Abstract
This study was planned to determine arsenic (As) (10 mg/kg body weight given through oral gavage) induced behavioral and cholinergic perturbations in three different age groups of rats; young (postnatal day 21), adult (3 months), and aged (18 months) at 7 days post-acute exposure (n = 6 for each of the four groups of all three age points). Further, we also evaluated the ameliorative effect of essential metal zinc (Zn; 0.02% through drinking water) and an antioxidant, α-tocopherol (vitamin E; 125 mg/kg body weight through oral gavage) against As-induced neurotoxicity. As exposure showed significant alterations in behavioral functions (open-field behavior, total locomotor activity, grip strength, exploratory behavior, and water maze learning). Cholinergic studies in three brain regions (cerebral cortex, cerebellum, and hippocampus) of different age groups also showed significant increase in acetylcholine levels and a decrease in acetylcholinesterase activity. These effects were more pronounced in hippocampus followed by cerebral cortex and cerebellum. Among the three different age points, aged animals were found to be more vulnerable to the As-induced toxicity as compared to young and adult animals suggesting that As neurotoxicity is age dependent. These As-induced alterations were significantly reversed following supplementation with Zn or vitamin E. However, vitamin E was found to elicit greater protection as compared to Zn in restoring the altered behavioral and cholinergic perturbations, providing evidence for As-induced oxidative damage.
Introduction
Arsenic (As), a naturally occurring metalloid, is widely distributed in the environment through natural, geochemical, and anthropogenic sources, and its contamination is associated with the global health concern which affects millions of people across the world. 1,2 Exposure to As at high levels can result in mild but persistent peripheral neuropathy, 3,4 sensory deficits, 5,6 and impairment of higher neurological function. 7 In rodents, As exposure is associated with altered neurotransmitter levels and operant learning deficits 8,9 as well as lowered levels of spontaneous activity. 10 –12 Neurochemical alterations involving multiple neurotransmitter systems have been reported following As exposure in rats. 9,13,14 Since As exposure has been suggested to be a risk factor for Alzheimer’s disease, 15 –17 much attention has been given to understand its impact on catecholaminergic and cholinergic mechanisms. It is generally believed that adults are more resistant than young and aged to the toxic effects of As. However, whether such resistance is there across the life span or not is yet to be clearly defined. Thus, age-related toxicity of As needs to be characterized as in the case with other metals.
Despite the well-established toxicity, yet no safe and effective therapeutic management of arsenicosis is available. Essential trace metals such as zinc (Zn) have been studied for their protective values against As. Earlier reports from our laboratory have established the protective role of essential metals in reducing the impact of Pb- and As-induced developmental neurotoxicity. 18 –20 Essential metals such as Zn, Ca, and so on can affect the toxicity of metals by interacting with them at several points in the body.
Alpha (α) tocopherol is the most abundant and active form of vitamin E in vivo 21,22 and is an important lipophilic radical scavenging antioxidant. α-Tocopherol is a term that encompasses a group of potent, lipid soluble, chain-breaking antioxidants that prevents the propagation of free radical reactions. A number of studies have been carried out to determine the protective effects of vitamin E in different biological models of injury. 23,24 Since cholinergic system plays a vital role in the regulation of cognitive process and motor functions, 25 the evaluation of the alterations induced by As to this system was considered to be the first neurochemical parameter to be investigated during As exposure along with behavioral alterations.
We hypothesized that As-induced neurobehavioral alterations are age dependent and driven by oxidative damage. Therefore, antioxidants and essential metals will provide protection against As-induced neurotoxicity. In view of this, the present study was designed to study the impact of age on As-induced alterations in behavior and cholinergic system in albino rats at three different age points (postnatal day (PND) 21: young; 3 months: adult; 18 months: aged) and to confirm the presence of an association between the cholinergic alterations and behavioral perturbations. Further, the present study was extended to compare the ameliorative effect of essential metal Zn and an antioxidant vitamin E against As-induced behavioral and cholinergic perturbations.
Materials and methods
Procurement and maintenance of animals
Three months (adult) and 18 months (aged) old male albino rats (Wistar) were purchased from Sri Venkateswara Traders (Bangalore, India), whereas PND21 (young) rats were obtained from our animal house by maintaining pregnant rats. Animals of all age groups were maintained in the animal house of Sri Venkateswara University, Department of Zoology, Tirupati for at least 1 week for acclimatization before using for treatments. All the rats were kept in well cleaned, sterilized polypropylene cages lined with paddy husk (18″ ×10″ × 8″). The animals were maintained under a regulated light: dark 12 h (7:00–19:00) scheduled at 24 ± 1°C and relative humidity of 55 ± 15%. Rats were provided standard rat chow (Sai Durga Feeds and Foods, Bangalore, India) and water ad libitum. The experiments were carried out in accordance with the guidelines of the Committee for the Purpose of Control and Supervision on Experiments on Animals, Government of India (CPCSEA, 2003) and approved by the Institutional Animal Ethical Committee (resolution no. 09/2012-2013/i/a/CPCSE/IAEC/SVU/GRR-MRKdt.02-07-2012) at Sri Venkateswara University, Tirupati, India.
