), lactate dehydrogenase (LDH), blood urea nitrogen (BUN) and creatinine (CRE), and induced inflammatory reactions including intra-alveolar edema and hemorrhages, as well as resulted in the target organ concentration in lungs at the level of about 1.0 μg/g and maintained for more than 1 week, when administered intratracheally in rats. The cytotoxic action of CHX might induce those detrimental reactions in rats.
Introduction
Chlorhexidine (CHX) is a symmetrical cationic molecule containing two 4-chlorophenyl rings and two biguanide groups connected by a central hexamethylene chain. The presence of two symmetrically positioned basic chlorophenyl guanide groups attached to a lipophilic hexamethylene chain aids in rapid absorption through the outer bacterial cell wall, causing irreversible bacterial membrane injury, cytoplasmic leakage and enzyme inhibition. 1 Due to its wide spectrum of bactericidal and antiviral activity, CHX is used as a common ingredient in various formulations ranging from skin disinfectants in healthcare products to antiplaque or anticariogenic agents, both in human and veterinary medicine. 2,3 In human use, CHX is applied to prevent and treat the redness, swelling and bleeding gums associated with gingivitis. In veterinary medicine, CHX is used as a general purpose disinfectant for cleansing wounds, skin, instrument, and equipment. Because of its antiseptic properties and low potential for systemic or dermal toxicity, CHX has been incorporated into shampoos, ointments, skin and wound cleansers, teat dips, surgical scrubs, etc. 4
Many cases of poisoning have occurred in Japan recently and are involved in disinfectants and antiseptics because they are easily accessible. Accidental ingestion by children or the elderly and suicidal ingestion of these compounds occasionally occurred. 5 –11 Two cases of accidental intravenous injection to patients, 7,8 a fatal case of suicidal injection, 9 a survival case of suicidal ingestion 10 and a fatal case of accidental ingestion 11 have been reported. The development of acute respiratory distress syndrome (ARDS) was reported after accidental intravenous injection of CHX 7 and accidental ingestion of CHX. 11 Although it is highly possible that direct pulmonary damage might be the cause of ARDS, there is no preclinical information about the pulmonary toxicity of CHX.
In our laboratory, great endeavors have been made for the determination of disinfectants or surfactants in biological samples and evaluation of toxicological characteristics of these compounds for a forensic purpose. We have previously reported a sensitive high-performance liquid chromatographic (HPLC) method to determine benzalkonium chloride, a cationic surfactant used as a disinfectant in blood and tissue samples, 12 and also reported a specific determination of linear alkylbenzenesulfonates, anionic surfactants used widely in household products in the whole blood by HPLC method. 13 The kinetic characteristics and toxic effects of benzalkonium chloride following various routes of administration in rats were also investigated. 14,15 Determination of CHX in the whole blood by HPLC method with solid phase extraction and the kinetic characteristics following an intravenous injection were also previously reported. 16 The aim of this study was to investigate the acute pulmonary toxic effects following an intratracheal instillation of CHX. The way of intratracheal instillation has been used usually to examine direct pulmonary toxicities of a chemical or toxin. 17,18 The hematological examination and biochemical analysis of serum, as well as the histopathological examination were carried out. Blood and tissue CHX concentrations in lung, liver and kidney were determined following an intratracheal instillation of a CHX product, Maskin® solution, using our previous reported method. 16
Materials and methods
Chemicals
A chlorhexidine digluconate solution (a commercially available product of CHX, Maskin® solution [20 w/v% CHX digluconate in water], which is one of the most common disinfectants in Japan) was purchased from Maruishi Pharmaceutical Ltd. (Osaka, Japan). The drug was diluted with distilled water to obtain solution of 0.02% and 0.2% for the test. Chlorhexidine digluconate (20% solution in water) used as a standard and chlorpromazine hydrochloride used as an internal standard for CHX assay were purchased from Sigma-Aldrich (St. Louis, MO, USA) and ICN Biomedicals (Solon, OH, USA), respectively. The reagents for mobile phase were of HPLC grade and all other reagents were of analytical grade.
