Abstract
Background:
We investigated the association between serum levels of glial fibrillary acidic protein (GFAP) and stroke functional outcomes in a cohort of 286 patients with acute ischemic stroke (AIS).
Methods:
We prospectively studied 286 patients with AIS who were admitted within 24 h after the onset of symptoms. Serum levels of GFAP and National Institutes of Health Stroke Scale (NIHSS) were measured at admission. The primary end point was stroke functional outcome among 1-year after stroke onset. We used logistic regression models to assess the relationship between GFAP levels and stroke outcomes.
Results:
The GFAP level was obtained with a median value of 0.18 (interquartile ranges (IQRs): 0.09–0.28) ng/ml. In multivariable models adjusted for age, gender, and other risk factors, GFAP levels were associated with an increased risk of a NIHSS>6 (odds ratio (OR) = 1.55; 95% confidence interval (CI): 1.16–1.89; p = 0.012). The poor outcome distribution across the GFAP quartiles ranged between 12.7% (first quartile) and 70.4% (fourth quartile). After adjusting for other established risk factors, in multivariate models comparing the Q3 and Q 4 quartiles against the Q1 of the GFAP, the levels of GFAP were associated with poor outcome, and the adjusted risk of poor outcome increased by 211% (3.11[1.80–5.05], p < 0.001) and 522% (6.22[2.98–11.83], p < 0.001), respectively. Interestingly, GFAP improved the ability of NIHSS score to diagnose poor outcomes (area under the curve [AUC] of the combined model 0.82; 95% CI: 0.77–0.88; p = 0.02).
Conclusion:
GFAP levels are a novel and complementary biomarker to predict functional outcome 1 year after AIS
Introduction
In China, the annual stroke mortality rate is approximately 1.6 million, and stroke had been recognized as a leading cause of serious long-term disability in adults. 1 In those patients, ischemic stroke (IS) constituted nearly three-quarters. 2 The mechanisms underlying the pathophysiology of stroke are not fully understood but there is increasing evidence demonstrating the contribution of inflammation to the drastic changes after cerebral ischemia. 3
Glial fibrillary acidic protein (GFAP) is considered to be highly brain specific. As a structural protein, it is not released from cells under physiologic conditions, and healthy individuals do not show detectable GFAP levels in their blood stream. 4 Previous studies have suggested that GFAP is related with intracerebral hemorrhage (ICH), 5 traumatic brain injury, 6 autoimmune diabetes, 7 and autism spectrum disorders. 8
Furthermore, some studies reported that it is released with some delay in IS. 4,9 Recent studies have proposed that GFAP serum concentrations distinguish between ICH and IS shortly after symptom onset. 10 Katsanos et al. 11 also found that plasma GFAP seems to be a sensitive and specific biomarker for the differentiation of ICH from both AIS and other acute neurological disorders. Thus, we hypothesize that serum GFAP concentration at admission might be associated with future functional outcomes events after IS. In this study, we investigated the association between serum levels of GFAP and stroke functional outcomes in a cohort of 286 patients with acute ischemic stroke (AIS).
Material and methods
Methods
From January 2015 to December 2015, consecutive first-ever AIS patients admitted to the Department of Emergency of the First Affiliated Hospital of Kunming Medical University, China, were identified. The study population was exclusively Chinese. IS was defined according to World Health Organization recommendations (defined stroke as a “neurological deficit of cerebrovascular cause that persists beyond 24 hours or is interrupted by death within 24 hours”). 12 The clinical diagnoses were validated on the basis of computed tomography (CT) and/or magnetic resonance imaging (MRI; blindly assessed by Geng). The exclusion criteria were malignant tumor, ICH (including the strokes that were initially ischemic but later underwent hemorrhagic conversion), renal insufficiency (creatinine >1.5 mg/dl), head trauma, severe edema, febrile disorders, and autoimmune diseases. Patients without informed consents, lost blood samples, and follow-up were also excluded. The present study has been approved by the ethics committee of the First Affiliated Hospital of Kunming Medical University. Written informed consents were obtained from participants or their relatives.
