Abstract
Background:
Bronchial asthma is one of the top disabling diseases in pediatrics. Limited research has been studied the association of the widely used plastic monomer bisphenol A (BPA) with childhood asthma.
Objective:
To compare the levels of urinary BPA in asthmatic and control children and to investigate the implication of BPA among other risk factors for the development of asthma.
Subjects and methods:
This case–control study included 97 children (45 asthmatic and 52 healthy controls) aged 3–8 years. Asthmatic children were diagnosed according to Global initiative for asthma (GINA) guidelines. Sociodemographic factors were assessed and urinary levels of BPA were determined in spot urine samples using high-performance liquid chromatography. The contribution of BPA among predictors for developing asthma was studied in asthmatic children.
Results:
Median total urinary BPA levels were significantly higher in asthmatic children than in control group (1.56 ng/mL in asthmatic children compared to 0.790 ng/mL in control group, p = 0.001). Children who had total urinary BPA levels
Conclusion:
Association of higher levels of urinary BPA with the diagnosis of asthma in children may indicate the potential risk of BPA exposure in the precipitation of bronchial asthma. Further clinical and biochemical research are needed to clarify the proper mechanism explaining this association.
Keywords
Introduction
The prevalence and morbidity of childhood asthma have been rising evidently during recent decades throughout the world 1 –3 According to Centers for Disease Control and Prevention reports in 2011, the rate of childhood asthma is about 1 in 10. Although genetic and environmental factors are major risk factors for asthma and allergic diseases, 4 the reasons for the increased prevalence of asthma remain poorly understood. 5,6 The increase in global asthma prevalence has occurred within approximately the same time frame as the widespread use of industrial chemicals like bisphenol A (BPA). 7
BPA is one of the chemicals produced intensively worldwide. Annually, greater than 15 billion pounds of BPA are produced and more than 100 tons are released into the atmosphere. 8 It is used in the production of polycarbonate plastics and epoxy resins. Polycarbonate plastics are involved in manufacturing products such as water bottles, toys, dental sealants, and compact discs, whereas epoxy resins are used to coat the food and beverage cans from insides. 8,9 Human exposure to BPA is widespread; and about 95% of the Americans have detectable BPA concentrations in urine. 10
Recent studies have showed a potential correlation between BPA exposure and asthma. Animal studies have reported that BPA may promote allergic immune responses and then affect the development of asthma-related conditions, respectively. 11 –15 BPA has also been shown to promote eosinophilic bronchial inflammation and airway responsiveness in mice. 16 Animal models have identified an association of prenatal BPA exposure with the development of an experimental model of asthma, and one study noted that rhesus macaques exposed to BPA had accelerated development of secretory cells in the proximal airways. 16 –18 However, in other animal study, the authors reported that maternal exposure to BPA has only subtle effects on allergic inflammation which did not lead to significant airway responsiveness. 19 Some investigators have suggested that exposure to BPA may lead to the development of asthma in children. 20 –22
However, to the best of our knowledge, there is a limited evidence regarding the association of BPA exposure and the development of asthma in Egyptian children.
The aim of this case–control study is to compare the level of urinary BPA in asthmatic and control children and to investigate the implication of BPA among other risk factors for the development of asthma.
Subjects and methods
Subjects and study setting
This case–control study included 97 children chosen randomly from patients attending Abu El Reesh Specialized Pediatric Educational Hospital, Kasr El Aini Faculty of Medicine, Cairo University. At the Respiratory Diseases Department, 45 children were chosen randomly out of patients who were diagnosed as having bronchial asthma with varying severity according to Global Initiative for Asthma Guidelines. 23
The control group was 52 apparently healthy children of matched age and gender who were chosen randomly from the Children Daycare Center, caring for children of the National Research Center employees.
Chronic asthmatic children in remission or exacerbation according to clinical picture were considered eligible.
Exclusion criteria for both cases and control groups: (1) History of congenital malformation causing narrowing of the intrathoracic airways. (2) History of congenital heart disease. (3) Recurrent lower respiratory infection (4) associated with liver or kidney disease. (5) Febrile children with chest infection.
