Abstract
Virgin coconut oil (VCO) is an unrefined kernal oil, prepared from
Introduction
Cyclophosphamide (CTX) is a widely accepted chemotherapeutic agent used in the treatment of several malignancies and also as a prophylactic to metastatic tumours. 1 However, the severe adverse reactions including immunosuppression, cardiotoxicity and liver and kidney toxicity observed in CTX treatment pose great challenges. 2 Most of these secondary toxicities attributed to CTX administration are due to secondary radicals formed during metabolic conversion that generate oxidative stress. 3
Symptomatic treatment and use of cytoprotective agents as adjuvants are the generally accepted approaches. Widely used cytoprotective agents like amifostine, aminoguanidine and thymoquinone are efficient free radical scavengers. These molecules, however, are also known to exert widespread secondary toxicities such as nausea and vomiting. 4,5 Exploration for more potent non-toxic cytoprotective agents is therefore essential to improve cancer therapy. Accordingly, natural products that have potential to be both safe and enhance immunity and vigour of humans are being much emphasized in drug development.
Virgin coconut oil (VCO) is obtained from fresh, mature kernel of the coconut palm (
Considering its wide spectrum of biological properties including antioxidant and metabolic stimulation, the present study evaluates the effect of VCO on CTX-induced systemic toxicity in mice, a prospect that has not been explored so far.
Materials and methods
Materials
5,5′-Dithiobis-2-nitrobenzoic acid (DTNB), reduced glutathione (GSH), riboflavin and nitro blue tetrazolium (NBT) were purchased from Sigma Chemical Co. Inc. (St Louis, Missouri, USA). Ethylenediaminotetraacetic acid was purchased from Sisco Research Laboratories Pvt. Ltd (Mumbai, Maharashtra, India). Thiobarbituric acid (TBA) was purchased from Hi Media Laboratories Pvt. Ltd (Mumbai, Maharashtra, India). Thin layer chromatography (TLC) plate (Silica gel 60 F254, DC Kieselgel) was purchased from Merck India Ltd. (Mumbai, Maharashtra, India). All other chemicals and reagents used in this study were of analytical grade and obtained from reputed Indian manufacturers.
Animals
Male Swiss albino and Balb/c (25–30 g) mice were purchased from Small Animal Breeding Station, Kerala Agricultural University, Mannuthy, Kerala, India. The animals were maintained under standardized environmental condition (22–28°C, 60–70% relative humidity, 12 h dark/12 h light cycle) and fed with standard rat feed (Lipton Lever Ltd., Pune, India) and water ad libitum. All animal experiments conducted had prior permission from Institutional Animal Ethics Committee and strictly followed guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals constituted by the Animal Welfare Division, Government of India.
Preparation of VCO
The VCO was prepared based on the method of Nevin and Rajamohan. 13 The grated coconut and its neutral water were mixed together and squeezed into viscous slurry until all creamy milk was expelled. This was kept at room temperature for 48 h until fermentation process took place. Three layers were obtained as follows: creamy mixture in the upper layer, the VCO in the middle and water in the lower layer. The oil was gently scooped out and filtered into a container. Same extraction procedure was followed for the preparation of three batches of VCO (VCO I, VCO II and VCO III).
TLC analysis of VCO preparations
Approximately 10 µL each of VCO I, VCO II, VCO III and CO was applied on to TLC plates (Silica gel 60 F254, DC Kieselgel from Merck). The plate was developed using the solvent system hexane:diethyl ether:acetic acid (35:15:0.15). The separated compounds were visualized under ultraviolet (UV) (234 nm) and iodine vapour.
Quantitative estimation of hydro peroxide and aldehydes in VCO
The lipid hydroperoxide content in the oil was quantitatively determined by Fox assay. 14 The aldehyde content of the oil was determined by TBA method 15 and expressed as nanomole malondialdehyde equivalent.
Effect of VCO on formalin-induced chronic inflammation
Male Swiss Albino mice (24 nos.) were divided into four groups of six animals each. Animals in all groups were injected with 0.02 mL of 1% formalin in the right hind paw to produce chronic inflammation. 16,17 Group I animals were kept as control without any drug treatment. The animals in group II received standard reference drug diclofenac at 10 mg/kg body weight (bwt) intraperitoneally. Group III and IV animals were administered orally with 4 and 8 g/kg bwt VCO, respectively. The standard drug and VCO were given 1 h prior to formalin injection and continued for six consecutive days. The paw thickness was measured using vernier callipers before formalin injection and thereafter for six consecutive days.
