Abstract
The performances of acute physiology and chronic health evaluation (APACHE) II and simplified acute physiology score (SAPS) II have previously been evaluated in acute organophosphate poisoning. We aimed to compare the performance of the SAPS III with those of the APACHE II and SAPS II, as well as to identify the best tool for predicting case fatality using the standardized mortality ratios (SMRs) in acute organophosphate poisoning. A retrospective analysis of organophosphate poisoning was conducted. The APACHE II, SAPS II, and SAPS III were calculated within 24 h of admission. Discrimination was evaluated by calculating the area under the receiver operating characteristic curve (AUROC). The SMRs were calculated as 95% confidence intervals (CIs). In total, 100 cases of organophosphate poisoning were included. The in-hospital case fatality was 19%. The median scores of the APACHE II, SAPS II, and SAPS III were 20.0 (10.0–27.0), 41.0 (28.0–54.8), and 53.0 (36.3–68.8), respectively. The AUROCs were not significantly different among the APACHE II (0.815; 95% CI, 0.712–0.919), SAPS II (0.820; 95% CI, 0.719–0.912), and SAPS III (0.850; 95% CI, 0.763–0.936). Based on these scores and in-hospital case fatality, the SMRs for the APACHE II, SAPS II, and SAPS III were 1.01 (95% CI, 0.50–2.72), 1.01 (95% CI, 0.54 -2.78), and 0.98 (95% CI, 0.33–1.99), respectively. The SAPS III provided a good discrimination and satisfactory calibration in acute organophosphate poisoning. It was therefore a useful tool in predicting case fatality in acute organophosphate poisoning, similar to the APACHE II and SAPS II.
Introduction
Three million people suffer from acute organophosphate (OP) poisoning every year, and 250,000 of these die, particularly in Asian countries. 1 –3 The common cause of death in acute OP poisoning is respiratory failure caused by respiratory muscle paralysis. Furthermore, even though there are several therapeutic strategies for acute OP poisoning, the associated case fatality remains high. 4 Hence, both case fatality prediction and proper management in high-risk groups are important in acute OP poisoning.
In this regard, several scoring systems have been developed to predict case fatality in critically ill patients. Of these, the acute physiology and chronic health evaluation (APACHE) II, the simplified acute physiology score (SAPS) II, and the SAPS III are commonly used to predict case fatality in various diseases. 5 –7 More specifically, several studies have assessed case fatality prediction in acute OP poisoning using these scoring systems. 8 –12 For instance, one previous study showed that the APACHE II and SAPS II were useful in predicting case fatality in acute OP poisoning. 8 Another study showed that the sequential organ failure assessment, APACHE II, and SAPS II were associated with in-hospital case fatality and evaluated the performance of the scoring systems in OP. The study then demonstrated that the SAPS II predicted case fatality poorly when the calculation was based on the standardized mortality rate (SMR). 9 To supplement the weak points of the SAPS II, the physiological data of the SAPS III include additional preadmission data, as well as information regarding the circumstances in 1 h before and after the patient has been admitted to the intensive care unit (ICU). Several recent studies have shown the usefulness of the SAPS III in the ICU setting. 7,13 –15 However, to the best of our knowledge, no studies have evaluated the usefulness of the SAPS III in acute OP poisoning.
The present study aimed to analyze and compare the performances of the APACHE II, SAPS II, and SAPS III in acute OP poisoning. Additionally, using the ratios of observed-to-expected deaths (SMR), we tried to identify the best tool for predicting case fatality in acute OP poisoning.
Materials and methods
Study design
We performed a retrospective observational cohort study involving patients who had been treated for acute OP poisoning at Chonnam National University Hospital (CNUH) between January 2011 and December 2015. During the study period, CNUH was a referral center for OP poisoning patients in Gwangju and a large rural area of Chonnam province. The study design was approved by the Institutional Review Board of Chonnam National University Hospital in Gwangju, Republic of Korea.
Subjects
Patients over 18 years of age who presented at our emergency department (ED) after OP ingestion were included in this study. The diagnosis of acute OP poisoning was based on (1) confirmation of OP ingestion by the patients or a witness, (2) typical clinical features, and (3) decreased butyrylcholinesterase (BChE) activity. The exclusion criteria were (1) uncertainty regarding exposure, (2) age under 18 years, (3) concomitant drug exposure, (4) transfer to another hospital before detoxification, (5) OP poisoning in a manner other than oral ingestion, and (6) missing data.