Clinical information was collected. Demographic data (age and sex), body mass index (BMI), and history of risk factors (hypertension, diabetes mellitus, atrial fibrillation, hyperlipidemia, smoking habit, and alcohol abuse) were obtained at admission. Pre-stroke therapy (oral anticoagulants, antiplatelet agents, and antihypertensive treatment) and acute treatment (Intravenous [IV] thrombolysis and/or mechanical thrombectomy) were recorded. Clinical severity was assessed at admission using the National Institutes of Health Stroke Scale (NIHSS) score. Strokes classification and the clinical stroke syndrome were obtained. 13
We followed the participants for a median of 1 year using a standard questionnaire, and telephone or household contact by physician investigators every 4 months after admission. The primary end point was stroke functional outcome among 1-year after stroke onset. Functional impairment was evaluated using the modified Rankin scale (mRS). A good functional outcome of stroke patient was defined as an mRS score of 0–2 points, while poor functional outcome was in the range of 3–6 points. 14 The secondary end point was death from any cause. All blood samples were collected on the first day of admission under fasting state. The serum GFAP was measured using a commercial ELISA kit (CSB-F201705; Cusabio, Wuhan, China) by a blinded coworker. Each GFAP measurement was performed in full calibration mode. The measuring range of the GFAP is between 0.03 ng/ml and 40 ng/ml (defined by the lower detection limit and the maximum of the master curve). The coefficients of variation for the intra- and inter-assay reproducibility were 4.0–7.5% and 5.0–9.0%, respectively. Results of the other blood analyses such as fasting blood glucose (FBG), homocysteine (HCY), and high-sensitivity C-reactive protein (Hs-CRP) were also measured using routine laboratory methods.
Statistical analysis
The results were expressed as percentages for categorical variables and as medians (interquartile ranges (IQRs)) for continuous variables. The Mann–Whitney U test and χ2 test were used to compare the two groups. Spearman’s rank correlation was used for bivariate correlations. The influence of GFAP levels on stroke outcomes was performed by binary logistic regression analysis, which allows adjustment for confounding factors (age, sex, BMI, stroke syndrome, stroke etiology, the NIHSS score, infarct volume, vascular risk factors, pre-stroke therapy, acute treatment, and serum levels of Hs-CRP, FBG, and HCY). We used crude models and multivariate models adjusted for all significant predictors and report odds ratios (ORs). For a more detailed exploration of the GFAP-stroke outcomes relationships, we also used multivariate analysis models for GFAP quartiles (with lowest GFAP quartile as reference). Further, the receiver operating characteristic (ROC) curves were used to test the overall accuracy of the GFAP and other markers to predict stroke outcomes. All statistical analysis was performed with SPSS for Windows, version 20.0 (SPSS Inc., Chicago, Illinois, USA). Statistical significance was defined as p < 0.05.
Results
We recorded 286 stroke patients. The GFAP level was obtained with a median value of 0.18 (IQR: 0.09–0.28) ng/ml. The characteristics of stroke patients according to GFAP4 levels are shown in Table 1.
Baseline characteristics of patients with stroke.

GFAP: glial fibrillary acidic protein; IQR: interquartile range; BMI: body mass index; TIA: transient ischemic attack; IV: intravenous; NIHSS: National Institutes of Health Stroke Scale; FBG: fasting blood glucose; Hs-CRP: high-sensitivity C-reactive protein; HCY: homocysteine; MRI: magnetic resonance imaging.
aIn patients for whom MRI data were available (n = 193).
