Abstract
The aim of the study was to investigate the effect of astaxanthin and its possible mechanisms on acute cerebral infarction (ACI) in rat model. Male Sprague Dawley rats were randomly divided into sham group, model group, and astaxanthin-treated groups (20, 40, and 80 mg/kg). Neurological examination, the ratio of cerebral edema, and histopathology changes were assessed. Moreover, some oxidative stress markers were obtained for biochemical analysis, and the expression of neurotrophic factors gene was detected by real-time polymerase chain reaction (RT-PCR) method. The results showed that treatment with astaxanthin notably reduced neurological deficit scores and the ratio of cerebral edema compared with the model group. Meanwhile, astaxanthin increased the activity of catalase, superoxide dismutase, and glutathioneperoxidase as well as decreased the content of malondialdehyde in brain tissue. RT-PCR results showed that the expression of brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF) mRNA were increased with astaxanthin treatment. The results indicated that astaxanthin could ameliorate ACI followed by suppressing oxidative stress and upregulating the expression of BDNF and NGF mRNA.
Introduction
Acute cerebral infarction (ACI), as a common clinical cardiovascular, cerebrovascular, and neurological diseases, has high incidence and mortality and is becoming one of the major challenges to public health. 1,2 It is ischemic, a result of a transient or permanent decrease in cerebral blood flow which is only referred to the range of a major brain artery. 3 At present, the thrombolytic recombinant tissue plasminogen activator is recognized as the most effective agent in the clinical treatment of ACI, while it has lots of side effects. 4 Thus, an effective therapy remains to be found.
The pathological process and the corresponding mechanism of ACI is rather complex and not clear yet. Recent insight into the possible mechanism of ACI showed that the damage and inflammation caused by free radicals, neuronal apoptosis, the toxicity of excitatory amino acid, and the metabolism abnormal of brain tissues are all involved in the occurrence and development of ACI. 5,6 Accumulating data demonstrated that oxidative stress is a key mechanism of damage in ACI because the brain consumes a large quantity of oxygen. 7,8 Reactive oxygen species (ROS) was well known as one of the most important components of tissue injury after ischemic injury. 9 While antioxidant enzymes are the primary defense method by which neuronal cells protect themselves from toxic ROS. 10 Oxidative stress was considered as one of the major contributors in the pathogenesis of ischemia/reperfusion (I/R) injury and was an important reason leading to tissue damage following ischemic injury in the brain. In addition, animal studies have indicated that neurotrophic factors are also associated with the occurrence and development of ACI. 11,12 Brain-derived neurotrophic factor (BDNF) is a vital member of neurotrophin family, which not only facilitates neuronal survival and development but also modulates synaptic plasticity. 13 Nerve growth factor (NGF) is necessary for neuronal growth and survival. 14 Therefore, antioxidant and regulation of neurological factors therapies might become the two most important strategies for ACI treatment.
Astaxanthin, as a xanthophyll carotenoid, is widely present in Haematococcus pluvialis, seafood, quail, and some complex plants. It possesses multiple pharmacological properties including antioxidative, anti-inflammatory, antiapoptotic, and neuroprotective effects. 15,16 Previous study demonstrated that astaxanthin is a well-known antioxidant which has been shown to be 500 times more potent than α-tocopherol. 17 Because of the confirmed physiological activities, and the advantage of low toxic and side effects, astaxanthin is considered to be greatly potential as a therapeutic agent in cardiovascular and neurological disease.
However, the protective effect of astaxanthin on ACI has not been explored yet, and it is unclear whether astaxanthin could ameliorate ischemic injury by antioxidative activities. Therefore, in this study, we aimed to investigate the protective effects of astaxanthin by rat middle cerebral artery occlusion (MCAO) model, and further investigated whether these effects were closely related to the antioxidative activities and the expression of BDNF and NGF.
