Abstract
Objective:
Organophosphate insecticide (OPI) self-poisoning is a major medical problem in many countries. Several studies have demonstrated that the base deficit (BD) is a prognostic tool that is correlated with the severity of injury and predicted mortality, particularly in trauma patients. Here, we aimed to investigate the prognostic significance of BD in OPI poisoning.
Methods:
This retrospective observational study was conducted between January 1, 2006, and January 31, 2015, at a single emergency department (ED). The BD values were divided into quartiles according to the number of patients: 3 mEq/L or less, 3–5.9 mEq/L, 6–9.9 mEq/L, and 10 mEq/L or greater. Survival at 30 days from ED admission was estimated using the Kaplan–Meier survival analysis.
Results:
Among 154 patients, 31 died, yielding a mortality of 20.1%. The highest BD quartile (≥ 10 mEq/L) and the 6–9.9 mEq/L group were associated with an increased risk of 30-day mortality. Patients with a BD of 10 mEq/L or greater had a 5.85-fold higher risk of 30-day mortality and patients with a BD of 6–9.9 mEq/L had a 5.40-fold higher risk of 30-day mortality compared to patients with a BD of 3 mEq/L or less. The area under the curves of the BD and the Acute Physiology and Chronic Health Evaluation II score for mortality were 0.748 (95% confidence interval (CI), 0.660–0.835) and 0.852 (95% CI, 0.789–0.915), respectively.
Conclusions:
This study showed that the BD is a predictor of 30-day mortality in patients with OPI poisoning.
Introduction
Organophosphate insecticides (OPIs) are among the most widely used insecticides in agricultural areas worldwide. According to the 2013 American Association of Poison Control Centers annual report, nearly 3000 cases of OPI exposure are reported annually in the United States. 1 As OPI agent usage is phasing out, OPI exposure has declined in the United States. However, OPI poisoning in developing countries is still problematic. 2,3 Reliable prognostic factors have been identified for predicting the outcomes of OPI poisoning, including the poisoning severity score, the Glasgow Coma Scale (GCS), the Acute Physiology and Chronic Health Evaluation II (APACHE II) score, C-reactive protein levels, and electrocardiogram recordings (e.g. prolongation of heart rate-corrected QT interval). 4 –7
Base deficit (BD) represents tissue hypoperfusion and hypoxia and is defined as the amount of strong acid or base required to normalize the pH of 1 L of whole blood to 7.4, assuming a temperature of 37°C and a partial pressure of carbon dioxide of 40 mmHg. This assay was developed by Anderson and Engel in 1960 and is still used to measure metabolic acid–base activity. 8 Several studies have demonstrated that BD is a prognostic tool that correlates with the severity of injury and predicts mortality in trauma patients. 9 –13 Related to the latter, BD has also been identified as a prognostic factor associated with mortality in cases of medical illness. 14–15
In light of these finding, we hypothesized that BD is a reliable prognostic factor in patients with pesticide poisoning. Therefore, the aim of this study was to determine the prognostic significance of BD in OPI poisoning.
Methods
Study setting
This retrospective observational study was conducted between January 1, 2006, and January 31, 2015, at a single emergency department (ED) in an 800-bed academic hospital with an annual ED census of 30,000 patients. This study followed the Declaration of Helsinki and was approved by the institutional review board at Gyeongsang National University Hospital, with an exemption from informed consent requirements. The database was derived from and based on our previous study, which showed that red blood cell distribution width was a predictor of mortality in OPI poisoning. 16 Since the publication of those results, we have continued compiling data in the OPI poisoning database. Furthermore, we reviewed the electronic charts of patients who were treated prior to the previous study to record additional cases in the database.
We analyzed 154 patients with a diagnosis of OPI poisoning who were at least 16 years of age. OPI poisoning was diagnosed through the patients’ clinical history of intentional ingestion of OPIs and specific signs and symptoms of OPI poisoning, such as salivation, lacrimation, urination, defecation, gastric emesis, bronchorrhea, bronchospasm, and bradycardia. Of 202 total patients, 154 were included in this study. Forty-eight patients were excluded, including 16 patients who co-ingested other agents, 11 patients who were transferred to other hospitals, 12 patients who were discharged against medical advice, 6 patients who experienced prehospital cardiac arrest, and 3 patients who were unintentionally poisoned.
