Abstract
Objectives:
Oxcarbazepine (OXC) is a 10-keto analogue of carbamazepine used in patients with partial and secondary generalized seizures. We evaluated ingestions of OXC reported to US poison centers for adverse effects from supratherapeutic doses and/or overdose.
Method:
Retrospective analysis of data reported to National Poison Data System from single-substance OXC ingestions between January 2000 and December 2012.
Results:
There were 18,867cases with a mean of 1451 exposures/year. The patients were predominantly adults with 5464 exposures in children <6 years (29%). The most commonly reported clinical effects were drowsiness (n = 4703, 25%), vomiting (n = 1559, 8%), tachycardia (n = 590, 3%), agitated (n = 342, 1.8%), hypotension (n = 178, 0.9%), electrolyte disturbance (n = 153, 0.8%), coma (n = 156, 0.8%), and seizures (n = 121, 0.6%). There were 176 patients with a major effect of which 31 involved were children and 1728 (9%) patients with moderate effects of which 300 involved were children. Five deaths were reported in adults. Intentional exposure (e.g. suicide) was the reason for exposure in 68% of patients with major effects and in all fatalities. Fifty-three percent of adults and 38% of children were managed in a health-care facility (HCF). HCF utilization levels remained consistent.
Discussion:
Severe outcomes appear to be infrequent (<1%). Unlike other anticonvulsants OXC does not appear to be proconvulsant in overdose.
Conclusion:
Serious outcomes for OXC overdoses are unlikely in the pediatric patient. With only mild symptoms likely, observation at home may be appropriate for the majority of cases. In the adult population there appears to be few neurologic and cardiovascular complications even in the intentional exposure.
Introduction
Oxcarbazepine (OXC) is a 10-keto analogue of carbamazepine approved by the US Food and Drug Administration in 2000 as monotherapy or adjunctive therapy in patients with partial and secondary generalized seizures. A major advantage of OXC over carbamazepine is suggested to be tolerability and reduced toxicity. 1 The hypothesis for the reduced toxicity is that OXC, unlike carbamazepine, is metabolized by cystolic ketoreductase to the active monohydroxy metabolite 10-hydroxy-carbazepine (MHC). This provides several advantages over carbamazepine, which is metabolized via cytochrome P-450 3A4 (CYP 3A4), including lack of a toxic epoxide metabolite, lack of auto-induction of CYP 3A4, and an absence of adverse drug reactions with other drugs involved in CYP 3A4.
The metabolite MHC retains the anticonvulsant properties of blocking voltage-gated sodium channels but without many of the toxic properties (i.e. anticholinergic and cardiac sodium channel poisoning) of carbamazepine or the epoxide metabolite of carbamazepine. There are limited published data regarding OXC toxicity in the 15 years since it has been widely available, primarily involving poly-substance ingestions. 2 –7 The few reports available appear to support the assertion that OXC is significantly less toxic than carbamazepine. 2,6,8 It has been theorized that the conversion of the prodrug OXC to the active MHC is a saturable process and this rate-limiting step may be partially responsible for the limited toxicity seen, even after massive overdose.
We analyzed data reported to National Poison Data System (NPDS) of the American Association of Poison Control Centers (AAPCC) on all individuals with ingestions of OXC with the goal of better characterizing adverse effects from supra-therapeutic doses and/or overdoses of OXC.
Method
This study was a retrospective analysis of data reported to NPDS from single-substance OXC ingestions between January 2000 and December 2012. The NPDS collects data in near real time for every call to the nation’s regional poison centers, which serve the entire population of the United States and its territories. Data are recorded at the time of the occurrence of the case by trained specialists (nurses, pharmacists, and physicians) during the routine management of the exposure. Inclusion criteria included single-substance OXC exposure in a human. Exclusion criteria included polysubstance exposure, animal exposure, information calls, and confirmed non-exposures. Data were analyzed using Epi Info and statistics were descriptive based on the retrospective nature of the dataset. Year-to-year means were compared using a student’s t-test, annual trend lines (R 2) for health-care utilization were compared. Data analyzed included demographics (age and gender), clinical effects, reason for exposure, medical outcome, therapies, and health-care facility (HCF) utilization. Medical outcomes and clinical effects were classified by poison center specialists at the time the case was being managed according to standard NPDS definitions. The definitions for medical outcome were the standard definitions used by NPDS as follows: no effect (no signs or symptoms as a result of the exposure), minor effect (signs or symptoms were minimally bothersome and resolved rapidly), moderate effect (signs or symptoms were more pronounced, more prolonged, or more systemic in nature, usually requiring treatment but not life-threatening), major effect (signs or symptoms were life threatening or resulted in significant residual disability), or death (death resulted from the exposure or direct complication of the exposure). Clinical effects may be coded by the poison center specialist as related, unknown if related, and unrelated. For the purposes of this study, only the clinical effects coded as related were included for analysis.
