Abstract
Background:
Nitric oxide (NO) is an effective mediator of ischemic preconditioning (IPC)-induced cardioprotection. Atrial natriuretic peptide (ANP) is downregulated after ovariectomy, which results in reduction in the level of NO. The present study deals with the investigation of the role of ANP in abrogated cardioprotective effect of IPC in the ovariectomized rat heart.
Methods:
Heart was isolated from ovariectomized rat and mounted on Langendorff’s apparatus, subjected to 30 min of ischemia and 120 min of reperfusion. IPC was given by four cycles of 5 min of ischemia and 5 min of reperfusion with Krebs–Henseleit solution. The myocardial infract size was estimated employing triphenyltetrazolium chloride stain, and coronary effluent was analyzed for creatine kinase-MB (CK-MB) and lactate dehydrogenase (LDH) release to consider the degree of myocardial injury. The cardiac release of NO was estimated by measuring the level of nitrite in coronary effluent.
Results:
IPC-mediated cardioprotection was significantly attenuated in ovariectomized rat as compared to normal rat, which was restored by perfusion with ANP. However, this observed cardioprotection was significantly attenuated by perfusion with L-NAME, an endothelial nitric oxide synthase inhibitor, and Glibenclamide, a KATP channel blocker, alone or in combination noted in terms of increase in myocardial infract size, release of CK-MB and LDH, and also decrease in release of NO.
Conclusion:
Thus, it is suggested that ANP restores the attenuated cardioprotective effect of IPC in the ovariectomized rat heart which may be due to increase in the availability of NO and consequent increase activation of mitochondrial KATP channels.
Introduction
Estrogen deficiency is one of the major risk factors of ischemic heart disease (IHD). It is well documented that men are more susceptible to the risk of IHD than women. But after menopause, the risk of IHD in women reached to the same level as in men of the same age. 1,2 IHDs have been remarked as major cause of morbidity and mortality. 3 Reperfusion of an ischemic myocardium is essential for the restoration of normal functioning of the myocardium. 4 However, abrupt reperfusion of an ischemic heart produces further damage of myocardium, described as ischemia reperfusion injury (I/R injury). 5,6 Ischemic preconditioning (IPC) is a potent endogenous protective happening in response to brief and multiple cycles of myocardial ischemia followed by reperfusion to protect the heart against more prolonged ischemic insult. 7 Hence, a short duration of ischemia and the following reperfusion render the heart more resistant to prolonged ischemic insult and reduce the infract size. IPC produces cardioprotection by activating G-protein coupled receptors that in turn activates phosphoinositide-3-kinase (PI3K)/serine/threonine kinase (Akt) leading to phosphorylation of endothelial nitric oxide synthase (eNOS) and opening of mitochondrial KATP channels. 8 –11 However, cardioprotective effect of IPC gets attenuated in certain pathological conditions such as hypertension, 12,13 aging, 14 heart failure, 15 diabetes mellitus, 16,17 hyperlipidemia, 18 and estrogen deficiency. 19
Atrial natriuretic peptide (ANP), a circulating hormone of cardiac origin, is polypeptide in nature and has vasodilator property. 20 Three types of ANP receptors (ANPR) have been identified, namely natriuretic peptide receptor A (NPR-A), natriuretic peptide receptor B (NPR-B), and natriuretic peptide receptor C (NPR-C). The ANPR are biologically active and work via activation of guanylate cyclase that is found to protect against reoxygenation-induced hypercontracture in isolated cardiomyocytes by increasing cGMP synthesis. 21,22 ANP triggers the eNOS and consequently increases the production of NO. 23 I/R injury after regional or global ischemia involves in injury to the endothelial cell, cardiomyocytes, and vascular smooth muscle, but perfusion of ANP at the time of reperfusion prevents the myocardium from the I/R injury. 22 IPC produces cardioprotection through the perfusion of ANP, which is responsible for the release of NO and subsequently activation of eNOS. 23
The cardioprotective effect of IPC is attenuated in estrogen deficiency. 19 Estrogen deficiency has been reported to downregulate the level of ANP which leads to decrease in the release of NO and the NO has been noted to produce IPC-mediated cardioprotection. 16,24 Therefore, the present study has been designed to investigate the role of ANP in modulation of cardioprotective effect of IPC in the ovariectomized rat heart.
