Abstract
Objective:
This study was designed to evaluate prolonged methylprednisolone (MP) treatment after pulse therapy for paraquat (PQ)-intoxicated rats.
Method:
Acute PQ toxicity was induced by intraperitoneally injecting single toxic dose of 25 mg/kg of body weight. Rats were divided into four groups: control group (saline solution for 15 days), PQ group (saline solution for 15 days after PQ toxicity), pulse group (15 mg·kg−1·day−1 MP for 3 days after PQ toxicity and then saline solution for 12 days) and pulse + prolonged group (15 mg·kg−1·day−1 MP for 3 days after PQ toxicity; dosage was subsequently reduced by half every 2 days, and MP was terminated until 0.47 mg·kg−1·day−1). Hydroxyproline (HYP) content in lung tissues was evaluated through enzyme-linked immunosorbent assay, and lung fibrosis was examined using a semiquantitative scoring system (Ashcroft staging criteria). Lung wet-to-dry weight (W/Dc) ratio and 15-day survival rates of the rats were also analysed.
Results:
Similar survival rates (55.0 vs. 65.0%) were obtained for the pulse group and the pulse + prolonged group. The W/Dc (4.79 ± 0.42 vs. 5.29 ± 0.35), HYP content in the lung tissues (3.23 ± 0.24 vs. 3.72 ± 0.23 μg/mg) and lung fibrosis scores (2.69 ± 0.74 vs. 3.12 ± 0.60) of the pulse + prolonged group were lower than those of the pulse group.
Conclusion:
Prolonged MP treatment after pulse therapy could effectively ameliorate PQ-intoxicated acute lung injury in rats. However, further studies should be performed to verify our findings.
Introduction
Paraquat (PQ) is a strong and widely used herbicide that has caused thousands of deaths worldwide, particularly in agrarian and undeveloped countries. This substance is also a highly toxic compound for humans and animals, and many cases of acute poisoning and death have been reported. PQ poisoning is alleviated through several management strategies, including extracorporeal elimination and immunosuppressant and antioxidant treatments. 1,2 However, antidotes or effective therapies have yet to be developed to help avoid serious lung injury and death. The cytotoxic effects of PQ are caused by the generation of superoxide radicals that stimulate inflammation. Thus, the possible lethal effects of PQ may be prevented by inhibiting inflammation.
Case reports 3 –5 and non-randomized trials with a small sample size 6,7 in patients with severe PQ poisoning have indicated a high efficacy of early large-dose methylprednisolone (MP) combined with prolonged glucocorticoid administration. Although prolonged therapy has provided potential benefits, spontaneous improvement has been rarely achieved in the cases of PQ ingestion, especially at a sublethal dose. With insufficient studies in this field, the beneficial effects of its prolonged administration remain unknown.
Therefore, this study aimed to evaluate the prolonged treatment after pulse as a potential therapy for the alleviation of PQ-induced lung injury in rat models. This study also aimed to observe its potential therapeutic effects on several indexes, such as survival rates, wet-to-dry weight (W/Dc), hydroxyproline (HYP) content and lung fibrosis score.
Materials and methods
Chemicals
HYP was purchased from Sigma-Aldrich Chemical Company (St. Louis, Missouri, USA). PQ was provided by Syngenta Chemical (Jiangsu, China). MP was obtained from Pfizer, Inc. (New York, New York, USA).
Animals
Eighty Wistar rats, weighing 200–250 g, were purchased from the Experimental Animal Centre of Cangzhou Medical College and provided with free access to food and water. The animals were kept under standard conditions (temperature, 23 ± 2°C; 12-h light/12-h dark cycle) and acclimatized to our laboratory environment for 7 days. The whole study was approved by the Ethics Committee of Cangzhou Central Hospital, China. Treatments and care were performed in accordance with the recommendations of the Cangzhou Central Hospital Committee Guidelines, which comply with the international guidelines for the use of experimental animals.