Effect of VCO on CTX-induced systemic toxicity
Twenty-four male Balb/c mice were divided into four groups of six animals each. The first group animals were kept as normal, without any treatment. All other groups were given CTX (International Nonproprietary Names, trade names Endoxan, Cytoxan, Neosar, Procytox and Revimmune) at a dose of 25 mg/kg bwt orally for 20 days. 18 The second group was maintained as control. Everyday, third and fourth group animals received VCO at doses of 4 and 8 g/kg bwt, respectively, for 20 days. Every 5th day, body weight and haematological parameters such as white blood cell (WBC) count and haemoglobin level were assessed. On the 21st day, all the animals were killed following an overnight fasting.
Activities of serum marker enzymes such as serum glutamate oxaloacetate transaminase (SGOT), serum glutamate pyruvate transaminase (SGPT), 19 alkaline phosphatase (ALP), 20 serum creatinine and urea levels were estimated using commercially available kits obtained from Span Diagnostics Ltd (Surat, Gujarat, India).
The liver and kidney tissue was excised and freed off blood and adipose tissue and tested for catalase (CAT), superoxide dismutase (SOD) and glutathione peroxidase (GPx) activities and GSH and TBA reactive substances (TBARs) levels. The CAT activity was measured as a decrease in UV absorption of hydrogen peroxide at 240 nm. 21 The superoxide anion radical scavenging activity was measured as a decrease in NBT reduction by riboflavin observed at 560 nm. 22 GSH was estimated using freshly prepared DTNB solution, and the intensity of the yellow colour formed was read at 412 nm. 23 GPx activity was detected based on the degradation of hydrogen peroxide in the presence of reduced GSH and consecutively measured using DTNB reagent at 412 nm. 24 TBARs levels were measured using TBA reagent at 532 nm. 15
Statistical analysis
In vitro biochemical assays were repeated at least thrice and the data expressed as mean ± SD. In animal experiments, data presented are mean ± SD of six animals per group. Significance was calculated by one-way analysis of variance using GraphPad InStat software and
Results
Yield of VCO and its peroxide and aldehyde content