This study was reviewed by the human studies committee Internal Review Board of the authors’ institution and judged to be exempt.
Results
Over the 13-year study period, there were 18,867 cases with a mean of 1451 exposures/year. The patients were predominantly pediatric. There were 7198 (38%) exposures in adults aged >18 years, 6205 (33%) exposures in children aged 6–18 years, and 5464 (29%) exposures in children aged <6 years. There were 10,343 females (55%).
The most commonly reported clinical effects were drowsiness, vomiting, dizziness, tachycardia, and agitation (see Table 1). Serious outcomes were infrequent, even with intentional attempts at self-harm. There were 176 patients with a major effect (0.9% of all cases), of which 31 involved were children <6 years (0.57% of children <6 years), and 1728 patients (9% of all cases) with moderate effects of which 300 involved were children <6 years (5.5% of children <6 years). There were five adult and no pediatric fatalities. In the cases with a major effect, the predominant clinical effects were lethargy (n = 81, 46%), coma (n = 66, 38%), tachycardia (n = 44, 25%) and seizures (n = 43, 24%), respiratory depression (n = 27, 15%), agitation (n = 20, 11%), confusion (n = 16, 9%), and hypertension (n = 11, 6%). In a smaller number of cases with a major effect hypotension (n = 18, 10%) and bradycardia (n = 11, 6%) were reported. Therapeutic interventions were needed infrequently (<2% of patients) and were primarily supportive in nature, with occasional use of sedation and anticonvulsants (see Table 2).
Clinical effect reported.

Therapies administered.

The most common reasons for exposure were therapeutic error (n = 7679, 41%), suicide (n = 5142, 27%), and unintentional general (n = 4029, 21%). Intentional exposure (e.g. suicide) was the reason for exposure in 68% of patients with a major effect and in all fatalities. This is likely due to the larger doses generally ingested with intentional exposures.
A mean of 53% of adults and 38% of children <6 years were managed in an HCF (see Figures 1 and 2). HCF utilization levels remained consistent over the 13-year period for children <6 years old and for adults aged >18 years. There was a nonsignificant decrease in HCF utilization of the total OXC exposure group from 56% to 49%, primarily due to the children
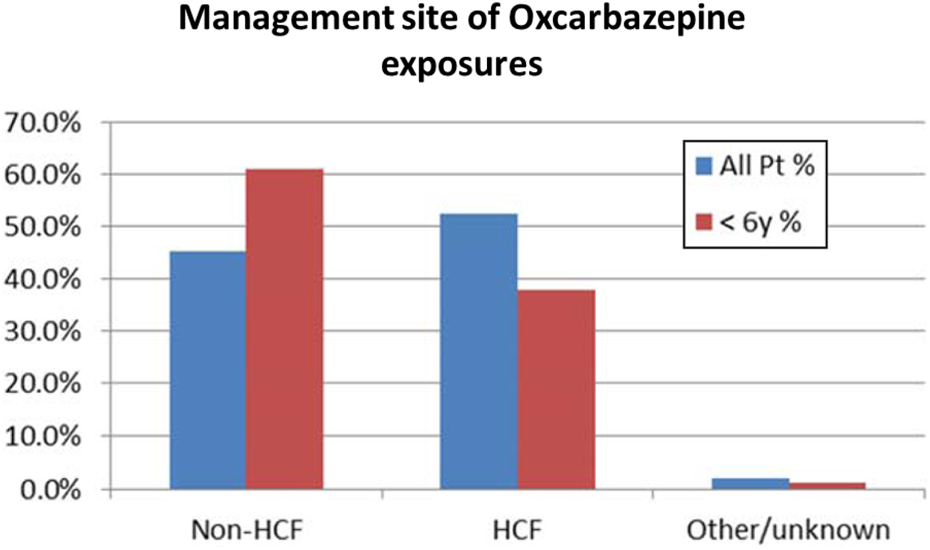
Patient management site by age group.