Animals
The experimental protocols were approved by Southeast University Faculty of Medicine Animal Experimental Committee. Thirty 7-week-old male Sprague-Dawley (SD) rats (provided by the Experimental Animal Center in Zhejiang, Hangzhou, China) were used in this study. Three or four rats each were housed in individual plastic cages under following conditions: 23 ± 2°C, 60% ± 10% humidity and 12-h light and dark cycles in a controlled room. They were fed pellet chow and water ad libitum.
The rats were divided into three groups at random (10 rats in each), receiving either distilled water (control group) or CHX at concentrations of 0.02% (low-dose group) and 0.2% (high-dose group). Under ether anesthesia, they were intratracheally instilled with the drug solution at a volume of 500 μl/kg b.w. The experimental doses were selected based on the results of a preliminary study and the solutions of CHX at concentrations of 0.2% and 0.02% were the most common diluted liquids used in medicinal purposes and in daily life. Toxic effects were investigated following administration. Five rats in each group were subjected to necrosis at 1 day and 7 days after administration. Blood and tissue samples were collected for hematological examination, histopathological studies and concentration determination. Male SD rats were used in this study because (1) no sex-specific effect of CHX was reported, 19 (2) male rat was thought to be a little sensitive to CHX based on LD50 (21 μg/kg for male and 23 μg/kg for female in intravenously) or LC50 (0.30 μg/L for male and 0.43 μg/L for female in inhalation) values reported 19,20 and (3) animal-welfare concerns were considered. All experimental procedures conformed to the guide for the care and use of laboratory animals. 21
Hematology and biochemistry
Rats were sacrificed by exsanguinations under ether anesthesia at the first day or seventh day following an intratracheal administration. Before necropsy, blood was drawn from the femoral artery, 2 mL was anticoagulated for hematological examination, 2 mL was allowed to clot for a biochemical analysis of serum and 2 mL was stored in –60°C refrigerator for CHX concentration assay. Hemetological parameters, including white blood cell count (WBC), red blood cell count (RBC), hemoglobin concentration (Hb), platelet counts (PLT), percent of neutrophils (NE%), percent of lymphocytes (LY%), percent of eosinophils (EO%), percent of monocytes (MO%), mean corpuscular volume (MCV), hematocrit (HTC), mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC), were analyzed using a blood cell counter (Pentra 60, ABX company, French). Serum biochemical parameters, including albumin (ALB), alkaline phosphatase (ALP), blood urea nitrogen (BUN), creatinine (CRE), glucose (GLU), glutamic oxalacetic transaminase (GOT), glutamic pyruvic transaminase (GPT), lactate dehydrogenase (LDH), total cholesterol (T-CHO) and total protein (TP) were analyzed using a biochemical blood analyzer (Hitachi 7150, Hitachi, Japan).
Histopathology
After collecting the blood, the rats were sacrificed by cervical dislocation. The brain, heart, lungs, liver, spleen and kidneys were removed carefully. The organs were cut into two parts. One part of organs was fixed in a 10% formalin solution containing neutral phosphate-buffered saline. The tissues were routinely processed, embedded in paraffin, sectioned at 3–5μm and then stained with haematoxylin and eosin for microscopic examination. Another part of tissues was used for concentration determination.
The histopathological analysis was performed by two experienced pathologists independently. A blind study was conducted to measure the tissue outcome, observing the presence of neutrophils, lymphocytes, plasmocytes, eosinophils and macrophages. Based on these findings, the degree of pathological changes was defined as follows: –, not observed (when inflammatory cells were absent); ±, very slight (when inflammatory cells were sparsely present); +, slight (when inflammatory cells were present in very small groups); ++, moderate (when inflammatory cells were densely present or in some groups); +++, severe (when inflammatory cells were present in a large number, configuring great intenseness).
