Abstract
Paraquat (PQ) is one of the most widely used herbicides in the world and can cause pulmonary fibrosis in the cases with intoxication. Losartan, an angiotensin II type 1 receptor antagonist, has beneficial effects on the treatment of fibrosis. The aim of this study was to examine the effect of losartan on pulmonary fibrosis in PQ-intoxicated rats. Adult male Sprague Dawley rats (n = 32, 180–220 g) were randomly assigned to four groups: (i) control group; (ii) PQ group; (iii) PQ + losartan 7d group; and (iv) PQ + losartan 14d group. Losartan treatment (intragastrically (i.g.), 10 mg/kg) was performed for 7 and 14 days after a single i.g. dose of 40 mg/kg PQ. All rats were killed on the 16th day, and hematoxylin–eosin and Masson’s trichrome staining were used to examine lung injury and fibrosis. The levels of hydroxyproline and transforming growth factor β1 (TGF-β1), matrix metallopeptidase 9 (Mmp9), and tissue inhibitor of metalloproteinase 1 (TIMP-1) messenger RNA (mRNA) expression and relative expression levels of collagen type I and III were also detected. PQ caused a significant increase in hydroxyproline content, mRNA expression of TGF-β1, Mmp9, and TIMP-1, and relative expression levels of collagen type I and III ( p < 0.05), while losartan significantly decreased the amount of hydroxyproline and downregulated TGF-β1, Mmp9, and TIMP-1 mRNA and collagen type I and III expressions ( p < 0.05). Histological examination of PQ-treated rats showed lung injury and widespread inflammatory cell infiltration in the alveolar space and pulmonary fibrosis, while losartan could markedly reduce such damage and prevent pulmonary fibrosis. The results of this study indicated that losartan could reduce lung damage and prevent pulmonary fibrosis induced by PQ.
Introduction
Paraquat (1,1′-dimethyl-4,4′-bipyridinium dichloride; PQ) is one of the most widely used nonselective nitrogen herbicides for broadleaf weed control. It has been proven to be a safe commercial product when appropriately applied to eliminate weeds. However, over the past decades, numerous fatalities have been caused mainly by accidental and/or intentional ingestion of commercial liquid formulations of PQ. Many cases of acute poisoning and deaths have been reported in the past several decades and survivors of PQ poisoning may be left with a restrictive type of lung dysfunction in long-term follow-up. 1,2
PQ produces toxicity in humans, and the lung is one of the primary target organs. However, the mechanism of PQ toxicity has not been fully understood. The main potential mechanism is the structural similarity of PQ with lung diamines and polyamines such as putrescine, spermine, and spermidine, which could provoke the lung to accumulate PQ competitively. 3 There are two distinct stages in the development of pulmonary toxicity, namely, an early destructive stage characterized by damage and destruction of alveolar epithelial cells causing edema and hemorrhage, followed by an infiltration of myofibroblasts into the alveolar spaces and septa and differentiation in fibroblasts with the production of collagen and occurrence of massive pulmonary fibrosis. 4,5 Other studies also demonstrated that PQ can cause severe lung injury in humans and experimental animals, characterized by edema, hemorrhage, and interstitial inflammation. More seriously, PQ poisoning causes significant respiratory failure due to oxidative damage to the alveolar epithelium, which triggers an irreversible pulmonary fibrosis in the interstitium and alveoli. 6,7 Since there are no effective clinical antagonists for PQ, the treatment of PQ poisoning is limited to either decrease its absorption or enhance its elimination. 8 Unfortunately, despite the severity of the diseases caused by PQ, the remedy currently available for pulmonary fibrosis provides only minimal benefits and has significant negative effects.
Studies showed that angiotensin II (Ang II) has multiple biological activities which are mediated by stimulating the G protein-coupled Ang II type 1 receptor (AT1R). In addition to regulating blood pressure, aldosterone release, and sodium reabsorption, Ang II is also involved in the inflammatory process acting as a proinflammatory mediator. 9 Patients with idiopathic pulmonary fibrosis were found to have a high concentration of angiotensin-converting enzyme (ACE) in bronchoalveolar lavage fluid. In an animal model of radiation-induced pulmonary fibrosis, there were significant concentrations of ACE and Ang II in lung tissue homogenates. 10 Besides, Ang II and Ang receptors take part in the pathogenesis of experimental lung fibrosis by activating the AT1R, inducing fibroblast proliferation and interstitial collagen deposition, eventually leading to lung fibrosis. 11 Administration of an AT1R antagonist or ACE inhibitor significantly inhibited bleomycin- or radiation-induced pulmonary fibrosis in animal model. 12,13
The present study hypothesized that the AT1R antagonist (losartan) would attenuate PQ-induced lung fibrosis in rats. Thus, pathological reactions and hydroxyproline content of lung tissue were used to determine whether the pulmonary fibrosis model is successful in rats, and the expression of related messenger RNA (mRNA) and proteins were investigated to explore their possible involvements in pulmonary fibrosis induced by PQ. In particular, the intervention effect of losartan against lung fibrosis in male rats was also assessed.