Methods
Animal group
Male Sprague Dawley rats (7–8 weeks, 250–280 g) were supplied by Experimental Animal Center of Suzhou Aiermaite technology Co. Ltd (SPF grade, certificate no. SCXK20160017, Suzhou, China). All studies were in compliance with institutional guidelines of The Affiliated Yantai Yuhuangding Hospital of Qingdao University, and all experiments were approved by Animal Ethics Committee of The Affiliated Yantai Yuhuangding Hospital of Qingdao University. All the rats were housed in conventional cage and allowed free access to food and water with a 12-h light/12-h dark cycle at a temperature of 22 ± 1°C and a relative humidity of 50 ± 10%.
Fifty rats were randomly divided into five groups, with 10 animals in each group: sham group, model group, and astaxanthin-treated groups (20, 40, and 80 mg/kg). The MCAO model was induced by the Longa method as previously reported 18 with a few modifications. All rats were anesthetized with chloral hydrate (400 mg/kg) and placed in dorsal recumbency. Rectal temperature was maintained at 37.0 ± 0.5°C. The right common carotid artery was exposed through a midline neck incision, and then, silicone rubber was introduced into the external carotid artery (ECA) and gently advanced into the middle cerebral artery through the internal carotid artery (ICA) (approximately 17-20 mm) until a slight resistance appeared and a sharp decline in the blood flow trace was seen. The resistance showed that the filament had passed up the proximal segment of the anterior cerebral artery (ACA). At this moment, the intraluminal suture blocked the origin of MCA and occluded all sources of blood flow from ICA, ACA, and the posterior cerebral artery. After 1.5 h of the middle cerebral artery infarction, the filament was slowly withdrawn. Finally, the incisions were sutured, and the rats were returned to a warm cage. The rats in the sham group received all the surgical procedures except the filament insertion.
Astaxanthin with doses of 20, 40, and 80 mg/kg was dissolved in peanut oil and was administered orally once per day for continuous 7 days. Simultaneously, rats in the sham and model groups received an equal volume of peanut oil. After 24 h of the treatment, neurologic deficit was evaluated. Then, rats in each group were killed, and their brains were removed.
Measurement of neurological deficits
After 24 h of the treatment, neurological examinations were conducted in a blind fashion. The neurological function of rats was evaluated by using a 0–5-point scale neurological score, as follows: score 0 = no symptom of neurologic impairment; score 1= side front paw could not stretch completely; score 2 = rotated inward when walking; score 3= tilted inward when walking; score 4 = unable to spontaneously walk and loss of consciousness; and score 5= completely unable to walk.
Assessment of rat cerebral edema
The brain was harvested and weighed immediately on an electronic analytical balance to get the wet weight. Then, brains were dried in a 110°C oven for 24 h to obtain the dry weight. The ratio of cerebral edema = (wet weight – dry weight)/wet weight × 100%.
Histological evaluation
Brain tissue was fixed with 10% formalin solution and then embedded in paraffin; 4-μm thick sections were prepared and stained with hematoxylin and eosin (HE) solution. Pathological changes were observed under a light microscope.
Quantification of antioxidant enzyme activity and malondialdehyde content in brain tissue
The malonialdehyde (MDA) content and the activities of antioxidant enzyme, including superoxide dismutase (SOD), catalase (CAT), and glutathioneperoxidase (GSH-Px) in brain tissues were assayed by ELISA kits (Nanjing Jiancheng Co., Nanjing, China).
Quantitative real-time polymerase chain reaction for analysis of the expression of BDNF and NGF mRNA
For detection of the expression of BDNF and NGF mRNA in the brain, real-time polymerase chain reaction (RT-PCR) test was performed. First, total RNA was extracted from rat brain tissue by the method of RNase Miniprep Kit (Qiagen, Germany). Then, RNA was used for transcribed into cDNA by a reverse transcription kit (TaKaRa, Dalian, China). Quantitative polymerase chain reaction (qPCR) was conducted to detect the expression level of BDNF and NGF mRNA by using a 7900HT fast real-time PCR system (Applied Biosystems, California, USA). The levels of BDNF and NGF mRNA were normalized with respect to the data for the β-actin gene primers for RT-PCR were as follows: BDNF (189 bp): sense: 5′-AGTATTAGCGAGTGGGTC-3′, antisense: 5′-GTTCCAGTGCCTTTTGTC-3′; NGF (121 bp): sense: 5′-CAGCATGGTCGAGTTTTG-3′, antisense: 5′-GATAGAAAGCTGCGTCCT-3′.
