Abstract
Background:
Accidental drug overdose is a common problem in young children. We examined the influence of formulation and dose in enquiries for different gastro-oesophageal reflux disease treatments in children under 5 years to the UK’s National Poisons Information Service.
Methods:
Overdose characteristics with ranitidine, omeprazole or domperidone were compared with those of metoclopramide and the H-1 antagonist chlorphenamine, for the period 1 July 2007 to 30 June 2015.
Results:
There were a total of 1092 ranitidine, 618 domperidone and 1193 omeprazole cases; 669, 281 and 424, respectively, were single agent enquiries; of these 77% (517) of ranitidine, 52% (145) domperidone and 32% (135) omeprazole cases occurred in children <5 years. In comparison, 17% (34/424) of metoclopramide and 53% (533/1013) of chlorphenamine were <5 years; 79% (410/517) of ranitidine overdose enquiries in children <5 years were under 6 months of age, higher than domperidone (68/145, 47%; p < 0.05), omeprazole (8/135, 6%), chlorphenamine (13/553, 2%) or metoclopramide (1/34, 3%) (all p < 0.01). In children aged <6 months, 101 were 10-fold overdoses, 86 with ranitidine.
Conclusions:
Tenfold overdoses in children (<5 years) were a feature of ranitidine enquiries, likely due to the high concentration of the syrup. This has relevance to other liquid formulations used for non-licenced indications in young children. Such therapeutic errors cause significant carer anxiety and healthcare utilization. Assistance is needed from manufacturers and legislators in modifying formulation so that drugs can be safely used in young children. Education of prescribers and carers is also needed to reduce the incidence of such errors that cause significant carer anxiety and healthcare utilization.
Introduction
Problems in correctly dosing young children with medicines are well recognized, with 10-fold overdosing errors, a particular problem in very young children. 1 Overdose of drugs used for gastro-oesophageal reflux disease (GORD), notably cimetidine and metoclopramide, has been identified as a particular problem. 2
In the United Kingdom, ranitidine has generally replaced cimetidine for GORD because of concerns over drug interactions. It is commonly prescribed for children 3,4 although safety and efficacy in paediatric patients is uncertain 5 and oral ranitidine is not licensed in children under 3 years old. 6 Metoclopramide is not licenced in the United Kingdom for GORD in young children because of dystonic reactions, being licenced only for chemotherapy-induced vomiting in children. Instead, domperidone, an alternative pro-motility drug, is licenced for oral use in children 4,7 though with little evidence of efficacy 8 and recent UK Medicines and Healthcare products Regulatory Agency (MHRA) advice in 2014 has advocated short-term (<1 week) use only. 9
Review of these medications (Table 1) demonstrates the difficulty of dosing children due to licenced ages and hence the formulations available. The solid tablets are often too large for the doses required and liquid formulations are necessary. However, the high concentration of ranitidine syrup (15 mg/mL) in particular might make it difficult to calculate the correct volume and thus give the correct dose to young children. In addition, the individual volume dose for young infants is surprisingly small (<1 mL) for parents used to administering typical volume doses of 2.5–5 mL.
