Abstract
Nandina domestica is grown as an ornamental plant in the United States but has also been reported as an invasive plant in a number of states. Parts of the plant, particularly the berries, contain cyanogenic glycosides that convert to hydrogen cyanide when ingested. This investigation characterized N. domestica ingestions involving patients of age 5 years and less reported to Texas poison centers during 2000–2015. There were 875 total N. domestica ingestions. A seasonal pattern was observed with the highest proportion of ingestions occurring in March (18.5%) and April (14.7%). The patients were male in 55.0% of the cases; 40.8% of the patients were of age 1 and 37.0% of age 2. Berries were specifically mentioned in 709 ingestions, of which 57.3% involved one berry and 28.5% an unknown number of berries. The ingestion occurred at the patient’s own residence in 92.9% of the cases, and the patient was managed on site in 97.0%. The most frequently reported clinical effects were vomiting (3.7%), abdominal pain (1.0%), diarrhea (0.9%), and nausea (0.7%). In conclusion, N. domestica ingestions among young children generally do not result in serious outcomes and can be managed successfully outside of a healthcare facility.
Introduction
Nandina domestica, commonly known as nandina, sacred bamboo, and heavenly bamboo, is not a bamboo but a perennial shrub in the family Berberidaceae (barberry). The plant stands 2–3 m tall and 1–1.5 m in width, although dwarf cultivars are smaller. In early summer, N. domestica produces terminal conical clusters of white or pink flowers. In fall and winter, it produces bright red, spherical berries 5–10 mm in diameter. 1 –3
N. domestica contains cyanogenic glycosides with the highest concentration in the plant’s berries. When ingested, the cyanogenic glycosides convert to hydrogen cyanide. 4 –7 N. domestica berries are considered toxic to pets and grazing animals. 1,2 One study attributed the deaths of dozens of cedar waxwings (Bombycilla cedrorum) to the ingestion of toxic amounts of N. domestica. 8 According to the Pet Poison Helpline, which assists in the management of potentially adverse exposures involving animals in the United States, Canada, and the Caribbean, N. domestica is poisonous to cats and dogs, with ingestion potentially leading to vomiting, abdominal pain, dark red mucous membranes, increased temperature, tachycardia, hypertension, respiratory failure, and shock. 9
Although native to China, Japan, and India, in the early 1800s, N. domestica was introduced to the United States where it was used as an ornamental plant but has now escaped. 1,2 According to the US Department of Agriculture (USDA), the plant is considered an introduced or invasive species in nine states (Alabama, Florida, Georgia, Louisiana, Mississippi, North Carolina, South Carolina, Texas, and Virginia). 2,10 According to the Early Detection and Distribution Mapping System (EDDMapS), a web-based mapping system that documents the distribution of invasive species, N. domestica can be found as an invasive species in 16 states and the District of Columbia (Alabama, Arkansas, California, District of Columbia, Florida, Georgia, Kentucky, Louisiana, Maryland, Missouri, Mississippi, North Carolina, Oregon, South Carolina, Tennessee, Texas, and Virginia). 11
Review of the literature failed to identify information on N. domestica ingestions by humans. The intent of this study was to describe N. domestica ingestions by young children reported to Texas poison centers. In Texas, the USDA lists the plant as an invasive species in 12 of the 254 counties. 10 According to EDDMapS, N. domestica can be found as an invasive species in 55 of the Texas counties, including the 12 reported by the USDA. 11
Methods
Data for this retrospective investigation were obtained from the Texas Poison Center Network (TPCN), which is comprised of six poison centers that together service the entire state. In Texas, the estimated population age of 5 or less was 2.4 million in 2015. All poison centers in the TPCN use the same electronic database to collect information on calls in a consistent manner. The American Association of Poison Control Centers (AAPCC) standardized the data variables and coding used in this database. 12
Cases were N. domestica ingestions by patients of age 5 or less reported to the TPCN during 2000–2015. The study was limited to this age-group because preliminary analysis indicated that they represented 94% of all N. domestica ingestions. Cases were identified by identifying those records where the PoisIndex code was 2454033 or the verbatim substance field included various spellings of nandina, sacred bamboo, or heavenly bamboo. These records were reviewed to exclude those that did not appear to be about N. domestica. Ingestions involving substances in addition to N. domestica (n = 24) were included. Ingestions not followed to a final medical outcome also were included because they represented a majority of the cases.
