Abstract
Human serum albumin (HSA) is a soluble blood protein which binds to small molecules (such as drugs and toxins) and transfers them within the blood circulation. In this research, the interaction of diazinon, as a toxic organophosphate, with HSA was investigated. Various biophysical methods such as fluorescence, ultraviolet–visible (UV-vis), Fourier transform infrared spectroscopy, and molecular docking were utilized to characterize the binding properties of diazinon to HSA under physiological-like condition. The UV-vis spectroscopy showed that the absorption increased and the fluorescence intensity of HSA decreased regularly with regard to the gradual increases of the concentrations of diazinon. Due to the binding constant of (
Keywords
Introduction
Nowadays, massive amounts of the chemical materials are produced and stored in many different parts of the world. In spite of their high economic benefits, utilizing them is always associated with the health and environmental risks. Therefore, in recent years, the concerns about the direct and indirect effects of the chemical substances on human health and environment have been increased. Among the chemical compounds, insecticides can cause serious problems like wide range of acute intoxications. Diazinon (
Materials and methods
Materials
HSA was purchased from Sigma company with the molecular weight of 66,500 Da. HSA solution with the concentration of 0.45 mM (30 mg ml−1, 3% W/V) in phosphate-buffered saline solution (PBS, 50 mM, pH 7.4) was utilized for all experiments.
Diazinon solution (9 mM) was prepared, and then, it was diluted by PBS to obtain 11 original concentrations (Table 1). The molecular weight of diazinon is 304.35 g mol−1.
The 11 concentrations of diazinon soluion (pH 7.4) for the experiments.

D: diazinon.
HSA was incubated with diazinon in the presence of ethylenediaminetetraacetic acid (1 mM) and sodium azide (0.1 mM) for 35 days. All incubations were done in physiological-like conditions (pH 7.4, 37°C and darkness), and the measurements were repeated for three times.
Apparatus and methods
UV-vis spectroscopy
UV-vis absorption spectra were recorded by means of Dual-beam UV-vis Spectrophotometer (T90 + UV-Vis Spectrophotometer PG Instruments Ltd, England) and two quartz cells with a path length of 1 cm. The main spectrum was observed at 250–290 nm, but to study the other spectra, the wavelength was increased up to 400 nm. Because of the high concentration of samples, all samples were diluted before the measurements. The final concentrations of the fresh and modified HSA were 3 × 10−3 mM in PBS (pH 7.4).
Analysis of HSA-diazinon binding constant by UV–vis spectroscopy
The values of the binding constants


where
Assuming [HAS: diazinon] =

where
According to the Beer–Lambert law:
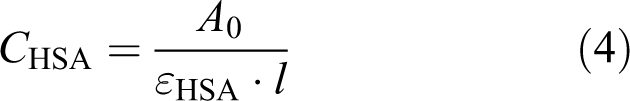