UK dose forms and advised paediatric dosage for the drugs used for GORD.a
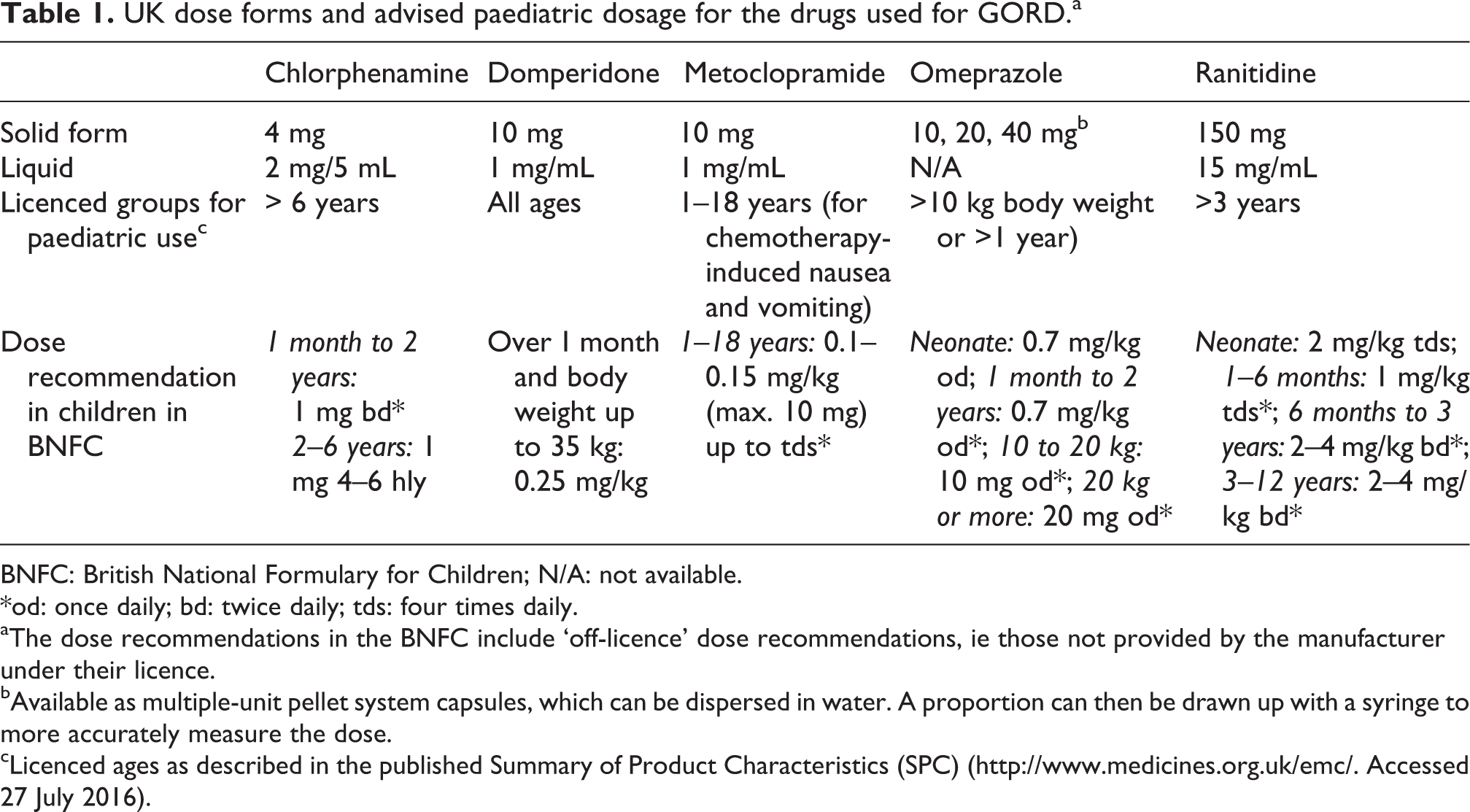
BNFC: British National Formulary for Children; N/A: not available.
*od: once daily; bd: twice daily; tds: four times daily.
aThe dose recommendations in the BNFC include ‘off-licence’ dose recommendations, ie those not provided by the manufacturer under their licence.
bAvailable as multiple-unit pellet system capsules, which can be dispersed in water. A proportion can then be drawn up with a syringe to more accurately measure the dose.
cLicenced ages as described in the published Summary of Product Characteristics (SPC) (http://www.medicines.org.uk/emc/. Accessed 27 July 2016).
We therefore tested the hypothesis that overdose with GORD medication in children might be related to formulation and dose. We therefore compared rates of enquiries to the UK’s National Poisons Information Service (NPIS) for different GORD treatments (ranitidine, domperidone, omeprazole) in children under 5 years old and also included chlorphenamine, a standard liquid antihistamine formulation, and an alternative pro-motility drug metoclopramide, which is not licenced for young children.