Permission to perform the study was granted by the Medical Ethics Committee of the National Research Centre. Written consents were obtained from parents or caregivers.
Study procedures
Parents or caregivers of asthmatics and controls were asked to fill out a form included demographic information such as gender, age, parental education, and occupation; potential risk factors such as passive smoking. Information about onset of asthma diagnosis, duration, and medication history were recorded in asthmatic group. Full clinical examination (general and local) was performed for asthmatic and control children. Plain X-ray chest and routine laboratory investigations were performed for asthmatic group or results were obtained from hospital files to confirm diagnosis. Urine samples were collected from children in the morning at 9 a.m. and stored at −70°C until assays.
Estimation of urinary BPA concentration
The concentration of total (free plus conjugated) species of urinary BPA was determined using high-performance liquid chromatography (HPLC)-tandem mass spectrometry according to Matsumoto et al. (2003). 24 The limit of detection was 0.3 ng/mL.
Urine (500 µL) was buffered with 30 µL of 2.0 M sodium acetate buffer (pH 5.0) and hydrolyzed enzymatically with β-glucuronidase/sulfatase (4,414/168 U/µL) for 3 h at 37°C in a shaking water bath. After hydrolysis, 100 µL of 2 N hydrochloric acid was added, and the hydrolysate was extracted once with 5 mL of ethyl acetate with 10 µg/L bisphenol B (internal standard). After centrifugation, 4 mL of supernatant was transferred to a new tube and evaporated with N2 gas. The residue was dissolved with 200 µL of 60% acetonitrile in water, and 40 µL of the solution was injected onto the HPLC system (Agilant 1100 series, Part No. G1311-90003 available at http://www.meadowshplc.com/sites/default/files/products/attachments/1100_pump.pdf). The total of conjugated plus unconjugated forms of BPA (total BPA) was measured by this procedure. The same procedure without β-glucuronidase/sulfatase was carried out in parallel to measure the unconjugated BPA (free BPA). The concentration of conjugated BPA was calculated by subtracting the amount of free BPA from the total BPA.
Statistical analysis
Data collected were tabulated and analyzed by SPSS (statistical package for the social science software) statistical package version 16. Statistical analyses included descriptive statistics, χ 2 test, and logistic regression. Urinary BPA levels were log-transformed to improve normality of the distribution. Quantitative data were expressed as mean and standard deviation (X ± SD) and analyzed by applying student t-test for comparison of two groups of normally distributed variables. Mann–Whitney test was used to compare between two groups not normally distributed. Qualitative data were expressed as number and percentage n% and analyzed by applying χ 2 test, OR (odds ratio), and 95% CI (confidence interval).
The main exposure of interest, urinary BPA, was categorized into two groups (<1.3 ng/mL and ≥1.3 ng/mL) and was associated and analyzed with asthmatic and control groups using the two-tailed χ 2 test, OR, and 95% CI. Multiple logistic regression analysis was done to detect the significant predictors of bronchial asthma. All tests were considered statistically significant when p < 0.05.
Results
Table 1 presents the characteristics of asthmatic and control children. Children in both groups were matched as regard to age, gender, and body mass index z-score. Comparison of sociodemographic variables revealed a statistically significant difference between the two groups, with lower maternal education and higher social level in the control group. With regard to exposure to risk environmental factors, it was found that the percentage of passive smoking exposure was significantly higher in the case group than that of the control group (58% in asthmatics vs. 42% in controls, p < 0.05). Admission to Neonatal intensive care unit (NICU) did not represent a risk factor as the percentage of children admitted to NICU in the asthmatic group was not significantly differed than that of the control group.
Sociodemographic variables of the asthmatic and control groups.

OR: odds ratio; CI: confidence interval.
a p < 0.05 is significant.
