Abstract
Purpose:
Alkaloids derived from Rhizoma Coptis (RC) has been widely applied to clinical treatments in China. However, the toxicity of RC and the alkaloids from RC remained controversial. The research is designed to clarify the cardiotoxic compounds found in RC.
Methods:
In this study, the real-time cellular analysis cardio system and the high-content analysis were applied to monitor the function of cardiomyocytes (CMs) in the treatment of nine alkaloids in RC. Luciferase-coupled adenosine triphosphate (ATP) assay was used to detect cell viability.
Results:
The results showed that berberine, palmatine, berbamine, and oxyberberine were cardiotoxic, which resulted in arrhythmia and cardiac arrest on CMs in a time- and dose-dependent manner. Meanwhile, berbamine and oxyberberine caused shrinkage and detachment on CMs at 10 μM. Cytotoxicity was induced by these two compounds with decline in cell index and ATP depletion. Cardiotoxicity or cytotoxicity was not observed in the other five alkaloids within 10 μM.
Conclusion:
For the first time, the cardiotoxicity of the nine alkaloids was evaluated to clarify the cardiotoxic components in RC. Furthermore, the experimental evidences were provided to support the safety of drug application.
Introduction
As a perennial herb, Rhizoma Coptis (RC) is the dried rhizome of coptis, triangular leaves or coptis teeta walls, which belongs to Ranunculaceae. 1 It has been first recorded in the “Shen Nong’s Herbal Classic,” the earliest pharmacopeia in the world, compiled more than 2000 years ago. Before the Song Dynasty of China, RC had been adopted in more than 32,000 prescriptions. In traditional Chinese medicine (TCM), RC has been widely used in a great number of treatments because of its heat-clearing, damp-drying, pathogenic fire purging, and detoxification effects. 1 The antiinflammatory property of RC has been proved in treatment of intestinal infection, urinary infection, and inflammation in lipopolysaccharide-induced endotoxemia. 2 In addition, RC has been used in treatment of type 2 diabetes, 3 cardiovascular, 4 and cerebrovascular diseases. 5 Furthermore, the immune-enhancement effect of RC is recently confirmed by activating the mitogen-activated protein kinase signaling pathways. 6 The RC contains alkaloids as its main active ingredients. 7 In particular, RC includes 6–10% berberine, 0.53–2.85% of coptisine, 1.24–2.78% of palmatine, and 0.39–1.65% of epiberberine. 8 The quality control index of RC is determined by these four components. The proportion of other alkaloids such as berberrubine (0.006%), choline (0.007%), oxyberberine (0.005–0.008%), magnoflorine (0.209–0.213%), and berbamine (0.203–0.230%) are much less in RC (Figure 1). 9 –11 Due to the large variation in origin and distribution places, growing period, and processing methods, the contents of alkaloids in RC could be very different. 12

Chemical structures of nine alkaloids derived from RC. 11 RC: Rhizoma Coptis.
Up to 2010, 108 Chinese patent drugs have been approved by State Food and Drug Administration (FDA), and all of them take RC as their main ingredient. 13 The suggested oral dosage of RC is 2.0–5.0 g/day per adult. 1 However, as a non-prescription drug, RC has often been overdosed in practice. As a result, some clinical adverse drug reactions (ADRs) had been reported, including nausea, glucopenia, and diarrhea. 14 As RC is taken over long period or used with large dose, it may cause diminished function of the stomach in digestion and absorption. 15 In addition, RC extracts showed weak embryotoxicity. 16 The alkaloids were also perceived as the main toxic substances in RC. 7 Berberine was accepted to be the major toxic constituent in RC, which showed the highest content in the heart tissue of mice. 7 The basic researches on acute toxicity illustrated that the lethal dose 50 (LD50) of berberine from intravenous and intraperitoneal injection is 9.0386 and 57.6103 mg/kg, respectively, in mice. 17,18 Furthermore, berberine on rats and mice showed certain developmental toxicity. 19 Meanwhile, the clinical ADRs of berberine including constipation and other gastrointestinal reactions have been reported in recent years. 20,21 The toxicity of other alkaloids has been reported as well. The cytotoxic activity of palmatine, 22 coptisine, 23 epiberberine, 24 and berbamine 25 in human cancer cell lines were demonstrated. Moreover, berberrubine-induced specific nephrotoxicity and did not affect hepatic function. 26 Rare adverse responses regarding the other three alkaloids, such as choline, oxyberberine, and magnoflorine, were reported. There are still no systematic investigations on the cardiotoxicities of alkaloids from RC.