Determination of CHX in blood and tissues
CHX in the whole blood and tissue samples was extracted by solid phase extraction and determined by HPLC utilizing our previously described method. 16 Lung, liver and kidney were selected as tissue samples because: (1) these organs have been reported as affected tissues in human poisoning cases and in animal experiments 5,22 and (2) the detected CHX levels were higher than those in other tissues in our preliminary study (data not shown), which would allow quantification of lower CHX levels at later time.
Statistical analysis
The quantitative data were expressed as means and standard deviation. A multiple variance analysis and Duncan’s multiple range tests were used to compare the relative organ weights and results of the blood biochemistry and hematology for the two experimental groups with those for the control rats. The software used for the data analysis was SPSS 13.0 for Windows (SPSS Inc., Chicago, IL, USA). Comparisons within groups were made using repeated measures using one-way ANOVA. Incidence data were compared by chi-square analysis. p Values of less than 0.05 were considered significant.
Results
General effects of toxicities and gross observation
After administration of CHX, no abnormal behavior and clinical symptoms were found in rats in the low-dose group. Sneeze, respiratory rales and rapid breathing were observed in almost all rats in the high-dose group at the beginning of administration. The activities of rats were also shown to be slow. The rats usually recovered within 2 h after administration. In necropsy, lung edema and hemorrhage were observed in 3/5 rats at 1 day after administration in the high-dose group by naked eyes. Other organs looked normal in all experimental animals.