Materials and methods
Female Wistar rats weighing about 200–250 g kept in animal house and provided 12 h light and 12 h dark cycle were employed in this study. They were fed on standard chow diet (wheat flour 22.5%, roasted bengal gram powder 60%, skimmed milk powder 5%, casein 4%, refined oil 4%, salt mixture with starch 4%, and vitamin and choline mixture 0.5%) and provided water ad libitum. The experimental protocol used in the present study was approved by institutional animal ethics committee (GLAIPAR/CPCSEA/IAEC/2016/P.COL) in accordance with the national guideline used in use of laboratory animals.
Drugs and chemicals
ANP (0.1 μM/L) (Sigma Aldrich [P] Ltd., Bangalore, India) was dissolved in Krebs–Henseleit (K-H) buffer and then perfused the isolated heart in four cycles of reperfusion. L-NAME (100 μM/L) (Sigma Aldrich [P] Ltd.) and Glibenclamide (10 µM/L) (Medirose Drugs and Pharmaceutical Pvt. Ltd., Ghaziabad, Uttar Pradesh, India) were added to K-H buffer for perfusion. All other reagents used in this research were of analytical grade and always prepared freshly before use.
Induction of experimental ovariectomy
Female rats were anaesthetized using pentobarbitone (45 mg/kg i.p.) and placed on its dorsal surface. The area for surgery was shaved and cleaned with ethanol. Then a small transverse peritoneal incision of nearly 0.4–0.6 cm was made with surgical scalpel blade on the center part of the abdomen slightly toward right. After peritoneal cavity was accessed, adipose tissue was dislocated out until the fallopian tube and the ovary surrounded by fat were recognized. The ovary and associated fat were easily located and exteriorized by gentle retraction. After identification of ovary, suture was applied at the end of the uterus and the beginning of ovary. Then ovaries were removed, and the uterine horn was returned to the peritoneal cavity. This similar procedure was repeated for the second ovary through the same incision. The cut was closed in two layers (muscle and skin) by using absorbable suture (Figure 1). Highly aseptic condition was maintained throughout the procedure. Neomycin antibiotic powder was applied twice daily on wounds for 1 week and animals were allowed to recover for 4 weeks. 19,25 After surgery, rats were housed individually in cages, net clean and dry bedding up of sterilized fabric cotton was provided to prevent the contamination. Estrogen level was investigated 30 days after surgery in ovariectomized rats and sham-operated controls. Plasma estrogen levels and the absence of the ovaries verified the adequacy of ovariectomy. Bilateral ovariectomy produced an estrogen level of 6.8 pg/ml after 4 weeks.

Surgical procedure of ovariectomy.
Isolated rat heart preparation
Rats were injected heparin (500 IU/L, i.p.) (Gland Pharma Ltd., Hyderabad, India) before 20 min of sacrificing animal by cervical dislocation. The rat heart was quickly excised and immediately mounted on Langendorff’s apparatus. 26 The heart was enclosed in a double walled jacket and temperature was maintained at 37°C by circulating warm water. Isolated heart was retrogradely perfused at constant pressure of 80 mmHg and coronary flow rate of 7–9 ml/min with K-H buffer (NaCl 118 mM; KCl 4.7 mM; CaCl2 2.5 mM; MgSO4·7H 1.2 mM; KH2PO4 1.2 mM; C6H12O6 11 mM), pH 7.4, maintained at 37°C bubbled with 95% O2 and 5% CO2. IPC was produced by closing the inflow of K-H solution for 5 min followed by 5 min of reperfusion. Four such episodes were employed. Global ischemia was produced for 30 min by blocking the inflow of K-H solution which was followed by 120 min of reperfusion. Coronary effluent was collected before ischemia, immediately, 5 min, and 30 min after reperfusion for estimation of LDH, CK-MB, and nitrite release. 19,27
Assessment of myocardial injury
The myocardial infarction was assessed by the estimation of LDH and CK-MB in the coronary effluent and measurement of infarct size which determine the extent of myocardial injury. The assessment of myocardial infarct size was done by using triphenyltetrazolium chloride (TTC) staining method while LDH and CK-MB were estimated using commercial kits. 16,19 Values of LDH and CK-MB were expressed in international units per liter (IU/L).