Table 2 shows that the asthmatic children had higher concentrations of total, conjugated and unconjugated urinary BPA than the control group with highly significant difference (p < 0.05). Urinary BPA levels were log-transformed to improve normality of the distribution. The mean log total urinary BPA of the asthmatic group was significantly higher than that of the control group (p = 0.003). Table 3 shows that children with higher levels of urinary BPA (>1.3 ng/mL) were more likely to be asthmatics nearly three times more than those who had low urinary BPA (<1.3 ng/mL; OR = 2.842, 95% CI: 1.224–6.597, p = 0.015). No significant difference (p > 0.05) was detected between males and females and between social classes in asthmatic group as regard to log urinary BPA (Table 4). Table 5 shows the results of multiple logistic regression analysis for predictors of bronchial asthma; no gender effect could be detected in the prediction of asthma. Exposure to BPA (as indicated by urinary BPA level) and exposure to passive smoking were the predictors of asthma in the studied children. The results demonstrated the importance of higher levels of BPA (>1.3 ng/mL) as a more significant predictor than passive smoking (p = 0.006 for BPA categories vs. p = 0.049 for passive smoking).
Comparison of urinary BPA and its metabolites between asthmatic patients and controls.

BPA: bisphenol A; SD: standard deviation.
a p < 0.05 is significant.
Association between BPA and bronchial asthma.a

BPA: bisphenol A; OR: odds ratio; CI: confidence interval.
aOR = 2.842; 95% CI = 1.224–6.597; p = 0.015b.
b p < 0.05 is significant.
Comparison of log BPA according to sex and social class in asthmatic group.a

BPA: bisphenol A; SD: standard deviation.
a p < 0.05 is significant.
Multiple logistic regression analysis for predictors of bronchial asthma.
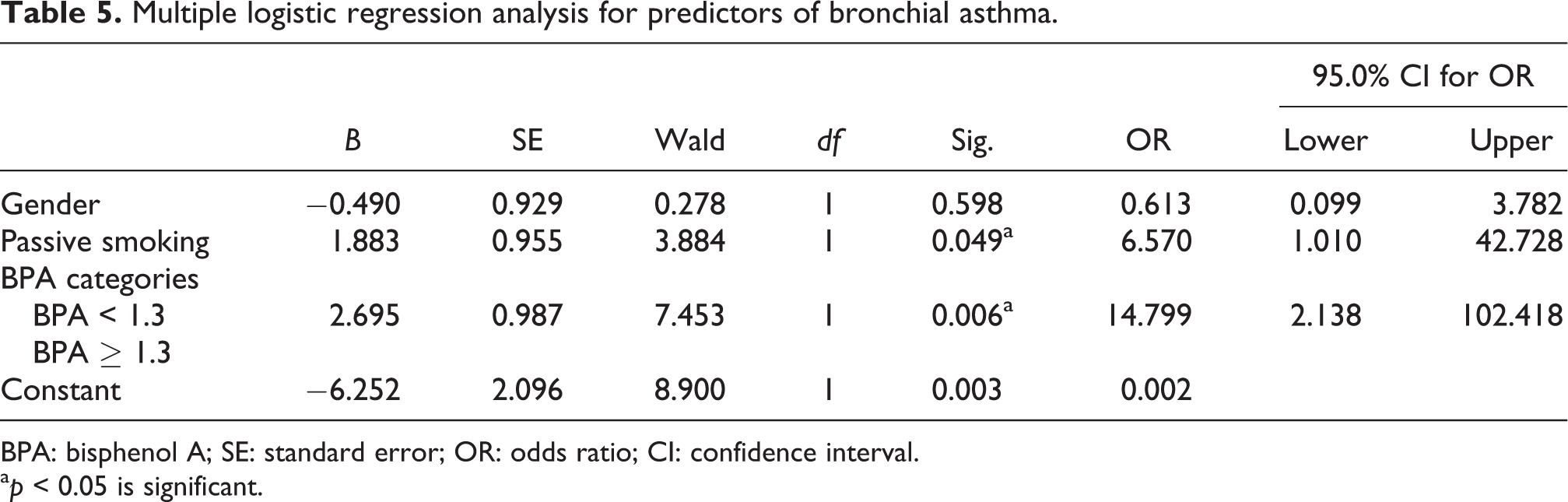
BPA: bisphenol A; SE: standard error; OR: odds ratio; CI: confidence interval.
a p < 0.05 is significant.