where
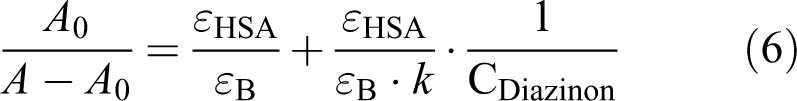
Therefore, the double reciprocal plot of 1/(
Fluorescence spectroscopy
The effect of diazinon on the third structure of HSA and its changes on the microenvironment of tryptophan (Trp-214) was studied by measuring the fluorescence intensity of Trp at the presence of diazinon using Carry-Eclipse spectrophotometer (Model 100, GMI, Inc. England). All samples were prepared at a concentration of 3 × 10–3 mM in PBS (pH 7.4). The prepared samples were excited at the wavelength of 290 nm, and the emission spectrum was recorded in the range of 300–600 nm by two quartz cells with a path length of 1 cm. The excitation slits were adjusted to 5 nm.
Spectroscopy in FTIR
Infrared spectra were recorded by means of the Thermo Nicolet FTIR spectrometer (Magna-IR 550). For FTIR experimental procedures, all solution was dried and used via tablets. 15 The spectra were considered to be at the range from 400 cm−1 to 4000 cm−1 with total scans of 100 and the resolution of 4 cm−1. The curve fitting analysis was performed by the GRAMS/AI version 7.01 software of the Galactic Industries Corporation. Therefore, the root mean square noise of every spectrum was calculated. By means of the second derivative in the spectral region (1600–1700 cm−1), eight major peaks for an HSA and the complexes of HSA with diazinon were resolved. For optimizing, the spectroscopy test was performed with a final protein concentration of 3.7 × 10−4 M (30 mg ml−1) and with diazinon concentrations of D1 to D11 (0.328, 1.64, 3.28, 4.92, 6.57, 8.21, 9.85, 11.5, 13.4, 14.7, and 16.4M) and the spectra of the dried tablets were recorded after some hours of incubation of HSA-diazinon solution on the tablets.
Docking calculations
The crystal structure of HSA (PDB ID: 1E78) was obtained through the PDB database and the 3D structure of ligand was achieved from PubChem Compound database. 16 AutoDock Tools 1.5.7rc1 (from MGL Tools software package) was utilized to prepare the receptor and ligand 3D structures. 17,18 For receptor, Kollman charges were added and nonpolar hydrogen atoms were merged, and for ligand, all partial charges were computed with Gasteiger method. Docking calculations were calculated by AutoDock Vina 1.1.2, 19 and to present the ligand–receptor poses, Viewer Lite 4.2 (Accelrys) and AutoDock Tools were used.
Results
UV-vis spectroscopy
The UV-vis absorption measurements were used to investigate the structural changes and the complex formations. The changes in absorbance of Trp-214 could be due to the structural changes of HSA. HSA has only one Trp-214 in its chemical structure. The absorption peak of HSA can normally be seen at 278/5–279 nm due to the aromatic amino acids (Figures 1 and 2). 20,21 Figure 3(a) and (b) shows the absorbance of free HSA and HSA-diazinon complexes (the modified HSA with mentioned concentrations) was increased, clearly after the first and 35 days of incubation. All experiments were done for three times.

The chemical structure of diazinon.
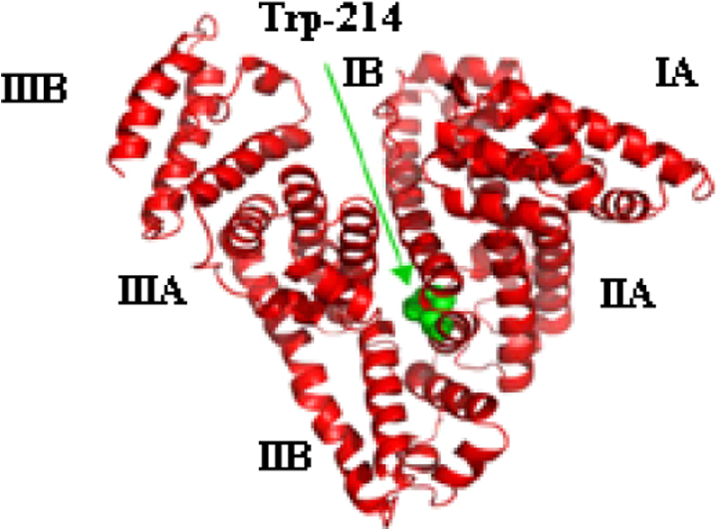
Structure of HSA. HSA: human serum albumin.

UV-vis absorption spectrum obtained from free HSA and HSA-diazinon complexes, in the PBS (pH 7.4): (a) first day and (b) 35 days. HSA: human serum albumin; UV-vis: ultraviolet visible; PBS: phosphate-buffered saline.
The most evident phenomenon in absorption curves of HSA and HSA-diazinon complexes in first and 35 days (Figure 3(a) and (b), respectively) is the hyperchromic effect of diazinon which is reflected in Figure 4 and it can be related to the mobility of the ligand around HSA molecule. Figure (4) shows that

The plot of 1/(
Analysis of fluorescence quenching of HSA by diazinon
The fluorescence method gives a wide range of protein structures and function. When the ligand (quencher) is sufficiently close to the Trp and/or Tyr residues, Trp is the major factor of protein intrinsic fluorescent. 22,23
The binding stoichiometry “
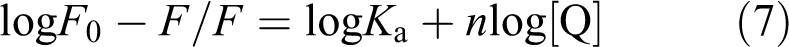
where
Two mechanisms are recognized via their different temperature dependence and their lifetime of excited states. 24 These two mechanisms (dynamic and static) can be studied by Stern–Volmer relationship which is mentioned in the following equation:

where

(a) The plot of log ((
By comparing two diagrams in Figure 6(a) and (b), the reduction in fluorescence emission on the first day and after 35 days of incubation can be seen.