Hematology
After 1 day and 7 days of exposure to CHX, a statistically significant increase in the WBC appeared in the high-dose group when compared to the control group. No significant changes in other hematological parameters were observed throughout the experimental period (Table 1 ).
Hematological values following intratracheal exposure to chlorhexidine in rats (n = 5) a
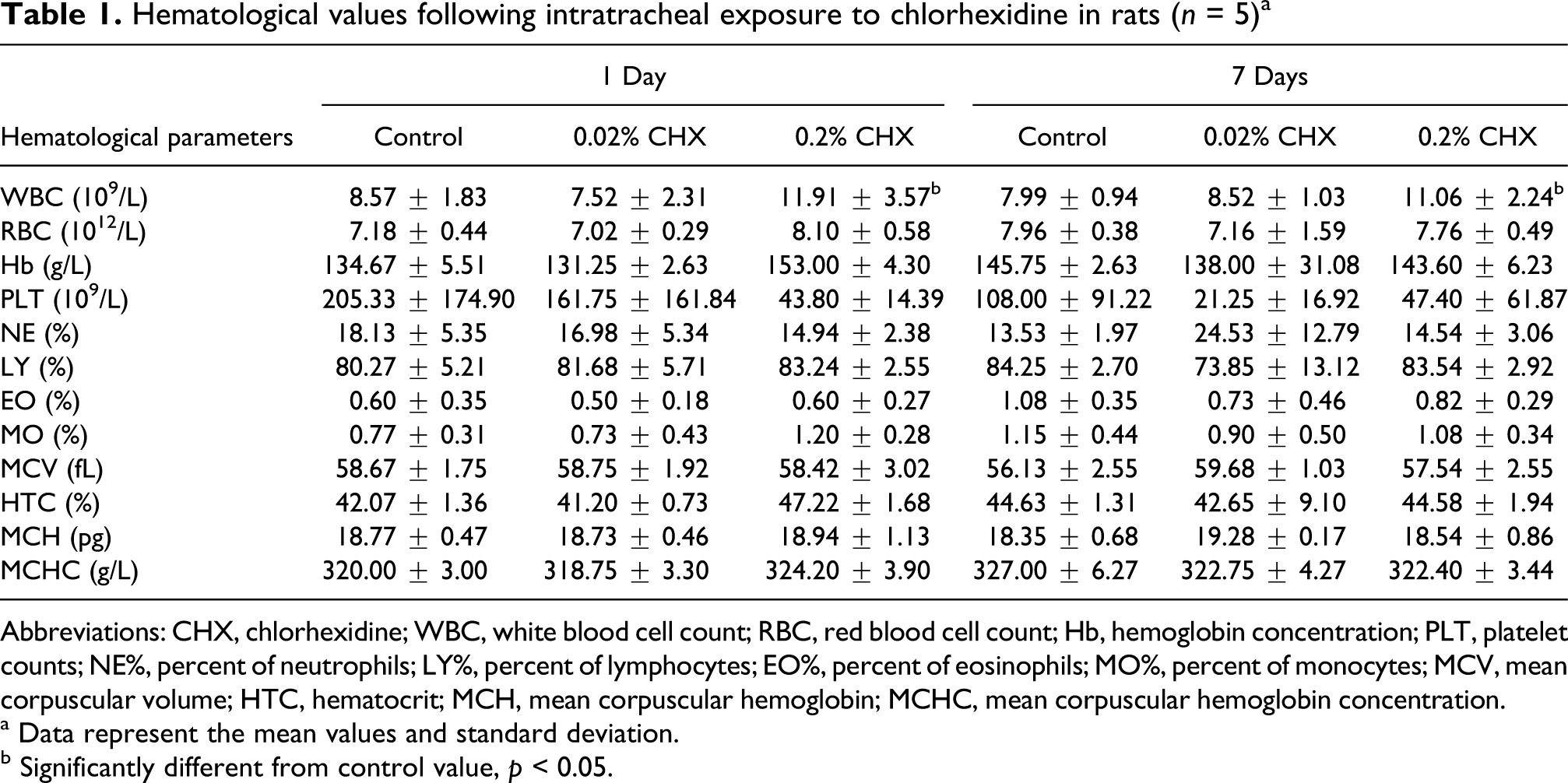
Abbreviations: CHX, chlorhexidine; WBC, white blood cell count; RBC, red blood cell count; Hb, hemoglobin concentration; PLT, platelet counts; NE%, percent of neutrophils; LY%, percent of lymphocytes; EO%, percent of eosinophils; MO%, percent of monocytes; MCV, mean corpuscular volume; HTC, hematocrit; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration.
a Data represent the mean values and standard deviation.
b Significantly different from control value, p < 0.05.
Serum biochemistry
After 1 day treatment, a statistically significant increase in the TP, BUN and CRE appeared in the high-dose group when compared to the control group. After 7 days exposure to CHX, a statistically significant decrease in the TP and ALB was noted in low-dose and high-dose group when compared to the control group. The LDH and CRE were significantly higher in the high-dose group than those in the control group after 7 days CHX treatment. Other serum biochemical parameters such as GPT, GOT, ALP, GLU and CHO did not show statistical differences from the control group (Table 2 ).