Chemicals
The chemicals used in this study were purchased from Sigma Chemicals (St. Louis, Missouri, USA) and Merck, Mumbai, Maharashtra, India. Sodium arsenite (NaAsO2, >99% purity) used in this study was purchased from Sigma-Aldrich (St. Louis, Missouri, USA) and dissolved in sterile distilled water to the desired concentrations.
Animal exposure
After a week of acclimatization, rats of all age groups (PND21: young; 3 months: adult; 18 months: aged) were randomly divided into four groups and were treated for a period of 1 week as follows:
Group II: Arsenic as sodium arsenite (10 mg/kg dissolved in deionized water, given orally through gavage) (n = 6).
Group III: As (10 mg/kg, orally) + zinc as ZnCl2 (0.02% through drinking deionized water) (n = 6).
Group IV: As (10 mg/kg, orally) + vitamin E (125 mg/kg body weight, orally through gavage) (n = 6).
The control animals which served as group I, received equivalent volumes of deionized water through oral gavage. The dose of sodium arsenic used in this study was based upon the reports of Itoh et al. 26 and Rodriguez et al. 12 in rats, and it represents one-fourth of the LD50 dose for rats. 27 Sodium arsenite solutions were prepared fresh daily and dissolved in deionized water. The vitamin E dose used in this study has no untoward effects and is considered a supplemental dose. 28 After completion of treatment, the open-field, locomotor, exploratory, grip strength, and water maze studies were performed continuously for a period of 4 days, and then, the animals were sacrificed through cervical dislocation, and the brain regions (cerebral cortex, hippocampus, and cerebellum) were quickly isolated and stored at −80°C for biochemical analysis. Synaptosomal fractions of the tissue samples were used to study the perturbations in cholinergic system.
Behavioral studies
Locomotor activity
Total locomotor activity of the rat was studied with Opto-Varimex mini (Columbus Instruments, Columbus, Ohio, USA). The apparatus consisted of a wooden box (42 × 42 × 30 cm) equipped with 15 infrared emitters (spaced at 2.65 cm intervals) located on the x axis 2–3 cm above the floor of the cage and an equivalent number of receivers located on the opposite walls. Each interruption of a beam generates an electric impulse scored by a digital counter. These data were recorded as total activity for 30 min duration to each animal with five session intervals for four consecutive days in control, As-exposed, As + Zn, and As + vitamin E animals of all the age points.
Open-field behavior
The open-field behavior of rats was assessed in a wooden box measuring 90 cm × 90 cm × 30 cm high. The floor of the arena is divided into 36 equal squares by black lines. Immediately after a rat was placed in the center of the open field, the movement of the animal was scored. The number of squares crossed with all paws (crossings); the standings on the hind legs (rearings); placing the nose against wall or floor (sniffings); wiping, licking, combing, or scratching of any part of the body (groomings) were counted in all sessions. Testing was carried out on four consecutive days in 5-min sessions in control, As-exposed, As + Zn, and As + vitamin E animals of all the age points. The number of crossings indicates the locomotor activity. 29
Exploratory behavior
Exploratory behavior was evaluated in the hole board. The apparatus was an open-field arena with four equally spaced holes (3 cm in diameter) on the floor. Each rat was placed individually in the center of the arena for 5 min, during which we recorded head-dip count and head-dipping duration, in seconds. A head dip was scored if both eyes disappeared into the hole. Head-dipping duration data were expressed as total duration during the 5-min session. The results for head dip were expressed as number of counts, and for head-dipping duration in seconds.
Grip strength
Grip strength was assessed by sensing the peak amount of force applied in grasping a pull bar connected to a force meter (1027MPB Grip Strength Meter, Columbus instruments, Columbus, Ohio, USA) as explained by Giniatullina et al. 30 Rats were allowed to grasp the pull bar five times with front limbs, followed by grasping five times with hind limbs. The median of each five repetitions was taken as grip strength. Testing was carried out on four consecutive days in control, As-exposed, As + Zn, and As + vitamin E animals of all the age points.
Morris water maze
The water maze is a circular water tank measuring 1.85 m in diameter and 0.7 m deep constructed according to a basic design similar to that of Morris. 31 Four points along the circumference of the water tank were designated arbitrarily north (N), south (S), east (E), and west (W), thus dividing the maze into four quadrants. The pool was filled to a depth of 30 cm with water maintained at room temperature and made opaque with white, nontoxic water-based paint. A circular submerged platform (diameter 12.5 cm) remained below the surface of water. Rats received one training block consisting of four trials in a day and were tested on four consecutive days. A trial was started by placing the rat in the pool facing the wall in one of four quadrants delineated by marks at the four cardinal directions. Rats were allowed to swim to the hidden platform and the escape latency (time to find the hidden platform) was recorded. If an animal did not escape within 90 s, it was manually guided to the escape platform by the experimenter. Three different phases including acquisition (stand (platform) placed in north direction), reversal (stand placed in south direction), and working memory (stand placed in all the four directions) were carried out on four consecutive days for control, As-exposed, As + Zn, and As + vitamin E treated rats of adult and aged groups. The average of four consecutive days’ data was considered as mean for each group. Since the young animals (PND21) were unable to swim, this task was not performed with them.