Therefore, our study was carried out to define the cardiotoxic components in RC. Real-time cellular analysis (RTCA) cardio system had been adopted by FDA for drug cardiotoxicity evaluation in 2012. It has been widely used in preclinical safety evaluation of pharmaceuticals since then. 27 –29 To validate the results, high-content analysis (HCA) were applied to analyze the cardiotoxicity with morphological change of cardiomyocytes (CMs). More attention should be paid to reasonable application of the main components of RC and its alkaloids because of their wide use in clinical treatments.
Materials and methods
Compounds preparation
The nine alkaloids, with the purities greater than 98%, were all purchased from the Chroma-Biotechnology Co., Ltd (Chengdu, China). Amiodarone was purchased from Sigma-Aldrich Chemicals (St Louis, Missouri, USA). All compounds were dissolved in dimethyl sulfoxide (DMSO) before diluted in media for cell treatments. A 0.1% DMSO was used as the vehicle control. The pharmacokinetic characteristics of alkaloids in rats were considered to design the doses of tested compounds in the current neonatal rat cardiomyocytes toxicity assay. In detail, the content of berberine occupied above 50% in total alkaloids, and the peak plasma concentration (C max) of berberine was 2.8 to 3.7 μg/mL (the average value is 7.9 μM). The C max values of the other alkaloids (such as palmatine and coptisine) were lower than that of berberine. 30 Therefore, we evaluated cardiotoxicity and cytotoxicity of all alkaloids at concentrations in the range of 0.03–10 μM.
Cell culture
Harvest and culture of neonatal rat CMs
CMs were prepared from 24 h old neonatal Sprague Dawley rats of both sexes according to protocols published previously. 31 The cell concentration was regulated to 2×105 cells/mL with high-glucose Dulbecco’s modified Eagle’s medium (DMEM) culture solution containing 10% fetal calf serum and transferred to xCELLigence E-Plate Cardio 96 (Roche Diagnostics GmbH, Mannheim, Germany) at a density of 17,000 per well. 32 The CMs were cultured in 37°C and 5% carbon dioxide (CO2) incubator for 72 h.
Cardiotoxicity evaluation by RTCA assay
Neonatal rat CMs were plated onto 1% matrigel-coated xCELLigence E-Plate Cardio 96 at a density of 17,000 per well. The media was changed every other day after plating using maintenance media. Media was exchanged 4 h before compound dilutions transferring to CMs on E-plate. Signals were collected every 20 s. Data collection was controlled and analyzed by the xCELLigence Cardio Software (Roche Diagnostics GmbH), which allows calculation of CMs beating parameters such as cell index, beating rate, amplitude, beating patterns, rising slope, and falling slope. 27
Morphology assessment by HCA assay
To assess changes in morphology following exposure to drug, CMs were treated for 24 h with 0.1% DMSO, berberine, palmatine, berbamine, or oxyberberine in different concentrations (0.03, 0.1, 0.3, 1.0, 3.0, and 10.0 μM) and stained for alpha actin. 33 Briefly, CMs were dosed and fixed in 4% formaldehyde, permeabilized using 0.1% Triton-X100 (Sigma-Aldrich) in phosphate buffer saline (PBS), blocked in 5% bovine serum albumin (BSA) (Sigma-Aldrich) in PBS, and incubated overnight at 4°C with the alpha actin antibody (Sigma-Aldrich). After washing, slides were incubated with DyLIGHT488-anti-mouse immunoglobulin M secondary antibody (Jackson Immuno Research, West Grove, Pennsylvania, USA) for 1 h and then stained with Hoechst 33342 (Sigma-Aldrich) for nuclear detection. Alpha actin stained cells were visualized and imaged using ImageXpress XLS (Molecular Devices, Sunnyvale, California, USA). Images (10×) are representative of four independent studies.