Patient management site of OXC exposure by year. OXC: oxcarbazepine.
Discussion
This is the first large case series of OXC exposures comprising data from 13 years. Severe outcomes appear to be infrequent (<1%). This is consistent with previous individual reports and a small case series of 55 patients and is distinctly different than outcomes after overdose of carbamazepine. 2,3,9 –11 OXC is a prodrug that requires in vivo conversion to the active monohydroxy metabolite MHC. It had been suggested that the rate-limiting step for the formation of the active metabolite may contribute to the relatively low toxicity in overdose. 1 Severe outcomes were infrequent in young children, which were nearly a third of the patients in the study. This may have been due to the small doses often ingested in exploratory behavior. Additionally, it may be due to the increased clearance of the monohydroxy metabolite in young children compared with older children or adults. 12 Younger children may need a higher milligram per kilogram dose to achieve equivalent serum concentrations of the active monohydroxy metabolite.
Several other features of interest were noted. The common clinical effects were mild central nervous system changes (e.g. drowsiness, agitation, and confusion) and mild gastrointestinal effects such as vomiting. Cardiovascular effects were infrequent with the most common being tachycardia and hypotension. Effects requiring clinical intervention were rare. Unlike a number of other anticonvulsants (e.g. carbamazepine or tiagabine) OXC does not appear to be significantly proconvulsant in overdose. 13 –15 This finding supports the suggestion that the monohydroxy metabolite MHC lacks much of the toxicity of carbamazepine and its epoxide metabolite. Electrolyte abnormalities were reported in 0.2% of patients <6-years old and 0.8% in the total group. The NPDS database does not allow us to specifically separate hyponatremia from other electrolyte abnormalities. Due to the previously reported hyponatremia from OXC, we believe these electrolyte abnormalities are likely hyponatremia. OXC can have an antidiuretic hormone-like effect, causing water retention and dilutional hyponatremia. 16 A second limitation on the reporting of electrolyte abnormalities is that it is unknown how many of 52% of our patients seen in an HCF had testing to look for hyponatremia.
Finally, HCF utilization appeared to remain consistently high despite predominantly benign outcomes. This is especially true in young children where <6% had a moderate or major outcome but nearly 40% were seen in an HCF.
There are a number of limitations of this study, including the inherent limitations of retrospective studies and the voluntary nature of reporting OXC exposures to a poison center. Another limitation is that there are no serum MHC concentrations in our study to verify that the ingestion occurred, especially in the large number of asymptomatic patients. NPDS does not record serum concentrations.
AAPCC (http://www.aapcc.org/) maintains the national database of information logged by the country’s regional poison centers. Case records in this database are from self-reported calls: they reflect only information provided when the public or health-care professionals report an actual or potential exposure to a substance (e.g. an ingestion, inhalation, or topical exposure, etc.), or request information/educational materials. Exposures do not necessarily represent a poisoning or overdose. The AAPCC is not able to completely verify the accuracy of every report made to member centers. Additional exposures may go unreported to poison centers, and data referenced from the AAPCC should not be construed to represent the complete incidence of national exposures to any substance(s).
Conclusion
Thirteen years of data and more than 18,000 patients reported to NPDS suggest serious outcomes in the pediatric patient after OXC ingestions are uncommon. With only mild symptoms likely, observation at home in consultation with a poison center may be appropriate for the majority of cases. In the adult population, there appears to be few neurologic and cardiovascular complications even in the intentional exposure. In the severe cases, therapeutic interventions were primarily supportive in nature with infrequent use of sedation and anticonvulsants.
Footnotes
Authors’ Note
This study was presented at the North American Congress of Clinical Toxicology in New Orleans, Louisiana, October 17–21, 2014.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