Neurochemical studies
Isolation of synaptosomal fractions
Synaptosomal fractions from brain homogenates were separated using Ficoll-sucrose gradients. 32 The three brain regions (cerebral cortex, cerebellum, and hippocampus) were isolated in cold conditions from the control, As-exposed, As + Zn, and As + vitamin E rats of all age points. The tissues were weighed and homogenized in 10 ml of ice-cold homogenizing buffer and the volume was brought up to 25 ml with homogenizing buffer. The homogenates were centrifuged at 750 × g for 10 min. The pellets were discarded. The supernatants were centrifuged at 17,000 × g for 20 min. The pellets were suspended in 10 ml 0.32 M sucrose and were layered on a two-step discontinuous Ficoll-sucrose gradient consisting 13 and 7.5% Ficoll and centrifuged at 65,000 × g for 45 min. A milky layer was formed at the interface of 13 and 7.5% Ficoll. The milky layer fraction was diluted with 9 volumes of 0.32 M sucrose and centrifuged again at 17,000 × g for 30 min. The supernatant was discarded and the pellet (synaptosomal fraction) was suspended in 0.32 M sucrose.
Estimation of acetylcholine
The acetylcholine (Ach) content was estimated by the method of Metcalf 33 as given by Augustinsson. 34 The synaptosomal fractions of cerebral cortex, cerebellum, and hippocampus were placed in boiling water for 5 min to terminate the acetylcholinesterase (AChE) activity and also to release the bound ACh. To the synaptosomal fractions, 1 ml of alkaline hydroxylamine hydrochloride and 1 ml of 50% hydrochloric acid were added. The contents were mixed thoroughly and centrifuged. To the supernatant, 0.5 ml of 0.37 M ferric chloride solution was added and the brown color developed was read at 540 nm against a reagent blank in a spectrophotometer. The ACh content was expressed as micromoles of ACh per gram of wet weight tissue.
Estimation of AChE activity
AChE activity was determined following the method of Ellman et al. 35 The reaction mixture contained 3.0 ml of phosphate buffer (pH 8.0), 20 µl of 0.075 M acetylthiocholine iodide (substrate), and 100 µl of 0.01 M 5,5-dithiobis-2-nitrobenzoic acid. The reaction was initiated with the addition of 100 µl of synaptosomal fraction. The contents were incubated for 30 min at room temperature and the color absorbance was measured at 412 nm in spectrophotometer (Hitachi, Model U-2000, Tokyo, Japan). The enzyme activity was expressed as micromoles of ACh hydrolyzed per milligram of protein per hour.
Statistical analysis of the data
Significance of each age group among different treatments was analyzed by one-way analysis of variance (ANOVA) followed by Bonferroni multiple comparisons test using Graphpad Prism 3.11 program software to compare the effects among various groups. The 0.05 level of probability was used as the criterion for significance.
Results
Our present findings showed that neurobehavioral functions such as open-field behavior, locomotor activity, exploratory behavior, and grip strength were significantly decreased in young (PND 21), adult (3 months), and aged (18 months) rats after exposure to As as compared to controls. Water maze learning studies were not performed in PND21 (young) rats, as the animals were very small and unable to swim in water maze. Exposure to As lead to a significant increase in escape latency of all the three phases (acquisition, reversal, and working memory) in adult (3 Month) and aged (18 months) rats. Neurochemical studies conducted in three brain regions (cerebral cortex, cerebellum, and hippocampus) at different age points showed significant increase in ACh content, accompanied by a decrease in the AChE enzyme activity levels.
Open-field behavior
Exposure to As altered the open-field behavioral tasks significantly, and these alterations were found to be greater in aged rats (70.83%—crossings; 71.70%—rearings; 68.37%—sniffings; 66.51%—groomings) followed by young rats (64.81%—crossings; 67.79%—rearings; 61.11%—sniffings; 60.89%—groomings) and adult rats (50.54%—crossings; 55.68%—rearings; 48.37%—sniffings; 50.51%—groomings). The observed decrease in open-field tasks of all the age points was significant at p < 0.05 (Figure 1(a) to (c)).

Effect of arsenic on open-field behavior tasks (crossings, rearings, sniffings, and groomings) and reversal responses to administration of Zn and vitamin E in (a) young, (b) adult, and (c) aged rats. Rats of all age groups were given deionized water (control), or arsenic (10 mg/kg orally), zinc (0.02% through drinking water), and vitamin E (125 mg/kg orally) for a period of 1 week. All the values are mean ± SD of six individual observations. The values marked with “a” are significant with respect to control, “b”—with respect to As, and “c”—with respect to As + Zn, whereas nonsignificant values are designated with hash (#) as evaluated by the ANOVA followed by Bonferroni multiple comparisons test (p < 0.05). SD: standard deviation; ANOVA: analysis of variance.