Experimental design
Acute poisoning was induced in rats by intraperitoneally injecting single toxic dose of 25 mg/kg 8,9 of body weight of PQ in 1 ml of saline, and the rats received saline or MP 1 h after PQ toxicity was induced. Because of encouraging reports of the use of pulse therapy with MP 1 g/day for 3 days in PQ-poisoned patients, 10 –13 MP pulse therapy in current experiment is defined as 15 mg·kg−1·day−1 for 3 days. Considering that drug therapy withdrawal rapidly negates the positive influence of glucocorticoids 14 and aggravates lung damage after glucocorticoid treatment is terminated, 3 we treated the rats with MP through tapered-dose prolonged administration after pulse therapy 15 : 15-mg·kg−1·day−1 MP was administered for 3 days and then reduced by half every 2 days; MP treatment was terminated until 0.47 mg·kg−1·day−1. Wistar rats were randomly divided into four groups and treated on the basis of their body weight. In the control group (n = 20), the rats received 1-ml/day saline for 15 consecutive days. In the PQ group (n = 20), the rats were given 1-ml/day saline for 15 consecutive days post-PQ toxicity. In the pulse group (n = 20), the rats were treated with 15 mg·kg−1·day−1 MP in 1 ml of saline for 3 consecutive days post-PQ toxicity. They were then administered with 1-ml/day saline for 12 consecutive days. In the pulse + prolonged group (n = 20), the rats received 15-mg·kg−1·day−1 MP in 1 ml of saline for 3 consecutive days post-PQ toxicity. The dosage was then reduced by half every 2 days, and MP treatment was terminated until 0.47 mg·kg−1·day−1. The rats were killed 15 days post-PQ toxicity.
Tissue collection
After 15 days, the surviving rats were anesthetized with pentobarbital sodium (30-mg/kg body weight, intraperitoneally). After the rats were subjected to midline sternotomy, their whole lung was dissected and washed with ice-cold saline to remove blood. The left lung lobes were transferred to 10% formalin solution for histopathological studies, and the right upper lung tissues were used for W/Dc ratio assay. The right lower lung tissues were frozen in liquid nitrogen for biochemical measurement.
W/Dc ratio assay
After blood and other contaminants were blotted, the wet weight of the right upper lung tissues was measured. The lungs were then dried in an oven at 80°C for 72 h, and the W/Dc ratio was used to quantify the water (H2O) content of the lungs.
Measurement of HYP content
The HYP content of the lung tissues was determined by a previously described method. 16 In brief, 50 mg of lung tissues from each rat were hydrolysed with 1 ml of 6 N hydrochloric acid (HCl) by heating at 110°C for 12 h. The hydrolysed tissues were maintained at 60°C in beakers until HCl was completely evaporated. The tissues were then reconstituted in 20 ml of H2O, and pH was adjusted to 7.0 with sodium hydroxide. Standard samples (100 μl) were added to a 96-well plate. HYP oxidation was initiated by adding 50 μl of chloramine-T (0.05 M). The plate was shaken and then incubated for 20 min at room temperature. To develop colour, we added 50 μl of p-dimethylaminobenzaldehyde solution (Sigma-Aldrich Chemical Company) to each well and then incubated the plate at 60°C for 20 min. After cooling, we determined the absorbance at 550 nm using a spectrophotometer.
Histological studies
The left lung lobes were transferred to 4% paraformaldehyde solution for 24 h, embedded in paraffin, sliced to obtain butterfly shaped sections with 5-μm thickness and placed on slides stained with haematoxylin and eosin for histopathological analysis. The degree of fibrosis in randomly selected 0.5-mm2 alveolar regions was analysed using a semiquantitative scoring system from 0 (normal lung) to 8 (most severe fibrotic lung), as previously reported by Ashcroft et al. 17 Two pathologists blind to the study groups performed all the histological examinations.
Data analysis
Data were statistically analysed using SPSS version 20.0 (SPSS Inc., Chicago, Illinois, USA). The lung fibrosis score and HYP content were examined through Mann–Whitney U test and non-parametric statistical tests. W/Dc was examined through analysis of variance followed by Dunnett’s post hoc t-test. Data were presented as mean ± standard deviation, and statistical significance was considered at p < 0.05.
Results
Survival rate of rats
Figure 1 shows the cumulative survival of the four groups for the 15-day experiment. At 15 days post-PQ intoxication, the survival rates were 100, 25, 55 and 65% in the control, PQ, pulse and pulse + prolonged groups (Figure 2(a)). The survival rates in the pulse + prolonged group were similar to that in the pulse group (p > 0.05).

Percentage of rat survival in the control, PQ, pulse and pulse + prolonged groups. PQ: paraquat.