At admission, 115 patients (40.2%) had a minor stroke (NIHSS≤5). In these patients, the median GFAP4 level was lower than that observed in patients with moderate-to-high clinical severity (0.12[0.05–0.19] ng/ml vs. 0.25[0.16–0.34] ng/ml; p < 0.001). In multivariable models adjusted for age, gender, and other risk factors, GFAP levels were associated with an increased risk of a NIHSS>6 (OR = 1.55; 95% CI: 1.16–1.89; p = 0.012). In addition, positive correlations between GFAP and Hs-CRP(r [spearmen] = 0.139, p = 0.019) and age(r = 0.155, p = 0.011) were obtained. In patients for whom MRI data were available (n = 193), GFAP4 levels increased with lesion size (r = 0.196, p = 0.001).
At follow-up, a poor functional outcome was found in 109 patients (38.1%; 95%CI: 32.5–43.7%) with a median mRS score of 4 (IQR, 3–6). The poor outcome distribution across the GFAP quartiles ranged between 12.7% (first quartile) and 70.4% (fourth quartile). GFAP levels in patients with a good outcome were significantly lower than those in patients with a poor outcome (0.13 [IQR, 0.06–0.21] ng/ml vs. 0.26 [IQR, 0.18–0.39] ng/ml; Z = 8.136; p < 0.001; Figure 1). In univariate logistic regression analysis, comparing the second (Q2), third (Q3), and fourth (Q4) quartiles against the first quartile (Q1) of the GFAP, levels of GFAP were associated with poor outcome, and the unadjusted risk of poor outcome increased by 147% (OR = 2.47 [95% CI: 1.03–5.92]; p = 0.039), 421% (5.21[2.25–12.07]; p < 0.001), and 1540% (16.40[6.91–38.96]; p < 0.001), respectively (Table 2). After adjusting for other established risk factors, in multivariate models comparing the Q3 and Q 4 quartiles against the Q1 of the GFAP, levels of GFAP were associated with poor outcome, and the adjusted risk of poor outcome increased by 211% (3.11[1.80–5.05], p < 0.001) and 522% (6.22[2.98–11.83]; p < 0.001), respectively (Table 2).