Methods
The NPIS operates a 24-h poisons information service for health professionals. Most information is currently accessed online via the TOXBASE database. Where more support is required, healthcare workers can call a 24-h telephone information service staffed by specialists in poisons information (SPIs) with consultant clinical toxicology support. 10 Approximately 10% of NPIS enquiries come by phone. Details of the calls are recorded in the UK Poisons Information Database (UKPID) held on a central server. The anonymized information held includes patient details (including features, investigation results and treatments already given), agent information (including dose, time of exposure, route of exposure and circumstances) and advice then given to the enquirer. A free text field for comments allows the SPIs to record any other information. The Poisoning Severity Score (PSS), indicating features at the time of the call, and the maximum PSS (MAXPSS, reflecting the most severe features experienced from the time of exposure until the time of the call), are also recorded. 11
Telephone enquiries were analysed for all cases referred over 8 years between 1 July 2007 and 30 June 2015 in which an age was recorded. Those involving children less than 5 years of age and ingestions of ranitidine, omeprazole, domperidone, metoclopramide or chlorphenamine were extracted. Particular attention was paid to enquiries involving a single agent as this is where medication errors are almost always recorded. Data for all single agent overdoses, and large overdoses (10 fold and greater), were studied. The overdose multiples were calculated based on the advised dose per kilogram body weight, or age of the child, as specified in the British National Formulary for Children (BNFC) (Table 1). 6
Demographic and clinical characteristics were described and summarized using counts (percentages) for categorical variables and the median [interquartile range (IQR)] for non-normally distributed variables and compared using the Wilcoxon test. The chi-square test was used to explore differences between proportions and the Fisher exact test used to compare individual drugs.
This study did not require approval by a UK Research Ethics Committee as the UK Health Research Authority has declared that ethical approval is not needed for research studies that use information collected routinely by any UK administration (England, Wales, Scotland and Northern Ireland) as part of usual clinical care, provided this information is passed to the researchers in a fully anonymized format.
Availability of data and materials
The data sets generated during and analysed during the current study are not publicly available as they are held within a confidential database that although anonymized may potentially contain patient identifiable data, and our ethical permissions do not permit release. They are available from the corresponding author on reasonable request.
Results
Between 1 July 2007 and 30 June 2015, for the drugs studied NPIS received most telephone enquiries for chlorphenamine (1606) and least for metoclopramide (491). When only children under 5 years of age were considered there were significant differences in the proportion of single agent enquiries for different drugs (χ 2 = 110.74, p < 0.001), the largest proportion of single agent calls involved ranitidine (517/669, 77.3%) and the lowest omeprazole (135/424, 31.8%) (Table 2); the proportion of such calls was significantly greater with ranitidine than with other agents (Fisher exact test p < 0.05 for ranitidine vs. domperidone; p < 0.001 for ranitidine vs. all others). The median age [25–75% IQR] of children aged less than 5 years was lowest for ranitidine (0.25 [0.15–0.42] years), followed by domperidone (0.66 years [0.20–2.00]), omeprazole (2 years [1.04–2.50]), chlorphenamine (2 years [1.5–3.00]), with metoclopramide having the greatest median age (2 years [1.13–3.00]) (Wilcoxon, p < 0.05) (Figure 1). The majority of single-agent ranitidine calls were in children under 6 months (412/517, 79.7%; Figure 1); this proportion was significantly greater than that any other agents studied (ranitidine vs. domperidone Fisher exact test, p < 0.05 and ranitidine vs. all others p < 0.01) (Table 2).
Total telephone enquiries, telephone enquiries involving single agents, and single-agent telephone enquiries in children under 5 years.a

IQR: interquartile range; NPIS: National Poisons Information Service.
aThe proportion of single agent calls in those under 5 years was significantly greater for ranitidine than all other drugs. (Fisher’s exact test p < 0.05 for ranitidine vs. domperidone; p < 0.01 for ranitidine vs. all others.)