Locomotor activity
In control rats, locomotor activity increased with age (PND21 < 3 months) and decreased in aged rats. Exposure to As exhibited significant decrease in the locomotor activity at all the age points studied and the decrease was more pronounced in aged rats (69.56%) than young rats (63.7%) and adult rats (50.38%). The observed decrease in locomotor activity of all the three age groups of rats was significant at p < 0.05 (Figure 2).

Effect of arsenic on total locomotor activity and reversal responses to administration of Zn and vitamin E in young, adult, and aged rats. Rats of all age groups given with deionized water (control), or arsenic (10 mg/kg orally), zinc (0.02% through drinking water), and vitamin E (125 mg/kg orally) for a period of 1 week. All the values are mean ± SD of six individual observations. The values marked with a are significant with respect to control, b—with respect to As, and c—with respect to As + Zn, whereas nonsignificant values are designated with hash (#) as evaluated by the ANOVA followed by Bonferroni multiple comparisons test (p < 0.05). SD: standard deviation; ANOVA: analysis of variance.
Exploratory behavior
The alterations in exploratory behavior were examined in hole board. A significant decrease in head-dip counts and head-dip duration was observed in the rats of all the age groups exposed to As, and the decrease was found to be greater in aged rats (65.84%—dipping counts; 69.77%—dipping duration) than young rats (56.76%—dipping counts; 63.46%—dipping duration) and adult rats (52.009%—dipping counts; 53.52%—dipping duration). The observed decreases in head-dip counts and head-dip duration of all the age points were significant at p < 0.05 (Figure 3(a) and (b)).

Effect of arsenic on exploratory behavior and reversal responses to administration of Zn and vitamin E in young, adult, and aged rats. (a) Number of head dips and (b) dipping duration. Rats of all age groups were given deionized water (control), or arsenic (10 mg/kg orally), zinc (0.02% through drinking water), and vitamin E (125 mg/kg orally) for a period of 1 week. All the values are mean ± SD of six individual observations. The values marked with a are significant with respect to control, b—with respect to As, and c—with respect to As + Zn, whereas nonsignificant values are designated with hash (#) as evaluated by the ANOVA followed by Bonferroni multiple comparisons test (p < 0.05). SD: standard deviation; ANOVA: analysis of variance.
Grip strength
The average grip strength of forelimb and hind limb increased with age in control and experimental rats. A significant decrease was observed in both forelimb and hind limb grip strength following As exposure at all the age points, and the decrease was more pronounced in aged rats (62.16%—forelimb; 68.36%—hind limb) followed by young rats (56.99%—forelimb; 60.01%—hind limb) and adult rats (43.41%—forelimb; 49.52%—hind limb). The observed decrease in forelimb and hind limb grip strength of all the age points was significant at p < 0.05 (Figure 4(a) and (b)).

Effect of arsenic on grip strength and reversal responses to administration of Zn and vitamin E in young, adult, and aged rats. (a) Forelimb and (b) hind limb. Rats of all age groups were given deionized water (control), or arsenic (10 mg/kg orally), zinc (0.02% through drinking water), and vitamin E (125 mg/kg orally) for a period of 1 week. All the values are mean ± SD of six individual observations. The values marked with a are significant with respect to control, b—with respect to As, and c—with respect to As + Zn, whereas nonsignificant values are designated with hash (#) as evaluated by the ANOVA followed by Bonferroni multiple comparisons test (p < 0.05). SD: standard deviation; ANOVA: analysis of variance.
Water maze
Behavioral assessments on water maze confirmed the impairment in performance of the water maze acquisition, reversal, and working memory tasks in As-exposed animals (Figure 5(a) to (c)). All the rats easily learned to find the submerged platform except for the first day of training when all subjects performed at chance level. Exposure to As leads to a significant increase in escape latency compared with control animals to find the hidden platform. As-exposed rats exhibited the tendency of peripheral swimming, thereby took longer time to reach the platform. The escape latency however greatly reduced in As-exposed rats supplemented with Zn/vitamin E.