Estimation of LDH and CK-MB
LDH was estimated in samples of coronary effluent collected before stabilization, immediately and 30 min after reperfusion using commercially available kit (Span Diagnostics Ltd., Surat, Gujarat, India) spectrophotometrically at 340 nm while CK-MB release was estimated before stabilization, immediately and 5 min after reperfusion using commercially available kit (Coral Clinical Systems, Goa, India) spectrophotometrically at 340 nm. 19
Estimation of myocardial infract size
The heart was removed from Langendorff’s apparatus. Both the atria and roots of aorta were excised and ventricles were reserved overnight at −4°C. Frozen ventricles were sliced into regular sections of about 1–2 mm thickness. The slices were incubated at 37°C for 30 min in 1% w/v TTC stain in Tris-chloride buffer pH 7.4. 28 The viable cells were stained brick red due to conversion of TTC to red formazone pigment by NADH and dehydrogenase enzyme, 29 while the infracted portion remained unstained (Figure 2). Infract size was expressed as percentage of average ventricular volume. 30,31

Images of the TTC-stained sections of heart. TTC: triphenyltetrazolium chloride.
Nitrite estimation
Nitrite is a stable nitrogen intermediate formed from the spontaneous degradation of NO. Unlike NO, nitrite can be measured without difficulty and nitrite concentration can be used to deduce the level of NO production. 32 Nitrite release in coronary effluent was measured. 33,34 Griess reagent 0.5 ml (1:1 solution of 1% sulfanilamide in 5% phosphoric acid and 0.1% N-(1-naphthyl) ethylenediamine dihydrochloride in water) was added to 0.5 ml of coronary effluent. The optical density at 550 nm was measured using spectrophotometer. Nitrite concentration was calculated by assessment with spectrophotometer reading of standard solution of sodium nitrite prepared in K-H buffer. 19 Result was expressed as micromoles per liter (µM/L).
Experimental protocol
The present study was conducted on eight groups and each group contained six rats. The diagrammatic presentation of experimental protocol is shown in Figure 3.

Diagrammatic representation of experimental protocol. S, P, I, R, ANP, L-NAME, and GLI denote stabilization, perfusion, ischemia, reperfusion, atrial natriuretic peptide, N-nitro
Group I: (Sham control; n = 6): Isolated rat heart preparation was allowed to stabilize for 10 min and then continuously perfused with K-H buffer for 190 min without subjecting them to global ischemia.
Group II: (Ischemic reperfusion control; n = 6): Isolated rat heart preparation was allowed to stabilize for 10 min and then subjected to 30 min of global ischemia followed by 120 min of reperfusion.
Group III: (IPC control; n = 6): Isolated rat heart preparation was allowed to stabilize for 10 min and then subjected to four cycles of IPC, each cycle comprising of 5 min of ischemia and 5 min of reperfusion with K-H solution. Then the preparation was subjected to 30 min global ischemia followed by 120 min of reperfusion.
Group IV: (IPC in ovariectomized rat heart; n = 6): Isolated heart preparation from ovariectomized rat was allowed to stabilize for 10 min and subjected to four cycles of IPC as described earlier in group III.
Group V: (IPC in ANP (0.1 µM/L) perfused ovariectomized rat heart; n = 6): Isolated rat heart preparation from ovariectomized rat was allowed to stabilize for 10 min and perfused with ANP (0.1 µM/L K-H buffer) in each cycle of 5 min reperfusion. Rest of the protocol was the same as described in group III.