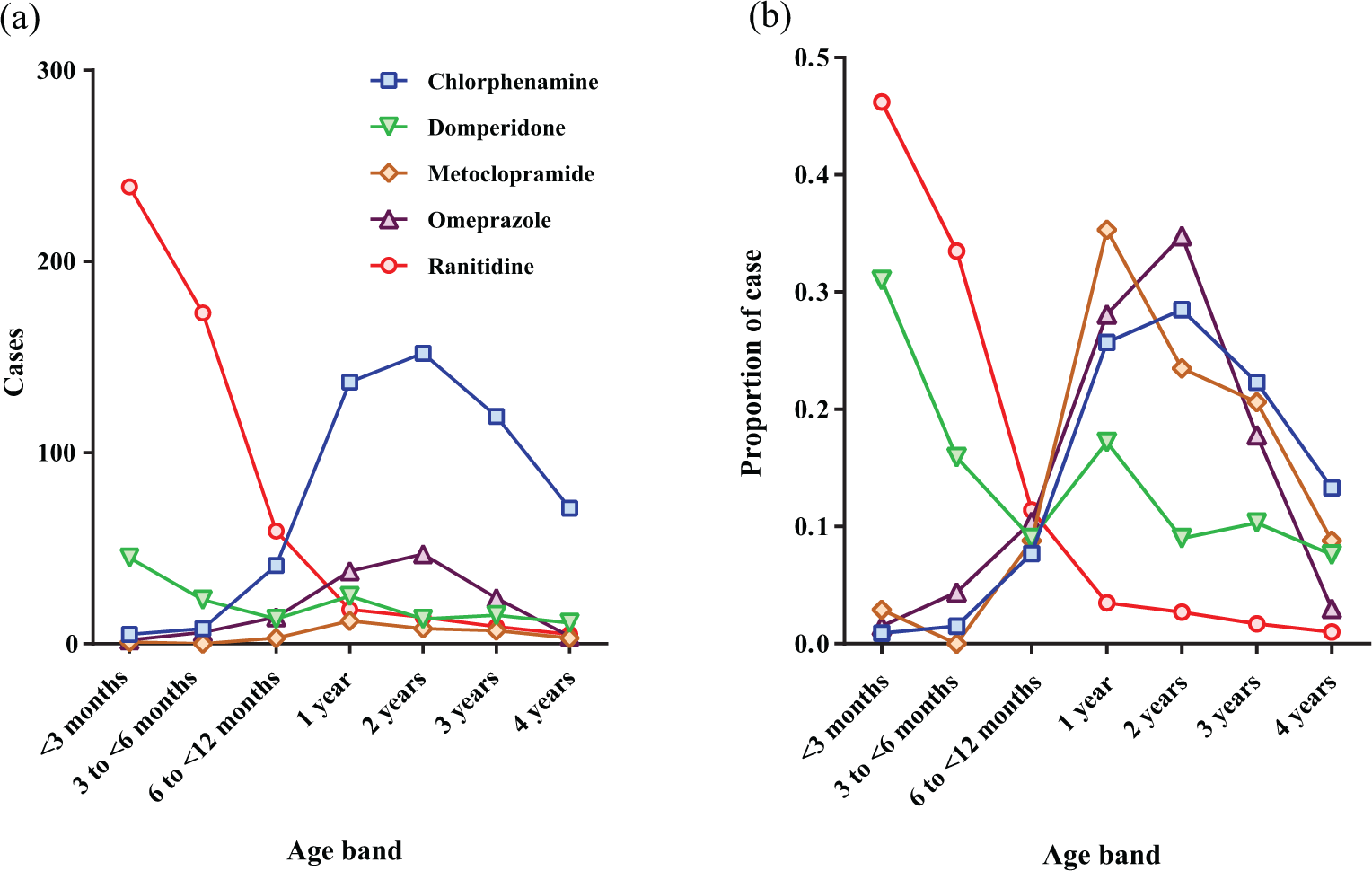
Age distribution of children under 5 years old involved in telephone enquiries for a single-agent ingestion by age band. The age was not known/recorded for 179 of all 5000 calls evaluated, of which only 3 were specifically recorded as children, with almost all the remainder being termed ‘adult’.