At the ED, treatment for OPI poisoning begins with airway management and adequate oxygen. Endotracheal intubation and mechanical ventilation are performed when patients have moderate-to-severe poisoning and either respiratory or hemodynamic compromise. Decontamination with gastric lavage and activated charcoal administration is conducted when indicated. Antidotal therapy with atropine and pralidoxime is administered in every case of acute OPI poisoning. 17
Data collection
All patient-related data were obtained from a retrospective chart review. Demographic variables (e.g. age and gender), initial vital signs, and GCS data were collected. The initial laboratory findings, including tests for arterial pH, HCO3, white blood cell (WBC) count, hematocrit, platelet count, levels of blood urea nitrogen (BUN), creatinine, albumin, creatinine kinase, and butyrylcholinesterase, were obtained. The arterial blood gas test including arterial pH, HCO3, and BD were determined using the Siemens® Rapid lab 1265 blood gas analyzer (Siemens, Germany). Data related to atropine usage and intubation duration were also collected. Initial APACHE II scores of patients in the ED were also calculated using values from 12 routine physiological measurements, including initial vital signs, laboratory findings, patient age, and previous health status. The primary outcome of this study was hospital mortality within 30 days after ED admission for OPI poisoning. The secondary outcome was a comparison of the predictive accuracy of patients’ BD and APACHE II scores for 30-day hospital mortality.
Statistical analysis
Continuous variables were expressed as the mean with standard deviation and were compared using the student’s t-test. Categorical variables were described as percentages based on their frequency of occurrence and were compared using the χ 2 test. The BD values were divided into quartiles according to the number of patients: 3 mEq/L or less (n = 40), 3–5.9 mEq/L (n = 34), 6–9.9 mEq/L (n = 37), and 10 mEq/L or greater (n = 43). Thirty-day survival (from ED admission) was estimated using the Kaplan–Meier survival analysis, and comparisons of the BD quartiles were performed using the log-rank test. Univariate and multivariate Cox proportional hazards regression analyses were applied to investigate the predictors of 30-day hospital mortality. Selected variables in the univariate analysis (p < 0.1) were included in the multivariate model. Receiver–operator characteristic (ROC) curves were drawn, and the area under the curves of the BD and the APACHE II scores for mortality were calculated. Furthermore, the optimal cut-off points of the BD and the APACHE II scores for 30-day hospital mortality were determined using the Youden’s J statistic and ROC curves. All of the p values were two sided, and p < 0.05 was considered to be statistically significant. We used STATA 12.0 software (Stata Corporation, College Station, Texas, USA) to conduct all of the statistical analyses.