Fluorescence emission spectra of HSA in the absence and presence of diazinon (D1–D11) at pH7.4, (a) first day and (b) 35 days. HSA: human serum albumin.
When HSA excited at wavelength of 290 nm, tryptophan was affected and changed third structure of protein. In Figure 6(a) and (b), fluorescence emission intensity decreased by increasing the concentration of diazinon. It seemed that the presence of diazinon in the micro-environment of tryptophan led to its shift to the nonpolar environment, which made Trp-214 residue to leave the hydrophobic environments and to transmit to more inner hydrophobic environment. The decrease in intrinsic fluorescence confirmed the interaction of diazinon with HSA. The Figure 6(b) shows HSA at high toxic concentration of diazinon. After 35 days of incubation, the emission intensity of HSA decreased and the third structure of protein was changed. For better understanding of the forces which were involved in HSA and diazinon interaction and their determination, the following equations were used:


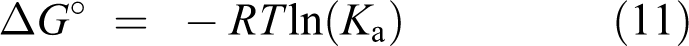

where
Briefly, the model of interaction can be summarized as follows on the basis of the thermodynamic data:
Δ
The binding constant

HSA: human serum albumin.
Transfer of energy between diazinon and HSA
During Forster’s resonance energy transfer (FRET), donor fluorophore in its excited state transfers energy to an acceptor molecule through nonradiative dipole–dipole coupling.
25
In this context, the energy transfer can be used to determine the distance from a Trp residue to a complexed diazinon. According to FRET theory, the efficiency of energy transfer (

where

where ϕ,
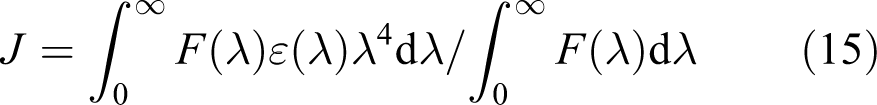
where
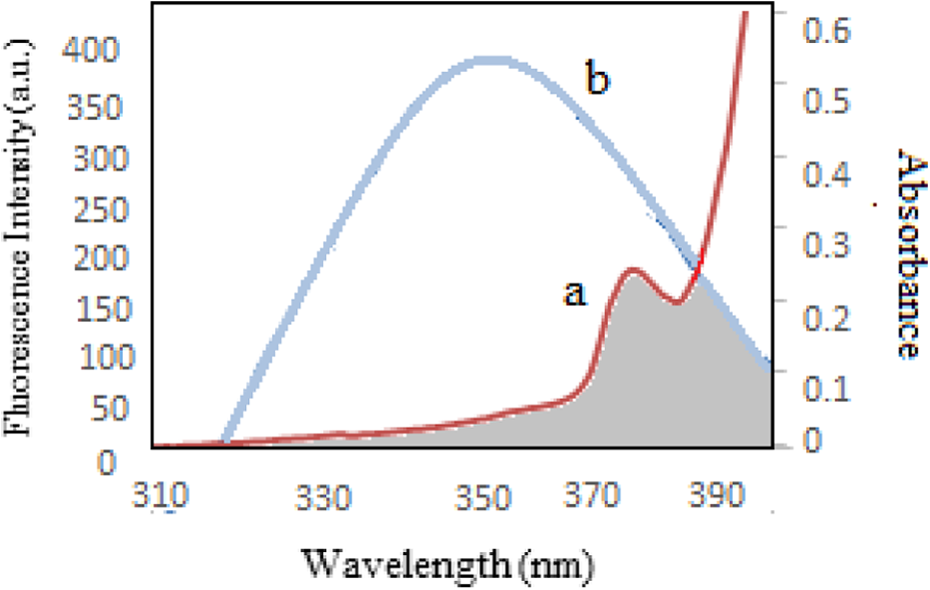
Spectral overlap between the absorption spectrum of diazinon (a) and fluorescence emission spectrum of HSA (b). HSA: human serum albumin.
According to equations (13), (14), and (15), the values of the parameters were obtained to be
Calculated values of energy transfer efficiency (