ATP depletion assay
CMs were seeded at 1.7 × 104 per well into Plate and incubated at 37°C in 5% CO2. Different concentrations of the compounds (0.03, 0.1, 0.3, 1.0, 3.0, and 10.0 μM) were added into Plate and incubated in 5% CO2 at 37°C for 24 h. Luminescence was read by Envision 2100 multilabel reader (PerkinElmer, Waltham, Massachusetts, USA) to detect cell viability following incubation with 100 μL CellTiter-Glo reagent (Promega, Madison, Wisconsin, USA) to 100 μL of medium containing cells each well for 10 min. 34 Each experiment was performed with three replicates.
Statistical analysis
Data were analyzed using xCELLigence Cardio Software and further processed by GraphPad Prism 6.0 (Graphpad Software, San Diego, California, USA). Data were expressed as mean ± SD, and statistical analysis was performed using analysis of variance with least significant difference (LSD) test. The value of p < 0.05 was considered statistically significant.
Results
Effect of RC alkaloids in RTCA and HCA cardiotoxicity assay
Beating patterns changes observed following berberine, palmatine, berbamine and oxyberberine treatment
To screen the cardiotoxicity of RC alkaloids, amplitude, beating rate, and beating patterns of CMs were monitored by RTCA cardio system. Amiodarone was used as the positive control. 35 Beating rate and amplitude were totally inhibited by berberine, palmatine, berbamine, and oxyberberine at 10 μM, compared with those of vehicle control group (p < 0.01). Representative beating patterns were captured at selected time points (0, 0.75, 1.5, 3.0, and 6.0 h). Cardiac arrest occurred in treatment of the four alkaloids. However, the parameters of CMs did not show obvious change in the treatment of the other five alkaloids (Figure 2).

Representative contraction profiles of the RTCA analysis of CMs treated by nine alkaloids in RC. (a) Beating rate of CMs treated by alkaloids. (b) Amplitude of CMs treated by alkaloids. CMs treated by the alkaloids at 10 μM were detected continuously for 6 h, and changes were normalized to that of the time-matched vehicle control group. The values were showed at selected time points (0, 0.75, 1.5, 3.0, and 6.0 h). (c) Representative beating traces captured at selected time points illustrate the cardiac arrest and rapid onset of beating rate reduction coursed by alkaloids. The real-time data from each negative peak to the following positive peak is recorded and presented with a unitless parameter called amplitude. With the vehicle group as the standard, the amplitude was 0.05, the time for each segment was 2 s. All the beating data are derived from the xCELLigence Cardio Software and adjusted to a unified scale, so that the data at different time points can be compared. Data were presented as mean ± standard deviation, n = 2. RTCA: real-time cellular analysis; CM: cardiomyocyte; RC: Rhizoma Coptis.