Results
Thirty-one (20.1%) of 154 patients died within 30 days. The causes of death were pneumonia (9, 29%), acute renal failure (5, 16%), respiratory arrest (4, 12%), cerebral vascular accident (2, 6%), or unknown (11, 35%). Table 1 summarizes the baseline demographics and clinical characteristics of the study subjects stratified by BD quartile. GCS, systolic blood pressure, arterial pH, HCO3 levels, WBC count, BUN level, creatinine, and APACHE II scores differed significantly. In addition, 30-day hospital mortality increased concomitantly with the increase in BD quartile. The Kaplan–Meier survival analysis with a log-rank test indicated that increased BD quartiles were associated with a significantly higher risk of 30-day hospital mortality (p < 0.001; Figure 1).
Patient characteristics stratified by BD quartile.a
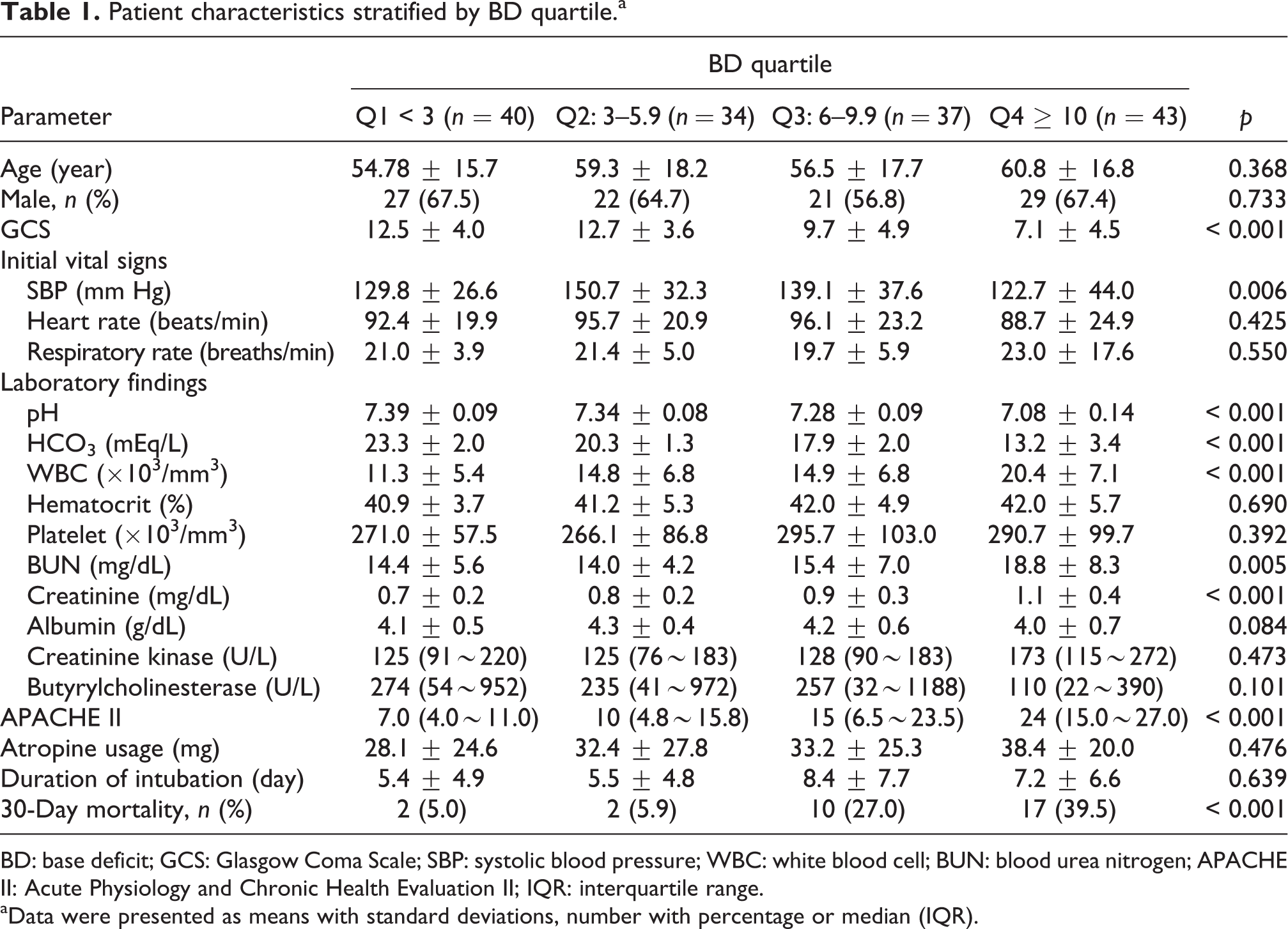
BD: base deficit; GCS: Glasgow Coma Scale; SBP: systolic blood pressure; WBC: white blood cell; BUN: blood urea nitrogen; APACHE II: Acute Physiology and Chronic Health Evaluation II; IQR: interquartile range.
aData were presented as means with standard deviations, number with percentage or median (IQR).

Kaplan–Meier survival curve of patients with OPI poisoning. p < 0.001 by the log-rank test.
The univariate and multivariate Cox regression analyses are shown in Table 2. In the multivariate analysis, the 6–9.9 mEq/L quartile and the highest BD quartile (≥ 10) were associated with an increased risk of 30-day hospital mortality. Compared with patients with a BD of 3 mEq/L or less, those with a BD of 6–9.9 mEq/L had a 5.40-fold higher risk of 30-day hospital mortality and those with a BD of 10 mEq/L or greater had a 5.85-fold higher risk of 30-day hospital mortality. In addition, the continuous values of BD levels and the probability of 30-day hospital mortality are presented by locally weighted scatterplot smoothing (LOWESS) (Figure 2). The optimal cut-off points of the BD and APACHE II scores for predicting 30-day hospital mortality were identified from the ROC curve analysis. The cut-off point for BD was more than 7.4, and the area under the ROC curve was 0.748 (95% confidence interval (CI), 0.660–0.835). When the BD was more than 7.4, the sensitivity was 87.1% and the specificity was 66.7%. The cut-off point for APACHE II scores was more than 17.0, and the area under the ROC curve was 0.852 (95% CI, 0.789–0.915). When the APACHE II score was more than 17.0, the sensitivity was 87.1% and the specificity was 71.5% (Figure 3).
Cox regression analysis of risk factors for mortality within 30 days.

HR: hazard ratio; CI: confidence interval; GCS: Glasgow Coma Scale; SBP: systolic blood pressure; WBC: white blood cell; BD: base deficit.