Statistical analysis
Statistical analysis was implemented using SPSS18.0 for windows. All data were reported as the mean ± SD, and differences between groups were evaluated by one-way analysis of variance followed by Tukey’s multiple range test. The level of statistical signification was set at p < 0.05.
Results
Effects of astaxanthin on neurological function
As shown in Figure 1, the neurological deficit score increased notably in model group compared with the sham group (0 vs. 4.31 ± 0.30, p < 0.01). However, the neurological deficit score of rats in the astaxanthin-treated group decreased notably compared with the sham group (4.31 ± 0.30 vs. 3.72 ± 0.13, 3.14 ± 0.24 and 2.03 ± 0.22, p < 0.05). In addition, the results showed a markedly downward trend with gradual increases in the doses of astaxanthin.

Effects of astaxanthin on neurological function. Data are presented as mean ± SD. **p < 0.01 versus sham group; #p < 0.05 versus model group, ##p < 0.01 versus model group.
Effects of astaxanthin on cerebral edema
As shown in Figure 2, the ratio of cerebral edema in the model group was increased significantly than that in the sham group (75.31 ± 3.27 vs. 85.33 ± 4.09, p < 0.01). However, administration with astaxanthin at different doses significantly reduced the ratio cerebral edema of rats compared with the model group (85.33 ± 4.09 vs. 81.23 ± 3.82, 79.62 ± 3.67, 77.22 ± 3.36, p < 0.05).
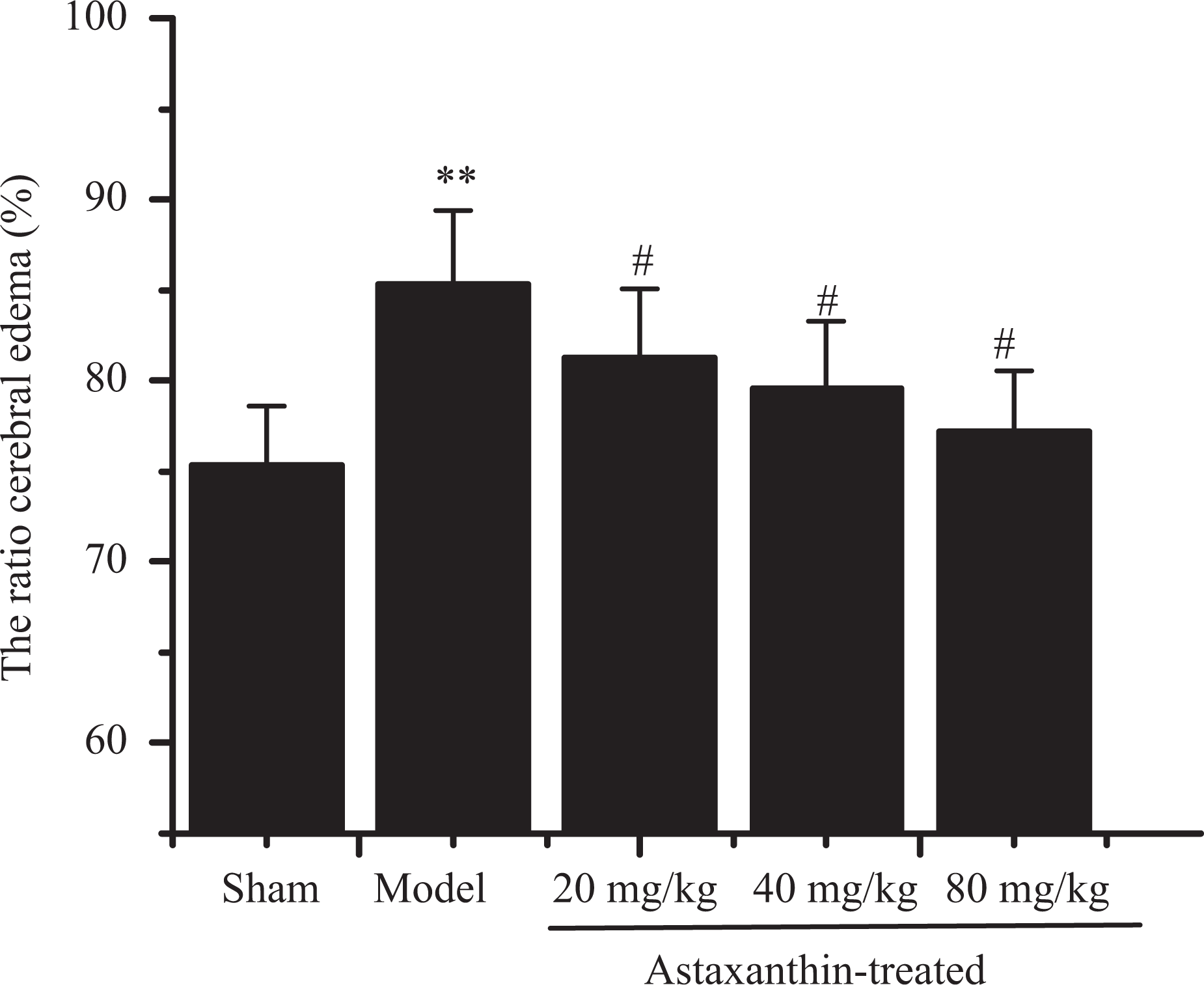
Effects of astaxanthin on the ratio of cerebral edema. Data are presented as mean ± SD. **p < 0.01 versus sham group; #p < 0.05 versus model group.
Histopathology analysis
To assess the effects of astaxanthin on histopathology in MCAO rats, HE staining was used. As shown in Figure 3(a), in sham group, the neurons with abundant cytoplasm and clear nucleolus were arranged orderly – there is no histopathological abnormalities observed. While in the model group, brain tissues were obviously damaged, and a large number of neurons were shrunken with eosinophilic cytoplasm and triangulated pyknotic nuclei. Edema of the neuropile was observed in the damage area. The infarct core was surrounded with ischemic injured neurons (Figure 3(b)). However, treatment with astaxanthin after MCAO ameliorated the cerebral injury, and the ameliorations in brain are revealed in a dose-dependent manner (Figure 3(c) to (e)).