Serum biochemical values following intratracheal exposure to chlorhexidine in rats (n = 5) a
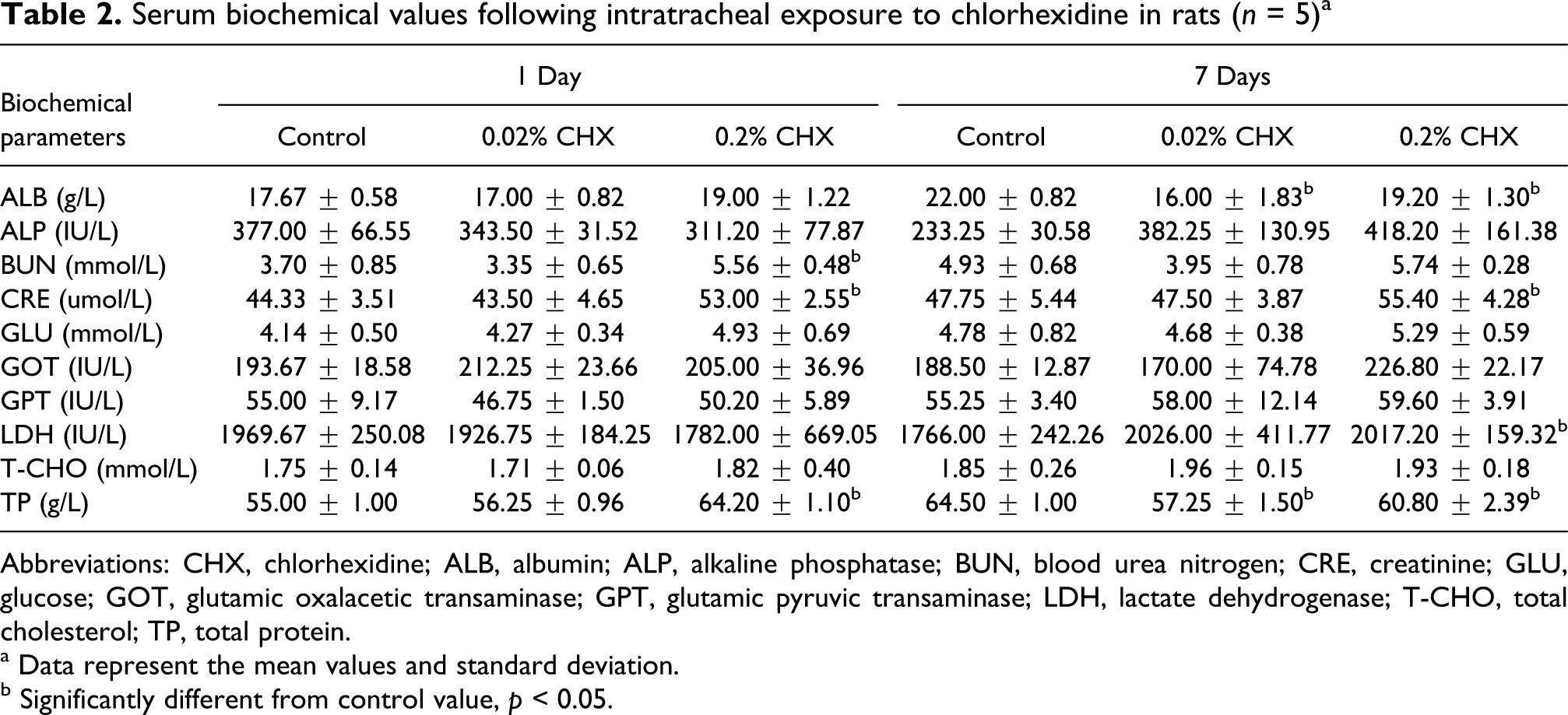
Abbreviations: CHX, chlorhexidine; ALB, albumin; ALP, alkaline phosphatase; BUN, blood urea nitrogen; CRE, creatinine; GLU, glucose; GOT, glutamic oxalacetic transaminase; GPT, glutamic pyruvic transaminase; LDH, lactate dehydrogenase; T-CHO, total cholesterol; TP, total protein.
a Data represent the mean values and standard deviation.
b Significantly different from control value, p < 0.05.
Histopathology
Histopathological observations of tissues were conducted (Table 3 , Figure 1 ). Lungs from high-dose CHX exposure showed severe edema and congestion to the alveoli and capillaries, and infiltration of focal inflammatory cells at 1 day, but these changes had decreased by 7 days after exposure. Slight interstitial edema and infiltration of inflammatory cells were observed in lungs in low-dose group which compared to slight and negligible changes in the control group. Slight edema on liver cells was found in all CHX-treatment rats. No infiltration of inflammatory cells was present on liver cells in all rats. There were no remarkable histophathological changes in brain, heart, spleen and kidney in all groups.