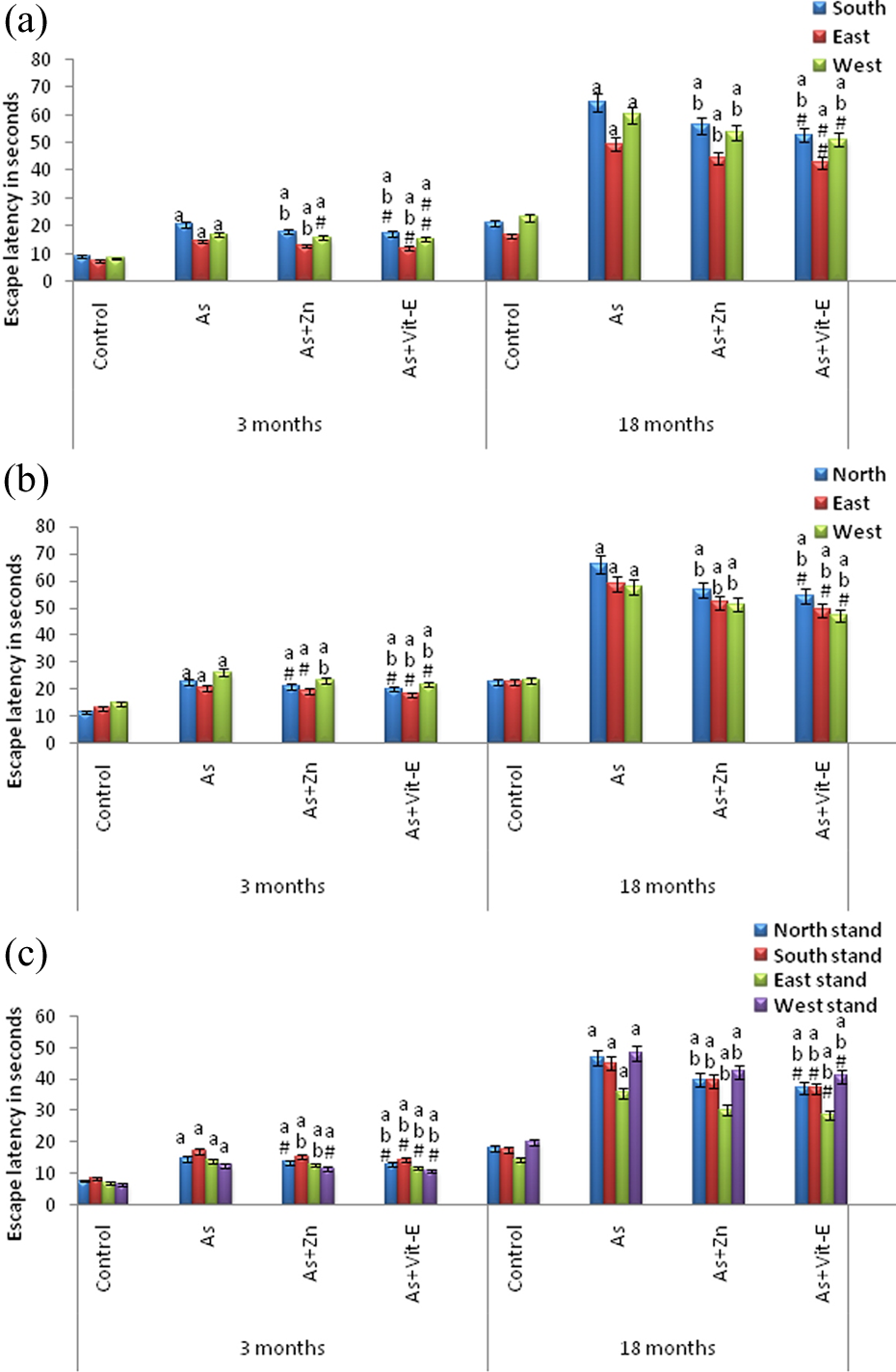
Effect of arsenic on cognitive functions of rats and reversal responses to administration of Zn and vitamin E in young, adult, and aged rats. (a) Water maze acquisition phase, (b) reversal phase, and (c) working memory phase. Rats of all age groups were given deionized water (control), or arsenic (10 mg/kg orally), zinc (0.02% through drinking water), and vitamin E (125 mg/kg orally) for a period of 1 week. All the values are mean ± SD of six individual observations. The values marked with a are significant with respect to control, b—with respect to As, and c—with respect to As + Zn, whereas nonsignificant values are designated with hash (#) as evaluated by the ANOVA followed by Bonferroni multiple comparisons test (p < 0.05). SD: standard deviation; ANOVA: analysis of variance.
From Figure 5(a), it is evident that As exposure significantly impaired the water maze acquisition performance where the submerged platform was placed in north quadrant. Control rats of adult and aged groups quickly proceeded to the north quadrant and identified the hidden platform, whereas As-treated rats took longer time to reach the hidden platform, that is, the escape latency was increased in the animals exposed to As at both the age groups, and the increase was more pronounced in aged rats (205.80%—south; 198.55%—east; 160.68%—west) followed by adult rats (128.69%—south; 94.40%—east; 102.52%—west). The observed increase in escape latency in As-exposed rats at both the ages was significant at p < 0.05 (Figure 5(a)).
During the spatial discrimination of reversal task, the submerged platform was shifted toward south quadrant. All the groups initially spent more time in north quadrant and then reached toward south quadrant. From Figure 5(b), it can be observed that the As-treated rats of both the age groups exhibited an increase in the escape latencies compared to control rats, and the increase was more pronounced in aged rats (189.47%—north; 162.33%—east; 148.66%—west) than the adult rats (97.31%—north; 58.28%—east; 78.82%—west). The observed increases in escape latency in As-exposed rats at both the ages were significant at p < 0.05 (Figure 5(b)).