VCO was prepared in three batches. The average percent yield obtained with the three batches was found to be 62.5 ± 2.1%.
TLC analysis of VCO
Figure 1(a) shows the TLC analysis performed on the three samples of VCO and CO. Under iodine exposure, VCO revealed three yellowish brown colour bands with retention factor (Rf) values 0.36 (band 1), 0.41 (band 2) and 0.091 (band 3), respectively. CO also revealed the same banding pattern with the same Rf values (0.36, 0.41 and 0.091).
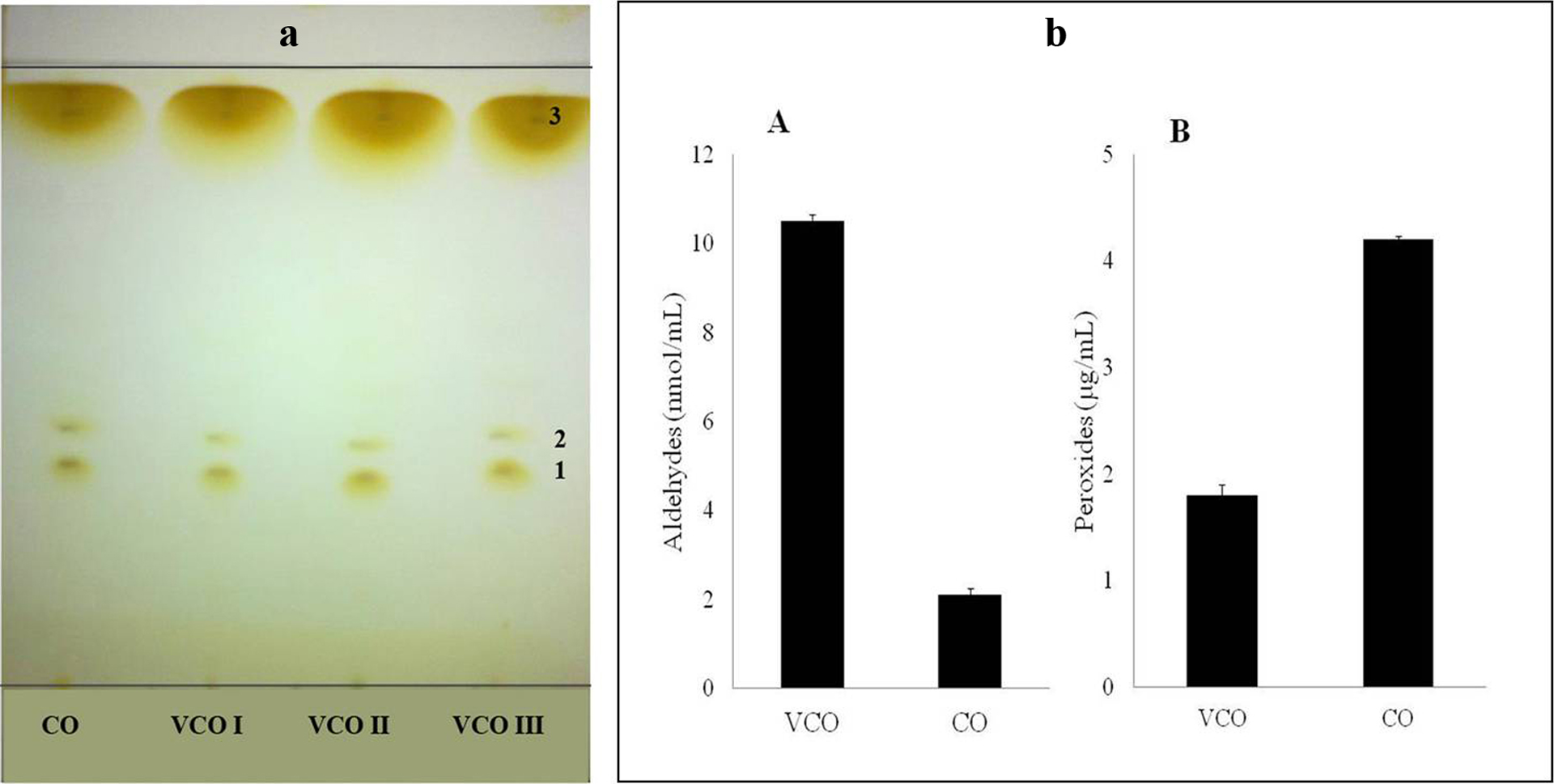
(a) TLC analysis of CO and different batches of VCO (I, II and III). (b) Estimation of aldehydes (A) and peroxides (B) in VCO and CO. TLC: thin layer chromatography; VCO: virgin coconut oil; CO: copra oil.
Aldehyde and peroxide contents in VCO
An average aldehyde content of 10.5 ± 0.35 nmol/mL and peroxide content of 1.8 ± 0.14 µg/mL oil were recorded in VCO samples. In CO, comparatively lesser amount of aldehyde (2.1 ± 0.39 nmol/mL oil) and higher content of peroxides (4.2 ± 0.08 µg/mL) were present (Figure 1(b)).
Effect of VCO on formalin-induced paw oedema
The intra-plantar injection of formalin on the hind leg of mice produced oedematous inflammatory response that showed an increase of 1.06 ± 0.30 mm in paw thickness on the 6th day. In animals treated with VCO 4 and 8 g/kg bwt, paw thickness reached only up to 0.91 ± 0.30 mm and 0.93 ± 0.38, respectively. Diclofenac, used as positive control in this experiment, had a marked decrease in paw oedema formation with the oedema size recorded to be 0.83 ± 0.06 mm on the 6th day (Table 1).
Effect of VCO on chronic pedal oedema in mice induced by formalin.a

VCO: virgin coconut oil.
aValues are mean ± SD of six animals in each group.
b
c
Effect of VCO on CTX-induced systemic toxicity in mice
There was significant decline in the total WBC counts (from 20,413 ± 524 cells/mm3 to 10,966 ± 202 cells/mm3) and haemoglobin content (17.2 ± 0.9 g/dL to 14.0 ± 0.25 g/dL) in animals that received CTX (25 mg/kg bwt) orally over a period of 20 days. However, no significant change in WBC count and haemoglobin level was noticed in both 4 and 8 g/kg bwt VCO-treated groups (Table 2).