The histophathological changes following intratracheal instillation of distilled water (control), 0.02% chlorhexidine (CHX) and 0.2% CHX (×100). Hematoxylin and eosin stains of lung at 1 day (a), 7 days (b) and liver at 1 day (c) and 7 days (d) after exposure.
Histopathological score of tissues following intratracheal exposure to chlorhexidine in rats a

Abbreviation: CHX, chlorhexidine.
a The degree of pathological changes: –, not observed; ±, very slight; +, slight; ++, moderate; +++, severe.
Determination of CHX in blood and tissues
Using our previous reported method, 16 blood, lung, liver and kidney were purified with solid phase extraction and the concentration of CHX in blood and tissues were determined by HPLC method. As shown in Table 4 , after 1 day exposure the concentration of CHX in lung was almost 10 times of in kidney and about 20 times of in liver, especially for the rats in high-dose group. At 7 days after exposure, the concentration of CHX in lung was even higher than that at 1 day, and was as high as 30 times of in kidney. However, the levels of CHX in kidney and liver were decreased at 7 days after exposure. The CHX level in liver after 7 days exposure in low-dose group was so low that it was under the detection limit. In the meanwhile, CHX was not detected in the blood in all groups.
Concentrations of chlorhexidine in tissues following intratracheal exposure to chlorhexidine in rats (ng/g; n = 5) a
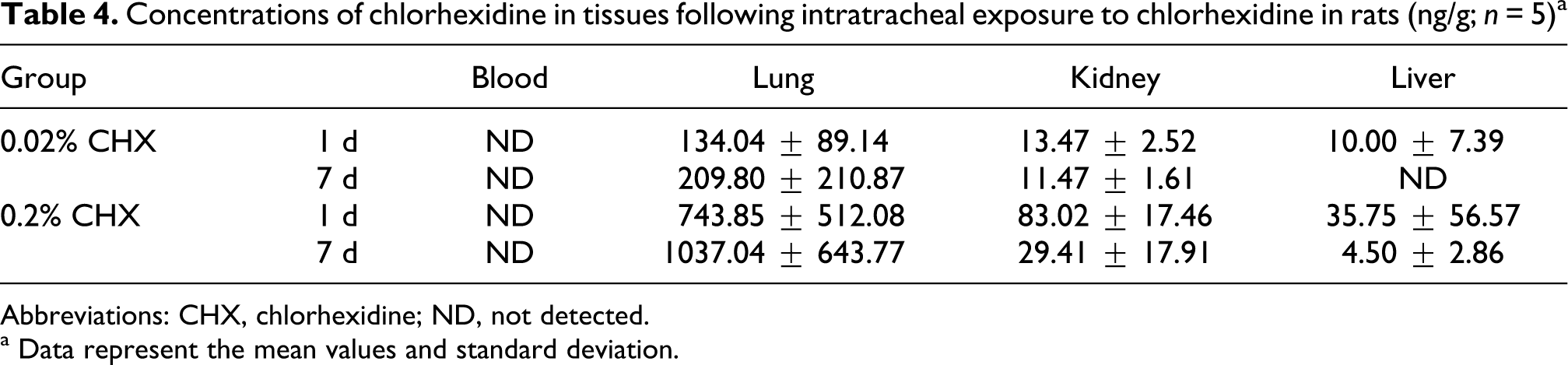
Abbreviations: CHX, chlorhexidine; ND, not detected.
a Data represent the mean values and standard deviation.