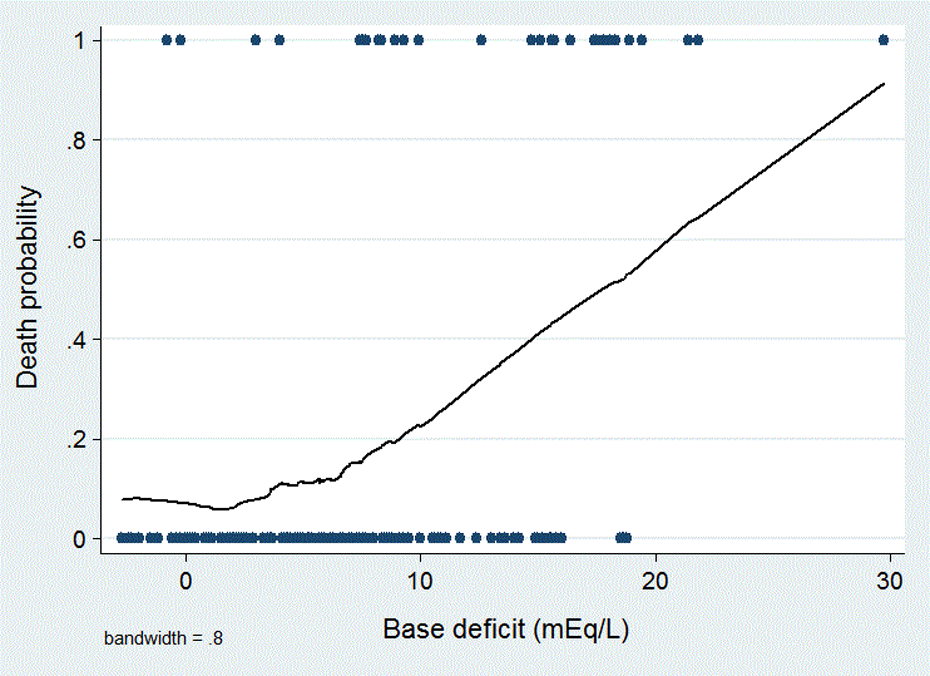
Linear relationship of BD levels and the probability of 30-day mortality in LOWESS analysis. BD: base deficit. LOWESS: locally weighted scatterplot smoothing.

Comparison of area under the ROC curve for predicting 30-day mortality during OPI poisoning at BD and APACHE II. ROC: receiver–operator characteristic; OPI: organophosphate insecticide; BD: base deficit; APACHE II: Acute Physiology and Chronic Health Evaluation II.
Discussion
In the present study, we observed that BD of nonsurvivors was higher than that of survivor in patients with OPI poisoning, and there was a graded association between BD and 30-day mortality. In addition, BD had a relationship with the severity of the patients and was a prognostic factor of 30-day mortality in patients with OPI poisoning.
BD is defined as the amount of base requires to return the blood pH to homeostatic levels. The normal range of BD is −2 to +2 mEq/L, and a high BD, along with lactate, correlates with metabolic acidosis, hypoperfusion, and oxygen debt. BD has been found to be a prognostic risk factor in patients with multiple traumas and burns. 9 –13,18,19 Furthermore, BD has also been used to guide volume resuscitation and to predict transfusion requirements and complication risks in patients with trauma. 20,21
Annually, the number of mortalities due to OPI poisoning has been reported to be approximately 200,000 cases worldwide. 22 This high number of deaths presents a major public health problem in developing countries. The effects of OPI poisoning include severe cholinergic toxicity due to the accumulation of acetylcholine (ACh) induced by overstimulation of the cholinergic nerve terminals. This concentration increase leads to the irreversible inhibition of acetylcholinesterase. The clinical features of OPI poisoning present as symptoms and signs of muscarinic, nicotinic, and central nervous system (CNS) effects. The muscarinic effects are described using the following two mnemonics; SLUDGE (salivation, lacrimation, urination, defecation, gastrointestinal motility, and emesis) and DUMBELS (diaphoresis/diarrhea, urination, miosis, bronchorrhea/bronchospasm/bradycardia, emesis, lacrimation, and salivation). The nicotinic effects include muscle weakness, cramps, fasciculation, and paralysis, whereas the CNS effects present as anxiety, confusion, tremor, seizure, and coma.
The primary cause of death due to OPI poisoning is believed to be respiratory insufficiency resulting from a combination of pulmonary cholinergic effects (e.g. bronchoconstriction and massive bronchial secretions), nicotinic effects leading to respiratory muscle weakness, and CNS cholinergic effects that inhibit the respiratory centers. 23 In addition to respiratory failure, OPI poisoning can also induce cardiac toxicity, including heart blocks and heart rate-corrected QT interval prolongation. 24 The mechanisms of cardiac toxicity by OPI poisoning are not fully understood. However, possible mechanisms are related to increased sympathetic and parasympathetic activity, hypoxemia, acidosis, electrolyte abnormalities, and direct myocardial damage induced by OPI compounds. 25 In addition, lethal arrhythmias could be induced by atropine itself, which is critical in the treatment of OPI poisoning. 26
Another mechanism of severe OPI poisoning, reduced systemic peripheral vascular resistance, induces lethal distributive shock, which has the same pathological progression observed in the cases of severe sepsis or septic shock. 27 A decrease in systemic vascular resistance can develop from excessive ACh, which stimulates muscarinic receptors and dilates peripheral vessels. When ACh binds to muscarinic receptors in the arterial endothelium, nitric oxide (NO)-dependent and NO-independent vasodilation pathways are activated.