HSA: human serum albumin.
The results of FTIR
FTIR spectroscopy is a powerful technique to study the hydrogen bonding and to determine the protein secondary structure at different physiological systems. 32 The information of protein secondary structures comes from the changes in amide band’s vibrations when small molecules bind to a globular protein like HSA. 33,34 The band of amide (I) appeared in the range of 1600 to 1700 cm–1, and it was principally related to the C=O (70–85%) stretching. 35,36 The bond of amide (II) primarily was related to N–H (40–60%) binding with contribution of C–N (18–40%) stretching vibrations. The bond of amide (II) was appeared at the range of 1510–1580 cm–1. 37,38 This section deals with the effect of diazinon as a chemical compound. The band of amide I in protein is sensitive to the changes of the protein secondary structures, and HSA has been generally utilized to extract information about this kind of structure. The results of the first and 35 days of incubation are shown in Tables 4, 5, 6, and 7. At the first day, the α-helix structure declined from 66.09% to 55.4%, and other structures such as turn increased from 16.99% to 24.4%, β-sheet from 4.32% to 5.93%, β-anti from 2.3% to 2.79%, and r-coil from 11.28% to 11.52%. Samples that were incubated for 35 days showed the decrement in α-helix structure from 59.99% to 46.48%, and other structures such as turn increased from 22.14% to 32.46%, β-sheet from 4.6% to 10.88%, β-anti from 1.44% to 2.47%, and r-coil from 8.63% to 11.5%. The changes in secondary structures in 35 days were more than in first day incubated samples. HSA lost its second structure clearly at the higher concentrations of diazinon in 35 days of incubation. Figure (8) shows the fitted curve of amide (I) and secondary structure of free HSA and modified HSA in the presence of diazinon after 35 days of incubation.
The secondary structure percentage of HSA at the absence and presence of diazinon, the FTIR spectrum for the first day.

HSA: human serum albumin; FTIR: Fourier transform infrared.
Continuation of the FTIR spectrum for first day.
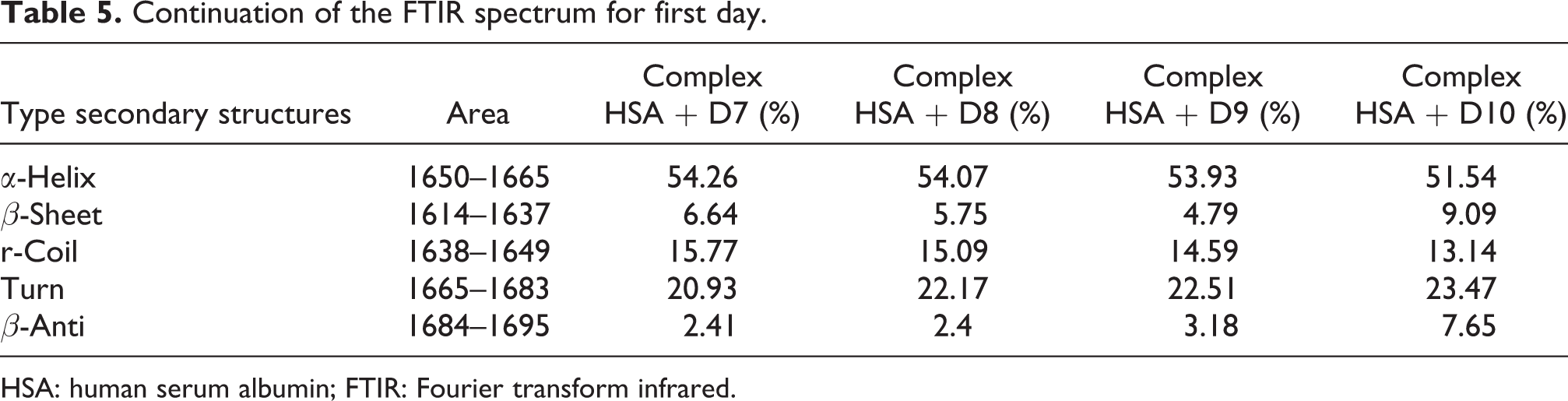
HSA: human serum albumin; FTIR: Fourier transform infrared.
The secondary structure percentage of HSA at the absence and presence of diazinon, the FTIR spectrum for the 35 days.