As the platform was changed to different quadrants in working memory, the escape latencies were longer than acquisition and reversal phases. As in the case of acquisition and reversal phases, As-exposure exhibited impairments in working memory with increased escape latency more in aged rats (156.75%—north stand; 155.36%—south stand; 149.05%—east stand; 138.31%—west stand) compared to adult rats (92.15%—north stand; 97.55%—south stand; 101.70%—east stand; 95.37%—west stand). The observed increases in escape latency in As-exposed rats at both the ages were significant at p < 0.05 (Figure 5(c)).
Supplementation with Zn/vitamin E, significantly reversed the As-induced alterations in behavior (open field, total locomotor activity, exploratory behavior, grip strength, and water maze) of young (PND 21), adult (3 months), and aged (18 months) rats (Figures 1(a) to 5(c)). However, between Zn and vitamin E, vitamin E produced greater recovery against As-induced behavioral perturbations than Zn.
ACh content
In the present investigation, the ACh content was measured in both control and experimental rat brain regions as it is a principal cholinergic neurotransmitter for nervous system function. In control brain regions, the levels of ACh content was found to be more at 3 months (adult) age, followed by PND21 (young) and 18 months (aged) with the hippocampus documenting the highest ACh content followed by cerebral cortex and cerebellum at 3 months age.
Exposure to As exhibited a significant increase in the brain regional ACh levels, and it was found to be greater in aged rats (66.94%—cortex; 63.43%—cerebellum; 68.18%—hippocampus) followed by young rats (60.13%—cortex; 58.90%—cerebellum; 63.70%—hippocampus) and adult rats (46.39%—cortex; 45.73%—cerebellum; 47.82%—hippocampus). From the results, it was found that hippocampus exhibited maximum increase in ACh levels followed by cerebral cortex and cerebellum. The observed increases in ACh levels in different brain regions at all the age points were significant at p < 0.05 (Figure 6(a) to (c)).

Effect of arsenic on acetylcholine content in three different brain regions: (a) cerebral cortex, (b) cerebellum, and (c) hippocampus and its response to administration of Zn and vitamin E in young, adult, and aged rats. Rats of all age groups were given deionized water (control), or arsenic (10 mg/kg orally), zinc (0.02% through drinking water), and vitamin E (125 mg/kg orally) for a period of 1 week. All the values are mean ± SD of six individual observations. The values marked with a are significant with respect to control, b—with respect to As, and c—with respect to As + Zn, whereas nonsignificant values are designated with hash (#) as evaluated by the ANOVA followed by Bonferroni multiple comparisons test (p < 0.05). SD: standard deviation; ANOVA: analysis of variance.
AChE activity
The AChE activity in control rats was increased with age documenting maximum activity in adult rats compared with young rats. However, the activity was decreased in aged rats (18 months). Among the different brain regions studied, the hippocampus showed greater AChE activity followed by cerebral cortex and cerebellum (Figure 7(a) to (c)).

Effect of arsenic on AChE activity in three different brain regions: (a) cerebral cortex, (b) cerebellum, and (c) hippocampus and its response to administration of Zn and vitamin E in young, adult, and aged rats. Rats of all age groups were given deionized water (control), or arsenic (10 mg/kg orally), zinc (0.02% through drinking water), and vitamin E (125 mg/kg orally) for a period of 1 week. All the values are mean ± SD of six individual observations. The values marked with a are significant with respect to control, b—with respect to As, and c—with respect to As + Zn, whereas nonsignificant values are designated with hash (#) as evaluated by the ANOVA followed by Bonferroni multiple comparisons test (p < 0.05). AChE: acetylcholinesterase; SD: standard deviation; ANOVA: analysis of variance.
Exposure to As significantly decreased the specific activities of AChE in all the three brain regions. Decrease in the AChE enzyme activity was more pronounced in aged rats (67.36%—cortex; 64.67%—cerebellum; 68.16%—hippocampus) followed by young (61.77%—cortex; 60.43%—cerebellum; 61.25%—hippocampus) and adult rats (47.72%—cortex; 46.12%—cerebellum; 51.75%—hippocampus) with hippocampus documenting maximum decrease in enzyme activity than cerebral cortex and cerebellum. The observed decrease in AChE activity levels in different brain regions at all the age points was significant at p < 0.05 (Figure 7(a) to (c)).
Tissue as levels
Brain regional accumulation of As at different age points was shown in Figure 8. As exposure led to a significant increase in As content in all the three brain regions at all age points. Maximum increase in As content was observed at PND21 (young) followed by aged and adult rats. Among the brain regions, maximum increase in As levels was found in cerebral cortex compared to cerebellum and hippocampus. The observed increase in As levels in different brain regions at all the age points was significant at p < 0.05 (Figure 8). However, the increase was marginal and nonsignificant in the cerebellum of As + Zn and As + vitamin E groups in 18 months old animals

Arsenic levels in three different brain regions: (a) cerebral cortex, (b) cerebellum, and (c) hippocampus of young, adult, and aged rats. Rats of all age groups were given deionized water (control), or arsenic (10 mg/kg orally), zinc (0.02% through drinking water), and vitamin E (125 mg/kg orally) for a period of 1 week. All the values are mean ± SD of six individual observations. The values marked with a are significant with respect to control, b—with respect to As, and c—with respect to As + Zn, whereas nonsignificant values are designated with hash (#) as evaluated by the ANOVA followed by Bonferroni multiple comparisons test (p < 0.05). SD: standard deviation; ANOVA: analysis of variance.