Group VI: (IPC in L-NAME (100 µM/L) and ANP (0.1 µM/L) perfused ovariectomized rat heart; n = 6): Isolated rat heart preparation from ovariectomized rat was perfused with L-NAME (100 µM/L K-H buffer) during 10 min of stabilization. Rest of protocol was the same as described in group V.
Group VII: (IPC in Glibenclamide (10 µM/L) and ANP (0.1 µM/L) perfused ovariectomized rat heart; n = 6): Isolated rat heart preparation from ovariectomized rat was perfused with Glibenclamide (10µM/L K-H buffer) during 10 min of stabilization. Rest of the protocol was the same as described in group V.
Group VIII: (IPC in L-NAME (100 µM/L), Glibenclamide (10 µM/L), and ANP (0.1 µM/L) perfused ovariectomized rat heart; n = 6): Isolated rat heart preparation from ovariectomized rat was perfused with Glibenclamide (10 µM/L K-H buffer) and L-NAME (100 µM/L K-H buffer) during 10 min of stabilization. Rest of the protocol was the same as described in group V.
Statistical analysis
All values were expressed as mean ± standard deviation. The data obtained from a various groups were statistically analyzed using one-way analysis of variance followed by Tukey’s multiple comparison test. A p value of less than 0.05 was considered to be statistically significant.
Results
Effect of ovariectomy on estrogen level
Ovariectomy significantly decreased the estrogen level as compared to sham control but did not become zero because the adrenal gland also produces little amount of sex hormone (Figure 4).
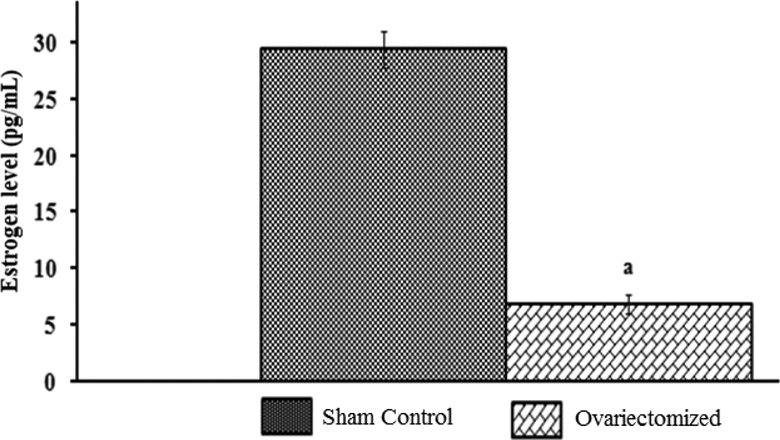
Effect of ovariectomy on estrogen level. Values are expressed as mean ± standard deviation.
Effect of IPC and pharmacological interventions on myocardial injury
Global ischemia of 30 min followed by reperfusion of 120 min significantly increased the myocardial infarct size and the release of LDH and CK-MB in coronary effluents as compared to sham control group. Four cycles of 5 min ischemia and 5 min reperfusion were sufficient to markedly prevent the I/R-induced increase in infarct size and the release of LDH and CK-MB in normal rat heart but not in ovariectomized rat heart. Moreover, perfusion of ANP significantly restored the IPC-induced reduction in myocardial injury in ovariectomized rat heart. Further, perfusion with L-NAME and Glibenclamide alone or in combination in stabilization phase significantly attenuated the decrease of myocardial infarct size, LDH and CK-MB in further ANP perfused ovariectomized rat heart (Figures 5 to 7).