Effect of VCO on total WBC count (cells/mm3) and haemoglobin content (g/dL).a

VCO: virgin coconut oil; WBC: white blood cell; Hb: haemoglobin; bwt: body weight.
aValues are mean ± SD of six animals in each group.
b
cNot significant when compared with CTX (at 25 mg/kg bwt) alone treated group.
d
e
The normal level of activity of serum marker enzymes such as SGOT, SGPT and ALP was 114.0 ± 20.5, 54.4 ± 12.3 and 65.5 ± 1.7 IU/L, respectively. The levels of activities of these enzymes were marginally elevated in animals treated with CTX alone. The respective values were 139.6 ± 12.5, 74.4 ± 8.8 and 71.80 ± 1.6 IU/L. The SGOT, SGPT and ALP activities in VCO-supplemented groups did not show such an increase. In the 4 g/kg bwt VCO-treated groups, the level of enzyme activities was 122 ± 9.6, 70.8 ± 5.3 and 69.24 ± 2.3 IU/L, respectively, and in 8 g/kg bwt treated animals 115.4 ± 11.4, 56.2 ± 8.6 and 64.40 ± 2.8 IU/L, respectively (Table 3).
Effect of VCO on serum parameters in CTX-treated mice.a
VCO: virgin coconut oil; CTX: cyclophosphamide; SGOT: serum glutamate oxaloacetate transaminase; SGPT: serum glutamate pyruvate transaminase; ALP: alkaline phosphatase; bwt: body weight.
aValues are mean ± SD of six animals in each group.
b
c
dNot significant when compared with CTX (at 25 mg/kg bwt) alone treated group.
e
f
A significant increase was noticed in serum urea (from 11.02 ± 0.67 mg/dL to 34.52 ± 0.94 mg/dL) and creatinine (0.37 ± 0.06 mg/dL to 2.14 ± 0.52 mg/dL) level in the CTX-alone treated animals. However, significantly lower value of urea (12.06 ± 0.42 mg/dL) and creatinine (0.33 ± 0.17 mg/dL) was recorded in the VCO (8 g/kg bwt) treated groups (Table 2).
Table 4 depicts the level of liver and kidney antioxidant enzyme activities and the GSH and TBARS levels in the various treatment groups. The antioxidant enzyme activities were found to be decreased dramatically in CTX-treated groups, whilst the animals that received VCO at 8 g/kg bwt had enzyme activities near normal values. Similarly the amount of TBA reactive species was decreased significantly in VCO-treated groups compared with that of CTX group in both kidney and liver.
Effect of VCO on antioxidant enzymes and lipid peroxidation in kidney (K) and liver (L) of cyclophosphamide-treated mice.a

VCO: virgin coconut oil; CTX: cyclophosphamide; CAT: chloramphenicol acetyltransferase; SOD: superoxide dismutase; GSH: glutathione; GPx: glutathione peroxidase; LPO: lactoperoxidase; bwt: body weight.
aValues are mean ± SD of six animals in each group.
b
c
d
eNot significant when compared with CTX (at 25 mg/kg bwt) alone treated group.
f
g
Discussion and conclusions
In the present study, VCO, a fermented product of coconut kernel, has shown substantial reduction in paw oedema in formalin-induced chronic inflammation model in Balb/c mice. In albino mice, the CTX induced decrease in total WBC count and haemoglobin level has been restored to near normal levels and reduced liver and kidney toxicity. This protective effect of VCO is thought to be mediated through the maintaining of the normal cellular redox status.
Inflammation is common in tissue injury and chronic inflammation is known to be the prevailing factor in many degenerative human ailments. The reduction of the oedematous response to formalin observed in the present study is comparable to standard drug diclofenac. Formalin-induced paw oedema is due to increased accumulation of inflammatory cells and cytokines in response to histamine. 25 Therefore, it is likely that VCO could inhibit histamine and the subsequent inflammatory cells/molecules that induce chronic inflammation. Zakaria et al. has reported the anti-inflammatory potential of VCO in carrageenan-induced acute inflammatory model. 26 The present study which used a chronic inflammatory model system further confirms the anti-inflammatory potential of VCO.
Another notable observation in this study is that VCO could maintain the levels of WBC and haemoglobin near normal levels against CTX-induced decrease. Myelosuppression often associated with CTX and other chemotherapeutic drugs is manifested by reduced WBC and haemoglobin level. The immunostimulatory activity of VCO has been already reported. 27 This might be the reason for the pattern of myeloprotection observed in this study also.