Distribution of GFAP in stroke patients with poor functional outcomes and good functional outcomes. Horizontal lines represent medians and IQR. P values refer to Mann–Whitney U tests for differences between groups. Poor functional outcome was defined as an mRS in 3–6. GFAP: glial fibrillary acidic protein; IQR: interquartile ranges; mRS: modified Rankin scale.
Multiple logistic regression analysis showing GFAP in quartiles independently associated with the stroke poor outcomes at baseline.
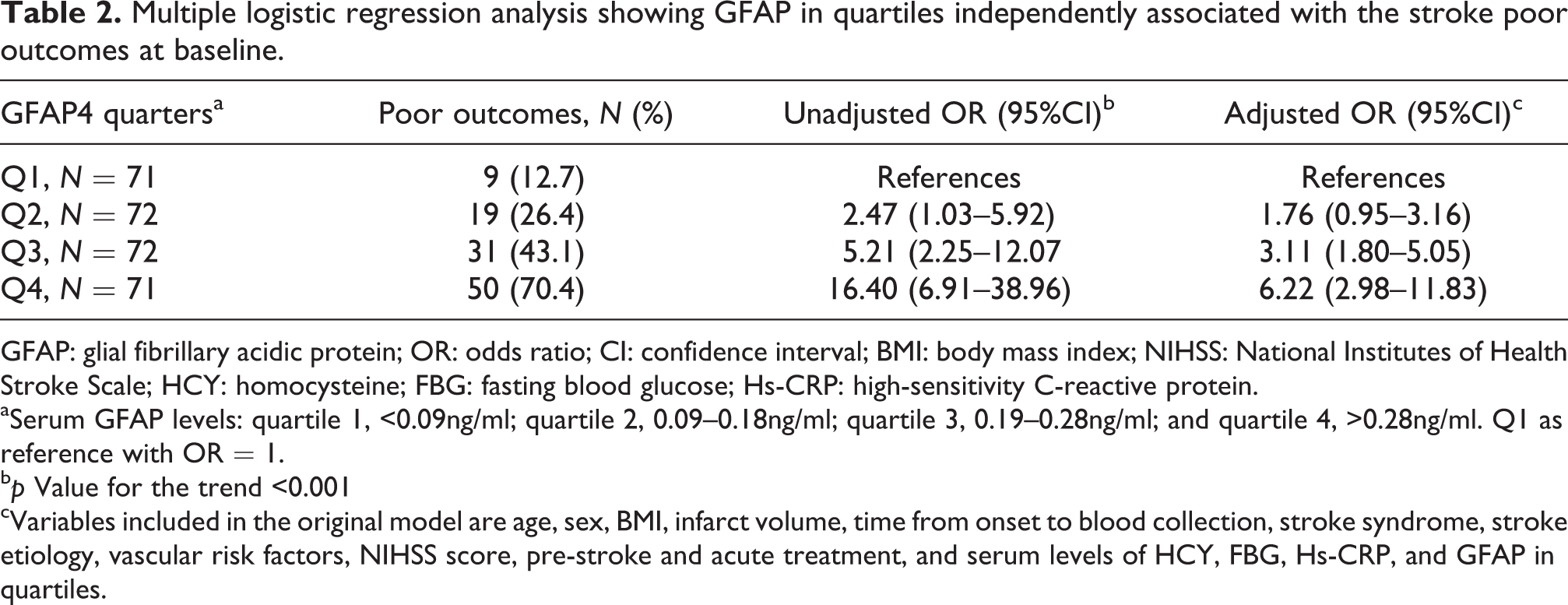
GFAP: glial fibrillary acidic protein; OR: odds ratio; CI: confidence interval; BMI: body mass index; NIHSS: National Institutes of Health Stroke Scale; HCY: homocysteine; FBG: fasting blood glucose; Hs-CRP: high-sensitivity C-reactive protein.
aSerum GFAP levels: quartile 1, <0.09ng/ml; quartile 2, 0.09–0.18ng/ml; quartile 3, 0.19–0.28ng/ml; and quartile 4, >0.28ng/ml. Q1 as reference with OR = 1.
b p Value for the trend <0.001
cVariables included in the original model are age, sex, BMI, infarct volume, time from onset to blood collection, stroke syndrome, stroke etiology, vascular risk factors, NIHSS score, pre-stroke and acute treatment, and serum levels of HCY, FBG, Hs-CRP, and GFAP in quartiles.
With an AUC of 0.78 (95% CI: 0.73–0.84), GFAP showed a significantly greater discriminatory ability as compared with Hs-CRP (AUC, 0.70; 95% CI: 0.67–0.75; p = 0.009), age (AUC, 0.57; 95% CI: 0.52–0.62; p < 0.001), BMI (AUC, 0.61; 95% CI: 0.56–0.66; p < 0.001), and HCY (AUC 0.68; 95% CI: 0.61–0.74; p < 0.001), while was in the range of NIHSS score (AUC 0.77; 95% CI: 0.70–0.83; p = 0.55) to diagnose poor outcomes. Interestingly, GFAP improved the ability of NIHSS score to diagnose poor outcomes (AUC of the combined model 0.82; 95% CI: 0.77–0.88; p = 0.02).
GFAP levels in 63 patients (22.0%) who died were more than 2 times greater as compared with patients who survived (0.31; IQR, 0.18 to 0.43 ng/ml vs. 0.15; IQR, 0.08 to 0.23 ng/ml; Z = 6.416, p < 0.001), Figure 2. After adjusting for all other significant outcome predictors (such as age, NIHSS score, and Hs-CRP), GFAP4 level (Q4 vs. Q1–3) remained an independent predictor for mortality with an OR of 3.03(95% CI: 1.93–5.98; p = 0.002).