Effect of I/R on myocardial infarct size, effect of IPC on myocardial infarct size in normal and ovariectomized rat heart, effect of ANP perfusion, effect of L-NAME and ANP perfusion, effect of Glibenclamide and ANP perfusion, and effect of L-NAME, Glibenclamide, and ANP perfusion on myocardial infarct size in ovariectomized rat heart. Values are expressed as mean ± standard deviation, a = p < 0.05 vs. sham control; b = p < 0.05 vs. I/R control; c = p < 0.05 vs. IPC in normal rat heart; d = p < 0.05 vs. IPC in ovariectomized rat heart; e = p < 0.05 vs. IPC in ovariectomized rat heart with perfusion of ANP. I/R: ischemic reperfusion; IPC: ischemic preconditioning; OVX: ovariectomy; ANP: atrial natriuretic peptide; L-NAME: N-nitro
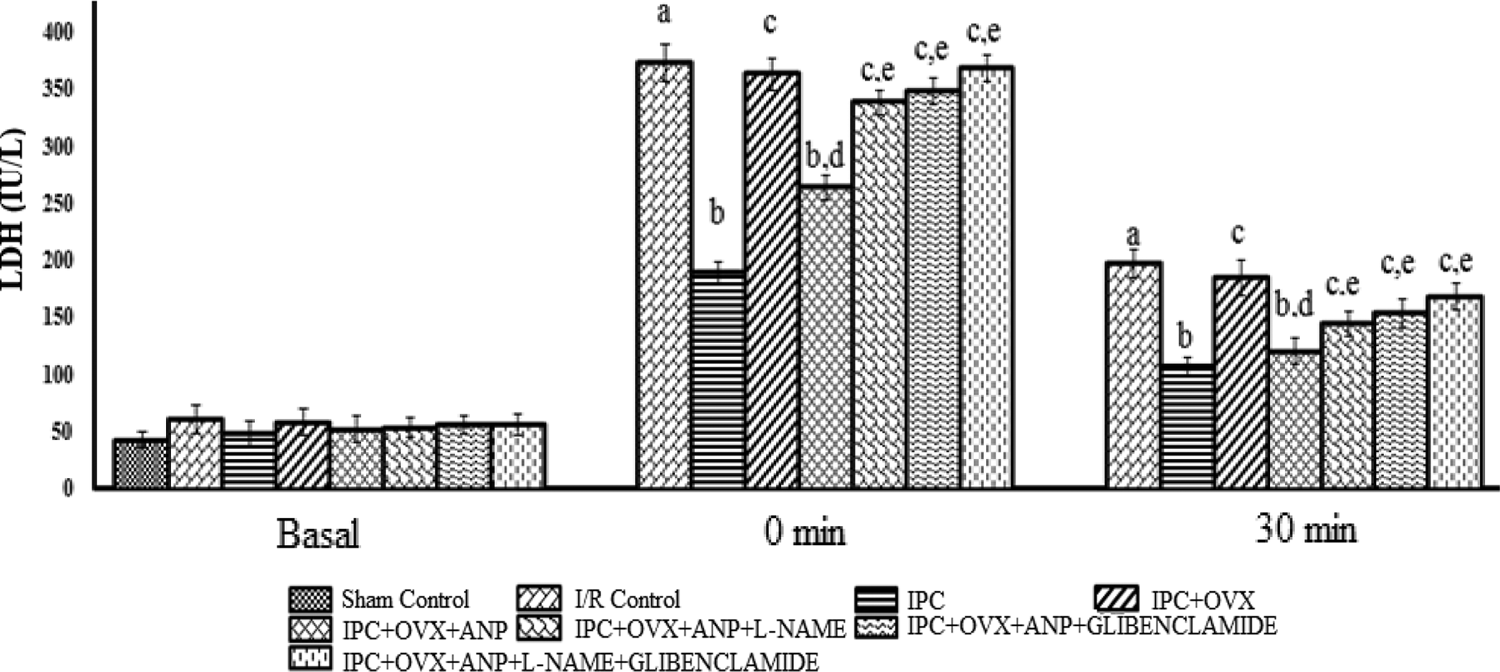
Effect of I/R on release of LDH, effect of IPC on release of LDH in normal and ovariectomized rat heart, effect of ANP perfusion, effect of L-NAME and ANP perfusion, effect of Glibenclamide and ANP perfusion, and effect of L-NAME, Glibenclamide, and ANP perfusion on release of LDH in ovariectomized rat heart. Values are expressed as mean ± standard deviation, a = p < 0.05 vs. sham control; b = p < 0.05 vs. I/R control; c = p < 0.05 vs. IPC in normal rat heart; d = p < 0.05 vs. IPC in ovariectomized rat heart; e = p < 0.05 vs. IPC in ovariectomized rat heart with perfusion of ANP. I/R: ischemic reperfusion; IPC: ischemic preconditioning; OVX: ovariectomy; ANP: atrial natriuretic peptide; L-NAME: N-nitro

Effect of I/R on release of CK-MB, effect of IPC on release of CK-MB in normal and ovariectomized rat heart, effect of ANP perfusion, effect of L-NAME and ANP perfusion, effect of Glibenclamide and ANP perfusion, and effect of L-NAME, Glibenclamide, and ANP perfusion on release of CK-MB in ovariectomized rat heart. Values are expressed as mean ± standard deviation, a = p < 0.05 vs. sham control; b = p < 0.05 vs. I/R control; c = p < 0.05 vs. IPC in normal rat heart; d = p < 0.05 vs. IPC in ovariectomized rat heart; e = p < 0.05 vs. IPC in ovariectomized rat heart with perfusion of ANP. I/R: ischemic reperfusion; IPC: ischemic preconditioning; OVX: ovariectomy; ANP: atrial natriuretic peptide; L-NAME: N-nitro
Effect of IPC and pharmacological interventions on the release of nitrite
Four episodes of IPC significantly increased the release of nitrite into coronary effluent of normal animals as compared to I/R group but not in isolated heart obtained from ovariectomized rat. Perfusion with ANP significantly increased the release of nitrite in ovariectomized rat heart subjected to IPC. However, perfusion with L-NAME and Glibenclamide alone or in combination in stabilization phase significantly attenuated the increase in release of nitrite in further ANP perfused ovariectomized rat heart (Figure 8).