Effects of astaxanthin on histopathology in MCAO rats (×200). (a) Sham group; (b) model group; (c) 20 mg/kg astaxanthin-treated group; (d) 40 mg/kg astaxanthin-treated group; and (e) 80 mg/kg astaxanthin-treated group. MCAO: middle cerebral artery occlusion.
Effect of astaxanthin on CAT, SOD, GSH-Px activity, and MDA content in brain tissue
In order to determine whether administration with astaxanthin could decrease oxidative damage, the antioxidant enzyme activity and MDA content in the brain tissues of rats were detected. As shown in Figure 4, the activities of CAT, SOD, and MDA in the model group were significantly lower than those in the sham group (p < 0.01), and MDA in the sham group were significantly higher than that in the model group (p < 0.01). After treating with astaxanthin at doses of 20, 40, and 80 mg/kg, the activities of CAT, SOD, and MDA showed a markedly increase compared with the model group (p < 0.05 or p < 0.01), while the content of MDA had a significant decrease (p < 0.01). Additionally, the effects of astaxanthin on oxidative stress were in a dose-dependent manner.

Effect of astaxanthin on CAT, SOD, GSH-Px activity, and MDA content in brain tissue: (a) CAT, (b) SOD, (c) GSH-Px, and (d) MDA content. Data are presented as mean ± SD. **p < 0.01 versus sham group; #p < 0.05, ##P≤0.01 versus model group. CAT: catalase; SOD: superoxide dismutase; GSH-Px: glutathioneperoxidase; MDA: malondialdehyde.
Effect of astaxanthin on the expression of BDNF and NGF mRNA
As shown in Figure 5, the expression of BDNF and NGF mRNA in model group was significantly higher than those in sham group (p < 0.01). After treated with astaxanthin, the expression of BDNF and NGF mRNA increased significantly compared with the model group (p < 0.05). The result showed astaxanthin significantly increased the expression of BDNF and NGF mRNA in a dose-dependent manner.