Exposure of cells to hydroxycyclophosphamide, the bio-activated form of CTX, induces lipid peroxidation, which reflects the presence of free radicals and peroxynitrite. 28 –31 It is also possible that, upon activation of CTX by cytochrome P450, there is production of reactive species such as superoxide anions. 32 This oxidative stress is thought to contribute to initiation and progression of liver and kidney damage. In this study, the level of TBARS produced in liver and kidney tissues is found to be increased in CTX alone treated animals; however, significantly reduced level of TBARS is noticed in animals treated with VCO. This suggests that peroxidative changes due to CTX administration are reduced by VCO intake.
It is thus expected that the antioxidant and anti-inflammatory properties of VCO might have been involved in reducing the secondary toxicity induced by CTX as evidenced by the reversal of CTX-mediated hike in serum marker enzymes such as SGOT, SGPT and ALP. Increased activity of SGPT along with SGOT and ALP clearly indicates hepatic injury. Present result thus indicates that VCO could protect from CTX-induced liver toxicity. Similarly, the increase in creatinine and urea level observed in CTX-intoxicated animals has been substantially reduced by VCO administration. Increased serum level of creatinine and urea indicates reduced glomerular filtration due to kidney damage. This further suggests that VCO can provide protection from CTX-induced kidney toxicity.
Reduced GSH is the most abundant intracellular antioxidant, with multiple and complex biological functions. GSH is usually considered in terms of its protective role in the toxicity of xenobiotics. 33 GSH reacts with electrophilic derivatives of alkylating agents and participates in their detoxification under the effect of glutathione S-transferase. 34 Reduced form of GSH and other compounds, such as amifostine or curcumin, have been shown to modulate various forms of short-term toxicity of CTX in animal models without reducing the antitumour efficacy. 35 –37 In the present study, the activities of antioxidant enzymes and molecules are decreased in liver and kidney associated with CTX administration. This observed decrease in antioxidant defence suggests that an oxidative type of stress has prevailed with CTX-induced hepatotoxicity and urotoxicity. The administration of VCO, however, increased the level of endogenous antioxidant molecules like GSH and enzymes like SOD, GPx and CAT activities in both liver and kidney. This indicates that VCO has maintained normal redox status in the animals and thereby substantially limited lipid peroxidative changes. The protective effect on liver and kidney from CTX toxicity revealed in this study thus could be partially explained. This assumption is supported by the fact that VCO has already been found to enhance antioxidant status in rats. 12
The difference between CO and VCO in terms of their observed antioxidant and anti-inflammatory activities has not been clarified yet. The constituent analysis of freshly prepared VCO and refined coconut oil used in this study is found to be the same with respect to fatty acid composition as evidenced by TLC analysis. Also, the peroxide content in VCO was observed to be lower than that of CO. This finding is similar to the gas chromatographic analysis report of fatty acid composition performed by Dayrit et al. on VCO and CO. However, there are reports indicating that in addition to medium and short chain fatty acids, VCO consists of polyunsaturated fats such as linoleic acids and oleic acids. 7 These essential fatty acids are known to possess cytoprotective effects. 38 It has been well documented that oxidation of polyunsaturated fats generates peroxides and aldehydes. 39 The hydroperoxides are unstable products that are further converted to aldehydes. Lipid-derived aldehydes are known to be toxic and are capable of acting as elicitors of various signalling pathways leading to inflammation. In the present study, the lower levels of hydroperoxide present in VCO as compared to CO make the use of VCO a better option as compared to CO. Caffeic acid and gallic acids known to be present in VCO might have reduced the oxidation of linoleic acid and oleic acid. 40 Further these unsaturated fatty acids and antioxidant molecules might also have contributed to the protective effect of VCO in CTX-induced toxicity.
The present study thus reveals, for the first time, the protective effect of VCO against CTX-induced liver and kidney toxicity and inhibition of chronic inflammatory response in murine models. VCO has been in use for a relatively long time as a dietary supplement due to its nutritional value, and no toxicity has been observed so far. It is thus expected that further studies with different experimental models may reveal the beneficial role of VCO in the treatment of chemotherapeutic drug-induced side effects.
Footnotes
Acknowledgement
We are indebted to Dr Ramadasan Kuttan, Research Director, Amala Cancer Research Centre for his support in carrying out the experiment.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