Effect of I/R on release of nitrite, effect of IPC on release of nitrite in normal and ovariectomized rat heart, effect of ANP perfusion, effect of L-NAME and ANP perfusion, effect of Glibenclamide and ANP perfusion, and effect of L-NAME, Glibenclamide, and ANP perfusion on release of nitrite in ovariectomized rat heart. Values are expressed as mean ± standard deviation, a = p < 0.05 vs. sham control; b = p < 0.05 vs. I/R control; c = p < 0.05 vs. IPC in normal rat heart; d = p < 0.05 vs. IPC in ovariectomized rat heart; e = p < 0.05 vs. IPC in ovariectomized rat heart with perfusion of ANP. I/R: ischemic reperfusion; IPC: ischemic preconditioning; OVX: ovariectomy; ANP: atrial natriuretic peptide; L-NAME: N-nitro
Discussion
In the present study, 30 min of global ischemia and 120 min of reperfusion increase the myocardial infarct size, release of LDH and CK-MB, and decreased the release of nitrite in coronary effluent of normal rat heart. However, four episodes of 5 min of ischemia and 5 min of reperfusion preceding 30 min of ischemia and 120 min of reperfusion were sufficient to significantly attenuate the I/R-induced increased myocardial infarct size, release of LDH and CK-MB, and decreased release of nitrite in coronary effluent of normal rat heart. These results are in accordance with earlier published report from our laboratory. 16,19
ANP is a 28-amino acid peptide that is secreted from the atria and possesses natriuretic, vasoactive, and renin-inhibiting actions. ANP, a cardiac hormone, exerts abundant of cardioprotective effect by NPR-A which activates the cGMP by the nitric oxide (NO) protein kinase C-dependent pathway. 20 –22 Increasing production of cGMP and NO by the ANP is pioneer to defend the myocardium from I/R injury. 21,35 This contention is supported by our earlier published report that the perfusion of ANP at the instance of reperfusion protects the myocardium from the I/R injury and minimizes the myocardial infract size to some extent and further perfusion of ANP with L-NAME in normal rat heart increased the myocardial infarct size in comparison to ANP perfused normal rat heart. These results show that ANP protects the myocardium from the I/R injury by facilitating the release of NO in normal rat heart. 36 However, in the ovariectomized rat heart, the level of ANP gets downregulated. 24 In the present study, IPC-induced cardioprotection and release of nitrite were significantly reduced in ovariectomized rat heart as compared to normal rat heart, which is supported by findings of our laboratory. 19 It may be due to decreased level of ANP and subsequent decrease in eNOS activity.