The distribution of cases was determined for month and season of ingestion, time between ingestion and contact of the poison center, whether the ingestion was known to involve the plant’s berries and, if so, how many, patient age and gender, circumstances of (reason for) the ingestion, ingestion site, geographic location, management site, medical outcome, clinical effect, and treatment. Season was defined as winter (January to March), spring (April to June), summer (July to September), and autumn (October to December). To identify whether the ingestion was known to involve berries and, if so, how many, the verbatim substance and notes fields were reviewed for mention of berries and the number of berries believed to have been ingested. A portion of the cases described the number of berries using terms such as “some” or “a bunch.” In these cases, the number of berries was categorized as unknown. For analysis of geographic location, the caller county was identified for each case. The proportion of cases reported from those Texas counties where the USDA and EDDMapS listed N. domestica as an invasive species was determined.
The medical outcome or severity of an exposure is assigned by the TPCN staff and is based on the observed or anticipated adverse clinical effects. Medical outcome is classified according to the following criteria: no effect (no symptoms due to exposure), minor effect (some minimally troublesome symptoms), moderate effect (more pronounced, prolonged symptoms), major effect (symptoms that are life-threatening or cause significant disability or disfigurement), and death. A portion of exposures are not followed to a final medical outcome because of resource constraints or the inability to obtain subsequent information on the patient. In these instances, the poison center staff record the expected outcome of the exposure. These expected outcomes are grouped into the following categories: not followed but judged as nontoxic exposure (symptoms not expected), not followed but minimal symptoms possible (no more than minor symptoms possible), and unable to follow but judged as a potentially toxic exposure. Another medical outcome category is unrelated effect where the exposure was probably not responsible for the symptoms.
The TPCN electronic database contains checkboxes for recording 130 specific adverse clinical effects (e.g. chest pain, rash, vomiting, headache, etc.). If a patient has a clinical effect that does not correspond to one of these checkboxes, it is assigned a checkbox for “other.” Likewise, there are 66 checkboxes for specific treatments or therapies and one checkbox for “other.”
The Texas Department of State Health Services institutional review board considers this analysis exempt from ethical review.
Results
Of 932 total N. domestica ingestions reported to the TPCN during 2000–2015, 875 (93.9%) involving patients of age 5 or less and were included in the study. (In comparison, during the same time period, there were a total of 1,282,909 ingestions among patients of age 5 or less reported to the TPCN, of which 38,436 involved ingestion of a plant of some type.) It was reported that the poison center was contacted within 1 h or less of ingestion in 567 (64.8%) of the cases. The distribution of ingestions by season was 381 (43.5%) in winter, 202 (23.1%) in spring, 73 (8.3%) in summer, and 219 (25.0%) in autumn. The months with the highest proportion of cases were March (18.5%) and April (14.7%); November to April accounted for 78.9% of the ingestions.
The patients were male in 481 (55.0%) of the cases and female in 394 (45.0%), or a male/female ratio of 1.22:1. The highest proportion of ingestions occurred by patients of age 1 followed by those of age 2 (Table 1). The mean age of male patients was 1.8 years and of female patients was 1.7 years.
Age distribution of Nandina domestica ingestions among patients of age 5 or less reported to the Texas Poison Center Network during 2000–2015.

Berries were specifically mentioned in 709 (81.0%) of the N. domestica ingestions. One berry was reported in 406 (57.3%) of the ingestions, two berries in 68 (9.6%), three berries in 18 (2.5%), four berries in 10 (1.4%), and five berries in 5 (0.7%); in 202 (28.5%) of the berry ingestions, the number of berries was unknown.
All of the ingestions were unintentional. The distribution by ingestion site was 813 (92.9%) own residence, 44 (5.0%) other residence, 13 (1.5%) school, 3 (0.3) public area, and 2 (0.2%) unspecified other location. The caller county was known for 860 of the cases. Of these 860 ingestions, 469 (54.5%) were on the USDA county list and 728 (84.7%) on the EDDMaps list of counties where N. domestica was reported as an invasive species.