Patients with acute OP poisoning were treated using general supportive care; this included treatment using atropine and pralidoxime, which were administered according to our hospital’s protocols. 16 If acute shortness of breath was detected, mechanical ventilation was provided at the clinician’s discretion.
Data collection
The following variables were obtained for each patient: age, gender, type of OP ingested, amount of OP ingested, World Health Organization (WHO) Classification of OP, time interval from ingestion to arrival at the first hospital, time interval from ingestion to arrival at our ED, total amount of atropine and pralidoxime administered during hospitalization, vital signs upon admission (systolic and mean arterial blood pressure (in millimeter of mercury), heart rate, respiratory rate, and body temperature), initial Glasgow Coma Scale (GCS), FiO2 (worst within 24 h of admission), laboratory data obtained within 24 h of admission (pH, PaO2, PaCO2, HCO3 −, white blood cell count, hematocrit, platelet count, sodium, potassium, creatinine, albumin, glucose, bilirubin, lactate, ammonia, red blood cell acetylcholinesterase (RBC AChE), and BChE activities), APACHE II score, SAPS II score, SAPS III score, length of stay, expected hospital case fatality, and discharge status (discharge home, transfer to another hospital, and in-hospital case fatality). The time interval from ingestion to arrival at the first hospital was estimated based on history obtained from patients or attendants. Amount of OP ingested was estimated based on history obtained from the patient or was calculated based on the amount of OP remaining in the bottle. The APACHE II, SAPS II, and SAPS III were calculated on the basis of the data collected within either the first hour or the first 24 h of admission. 5,6,17 The expected hospital case fatality was calculated according to the APACHE II, SAPS II, and SAPS III, as in previous studies. 5,6,17
Statistical analysis
Continuous variables are given as either mean ± standard deviation or median values with interquartile ranges, depending on the result of Shapiro–Wilk test. Categorical variables are presented as frequencies and percentages. Differences between the two groups were tested using the independent t-test or the Mann–Whitney U test for continuous variables. Fisher’s exact test or the χ 2 test was used to compare categorical variables, as appropriate. We used the area under the receiver operating characteristic curve (AUROC) to measure the three models’ discrimination of in-hospital case fatality. Calibration was assessed using Hosmer–Lemeshow goodness-of-fit C-statistics, with a p-value greater than 0.05 indicating good calibration. 18 The Brier score and SMR were also calculated. Data were analyzed using PASW/SPSS™ software, version 18 (IBM Inc., Chicago, Illinois, USA) and STATA software, version 14 (Stata Corporation, College Station, Texas, USA). A two-sided significance level of 0.05 indicated statistical significance.
Results
Study population
In total, 114 patients with OP poisoning were identified during the study period. All the patients intentionally ingested OP in a suicide attempt. In three patients, there was uncertainty regarding exposure; five patients had concomitantly ingested another drug, and six were transferred to another hospital before detoxification. Ultimately, 100 patients were included in this study.
Association between baseline or clinical characteristics and in-hospital mortality
Table 1 shows the baseline characteristics of the patients. There were 71 men (71%), and the median age was 61.0 (52.0–72.8) years. The median time from ingestion to the patients’ first hospital visit was 60.0 (30.0–100.0) min, and the median time from ingestion to the patients’ arrival at our hospital was 180.0 (120.0–292.5) min. The median amount of OP ingested was 150.0 (100.0–237.5) ml. The initial GCS score was 9.0 (3.0–15.0), and gastric lavage was performed on 88 patients (88%). The patients in the survivor group were younger, had ingested less OP, and were more likely to have a good GCS score. In addition, these patients had arrived at the first hospital significantly earlier than those in the nonsurvivor group (Table 1). The distribution of ingested OP according to WHO hazard classification was similar between survivors and nonsurvivors (p = 0.278). The in-hospital case fatality was 19% (19 patients); nine patients died within 2 days of ingestion. Most survivors (n = 79) had a complete recovery and were discharged home; two survivors were transferred to another hospital.
Baseline characteristics stratified according to survival discharge.