We also evaluated the protective effect of Zn/vitamin E against As-induced alterations in cholinergic system and brain As levels. Supplementation with Zn/vitamin E significantly reversed the As-induced alterations in cholinergic system (ACh levels and AChE activity) and As levels in all the three brain regions (cerebral cortex, hippocampus, and cerebellum) of young (PND 21), adult (3 months), and aged (18 months) rats (Figures 6(a) to 7(c)). However, among Zn and vitamin E, vitamin E produced greater recovery against As-induced behavioral and neurochemical perturbations compared to Zn.
Discussion
Arsenic is known to cross blood–brain barrier (BBB) and produce neurotoxic effects. 12,36 In the present study, arsenic-exposed rats showed a significant accumulation of arsenic in brain compared to controls leading to the observed neurotoxic and neurobehavior alterations. These increased As levels in rat brain regions exposed to As indicate that As and its metabolites are able to pass through the BBB, which is consistent with the results of Zheng et al. 37
The behavioral data showed that exposure to As at all the age groups significantly decreased the open-field behavior, locomotor activity, grip strength, and exploratory behavior with concomitant increase in the escape latency of all the three phases observed in water maze test. Altered motor coordination and locomotion could arise from As-induced aberrant cholinergic functioning. Apart from the disturbances in cholinergic system, damage to particular structure in the circuit or to connecting pathways in the developing nervous system, which is more susceptible than the adult nervous system, can also produce structural or functional changes that result in behavioral changes. 38,39
The open-field test provides simultaneous measures of both habituation and anxiety. Exposure to As leads to a significant decrease in open-field tasks like crossings, rearings, sniffings, and groomings at all the age points studied. Exposure to As also leads to a significant decrease in the locomotor activity at all the age groups (PND21, 3 months, and 18 months). The changes observed in locomotor behavior could be a result of the general malaise evident in these animals, or alternatively, it could reflect the deleterious effects of As on metabolism, particularly in the central nervous system. 40,41
In case of exploratory behavior, a significant reduction in head-dip counts and head-dip duration was observed in young, adult, and aged rats exposed to As. The observed alterations in exploratory behavior may be due to altered cholinergic system which was observed in the present study. 13,14 This decreased exploratory behavior, and impairment in learning and memory may also be attributed to the inhibitory effect of As on neurotransmitters in the brain regions. 42
We evaluated the alterations in motor strength by measuring the forelimb and hind limb grip strength in young, adult, and aged rats. Grip strength measurements are quite sensitive in detecting neuromuscular impairments. 43 We found a significant decrease in forelimb and hind limb strength in young, adult, and aged rats exposed to As when compared to their respective controls suggesting that impairment in motor coordination and muscle strength leads to impairments in motor functions. Similar to our findings, Yadav et al. 44 and Krishna and Ramachandran 45 also reported significant alterations in both forelimb and hind limb grip strength following As and Pb exposure.
In water maze test, As-exposed adult and aged rats showed a delay to find the hidden platform in all the phases (acquisition, reversal, and working memory) showing that As exposure lead to impaired functions in spatial reference memory and spatial working memory. The hidden platform acquisition is dependent on the function of hippocampus 46 ; therefore, the deficits indicate impairment of the hippocampus in As-exposed rats. These results show that structures involved in the swim tasks are affected by exposure to As. The striatum, the main nucleus of the basal ganglia, appears to be particularly involved in coding informational components required for navigation, such as rat’s location, heading direction, and execution of displacement movements. 47
Neurochemical studies showed that exposure to As altered the brain regional synaptosomal ACh content and AChE activity in all the three age points, and the more susceptible region being the hippocampus. Further, we observed that supplementation with Zn and vitamin E showed protection against As-induced alterations in cholinergic system. The clear mechanisms for how As decreases cholinesterase activity have not yet been documented. But still, As compounds are known as potent inhibitor of many enzymes 48 and the reaction between the As and free sulfhydryl groups of enzymes may underlie the mechanism of this inhibition. However, cholinesterase does not have the structural features suitable for binding with As. Cholinesterase has been found to contain cysteine only in the form of disulphide bridges and not as free thiol. 49 Therefore, As reduces the activity of cholinesterase probably due to other mechanisms rather than the direct binding to the enzyme. The decrease in AChE activity may accumulate ACh at synaptic regions leading to desensitization of the cholinergic receptors on the postsynaptic membrane rendering it ineffective to the transmitter action. 50
In addition, our results showed a significant decline in brain regional AChE activity in aged rats followed by young and adult rats. This age-related decline in cholinergic function is thought to be partially responsible for the memory disorders occurring during senescence. In support of the present results, decline in cholinergic indices such as choline acetyltransferase (ChAT), AChE, muscarinic, and nicotinic ACh receptors during normal aging process has been reported by Zhang 51 and Barnes et al. 52 ChAT is the rate-limiting enzyme of generating ACh, which is synthesized in cholinergic neuronal cell bodies and is often used in the studies of tissue localization and functional activity. A study by Butterweck et al. 53 showed decreased expression of ChAT immunoreactivity and Nissl stain in dentate gyrus area of hippocampal region suggesting that As affects the cholinergic circuitry in rats. Decreased AChE activity may also be due to the free radicals and increased oxidative stress. 54 It is therefore possible that the age-related decrease in brain AChE activity observed in the present study may be due to increased lipid peroxidation in aged rats. Previous studies in both rodents and humans have shown that there is a close correlation between age-related alterations in AChE and increased oxidative stress, 54 as well as between age-related losses of cognitive function with oxidative protein damage in the brain. 55 Therefore, in the present study, the impaired memory exhibited by the aged rats may be attributed to the observed decreases in AChE activity in brain.