The patient was managed on site (non-healthcare facility setting) in 849 (97.0%) cases, already at or en route to a healthcare facility in 18 (2.1%), referred to a healthcare facility by the poison center in 5 (0.6%), and managed at an unspecified other location in 3 (0.3%). The distribution by medical outcome was 305 (34.9%) no effect, 21 (2.4%) minor effect, 1 (0.1%) moderate effect, 163 (18.6%) not followed but judged as nontoxic exposure, 358 (40.9%) not followed but minimal clinical effects possible, 13 (1.5%) unable to follow and judged as a potentially toxic exposure, and 14 (1.6%) unrelated effect. No major effects or deaths were reported.
No specific adverse clinical effect was reported in 819 (93.6%) of the cases and one or more specific adverse clinical effects were reported in 56 (6.4%). The most frequently reported clinical effects were vomiting, abdominal pain, diarrhea, and nausea (Table 2). Of the 406 ingestions of one berry, 22 (5.4%) had one or more specific adverse clinical effects. Of the 101 ingestions of two or more berries, 5 (5.0%) had one or more specific adverse clinical effects. The most frequently reported treatments were decontamination by dilution (n = 617, 70.5%) and food or snack (n = 122, 13.9%).
Reported adverse clinical effects with Nandina domestica ingestions among patients of age 5 or less reported to the Texas Poison Center Network during 2000–2015.a
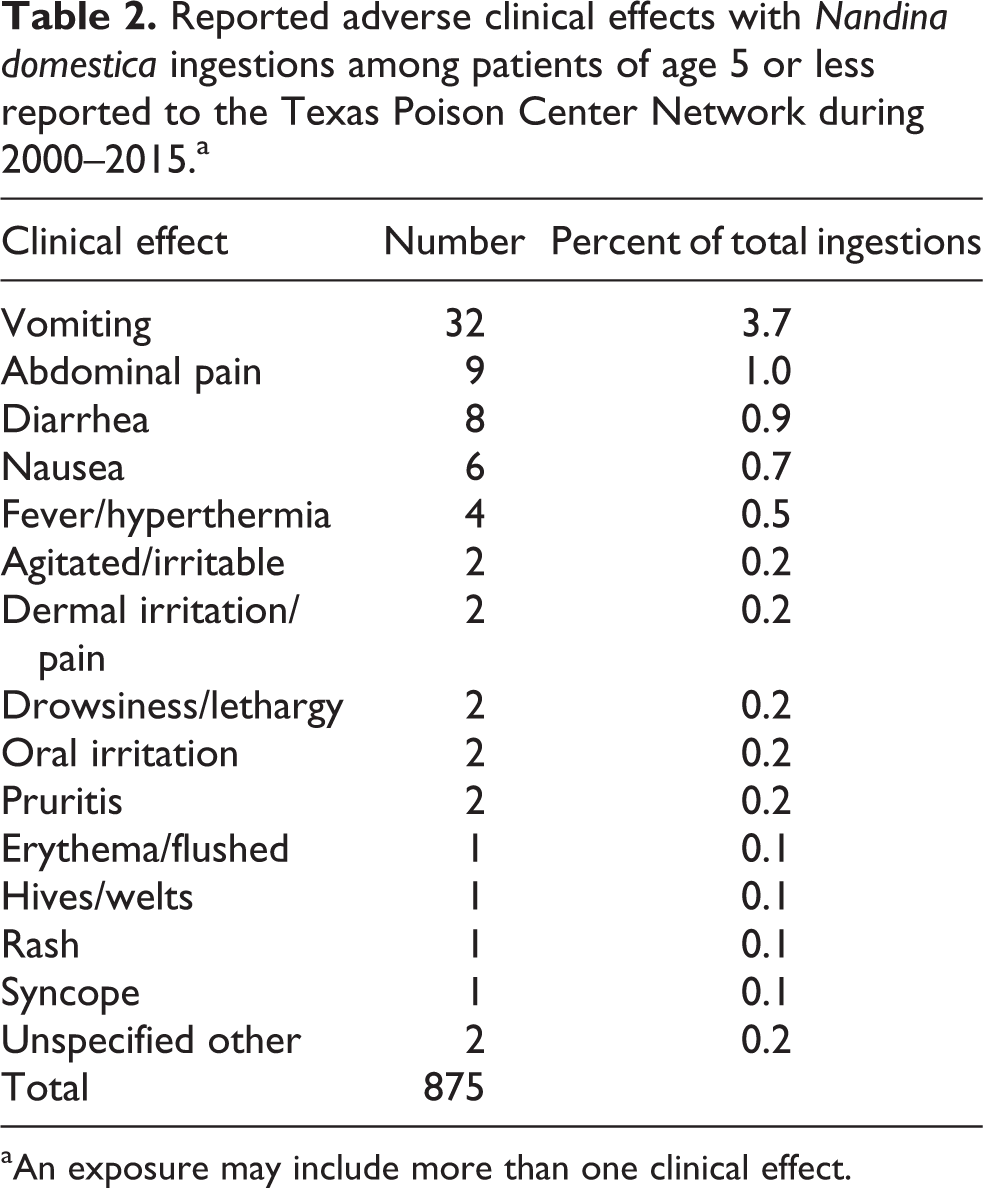
aAn exposure may include more than one clinical effect.