Distribution of GFAP in survivors and nonsurvivors of stroke. Horizontal lines represent medians and IQR. P values refer to Mann–Whitney U tests for differences between groups. GFAP: glial fibrillary acidic protein; IQR: interquartile ranges; mRS: modified Rankin scale.
Discussion
GFAP is the main component of intermediate filaments of the cytoskeleton of astrocytes. 15 Over more than four decades of fundamental and applied studies, GFAP achieved the status of the classical marker for glioblastoma. 16 Changes in GFAP expression and/or phosphorylation have been reported during brain damage or central nervous system degeneration. 17 In this prospective, observational study, we assessed serum level of GFAP at admission regard to their accuracy to predict stroke poor functional outcomes in patients with AIS within 1-year follow-up. GFAP was an independent marker for poor outcome events predicting and adds significant additional predictive information to the clinical score of the NIHSS. We also demonstrated that GFAP levels increased with lesion size, neurological deficit (assessed by the NIHSS), and reflecting the severity of the stroke.
Until now, little data are available for GFAP as a predictor of functional outcome in patients with IS.
10,11
However, from a pathophysiological point of view, GFAP appears to be an interesting biomarker candidate for further evaluation in this context. Lamers et al.
18
suggested that determination of GFAP in cerebral spinal fluid (CSF) and blood can be helpful to establish structural and/or functional brain damage to determine severity and prognosis of the disease process and to monitor treatment effects. Another study confirmed that serum GFAP can reliably detect ICH in the acute phase of stroke.
19
Papa et al.
20
suggested that GFAP breakdown products is detectable in serum within an hour of injury and is associated with measures of injury severity, including the GCS score, CT lesions, and neurosurgical intervention. In addition, GFAP levels were associated with a poor outcome in cardiopulmonary resuscitation patients,
21
and GFAP provides information about brain injury severity and outcome after aneurysmal subarachnoid hemorrhage.
22
In this study, we found that GFAP was associated with poor functional outcomes in patients with AIS. Furthermore, release kinetics of GFAP is suggested to be associated with patient
Stroke severity and volume of damaged brain are associated with stroke outcomes. GFAP associated with poor outcomes might be merely a marker of initial stroke severity and is proportional to volume of damaged brain. However, GFAP remained independently associated with poor outcomes even after adjusting for stroke severity and infarct volume. Thus, other mechanisms should be taken into account. First, GFAP as a component of intermediate filaments of the cytoskeleton plays a key role in the development of reactive astrocytosis, that is, of a typical response of the central nervous system (CNS) to injury. Second, Overexpression of GFAP or suppression of its biosynthesis reflect modifications of the functional activity of astrocytes related to damage to the nerve tissue, metabolic abnormalities, and development of neurodegenerative states. 15 Third, overexpression of GFAP might be a respond to proinflammatory cytokines. 24
Strengths of our study are its prospective design and the predictive value of serum levels of GFAP with respect to long-term outcome obtained. Some limitations should be taken into account. First, this is an observational study. It is difficult to draw any causal conclusion from this study. Second, the number of patients studied is still low considering the variability of stroke pathology. Third, this study measured GFAP in serum, not in CSF. It is still uncertain whether peripheral GFAP levels reflect similar changes in the CNS. However, a previous study suggested that serum biomarkers, particularly GFAP, may be correlative tools of brain protein changes and feasible theranostic markers of traumatic brain injury (TBI) progression and recovery. 25 Emerging evidence also suggests that, following traumatic brain and spinal cord injuries and stroke, GFAP and its breakdown products are rapidly released into biofluids, making them strong candidate biomarkers for such neurological disorders. 26 Lastly, without serial measurement of the circulating GFAP, this study yielded no data regarding when and how long biomarkers were elevated in these patients.
Conclusions
In summary, we have demonstrated that serum levels of GFAP at admission can predict long-term stroke prognosis in Chinese patients with first-ever IS.
Footnotes
Acknowledgements
We thank all the patients, nurses, and physicians who participated in this study and thereby made this work possible.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