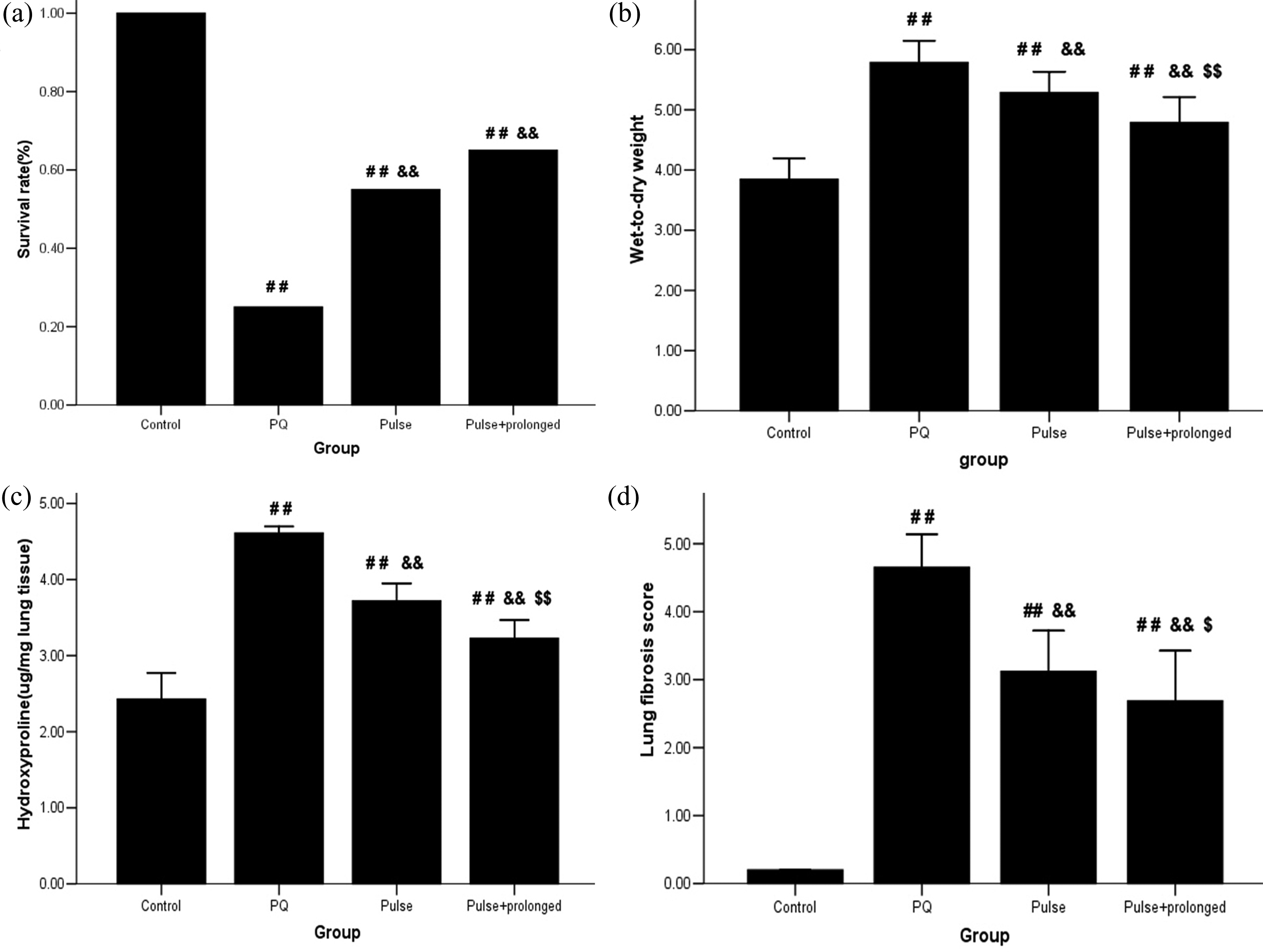
Effect of MP on survival rate, wet-to-dry weight ratio, hydroxyproline content and lung fibrosis score. # p < 0.05 and ## p < 0.01 compared with the control group; & p < 0.05 and && p < 0.01 compared with the PQ group; $ p < 0.05 and $$ p < 0.01 compared with the pulse group. PQ: paraquat. MP: methylprednisolone.
W/Dc ratio
After 15 days of treatment, the W/Dc ratio was significantly decreased by MP treatment. In particular, the W/Dc ratios of the pulse + prolonged group (p < 0.01) and the pulse group (p < 0.01) were 4.79 ± 0.42 and 5.29 ± 0.35, respectively. These values were significantly lower than that (5.79 ± 0.36) of the PQ group (Figure 2(b)). By comparison, the W/Dc ratio in the pulse + prolonged group was lower than that in the pulse group (p < 0.01).