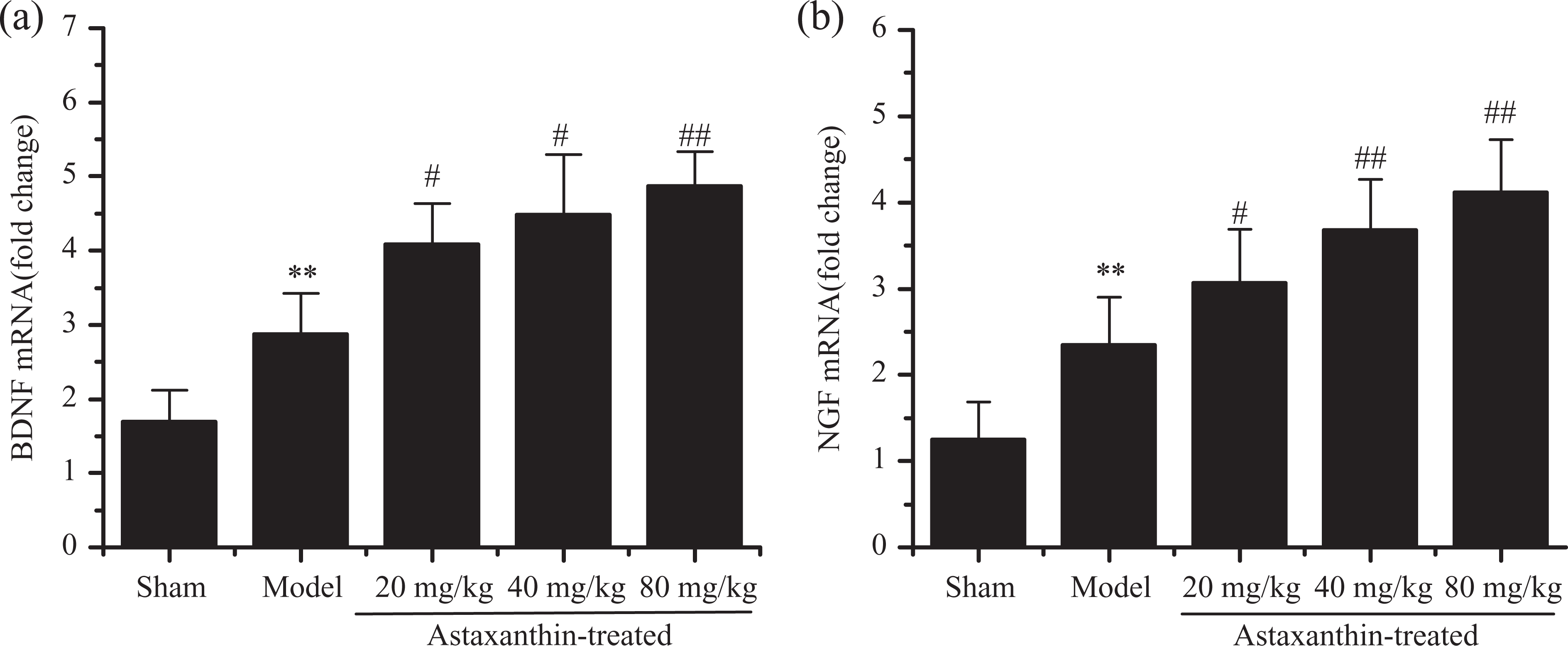
Effect of astaxanthin on the expression of BDNF and NGF mRNA in brain: (a) BDNF mRNA. (b) NGF mRNA. Data are presented as mean ± SD. **p < 0.01 versus sham group; #p < 0.05, ##p < 0.01 versus model group. BDNF: brain-derived neurotrophic factor; NGF: nerve growth factor.