To investigate the cardiotoxicity of berberine, palmatine, berbamine, and oxyberberine, different concentrations (0.03, 0.1, 0.3, 1.0, 3.0, and 10.0 μM) of the four alkaloids were added to wells. Beating rate of CMs under berberine treatment was declined to zero at 10.0 μM (p < 0.01), whereas that of other concentrations did not show significant changes comparing to vehicle control group (p > 0.05). Meanwhile, amplitude was dropped to zero at 10.0 μM and was obviously inhibited at 3.0 μM (p < 0.01). The IC50 of berberine in amplitude was 2.1 μM. Rising slope and falling slope were calculated to evaluate the effect on myodynamia of CMs, which showed an identical trend of amplitude. The beating of CMs was totally inhibited by berberine at 10.0 μM and did not recover in 24 h. Irregular beating occurred at 0.3, 1.0, and 3.0 μM, whereas the other groups did not show obvious change comparing to the vehicle control group. Palmatine inhibited the beating rate and amplitude of CMs to zero at 10.0 μM (p < 0.01), with IC50 of amplitude was 5.2 μM. Rising slope showed a similar trend of amplitude, whereas falling slope declined to a lower level at 3.0 μM (p < 0.05). The result represented that the diastolic function of CMs was inhibited in a greater degree. The beating of CMs was totally inhibited by palmatine at 10.0 μM and recovered in long time. Irregular beating was observed in 3.0 μM group, whereas the other groups showed normal beating. Time- and dose-dependent recovery was observed in berberine and palmatine treatment on CMs. Beating rate and amplitude of CMs were declined to zero at 3.0 and 10.0 μM in treatment of berbamine (p < 0.01). The IC50 of amplitude on CMs was 1.3 μM. Rising slope and falling slope showed an identical trend of amplitude in all concentrations. Berbamine resulted in cardiac arrest at 3.0 and 10.0 μM, which did not show recovery in 24 h. No significant change of beating showed in other concentrations treatment (p > 0.05). Oxyberberine totally inhibited the beating rate and amplitude of CMs to zero in 0.1, 0.3, 1.0, 3.0, and 10.0 μM (p < 0.01). In addition, the IC50 of amplitude was 39.6 pM. Cardiac arrest was brought out by the treatment of oxyberberine in concentrations with 0.1 μM and above, which persisted over 24 h. Irregular beating was observed in 0.03 μM group, compared with the vehicle control group (Figure 3).
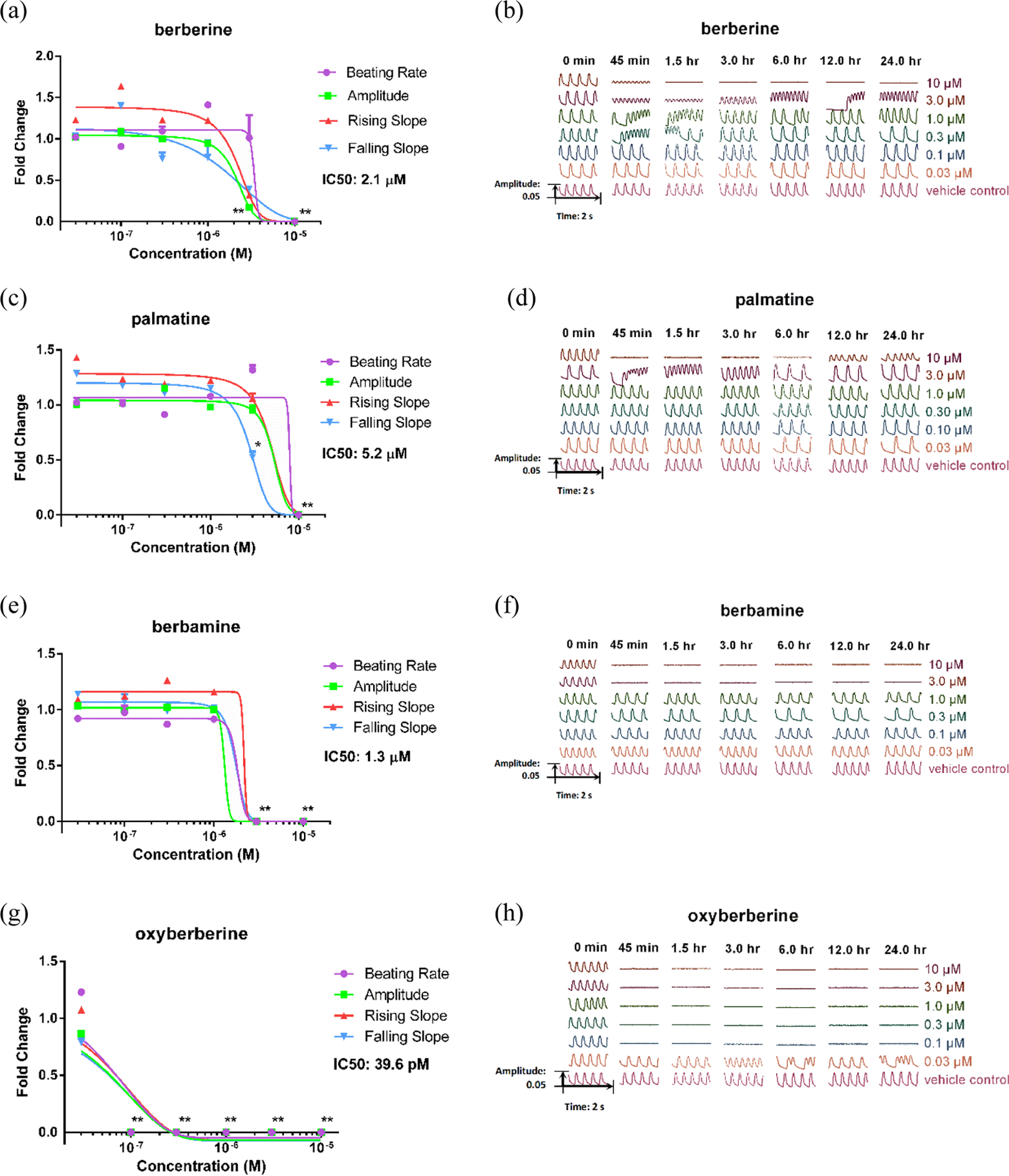