ED: emergency department; GCS: Glasgow Coma Scale; WHO: World Health Organization.
aTime to first hospital arrival: time interval from ingestion to arrival at the first hospital.
bTime to our ED: time interval from ingestion to arrival at our ED.
Table 2 shows the clinical characteristics of the patients. The patients in the survivor group had higher levels of basal excess and bicarbonate and lower levels of lactate, ammonia, blood urea nitrogen (BUN), and creatinine. They required higher doses of atropine and pralidoxime. Over 90% of patients in the survivor group received gastric lavage (gastric lavage was performed within 1 h of ingestion in 51 (63.0%) patients), while about 73% of patients in nonsurvivor group received gastric lavage (gastric lavage was performed within 1 h of ingestion in 3 (15.8%) patients). The scores of the APACHE II (18.0 (8.0–26.0) vs. 30.0 (21.0–36.0); p < 0.001), SAPS II (35.0 (25.0–51.0) vs. 63.0 (44.0–75.0); p < 0.001), and SAPS III (48.0 (34.0–62.5) vs. 74.0 (64.0–89.0); p < 0.001) were significantly lower in the survivor group.
Clinical characteristics stratified according to survival discharge.

BUN: blood urea nitrogen; BChE: butyrylcholinesterase; RBC AChE: red blood cell acetylcholinesterase; APACHE: acute physiology and chronic health evaluation; SAPS: simplified acute physiology score.
aBChE and RBC AChE: The normal ranges of BChE and RBC AChE activity were 4260–11,250 U/l and 11,188–16,698 U/l, respectively.
bNumber for analysis.
Performance of physiologic scoring systems
To evaluate the predictive value of the scoring systems, the AUROC was calculated. The AUROC was not significantly different among the APACHE II (0.815; 95% confidence interval (CI), 0.712–0.919), SAPS II (0.820; 95% CI, 0.719–0.912), and SAPS III (0.850; 95% CI, 0.763–0.936 (Figure 1)). These scoring systems showed good calibration based on the Hosmer–Lemeshow goodness-of-fit C-test results: χ2 = 6.755, p = 0.563 in the APACHE II; χ2 = 10.409, p = 0.237 in the SAPS II; χ2 = 4.575, p = 0.802 in the SAPS III (Table 3 and Figure 2). The median scores of the APACHE II, SAPS II, and SAPS III were 20.0 (10.0–27.0), 41.0 (28.0–54.8), and 53.0 (36.3–68.8), respectively. Based on these scores and on in-hospital case fatality, the SMRs for the APACHE II, SAPS II, and SAPS III were 1.01 (95% CI, 0.50–2.72), 1.01 (95% CI, 0.54–2.78), and 0.98 (95% CI, 0.33–1.99), respectively.

Receiver operating characteristic analyses of clinical scoring tools: The APACHE II, SAPS II, and SAPS III had no significant differences in performance. APACHE: acute physiology and chronic health evaluation; SAPS: simplified acute physiology score.
Area under the receiver operating characteristic curve, Hosmer–Lemeshow goodness-of-fit test, and standardized mortality ratios for the APACHE II, SAPS II, and SAPS III prognostic models in acute organophosphate poisoning.