Discussion
ACI may occur when the coronary artery becomes blocked. Interrupting blood flow through the vessels caused oxygen deficiency and ischemia, followed by infarct areas to be anaerobic and brain tissues to be depleted of energy, finally resulting in necrosis or malacia of brain tissues. Previous studies demonstrated that MCAO results in behavioral, neurochemical, and histological abnormality in rat brain, and it could mimic many features of the ACI in humans. 19,20 In this study, we show evidence that astaxanthin exhibits powerful neuroprotective activity on ACI by using MCAO model. The results confirmed that astaxanthin has potential as a therapeutic agent in ACI by inhibiting the deterioration of cerebral edema, oxidative stress damage, and regulating the expression of neurotrophic factors such as BDNF and NGF.
Cerebral ischemia is an important reason resulting in neurological impairment. 21 Neurological deficit score and cerebral edema are two common markers for evaluating the brain injury. 22 The results in our study show the notion that occlusion of rat middle cerebral artery can result in increased neurological deficit score and cerebral edema. Furthermore, in this study, it was found that treatment with astaxanthin at doses of 20, 40, and 80 mg/kg ameliorated the neurologic function and attenuated the development of cerebral ischemia. In addition, the histologic damage, including impaired neurons and edema of the neuropile were observed, while astaxanthin treatment could ameliorate the cerebral injury, shrink the infarct focus. These results demonstrated that astaxanthin significantly inhibited the cerebral edema and neurological injury was induced by MCAO in a dose-dependent manner. It showed a protective effect on cerebral ischemia.
Approximately 20% of the total oxygen supply of the body were used by the brain tissues. Considerable evidences also point that brain oxidative stress plays a vital role in the pathogenesis of ACI. CAT is one of the main agents to provide cellular protection against damage from oxygen-derived free radicals. 23 SOD is the primary line of defense against tissue that could quench the superoxide anion in mitochondrial matrix. 24 GSH-Px is an intracellular element which plays a crucial role in cellular protection against oxidant damage. 25 MDA is a crucial product of lipid peroxidation, which could indirectly reflect the production of intracellular. 26 Substantial and significant increases in antioxidant enzyme activities and decrease in lipid peroxide contents in ischemic cortex could represent an improvement of antioxidative ability of the brain. In our study, the activities of CAT, SOD, and GSH-Px were obviously increased by treatment with astaxanthin, while the content of MDA was remarkably reduced. These data suggest that astaxanthin is capable of reducing cerebral damage through inhibition oxidative stress.
Numerous literature revealed that neurotrophic factors are essential molecules which can promote early peripheral nerve regeneration. 27 They are central to some critical CNS functions, as well as growth, differentiation, maturation, synaptic transmission, and plasticity. 28 BDNF was well known to be the most abundant and widely distributed throughout the brain, and it plays a vital role in promoting neuron survival and neurite outgrowth. 29 In addition, the upregulation of BDNF expression could attenuate the oxidative damage because BDNF ameliorate mitochondrial function, which is one of the main producer of oxidative stress after ischemic injury. Studies have shown that NGF administration could improve cerebral blood flow, reduce infarct volume and brain edema, and also ameliorate neurologic function in MCAO rats. 30 In animals subjected to transient forebrain ischemia, NGF may decrease ischemic neuronal injury. In our study, we found that the expression of BDNF and NGF mRNA was upregulated by MCAO, and administration with astaxanthin increased the mRNA expression. After focal ischemic injury, BDNF and NGF participated in mediating nerve regeneration process, and obviously promoted the survival and growth of neurons after ischemia and prevented their death. Astaxanthin might promoted the production of endogenous BDNF and NGF against nerve damage. The results suggested that astaxanthin effectively prevented the nerve damage by upregulating the expression of nerve factor gene.
In conclusion, the present study demonstrated that astaxanthin provided significant neuroprotective effects on MCAO-induced ACI. The mechanisms may be attributed to suppressing oxidative stress and upregulating the expression of BDNF and NGF mRNA. All these results of the study disclosed that astaxanthin may be a new therapeutic agent for attenuating brain injury during ACI.
Footnotes
Author contribution
Yu Nai, Hong Liu, and Xizhuang Bi contributed equally.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health and Family Planning Commission of Shandong Province (no. 2016WS0717), Shandong Provincial Key Research & Development Project (no. 2017GSF218043), National Natural Science Foundation of China (no. 81501185), and Yantai Yuhuangding Hospital (no. 201624).