Of all enquiries for the 5 drugs in those under 5 years old, in 101 a 10-fold overdose was reported, and of these 86 (85%) involved ranitidine (Table 3). This represented 16.6% of all single-agent enquiries for ranitidine. This proportion was significantly greater with ranitidine than the two other agents used in GORD in children (domperidone 1.4%, omeprazole 1.5%) (Fischer exact test, p < 0.01) but similar to metoclopramide, although there were only 34 (17.6%) of enquiries for metoclopramide in this age group. When dose multiples greater than 10 were analysed there were a total of 87, and of these 58 were with ranitidine (11.2% of all ranitidine enquiries in those under 5 years), significantly greater than domperidone (0.7%) and omeprazole (3%) (Fishers exact test, p < 0.01).
Number and percentage of 10-fold and greater than 10-fold overdose enquiries by agent for single-agent enquiries.a

aThe proportion of 10-fold overdoses for ranitidine and metoclopramide were significantly greater than chlorphenamine, domperidone and omeprazole (Fisher’s exact test: p < 0.01 and <0.05, respectively). For >10-fold overdoses there was a significant difference between ranitidine and all other drugs (Fisher’s exact test p < 0.01).
Reports of symptoms relating to these high overdoses were rare. Only 25 of ranitidine single-agent ingestion calls in children under 5 years old were reported as symptomatic, but poisons severity scores were low, indicating little risk of adverse health impact. Almost all overdoses in young children were reported as accidental and all but 18 of the ranitidine enquiries occurred at home, emphasizing the involvement of parents and carers in the overdose.
Discussion
Previous work has shown the risks of 10-fold overdoses in young children, 1,12 especially involving liquid preparations. 2 The key finding of the current study is the frequency of 10-fold overdoses of ranitidine syrup. These errors usually occurred in the home and the majority involved parents and carers rather than healthcare professionals. Fortunately, these episodes are associated with few adverse effects, but they cause concern for both caregivers and healthcare providers as judged by the numbers of enquiries received. The reason for these mistakes is likely to be the high concentration of ranitidine syrup, which results in prescribed doses being far smaller than carers used to give for other paediatric medicines, such as paracetamol or chlorphenamine. The restriction of metoclopramide to cancer chemotherapy indications in the United Kingdom resulted in domperidone being the most commonly used pro-motility agent for children with GORD. This seems to be reflected by the profile of domperidone poisons enquiries in young children (Figure 1)
This case series highlights one of the problems that affects the use of prescription drugs in children, that of unlicenced use in a young paediatric population. Attention has been drawn to the problems of clinical trials in very young children. 13 Nevertheless, drugs are widely used outside licenced indications in children under 3 years old. Prescribers therefore have dosing recommendations in official publications such as summaries of product characteristics and national prescribing advice. The problem we highlight is the difficulty parents have in interpreting dose recommendations where concentrations are high in comparison to advised dose and concentrations are not in simple round numbers (Table 1). In addition, the variability of licenced age may affect the concentrations available in prescribed formulations. The more complex age/weight-based dosing regimen in the BNFC for ranitidine as compared to domperidone further illustrates a paradox in dosing recommendations in young children. The apparent safety of ranitidine in the cases reported to us throws doubt on the need for such a cautious approach to dosing.
Two case series of paediatric overdoses of ranitidine have previously been reported in abstract form from poisons centres in Connecticut 14 and Illinois. 15 In the former, 82 reports of ranitidine syrup overdose in children aged under 2 years (median age 5.3 months) were received over 4 years; 16% of the cases were 10-fold therapeutic errors. In the latter, 146 reports of ranitidine overdose in children under 6 years old (76% less than 1 year, 98.6% with syrup) were received over 3.5 years; 24.7% of cases were 10-fold therapeutic errors. Both results are remarkably similar to our findings.