Discussion
In the current study, a significant association was documented between urinary BPA concentration in children and asthma. Some investigators have suggested that exposure to BPA may be related to the development of asthma in children. 20 –22,25 Other studies note a similar correlation of BPA and asthma, but the timing of exposure and associated risks are conflicting. Spanier et al. previously reported that prenatal BPA exposure was associated with wheezing among children under the age of 3 years. 26 However, one study reported a postnatal association of BPA exposure with child asthma and wheeze but did not find an association of prenatal BPA exposure. 25 Using 2005–2006 National Health and Nutrition Examination Survey (NHANES) data, Vaidya reported an association of urinary BPA and allergic asthma primarily in females. 27
The interpretation of these findings is still unexplained; however, the oxidative stress pathway with BPA exposure could be suggested as a possible explanation. BPA is known to cause oxidative stress in rats 1,28 –30 and humans 15 and with the strong evidence indicates that oxidative stress and subsequent mitochondrial dysfunction are correlated to BPA-induced damage. 31,32 Increased production of reactive oxygen species and decreased anti-inflammatory capacity could reinforce the vulnerability to the stimuli, as air pollution, which results in chronic airway inflammation and asthma. 33,34
In this study, BPA concentrations were also higher in asthmatic children exposed to second-hand tobacco smoke (SHS). SHS has also been reported to be a significant predictor of BPA exposure in human children. 35,36 This is contributed to the composition of some cigarette filters is around 25–30% dry weight of BPA. 37 Free BPA within the cigarette filter, which attaches to combusted particulate matter of the cigarette, would be expected to deposit on the respiratory epithelium when inhaled.
Low social class and less maternal education were significantly associated with asthma in this study. Lee et al. (2017) and Gong et al. (2014) showed an inverse association between Socioeconomic status (SES) with asthma regardless of age. 12,38 The association between SES and asthma can be partially explained by exposure degree to allergens, gene by environmental interactions, malnutrition, and psychosocial factors. 39 –41 These factors are considered to affect the immune development through balance between T-helper 1 (TH1) and TH2 immune response.
Due to BPA’s ability to bind to estrogen receptors, BPA is classified as an endocrine-disrupting chemical. The results of the present study suggest that the effects of BPA on asthma-related outcomes might be mediated, at least in part, by an endocrine-disrupting mechanism. Comparison of log BPA between male and female in asthmatic group showed that log BPA was higher in girls (0.2837 ± 0.3387) than boys (0.1834 ± 0.3185) but the difference was not significant. Estrogen has been demonstrated to enhance TH2 polarization, class switching of B cells to the production of immunoglobulin E, and the degranulation of mast cells, 42 –44 shift in T-helper cell populations can alter immune responses to antigens and disease profile, contributing to allergic diseases (e.g. asthma). 45 The association of urinary BPA concentration with asthma adds information about the health hazards of BPA exposure in humans and supports public health initiatives to protect the health of a susceptible population such as children.
Future research should focus on defining the mechanism of action and the dose at which adverse effects of BPA exposure on childhood wheeze/asthma and the roles that BPA play in the rising incidence of allergy and asthma. Ultimately, if the research continues to support the existence of a relationship between BPA and immune deregulation, it will be necessary to determine methods to limit further exposure to BPA.
Conclusion
In the current study, a significant association was documented between urinary BPA concentration in children and asthma. Association of higher levels of urinary BPA with the diagnosis of asthma in children may indicate the potential risk of BPA exposure in the precipitation of bronchial asthma. Future research should focus on defining the mechanism of action and the dose at which adverse effects of BPA exposure on childhood wheeze/asthma, the effect of spacers and nebulizers as a risk factor in BPA exposure, and the roles that BPA play in the rising incidence of allergy and asthma. Ultimately, if the research continues to support the existence of a relationship between BPA and immune deregulation, it will be necessary to determine methods to limit further exposure to BP.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was a part of a project supported financially by National Research Centre Egypt (grant no. 10010403).