Discussion
CHX is a cationic biguanide compound that has been in use as an antiseptic since the early 1950s. It is widely available for disinfection of skin, mucous membranes, and medical instruments. Maskin® is a popular brand of disinfectant containing CHX and is in common use in Japan. It has been ubiquitous in hospitals and the community for over four decades. Accidental ingestion or suicidal injection seems to have occurred more frequently than previously reported. 5 –11 Although CHX has been considered ordinarily to have low toxicity due to its poor absorption via oral or dermal administration, its high toxic effects when given parenterally are not well characterized. The bioavailability of CHX via oral or dermal administration can be estimated to be quite low due to its ionic properties. However, a high pulmonary risk of CHX should not be overlooked because its high concentration can be reached by the direct aspiration into the lung. A fatal case caused by accidental ingestion of approximately 200 mL of Maskin® (5% CHX) was reported. 11 The patient developed hypotension and rapid deterioration of consciousness, and died of ARDS. The CHX was suspected to absorb through the pulmonary alveoli following aspiration, not from the gastrointestinal tract. Another case also developed ARDS by unintentional injection of CHX. 7 The pulmonary toxic effects of CHX have not been investigated since the products of CHX are usually developed for disinfection by topical use. The aim of this study was to investigate the acute pulmonary toxic effects of CHX in rats following an intratracheal exposure.
CHX is bactericidal via destruction of the cell membrane and shows strong action to biomembrane. Its antibacterial activity arises from its positive charge at physiological pH, which produces nonspecific binding to the negative-charged membrane phospholipids. In our preliminary study, dyspnea caused by widespread pulmonary dysfunction appeared when CHX was instilled into the trachea at a concentration of 0.5% or 0.3%. We were not able to make the appropriate observation of toxic effects because rats died soon after the exposure. Therefore, we exposed the rats with 0.2% and 0.02% of CHX, which were the common concentrations used in medicinal purposes and in daily life.
By exposure to a lower dose of 0.02% CHX, no abnormal behavior was observed and no significant changes in hematological and serum biochemical parameters were detected. Histopathological examination revealed the only slight interstitial edema and infiltration of inflammatory cells in lungs and the concentration of CHX in lungs was at the level of 0.1–0.2 μg/g, which would be too low to induce damage on lungs. By exposure to a higher dose of 0.2% CHX, respiratory rales, breathing speedup and slow down on activity were observed at the beginning of administration. Hematological and biochemical parameters such as WBC, TP, ALB, LDH, BUN and CRE were changed significantly; these changes would be resulted from the tissue injury on lungs, liver and kidneys caused by the cytotoxic action of CHX. Indeed, several studies have reported that CHX is a cytotoxic agent. 23–31 It has cytotoxic activity on cultured alveolar bone 25 and gingival epithelial cells 26 and induces a dose-dependent reduction of human gingival fibroblast proliferation and reduces both collagen and non-collagen protein production at concentrations with little effect on cellular proliferation. 27–29 Ribeiro et al. have observed that CHX at 0.12% induced a significant increase in the level of DNA strand breaks in rat leukocytes after oral administration. 30 Grassi et al. indicated that rat blood and kidney were particularly sensitive organs for DNA-damaging following CHX exposure. 31 Nephrotoxic and hepatotoxic effects of CHX in rat after oral administration were investigated. 22 BUN, GOT and GPT activities were elevated 24 h after administration of CHX, suggesting both liver and kidney toxicities. A suicidal case with a striking aminotransferase rise was also reported. 