HYP content in lung tissues
Lung fibrosis was assessed by measuring the HYP content in the lungs to indicate collagen accumulation. The HYP contents of the four groups are shown in Figure 2(c). The HYP content in the pulse + prolonged group was 3.23 ± 0.24 μg/mg, which was significantly lower than that of the PQ group (4.61 ± 0.09 μg/mg; p < 0.01) and the pulse group (3.72 ± 0.23 μg/mg; p < 0.01).
Histological studies
The lung tissues from each study group were histologically examined to evaluate the severity of PQ-induced lung fibrosis. After the rats were treated with MP for 15 days, the lung fibrosis score was significantly decreased by MP treatment. The following scores were obtained: 2.69 ± 0.74 in the pulse + prolonged group (p < 0.01), 3.12 ± 0.60 in the pulse group (p < 0.01) and 4.65 ± 0.49 in the PQ group (Figure 3). The lung fibrosis score in the pulse + prolonged group was significantly lower than that in the pulse group (p < 0.05).

Histological evaluation of H&E-stained rat lung specimens in the (a) control, (b) PQ, (c) pulse or (d) pulse + prolonged groups 15 days post-PQ injection (100× magnification). PQ: paraquat. H&E: haematoxylin and eosin.
Discussion
To the best of our knowledge, this study is the first to evaluate the beneficial effects of prolonged treatment after pulse therapy on PQ poisoning and a novel tapered-dose prolonged drug delivery method of MP. The survival rate of the pulse group was similar to that of the control group. The W/Dc, HYP content in lung tissues and lung fibrosis score of the former were lower than those of the latter. By comparison, these parameters were improved and more evident in the pulse + prolonged group, which exhibited a higher survival rate.
Chen et al. 18 compared three doses (30 mg/kg MP for 3 days) and one dose (30 mg/kg MP for 1 day) of treatment against PQ-induced lung injury in a rat model. The results showed that saturated phosphatidylcholine levels and surfactant pool size are increased, lung histology is improved and inflammatory cell infiltration is decreased by the three doses of MP treatment to a higher extent than by the one dose of MP treatment. Conversely, Wu et al. 19 reported that MP + prolonged dexamethasone treatment results in a higher number of survival days than MP pulse treatment does, but the former does not yield a significantly higher survival rate than the latter. Yu et al. 3 reported that a patient’s condition is aggravated after glucocorticoid treatment is terminated 2 months post-PQ poisoning, but lung injury was improved when glucocorticoid treatment is resumed. This finding indicates that glucocorticoids may play an important role in the treatment of lung injury caused by PQ poisoning. This potential effect of pulse + prolonged treatment may be related to the slow release and elimination of PQ from muscles; as a consequence, lung injury becomes concealed and persistent for weeks or months after poisoning. 20 –22
In addition to prolonged treatment, pulse treatment is a major issue in PQ research. Ghaffari et al. 23 and Seifirad et al. 24 reported that pulse treatment yields a similar survival rate and a lower histology score than PQ group does, and these results are similar to our findings. By contrast, Dinis-Oliveira et al. 25 reported that high-dose dexamethasone treatment decreases the pathological score and increases the survival rate of PQ-intoxicated rats. Lavagno et al. 26 reported that betamethasone aggravates the toxicity of PQ, decreases the survival rate and increases the lung wet/body weight ratio. However, the effects of variables, such as PQ dose and glucocorticoid dose and administration, were not completely comparable. Hence, further experiments should be performed to determine such effects.
Some limitations of this investigation should be noted. First, prolonged glucocorticoid treatment has induced adverse effects, including leucopenia, acne, hair loss and diabetes, 13 although such effects are likely transient because patients completely recover within several weeks. In our study, these adverse effects could not be monitored. Second, excessive PQ ingestion could damage organs, induce organ malfunction and trigger systemic diseases caused by respiratory failure. Additional evidence should also be obtained to determine whether prolonged administration is beneficial for other organs, such as kidney, liver and heart.
Conclusion
In conclusion, our study confirmed that prolonged treatment after pulse therapy could effectively ameliorate PQ-intoxicated acute lung injury in rats. However, further well-designed animal and clinical trials should be performed to assess the efficacy and safety of prolonged treatment after pulse therapy for the alleviation of PQ poisoning.
Footnotes
Acknowledgement
The authors are grateful for the excellent technical assistance provided by Professors Di Xie and Li Zhao.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