HSA: human serum albumin; FTIR: Fourier transform infrared.
Continuation of the FTIR spectrum for 35 days.

HSA: human serum albumin; FTIR: Fourier transform infrared.

Second derivative resolution enhancement and a curve fitted amide I and secondary structure determination of the free HSA and HSA–diazinon complexes for 35 days in the concentrations range from D1 to D11. HSA: human serum albumin.
HSA-diazinon docking study
The performed docking calculations for HSA as receptor and diazinon as ligand illustrated that there was just one binding site for diazinon in site IIA (Figure 9). In this calculation, the predicted binding energy was −28.42 kJ mol−1. Beside Trp-214, which is an indicator for site IIA of HSA, other near residues, which are in contact distance with diazinon, are included of Ile-264, Phe-223, Arg-222, Ala-291, Leu-219, Leu-260, Leu-234, Leu-238, Tyr-150, Tyr-214, and Lys-199 (Figure 10). Some of these residues have net charges in their side chains, and some others obtain their charges from hydrophobic residues. Due to this fact, it can be said that the interaction of diazinon with HSA consists of hydrophobic and electrostatic interactions, which are in good agreement with fluorescence spectroscopy results. In addition, the potential of formation of three hydrogen bonds between diazinon and Arg-222 is illustrated in Figure 11. These hydrogen bonds were formed between two of diazinon oxygen atoms and two H–Ns of guanidino group in this Arg. Our spectroscopic studies documented just one binding site for diazinon on HSA protein. The docking studies also predicted just one nearly strong binding site for diazinon on HSA protein.

HSA-diazinon docking pose which HSA illustrated IIA site for diazinon in our docking studies. CPK models of diazinon and Trp-214 are demonstrated in yellow and green colors, respectively. Red ribbons illustrated α-helixes, and the gray ones are mostly for random coils. HSA: human serum albumin.
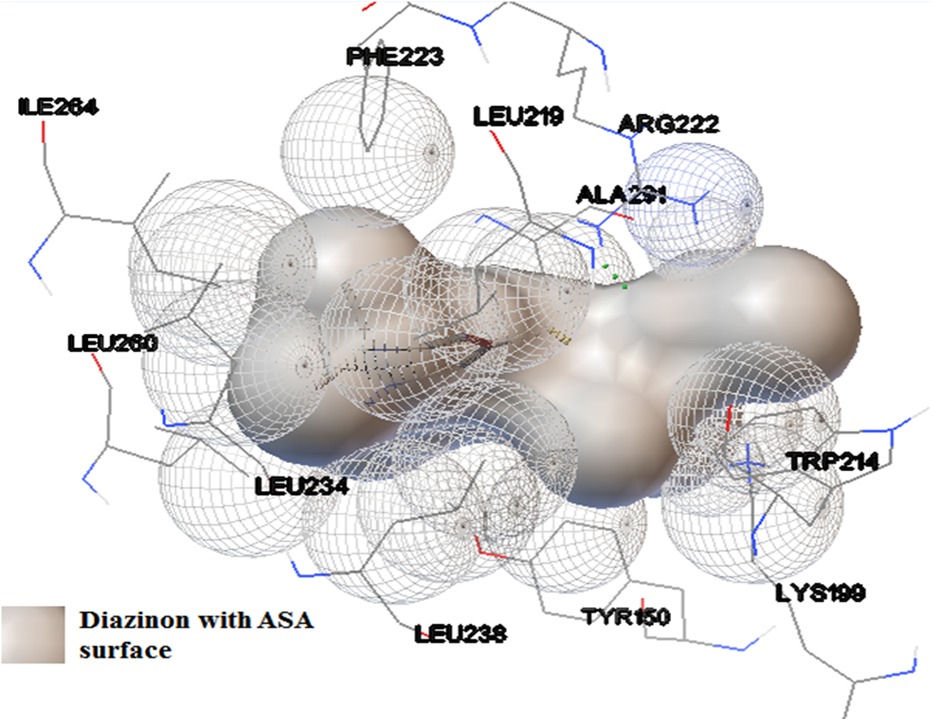
HSA-diazinon interactions pose with all residues of HSA in contact distance with diazinon. The gray colored surface is the solvent ASA for diazinon. The image of HSA was prepared with AutoDock tools. HSA: human serum albumin; ASA: accessible surface area.