Detailed parameters and beating patterns in the presence of berberine, palmatine, berbamine, and oxyberberine. Four CM beating parameters, including beating rate, amplitude, raising slope, and falling slope, were used for evaluation 1.5 h after compounds treatment. (a) Dose response of CMs to berberine treatment. (b) Representative beating traces captured at selected time points (0, 0.75, 1.5, 3.0, 6.0, 12.0, and 24.0 h) treated by various doses (0.03, 0.1, 0.3, 1, 3, and 10 μM) of berberine. (c) Dose response of CMs to palmatine treatment. (d) Representative beating traces treated by various dose of palmatine. (e) Dose response of CMs to berbamine treatment. (f) Representative beating traces treated by various dose of berbamine. (g) Dose response of CMs to oxyberberine treatment. (h) Representative beating traces treated by various dose of oxyberberine. Data were expressed as mean ± SD, n = 3. *p < 0.05; **p < 0.01. CM: cardiomyocyte.
Morphological changes observed following berbamine and oxyberberine treatment
To validate the cardiotoxicity of the four alkaloids analyzed by RTCA cardio system, morphological changes of CMs were detected by HCA. Alpha actin were staining following 24 h treatment with different concentrations (0.03, 0.1, 0.3, 1.0, 3.0, and 10.0 μM) of each compounds. Cardiac and muscle cell actin were stained by alpha actin, serving as a specific marker for CMs. Berbamine- and oxyberberine-treated (10 μM) cells demonstrated an elongated cellular morphology as well as condensed nuclei, and even apoptosis, which were similar to amiodarone at 10 μM as the positive control. Both of these morphological changes may be indicative of drug-induced cardiotoxicity. In contrast, berberine- and palmatine-treated CMs did not show significant morphological changes (Figure 4).

Representative images of the HCA analysis of CMs for the evaluation of morphology and cell count. (a) Typical images of CMs treated with 0.1% DMSO, amiodarone, and each alkaloids in 10 μM for 24 h. The cell nucleuses were stained by Hoechst 33342, overall cell morphology stained by alpha actin. Changes were indicated by white arrow. (b) Cell count under a various dose (0.03, 0.1, 0.3, 1, 3, and 10 μM) of berberine, palmatine, berbamine, and oxyberberine. Data were expressed as mean ± standard deviation, images (×10) were representative of n = 3 experiments. **p < 0.01. HCA: high-content analysis. CM: cardiomyocyte. DMSO: dissolved in dimethyl sulfoxide.