Alterations in the acid–base equilibrium are also caused by OPI poisoning. Systemic arterial pH is maintained within the range of 7.35–7.45 by extracellular and intracellular chemical buffering along with respiratory and renal regulatory mechanisms. The metabolic and respiratory components that regulate systemic pH are described by the Henderson–Hasselbalch equation. BD may be used as a substitute to detect metabolic acidosis because it has a strong linear correlation with serum HCO3. 28 Many authors have reported that OPI poisoning contributes to cardiac toxicity with ECG abnormalities and respiratory distress, which may result in hypotension or hypoperfusion as well as an electrolyte imbalance. These factors might be the primary source of metabolic acidosis and respiratory acidosis in acute OPI poisoning. Acidosis is a major predisposing factor that influences the health outcomes of patients with OPI poisoning. 29 The metabolic acidosis that develops secondary to OPI poisoning is corrected with sodium bicarbonate infusion, which in turn improves the protective effects of atropine and oxime therapy. The increase in blood pH resulting from the sodium bicarbonate treatment might also increase the hydrolysis of the ester moiety of the organophosphate molecule, thus decreasing its toxicity. 30
The APACHE II uses a score based on the initial values of 12 routine physiological measurements, including arterial pH, age, and previous health status, thereby providing a general measure of disease severity. An increasing score (range 0–71) closely correlates with mortality. 31 Additionally, BD may be used as a substitute to detect metabolic acidosis, since a relationship exists between BD and APACHE II scores with regard to mortality.
Thirty-one (20.1%) of the 154 patients died within 30 days of their ED admission due to various causes. Acute OPI poisoning is a highly fatal condition that respiratory disturbance and hemodynamic complications. Additionally, patients with severe OPI poisoning need to be treated with long-term mechanical ventilation. For these reasons, patients with OPI poisoning died from various health complications.
Our study focused on patients with OPI poisoning and provided support for the utilization of BD level at admission in initial patient management. In our analysis, higher BD values were associated with an increased risk of 30-day hospital mortality in patients with OPI poisoning.
Limitations
Some limitations of the present study must be acknowledged. First, our study was a retrospective review potentially limited by selection bias. Therefore, to eliminate potential selection bias, the study excluded patients who co-ingested other agents, were transferred to other hospitals, were discharged against medical advice, or experienced either prehospital cardiac arrest or unintentional poisoning. Additionally, this study is limited by its relatively small sample size. Therefore, large prospective multicenter studies should be considered in the future. Second, the amount of time between the patients’ poisoning and the intervention may have influenced our findings, as this time was not considered in this study. Third, we used only the initial recording of patients’ BD, which was measured upon admission to the ED, but subsequent BD measurements after the intervention were not collected in the study. At the present time, these subsequent BD measurements would be difficult to study because BD is not consistently measured; patients with severe OPI poisoning are often measured, while patients with a low level of poisoning are rarely measured. Patients who die within a short time after OPI poisoning are not measured. Additionally, subsequent values of BD are affected by sodium bicarbonate treatment. Fourth, this study did not consider treatments using endotracheal intubation, mechanical ventilation, or metabolic acidosis management. Furthermore, patients in the study were not treated by the same physician. However, the treatments were performed according to appropriate protocols by trained physicians. Although treatment modalities could greatly influence mortality, the initial BD value is important with regard to the patient’s clinical condition, and future studies should focus on the association between mortality in patients with OPI poisoning and corrected BD levels. Finally, our study did not consider long-term respiratory distress resulting from muscarinic effects, as respiratory distress may influence the acid–base balance. Despite these limitations, we are confident that BD could be a reliable prognostic factor in patients with OPI poisoning.
Conclusion
Acute OPI poisoning is a highly fatal condition that requires a rapid and precise diagnosis and adequate support and treatment. BD could be a reliable prognostic factor in patients with OPI poisoning because it is associated with an increased risk of 30-day hospital mortality in patients with OPI poisoning. Higher numerical values of BD in patients with OPI poisoning require quick and precise intensive care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