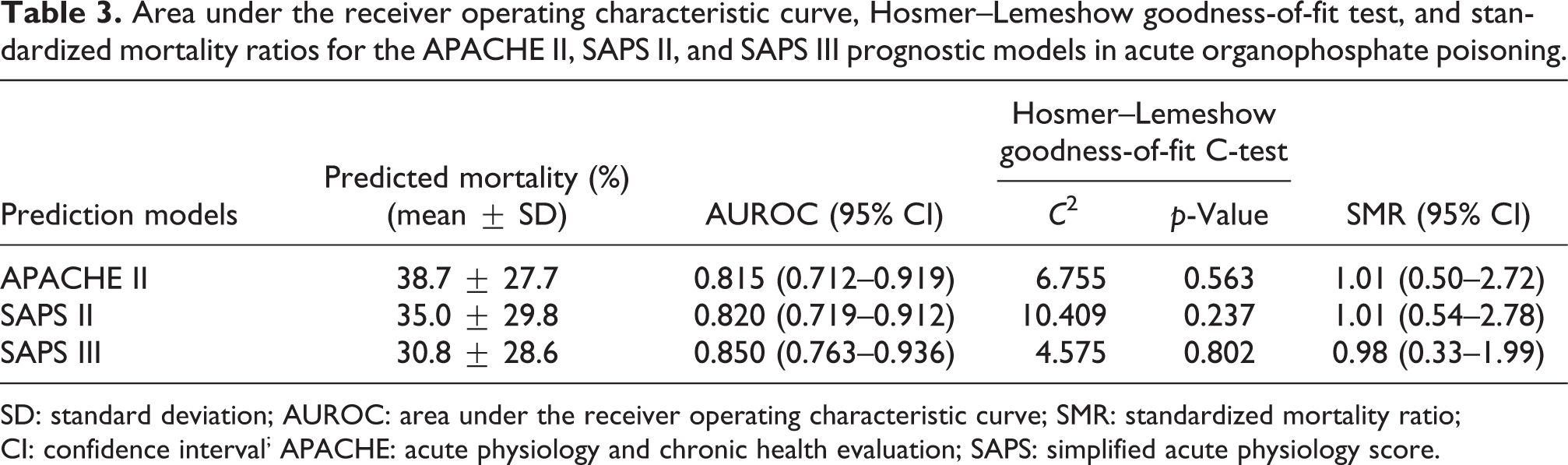
SD: standard deviation; AUROC: area under the receiver operating characteristic curve; SMR: standardized mortality ratio; CI: confidence interval; APACHE: acute physiology and chronic health evaluation; SAPS: simplified acute physiology score.

Calibration curves for the APACHE II, SAPS II, and SAPS III corresponding to number of patients per decile. APACHE: acute physiology and chronic health evaluation; SAPS: simplified acute physiology score.
Discussion
We found that the APACHE II, SAPS II, and SAPS III were associated with in-hospital case fatality in acute OP poisoning. Furthermore, all three scoring systems demonstrated acceptable discriminative performance. The APACHE II and SAPS II yielded SMRs over 1; that is, they underestimated case fatality. Meanwhile, the SAPS III overestimated case fatality—it yielded an SMR below 1. Nonetheless, the three scoring systems showed similar performances, insofar as their 95% CIs contained 1.
Scoring systems of this kind are used to predict case fatality in critically ill patients. 5,6,17,19 Furthermore, several previous studies have shown that such systems are useful in acute OP poisoning. 8 –10 One study involving 396 patients with OP poisoning showed that the APACHE II and SAPS II had AUROC values of 0.77; the study concluded that both scoring systems were useful in predicting case fatality in acute OP poisoning. 8 In other studies, the AUROC of the APACHE II ranged from 0.72 to 0.93, and that of the SAPS II ranged from 0.85 to 0.89. 9,10,12 In the present study, the APACHE II and SAPS II had 0.815 and 0.820 of AUROC values, respectively. Although the AUROC of the SAPS III was not significantly different from those of the APACHE II and SAPS II, the value was high (0.850). Hence, the SAPS III had good discrimination power—similar to the APACHE II and SAPS II.
In 1985, the APACHE II was developed by modifying the original APACHE; the score is calculated on the basis of 12 physiological variables. 2 However, the APACHE II is unreliable in cases of complex disease. Moreover, in a previous study, the APACHE II underestimated the case fatality in critically ill patients, 14,20 and it was created using the database from ICUs within the United States, so it may not be relevant to other countries.