The cell count in the various dose of the four alkaloids was analyzed. Berbamine and oxyberberine at 10 μM made the cell number decline comparing with the vehicle control group (p < 0.01).
Effect of RC alkaloids in RTCA and ATP depletion cytotoxicity assay
To evaluate cytotoxicity of the nine alkaloids, cell index was detected continuously for more than 20 h, which reflected the cell viability of CMs. The changes of cell index were normalized to that of the time-matched vehicle control group. The compounds were prepared at 10 μM in the primary screening, and amiodarone was used as the positive control. Cell index declined with treatment of berbamine, oxyberberine, and positive control, whereas that of the other seven alkaloids were similar to the vehicle control group.
To validate the cell viability of berbamine and oxyberberine, a luciferase coupled adenosine triphosphate (ATP) quantitation assay was used in the test. The cell ATP depletions were extremely obvious in treatment of berbamine at 10.0 μM (p < 0.05) as well as oxyberberine at 3.0 μM and 10.0 μM (p < 0.01; Online Supplementary Figure S2). These consequences were consistent with the results of RTCA assay. Two heat maps were provided to summarize the results of the cardiotoxicity evaluation (Figure 5).

The summary of the cardiotoxicity based on functional and structural evaluation. (a) Primary screening of the nine alkaloids by RTCA assay. The final concentration of compounds in the primary screening was 10 µM. (b) Cardiotoxicity and cytotoxicity evaluation of the four alkaloids by RTCA, HCA, and ATP depletion. RTCA: real-time cellular analysis. HCA: high-content analysis. ATP: adenosine triphosphate.
Discussion
To date, there is no research illuminating the cardiotoxicity of RC and its major constituents. Our study reports the potential cardiotoxic ingredients in RC with convincing evidence for the first time. Berberine is an isoquinoline alkaloid isolated from the plants of RC and Berberis, which has been used to treat diarrhea and gastroenteritis. 36,37 Previous studies reported that concomitant use of berberine with macrolides may require close monitoring because of potential drug toxicities, especially cardiotoxicity. 38 It was proved to block the open state of the human ether-à-go-go-related gene (hERG) channel with an IC50 of 3.1 ± 0.5 μM and decrease cytochrome P450 (CYP) 3A activities. 39 In our study, the IC50 of berberine on CMs amplitude was 2.1 μM, which was close to the previous study. Palmatine has a similar chemical structure to berberine, which is also an isoquinoline alkaloid existing in numerous medicinal plant species, such as RC and Phellodendron amurense. Previous studies have revealed the carcinogenesis of palmatine. Palmatine significantly increased the activity of CYP1A1 gene promoter, which was involved in metabolic activation of various procarcinogens. 40 Potent deoxyribose nucleic acid damage was induced by palmatine in human HepG2 cells through suppressing the activities of both topoisomerase I and II. 41 However, the cardiotoxicity of palmatine has always been neglected until our research. In the present study, palmatine as well as berberine showed cardiotoxicity in a dose- and time-dependent manner. In RTCA analysis, beating rate and amplitude of CMs were totally inhibited by berberine and palmatine at 10 μM. However, the morphology of CMs did not show significant change at the same concentration as well as cell count analyzed by HCA. The results indicated that CMs were out of the physiological function of contraction and relaxation, while keeping the cell morphology in the treatment of the two alkaloids. Actually, berberine and palmatine have been used in clinic application. For instance, berberine hydrochloride has been used in the treatment of diarrhea 42 and hydrochloride palmatine has been used in photodynamic therapy on colon adenocarcinoma. 43 Berberine was regarded as the main toxic component in RC all the time. In our study, palmatine was proved to be cardiotoxic, which takes the third largest proportion in total alkaloids in RC. The experimental evidences may provide references to the security application of RC and the two alkaloids in cardiovascular disease.