In our study, all but 14 of the single ranitidine overdoses occurred in children under 3 years old, the age group for which the oral route is unlicenced. 7 Overdoses in this age group are predominantly due to dosing error by carers. Unintended 10-fold overdoses in children are recognized 1,2 but have previously been reported almost exclusively for injectable medicines. 1,12,16 It is postulated that this is a direct consequence of the formulation of medications for an adult patient group. 17,18 We did not identify this problem with domperidone, also available as syrup, but with a much simpler age-based dosing regimen than ranitidine, or omeprazole, and available in a solid dosage form that may be readily made into a suspension (Table 1). Metoclopramide, an agent identified as a cause of overdose in young children in North America, is used far less commonly in the United Kingdom for children, being prescribed predominantly for cancer chemotherapy vomiting. Chlorphenamine is widely used as a liquid agent in children, but again, perhaps because of its simpler dosing regimen and concentration, it was also not associated with large excess doses in young children.
To avoid future harm, ranitidine should be formulated in a lower concentration, and ideally in a dose/millilitre that facilitates home administration for paediatric patients to reduce the incidence of medication errors and unnecessary presentation to healthcare providers as well as anxiety to parents. This may be complicated by the off-licence nature of this indication but the complexity of the advised doses in the BNFC may contribute. However, from a regulator’s perspective, we also understand the use of a Paediatric Use Marketing Authorization to encourage the production of an oral liquid formulation with a lower strength for children less than 3 years of age would be warmly welcomed. In the meantime, it is important that those who prescribe ranitidine syrup for use in infants take the time to explain clearly to carers the idiosyncrasies of the small dose volume.
This study shows the value of poison information centres for identifying pharmacovigilance signals. Detailed recording of the nature of the overdose can allow comparison with other products/cases. Daily review of cases also allows common errors/features to be picked up early and prospective studies initiated. While a primary physician may only see one case in her or his lifetime, the UK’s NPIS received more than 1 call per month reporting a 10-fold overdose over several years. The challenge for prescribers and carers is that many drugs are unlicenced for use in young children, even though recommended by national advisory sources (Table 1). From the standpoint of manufacturers and regulators, there is thus a need to consider the implications of off-licence use of widely used marketed products in young children.
There are a number of potential weaknesses in studies using poisons information centre data. They cannot be used to assess the incidence of poisoning and data are only available on the material collected at the time of the telephone call to the poisons centre. The rates of late onset adverse effects, not present at the time of the initial enquiry, may therefore be missed. Despite these potential limitations, it is unlikely they will differentially affect the products studied in respect of the main outcomes of this analysis.
Conclusion
In conclusion, we report a high incidence of paediatric overdoses resulting from off-label use of concentrated ranitidine syrup and a high proportion of 10-fold dosing errors. Even though there were no serious consequences from ranitidine in the enquiries that were received, they result in the use of healthcare resources and anxiety. When ranitidine solution is used, greater care is needed in informing carers about the small dose volumes required together with appropriate instructions and equipment for drawing these up. Dosing errors could be reduced by the use of a less concentrated ranitidine formulation or a simpler dosing regimen. While the clinical effects of overdose with ranitidine are generally not severe, the findings of this study have wider implications to other drugs used in adult formulation in children.
Footnotes
Authors’ Note
There were no specific funders of this work. The National Poisons Information Service is funded by Public Health England on behalf of the UK Health Departments. The funders had no role in data handling, analysis or manuscript production. The data set was extracted and initially sorted by CC. Data collection was facilitated by GJ, JT, GC, AV and SHLT. Further analysis was done by DNB who also drafted the manuscript, which was initially reviewed by ME and MA. All authors reviewed and contributed to the final submission.
Acknowledgements
NPIS is commissioned by Public Health England on behalf of the UK health departments. ME was a Scottish Senior Clinical Fellow and is a Lister Prize Fellow. We wish to thank the staff of the four NPIS units for their hard work collecting this data. We thank Dr Angeliki Siapkara and Dr June Raine of the MHRA for their comments on the original version of manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