Since the SAPS II, which includes 17 variables, was developed in 1993, it has been useful in predicting outcomes in critically ill patients. 6,19 In several studies, the system has been useful in predicting case fatality in cases of acute OP poisoning. 9 –12 However, several studies have shown—on the basis of the SMR—that the SAPS II underestimates the case fatality in acute OP poisoning. 9,12 In another study, the observed case fatality was 33.3%, while the predicted case fatality based on the SAPS II score was only 1.1%. 11 In the present study, based on the SMRs, the APACHE II and SAPS II underestimated case fatality, while the SAPS III overestimated it. Many authors have indicated that, no matter how good the calibration, underestimation by SMR-based prediction renders the system meaningless. 13,14,21 For example, Glance et al. insisted that if the SMR was statistically more than 1, the performance of the scoring system was low. 22 In the present study, the SAPS III overestimated case fatality. We propose that this prognostic discrepancy in SMR between the SAPS III and the APACHE II or SAPS II stems from differences among the scoring systems in terms of the variables used. Specifically, the SAPS III considers whether ICU admission was planned or unplanned, as well as the reason for ICU admission. 7,19 Additionally, the SAPS III gives more weight to preadmission data and admission circumstances, while the SAPS II emphasizes the physiological score. In other words, the SAPS III is based on severity at the time of admission; therefore, it probably reflects acute or unexpected severity. Indeed, several studies have revealed that the SAPS III reliably reflects acute or unexpected severity, such as trauma or recent surgery. 23,24 Most hospitalized patients with acute OP poisoning are admitted to ICU on an unplanned basis, since acute OP poisoning produces—in addition to respiratory failure—acid-base disturbance, myocardial injury, and altered mental state. 25 –27 Hence, the SAPS III reflects acute diagnosis and origin-of-outcome more reliably than the APACHE II and SAPS II in critically ill patients. It was probably for this reason that the SMR value of the SAPS III led to an overestimation of case fatality. Since the SAPS III overestimates mortality, it may be more effective in alerting physicians to closely monitor the cases likely to be lethal than the APACHE II or SAPS II, which underestimate mortality. Thus, applying the SAPS III may reduce case fatality in acute OP poisoning.
In acute OP poisoning, acute cholinergic crisis begins within a few minutes to several hours of exposure. 28 In acute cholinergic crisis, acute OP toxicity affects various systems, including the central nervous, cardiovascular, respiratory, gastrointestinal, and musculoskeletal systems; it can also cause cardiac arrest. 4 It takes at least 24 h to calculate the APACHE II and SAPS II scores, whereas the SAPS III score can be calculated within 1 h of admission. Interestingly, in the present study, nine patients died within 2 days. It is unclear, then, whether the APACHE II and SAPS II scores improve case fatality in OP poisoning. APACHE II and SAPS II scores require measurements over the course of the first 24 h of an ICU stay; therefore, they cannot be used for early screening. 5,6 However, patients evaluated using the SAPS III, which can be applied within 1 h of admission, can be quickly classified into high-risk groups; these groups can then be provided with intensive treatment during the critical period. 17
Patients with acute OP poisoning commonly require endotracheal intubation and mechanical ventilation to prevent or treat respiratory failure. Then, it is difficult to evaluate the GCS exactly. However, the GCS accounts for 16% of the total points in the APACHE II and SAPS II. 5,6 APACHE II and SAPS II give high weight to neurological points; they can underestimate case fatality. Loh et al. showed that the APACHE II underestimates case fatality—both in patients with vascular trauma and in those with nonvascular trauma. 29 On the other hand, the SAPS III gives less weight to neurological points, and the GCS accounts for 8% of the total points. 7 In this sense, the SAPS III estimates the severity of illness more accurately than the APACHE II and SAPS II.
Limitations
The present study had several limitations. First, it was retrospective and single-centered. Therefore, to assess generalizability and causation, further studies will be needed that include larger sample sizes, multiple centers, and a prospective design. Second, we could not completely correct selection bias. Because our study was conducted at a tertiary care hospital, then patients were mostly transferred and had more severe symptoms. Third, six patients were excluded because they were transferred to another hospital before detoxification, and two patients included in the present study were later transferred to another hospital. Therefore, we were not able to ascertain the overall case fatality rate due to OP poisoning, which could affect the SMR. However, we believe that the effect on SMR was minimal since most survivors were discharged following a complete recovery. Fourth, a number of patients suffered respiratory distress as a result of intubation and assisted ventilation, so it was difficult to measure the exact GCS score. This may have influenced the APACHE II, SAPS II, and SAPS III scores. Fifth, there were no significant differences between cases according to the WHO classification of OP poisoning in the present study. However, we could not differentiate toxicities according to individual OP compounds. Finally, potential confounders that were not evaluated in this study could affect the case fatality due to OP poisoning. Age, time to first hospital arrival, amount of OP ingested, and timing of gastric lavage were significantly different between survivors and nonsurvivors in the present study. Although these factors are not taken into consideration in the physiologic score, we believe that these factors would eventually affect physiologic score.
Conclusion
The SAPS III showed good discrimination and satisfactory calibration performance in acute OP poisoning. It is thus a useful tool in predicting case fatality in acute OP poisoning—similar to the APACHE II and SAPS II.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