Most of the studies of berbamine were designed to look for its beneficial effects, whereas rarely researches focused on the potential toxicity. Berbamine, a natural bis-benzylisoquinoline alkaloid, was identified from Berberis and used for the treatment of cancers and leukopenia. 44,45 It is worth mentioning that the concentration of berbamine suppressing the growth of A549 cells, the adenocarcinomic human alveolar basal epithelial cells, was higher than 10 μM in the previous researches. 46,47 In the present study, the IC50 value of berbamine inhibiting CMs amplitude was 1.3 μM. More attention should be paid to the cardiotoxicity of berbamine and its clinical adverse reactions. Oxyberberine was synthesized by air oxidation of berberine under basic conditions, with inhibitory dose (ID50) was 12 μM in vitro experiment. 48 In current study, the results showed that berbamine and oxyberberine exhibited obvious cardiotoxicity on CMs with unrecoverable cardiac arrest within 24 h. Shrinkage and detachment of CMs were observed at 10 μM in morphologic assay. Cell count was significantly lower than that of the vehicle control group. Moreover, cytotoxicity was induced by berbamine and oxyberberine with cell index decline and ATP depletion. The other seven alkaloids did not show cytotoxicity in the screening. The results demonstrated that berbamine and oxyberberine can make CMs out of the physiological function of systolic and diastolic, and even lead to death. The toxicity of the two alkaloids is always ignored for their small proportions in RC. However, RC is often overdosed in TCM decoctions in practice. Consequently, the toxicity of the alkaloids with tiny content in RC is correspondingly amplified. The results in the present study demonstrated that the ADRs caused by RC may relate to the toxicity of berbamine and oxyberberine.
As mentioned before, there are a variety of ADRs resulted from the applications of RC and the alkaloids. In previous studies, the toxicity of RC was attributed to the toxicity of berberine. However, the present study clearly demonstrated that palmatine, berbamine, and oxyberberine in RC exhibited cardiotoxicity as well. Meanwhile, berbamine and oxyberberine showed cytotoxicity. Accordingly, we presumed that the cardiac ADRs resulted from RC may attribute to berberine, palmatine, berbamine, and oxyberberine in it. The results of the present study provided preliminary evidence to improve the safety of medication. Further studies are needed to demonstrate the cardiotoxic mechanism of alkaloids in RC. Berberine was proved to reduce hERG membrane stability by disrupting of cavoline-1 and binding to the residues, aromatic tyrosine (Tyr652), and phenylalanine (Phe656) 49 , although the cardiotoxicity of other alkaloids in RC have not been reported before. Due to its similar structure to berberine, we hypothesized that the cardiotoxic mechanism of palmatine may be related to hERG potassium channel and its membrane expression. More future researches are required to validate these conjectures. Moreover, the method to attenuate the toxicity of them should be explored. As the drug tolerance with RC and alkaloids of people are different, individual prescription needs to be formulated based on more pharmacodynamics and toxicity experiments.
Conclusion
In this study, berberine, palmatine, berbamine, and oxyberberine showed cardiotoxicity, which resulted in arrhythmia, even cardiac arrest with time and dose dependency. Cytotoxicity of berbamine and oxyberberine manifested as morphologic shrinkage and ATP depletion of CMs. Coptisine, berberrubine, epiberberine, magnoflorine and choline manifested no cardiotoxicity or cytotoxicity in the screening. This research provided experimental evidences to clarify the toxic components of RC and an available method to evaluate the safety of drugs in vitro.
Footnotes
Authors’ note
SYL and HJY conceived and designed the project. MYZ designed and performed the experiments and wrote the article. YYY, SFW, and QZ participated in the experiment. HWW, JYW, and WY contributed to acquiring compounds and materials. SYL edited and revised the manuscript.
Acknowledgement
The authors are grateful to ACEABio Co., Ltd (Hangzhou, China) for technical assistance with the RTCA cardio system.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the State Key Program of National Natural Science Foundation of China (grant no. 81330086) and the National Program on Key Basic Research Project (973 Program) (grant no. 2015CB554406).
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