Moreover, AChE is a membrane bound enzyme with lipid dependence. 56 The effect of lipid peroxidation on cholinergic system was proposed by earlier investigations. 57 –59 In agreement with these reports, we observed a significant decrease in AChE activity following As exposure, and it was a correlated with an increase in lipid peroxidation and decrease in oxidative stress marker enzymes. 60 Hence, As may exert its neurotoxic effects via peroxidative damage to the membranes. 61
The results of the present study also revealed that the maximum activity of AChE was observed in hippocampus followed by cortex and cerebellum at all the age points. Regional variations in the density of cholinergic innervations in the brain could be one of the possible reasons for the differences in the level, synthesis, and turnover rate of ACh. 62,63 This could be the reason for the observed differences in AChE activities in different brain regions. Further, the cholinergic synapses were more in hippocampus as compared to cerebral cortex and cerebellum. 64 This may be another cause for higher AChE activity observed in hippocampal region as compared to cerebral cortex and cerebellum. Altered Ras-mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinases (ERK) signaling and synaptic regulation in the hippocampus could underlie behavioral deficits suggesting As-induced alterations in spatial learning and working memory. Even the glucocorticoid receptors present in hippocampus are sensitive to As leading to alterations in glucocorticoid signaling. 65 This could be another reason for its relative sensitivity to As compared to other regions.
The present investigation, however, showed that these alterations in behavioral and cholinergic system induced by As could be reversed by administration of essential metal, Zn or antioxidant, vitamin E. This may be attributed due to the fact that this essential trace elements can affect toxicity of heavy metals by interacting at the primary site of action and/or this trace element could reduce the gastrointestinal absorption of As. Zn could be competing for binding sites and effectively reducing the availability of binding sites for metal uptake. 19,66 Treatment with vitamin E reduced As-induced alterations, probably through its capacity to quickly and efficiently scavenge lipid peroxyl radicals before they attack membrane lipids. The reaction of α-tocopherol with free radicals generates tocopherol radicals, which can be reduced by vitamin C or glutathione (GSH), 67 thereby reducing reactive oxygen species (ROS). Since vitamin E is primarily located in cell membranes, it is possible that it modifies the kinetics of distribution of As in cell membranes, such that its delivery to neuronal cells may be impeded.
Administration of vitamin E/Zn to As-exposed rats reduced As burden in brain regions suggesting protection of vitamin -E/Zn from biochemical damage. The protective mechanism of vitamin E could be attributed to its antioxidant property or its location in the cell membrane and its ability to stabilize membrane by interacting with unsaturated fatty acid chain. 68,69 As-induced free radicals can attack the double bonds of polyunsaturated fatty acids, thereby initiating a chain reaction which can affect the membrane integrity and cellular function. Vitamin E inhibits them by reacting with free radicals and converting itself into α-tocopheroxyl radical that is not harmful and checks lipid peroxidation. 70
The reduced As levels in Zn supplemented group could be attributed to its antiperoxidative property. Zn deficiency reduces the activity of several enzymes in the brain that may disturb cell multiplication at a crucial period in morphogenesis. 71 Kreppel et al. 72 also reported reduction in As concentration in tissues of adult mice, when Zn was administered prior to As treatment. In view of these data, it can be concluded that vitamin E/Zn supplementation reduces As burden from gastrointestinal absorption, reduces entry through BBB, enhances scavenging of As, and thus provides protective effects against As-induced neurotoxicity. However, the precise underlying mechanisms of the observed vitamin E/Zn mediated regulation of enzymatic activities and reduction in As burden cannot be ascertained from this study and remains to be explored further.
In conclusion, among Zn and vitamin E, vitamin E supplementation produced significant reversal effect and found to be a better ameliorative agent against As-induced cholinergic and behavioral perturbations at all the age points. Our findings are consistent with the view that protective effects of vitamin E against high dose As exposure might be due to its antioxidant properties, and this may have a major impact on improving the quality of life of individuals suffering from neurotoxicity caused by As exposure. However, further studies have to be carried out to see whether this type of therapy can be advocated as safe and effective treatment for As-induced neurotoxicity at all the age points of life.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Indian Council of Medical Research, New Delhi (Grant no. 45/76/2012-PHA/BMS).