Three hydrogen bonds with green color between Arg-222 and diazinon in IIA binding site of HSA which was predicted via docking calculations. The green stick model shows Trp-214 as the indicator of IIA site. HSA: human serum albumin.
Discussion and conclusion
Feeding on the explosive growth of population in developing countries is the main global challenge. Simultaneously, the pests are one of the major causes of pre- and postharvest losses of crops, and human are having to use pesticides. Although the human body is well equipped with defense systems such as the detoxifying process through different enzymes and other mechanisms, prolonged exposure to these compounds has led to the growth and development of diseases like cancer. 39 The binding study of diazinon with HSA not only has toxicological importance but also showed the effect of this toxin on HSA conformational changes and consequent events. Our study reveals that the HSA fluorescence intensity is quenched by diazinon in various degrees. Also, the increment in absorbance intensity and the reduction in fluorescence intensity clearly revealed HSA conformational changes due to diazinon, especially for the modified HSA (HSA–diazinon) after 35 days of incubation. In comparison with the related free HSA, this toxin made the significant changes in the secondary structure of HSA in the first day and after the 35 days of incubation. In both cases (modified HSA with diazinon after first day of incubation and 35 days of incubation), the reduction in α-helix and increment in other secondary structures such as β-sheet, β-anti, and random coils were observed. Given the close relationship between secondary structure elements and the protein biological activity, the reduction in α-helix can lead to the decrease in HSA biological activity after interaction with diazinon. HSA treatment with a high toxic concentration of diazinon caused significant protein, partial unfolding (the tertiary structure) in modified HSA after 35 days of incubation. The results of secondary and third structural changes in HAS showed that HSA-diazinon binding site had good vicinity with some main amino acids of HSA, especially Trp214. The effects of diazinon on HSA structural changes are related to the effect of this compound on Trp214 residue. The results of molecular docking also confirmed that the ligand binding took place in Trp214. The binding pattern is determined by hydrophobic and electrostatic interaction of diazinon with HSA.The organophosphorus insecticides such as parathion and paraxon only interact with one type of binding sites of HSA. The primary binding site for methyl parathion with HSA and BSA is close to tryptophan residues 214 and 212, respectively. 40,41 Studies on the interactions of chlorpyrifos (CPF) with HSA and BSA showed that secondary structures of the proteins changed and also the binding site of the CPF on these two receptors are Trp-214 and Trp-212, respectively. 42 From this group of compounds, the dichlorvos binds to site (I) of HSA and this interaction induces changes in the secondary and tertiary structures of the receptor, similar to our work. 43 Also, in a similar study using fluorescence and docking, interaction of the imidacloprid with HSA revealed that Trp and Tyr residues existed in the environment of the binding site. 44 Also, the thiacloprid (TL) was located on the surface of the binding subdomain IIA in HSA. 45 On the other hand, the herbicides such as paraguat, glyphosate, 2,4-D, atrazine, and the fungicide tiophanate methyl (MT) induced changes in the secondary structure of HSA. 45,46,47,48 Dominant forces, that is, hydrophobic and electrostatic, of the interactions which are mentioned in the present study suggest that pesticides should have been targeted on Trp-214 of IIA site of HSA, and it could make changes in the structure of HSA even if it will be a small amount. Since all the studies in this present work have been done in in vitro condition and they are all have been dependent on laboratory conditions, in vivo studies could be a good suggestion for a future study. 49
This study suggests that prolonged exposure of human to some organophosphate insecticides such as diazinon can create serious modifications in HSA. Finding of the present study indicates that even the presence of the allowable value of diazinon in foods can be harmful.
Footnotes
Acknowledgements
The authors would like to thank the Institute of Biochemistry and Biophysics (IBB) of Tehran University and Islamic Azad University, Science and Research, Tehran Branch for support of this work
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