In the present study, perfusion of ANP in ovariectomized rat heart restored the attenuated cardioprotective effect of IPC and increased the release of NO. Further, perfusion of L-NAME, an NO synthase inhibitor with ANP restricted the IPC induced increase in release of NO, decrease in myocardial infarct size, and release of LDH and CK-MB in the coronary effluent of ovariectomized rat heart. It indicates that ANP protects the myocardium from I/R injury as indicated in our study by enhancing the release of nitrite in coronary effluent in the ovariectomized rat heart.
Opening of mitochondrial KATP channels protects the myocardium from I/R injury. 37 Various mediators, that is, adenosine, bradykinin, angiotensin, prostaglandins, and NO which are released by the stimuli of IPC produce cardioprotection through opening of mitochondrial KATP channel. 38,39 In the present study, perfusion of Glibenclamide, a KATP channel blocker with ANP, abolished the IPC- induced increase in release of NO, decrease in myocardial infarct size, and release of LDH and CK-MB in the coronary effluent of ovariectomized rat heart. It is suggested that the observed cardioprotective effect of IPC in normal rat heart and ANP perfused rat heart may be due to opening of mitochondrial KATP channel.
Moreover, perfusion of L-NAME and Glibenclamide with ANP in ovariectomized rat heart unable to produce any additional effect in comparison to individual drugs. These findings reflect that NO generated due to IPC in ANP perfused ovariectomized rat heart produces cardioprotection by opening of mitochondrial KATP channels.
These data indicate that perfusion of ANP with IPC can protect the myocardium from estrogen deficiency–induced myocardial injury. So these findings may have an important implication for the treatment of post-menopausal women undergoing cardiopulmonary bypass. Controlled reperfusion of ANP could be an adjunctive for the myocardial protection during open heart surgery.
Conclusion and future perspectives
On the basis of above discussion, it may be concluded that the perfusion of ANP restores the attenuated cardioprotective effect of IPC in ovariectomized rat heart which is due to increased availability of NO and consequent increased activation of mitochondrial KATP channels. This contention is supported by the fact that perfusion of L-NAME (an eNOS inhibitor) and Glibenclamide (KATP channel blocker) alone or in combination with ANP significantly attenuated the cardioprotective effect of IPC in the ovariectomized rat heart. This knowledge of the involved pathway would enhance and focus future applications of preconditioning and might enable pharmacological initiation of preconditioning cardioprotective pathways in post-menopausal women.
Limitation of the present study
There are several limitations in our study. Firstly, the proposed ANP level should have been assessed by RT-PCR. Secondly, additional studies are necessary to confirm whether ANP improves preconditioning induced cardioprotection in ovariectomized rat heart through other signaling pathways.
Footnotes
Acknowledgments
We are grateful to Shri Narayan Das Agrawal Ji, Chancellor, GLA University, Prof. D.S. Chauhan, Vice Chancellor, GLA University, Prof. Pradeep Mishra, Director, Institute of Pharmaceutical Research, GLA University, Prof. Meenakshi Bajpai, Head of Department, Institute of Pharmaceutical research, GLA University, Mathura for their praise worthy inspiration and constant support for this study.
Author contribution
All authors contributed equally to this work. J.K.G. (Supervisor) A.G. (Internal Supportive member of this study) and H.N.Y. (External Supportive member of this study) developed the concept and designed the experiment. H.N.Y. then moved to AIIMS, New Delhi, India. V.K.V. performed experiment on Langendorff’s apparatus. P.K.U. and J.K.G. carried out analysis of infarct size and data of other parameters (LDH, CK-MB, and nitrite). V.K.V. prepared the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