Discussion
This investigation characterized N. domestica ingestions involving patients age 5 years or less reported to Texas poison centers. Parts of the plant, particularly the berries, contain cyanogenic glycosides that convert to hydrogen cyanide when ingested. N. domestica is grown as an ornamental plant but has also been reported as an invasive plant in a number of states. Thus, it is possible that people, including young children, may ingest a potentially toxic plant. However, there is little information on such ingestions in the literature.
There was a strong seasonal trend in the N. domestica ingestions with almost 80% occurring in November to April, peaking in March and April. A Google Trends search of the term “nandina” limited to Texas likewise demonstrated an annual cycle with peaks in searches in March and April. Most of the patients were male and 78% were 1–2 years in age. The age pattern may be due to children of age 1 and 2 becoming more mobile and inquisitive and thus more likely to be able to reach the plant’s attractive bright red berries.
Berries were specifically mentioned in over 80% of the ingestions. It is likely that many of the remaining 20% also involved berries but this information was not included in the record. Of those ingestions where berries were mentioned, a single berry was reported in 57%. This would appear to indicate that when pediatric ingestions of N. domestica berries occur, they are most likely to involve a single berry. However, in almost 30% of these cases, the exact number of berries was not stated, with terms like “some” and “a bunch” being used. It should be noted that the number of berries ingested was based on the caller’s report and not independently verified. It could be that in a number of those ingestions reporting a single berry, more than one berry actually had been ingested.
The N. domestica ingestions predominantly occurred at the child’s own residence. However, TPCN records typically do not provide more detailed information on exactly where such exposures occurred, such as whether it was in a yard, garden, or wooded area. In any case, parents and other childcare providers may want to determine whether they have N. domestica on their property and take measures to prevent young children from ingesting the plant’s berries. Most of the ingestions were reported from those counties where N. domestica was listed as an invasive species by both the USDA (55%) and EDDMaps (84%). That a proportion came from other counties suggests that these maps may be incomplete, although it may also reflect counties where N. domestica is kept as an ornamental plant and has not escaped into the wild. This indicates that healthcare providers and educators should not rely solely on such maps when determining where to provide education and prevention activities.
The majority of patients were managed outside of a healthcare facility with only five patients referred to a healthcare facility by the poison center. This might be expected considering that few of the patients were known or expected to have serious outcomes. In addition, relatively few specific adverse clinical effects were reported among the pediatric N. domestica ingestions. Of those that were reported, the most common were gastrointestinal (vomiting, abdominal pain, diarrhea, and nausea). These and fever are consistent with those reported likely to occur among pets. 9 Furthermore, the most frequently reported treatments (dilution and food/snack) can be administered outside of a healthcare facility setting.
This investigation is subject to limitations. As noted previously, there may be uncertainty as to the number of N. domestica berries that were ingested. Moreover, the identification of the plant involved in the ingestion as N. domestica was usually made by the caller and not independently verified. Some of these ingestions may involve other plant species. Furthermore, reporting of N. domestica ingestions to the TPCN is not mandatory. Those that were reported may not represent all such ingestions that occur in Texas.
In summary, N. domestica ingestions among young children are more likely to occur in November to April, involve 1–2-year-olds, involve the plant’s berries (often just one), and occur at the child’s own residence. Such ingestions generally do not result in serious outcomes and can be managed successfully outside of a healthcare facility.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial, or not for profit sectors.