10 By intratracheal exposure to CHX in this study, GOT and GPT levels were elevated in the higher dose group but not significant when compared to the control group. The histopathological analysis observed only slight edema on liver cells and the concentrations of CHX in liver were correspondingly low. However, TP and ALB levels, which reflect the function of protein synthesis in liver, were changed significantly, suggesting some impaired liver function. Elevated BUN and CRE concentrations were observed, indicating the impaired renal function. However, the histopathological examination on kidneys revealed no remarkable changes. The amount of drug reaching the kidneys following 0.2% CHX intratracheal instillation was detected at 0.08 μg/g after 1 day exposure, and then decreased to 0.03 μg/g after 7 days exposure. These levels may not be enough to induce injury on kidneys obviously observed under the microscopic investigation. Grassi et al. 31 revealed DNA damage on kidney cells in rats treated with 3 mL of 0.12% CHX by gavage once a day for 8 days. The dose used by Grassi et al. was almost 15 times as that of ours. The issue regarding what level of CHX would be a crisis to induce damage in kidneys requires further study. Lungs would be the target organ following intratracheal instillation of CHX. The histopathological study confirmed that higher concentration of 0.2% CHX induced acute pulmonary inflammation including perivascular and intra-alveolar hemorrhages with infiltration of macrophages, neutrophils and red blood cells. Congestion to the capillaries and alveolar edema were also present. These characteristic changes have similarities to those in humans with ARDS. 32–34 Despite the fact that the ARDS-like histological changes were observed, rats recovered behaviorally from sickness almost within 2 h. The possible reason for the discrepancy on the clinical symptoms between rats and patients would be (1) species difference between animal and human and (2) the dose used in this study may be low due to the limitation of situation on rat survival. Orito et al. 35 examined the pathological changes by exposure CHX limited to the left lung in rats and revealed the same lung lesions under the microscopic investigation. The rats were reported to have general behaviors and activities during the experiment. The concentration of CHX in lungs was at about 1.0 μg/g and maintained for more than 1 week. Our previous study 16 on kinetics of CHX revealed that blood concentrations in rats were very high immediately after a caudal vein infusion at a dose of 15 mg/kg and declined quickly with the lapse of time. In this study, blood concentrations were too low to detect at either 1 day or 7 days after administration. The results of a pharmacokinetic study indicated that peak plasma CHX concentrations of 0.206 mg/mL were attained 30 minutes after ingestion of a 300 mg dose of CHX, however, the drug was undetectable 12 h after the dose. 36 Although CHX levels in kidneys and liver were much lower than those in lungs, they were detectable. Disposition of CHX following intratracheal instillation was not addressed in this study; however, systemic distribution from lungs to other organs may occur and CHX may have a tendency to accumulate in kidneys and liver, besides in lungs. These results suggest that lung could be a target organ of CHX following intratracheal instillation. Thus, it is highly possible that aspiration of CHX might induce ARDS when the target concentration in lungs is more than 1.0 μg/g. The fatal case 11 aspirating a 5% CHX solution would have been sufficient to induce ARDS.
The relationship between CHX and ARDS remains unclear so far. Ishigami et al. 7 reported a 67-year-old man who was unintentionally injected 0.8 mg of CHX and subsequently developed ARDS. Hirata and Kurokawa 11 reported an 80-year-old woman died of ARDS after being accidentally ingested approximately 200 mL of Maskin® (5% CHX). The direct lung injury following rapid pulmonary absorption of CHX should be considered. The destruction to biomembrane and cytotoxic action of CHX would cause a disruption of the epitherial cells and inflammatory reactions in lungs.
In conclusion, acute pulmonary toxic effects of CHX following an intratracheal instillation in rats were investigated. CHX at concentration of 0.2% caused changes in hematological and biochemical values including WBC, TP, ALB, LDH, BUN and CRE, and induced inflammatory reactions including intra-alveolar edema and hemorrhages, as well as resulted in the target organ concentration in lungs at the level of about 1.0 μg/g and maintained for more than 1 week, when administered intratracheally in rats. The cytotoxic action of CHX might induce those detrimental reactions in rats.
Footnotes
Acknowledgements
We would like to thank Professor Peilin Huang for his excellent technical assistance with histopathology analysis.
The author Yuying Xue received a Postdoctoral Fellowship for Foreign Researchers (ID No. P 05226) awarded by Japan Society for the Promotion of Science (JSPS). This work was supported by JSPS under Grant-in-Aid for Scientific Research (No. 17·05226) and also supported by National Basic Research Program of China (No. 2006CB705602 & No. 2011CB933404), National Natural Science Foundation of China (No. 30671782 & No. 30972504), Science Foundation of Southeast University (No. KJ2010440).