Methods
Chemicals and drugs
PQ was produced and supplied by Sigma Chemical Company (St Louis, Missouri, USA). Losartan was purchased from Merck Sharp & Dohme (Hangzhou, China). Hydroxyproline kit was obtained from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). The reverse transcription and quantitative real-time polymerase chain reaction (qRT-PCR) kits were purchased from TaKaRa Company (Dalian, China). The primer sequences of β-actin, transforming growth factor β1 (TGF-β1), matrix metallopeptidase 9 (Mmp9), and tissue inhibitor of metalloproteinase-1 (TIMP-1) were obtained from Generay Biotechnology (Shanghai, China). Goat anti-rat primary antibodies to β-actin, collagen type I and III, and horseradish peroxidase (HRP)-conjugated rabbit anti-goat secondary antibody were obtained from Santa Cruz Biotechnology (Santa Cruz, California, USA).
Animal grouping and surgical procedures
A total of 32 adult male albino Sprague Dawley rats weighing 180–220 g were purchased from the Experimental Animal Center of Lanzhou University (Lanzhou, China). All rats were housed in a temperature-controlled room and allowed to take food and tap water ad libitum. In addition, all animal experiments were performed following the institution’s criteria for the care and use of laboratory animals as adopted by the ethics committee of Lanzhou University. The animals were randomized into four groups and were given doses based on their body weight: (i) control group, n = 8, animals were treated with sterile saline followed by injecting daily intragastrically (i.g.) with sterile saline on the following day for 14 consecutive days; (ii) PQ group, n = 8, animals were intoxicated with a single dose of PQ (40 mg/kg i.g.) followed by injecting daily i.g. with sterile saline on the following day for 14 consecutive days; (iii) PQ + losartan 7d group, n = 8, animals were given daily losartan (10 mg/kg i.g.) in the following 7 consecutive days followed by daily injection of sterile saline i.g. until the 15th day after a single dose of PQ (40 mg/kg i.g.); and (iv) PQ + losartan 14d group, n = 8, animals were given daily losartan (10 mg/kg i.g.) in the following 14 consecutive days after a single dose of PQ (40 mg/kg i.g.). On the 16th day after PQ intoxication, rats were killed, and lung tissue samples were immediately harvested. The middle lobes of the right lungs were kept in 10% formaldehyde and the upper lobes of the left lung were kept in Bouin’s solution for histology studies under a light microscope. The remaining lung tissues were frozen in liquid nitrogen for biochemical measures. It should be emphasized that the dose of losartan (10 mg/kg) was based on previous studies. This dose has therapeutic strategy for preventing hyperoxia-induced lung fibrosis in newborn rats, 14 and the dose of PQ (40 mg/kg) used in our study has also been confirmed in a successful model. 3
Measurement of TGF-β1, Mmp9, and TIMP-1 mRNA expressions
To investigate whether losartan changed the TGF-β1, Mmp9, and TIMP-1 mRNA expressions after PQ poisoning, we quantified mRNA expression in lung tissue by qRT-PCR. Total RNA was extracted from lung samples using the TRIzol Reagent (Invitrogen Life Technologies, Paisley, UK) according to the manufacturer’s instructions. The quantity and quality of isolated RNA were evaluated using absorbance measurements at 260 and 280 nm wavelengths, and the ratio between 1.8 and 2.2 was used in the next step. Complementary DNA (cDNA) was synthesized with a First Strand cDNA Synthesis Kit for RT-PCR according to the manufacturer’s instructions, and the specific reaction steps were 37°C for 15 min, 85°C for 5 s, and 4°C for 5 min. All of the RT-PCR primers used in this study are listed in Table 1. The SYBR Green dye was used in RT-PCR, and the operating steps were performed according to the SYBR Premix Ex Tap II manufacturer’s instructions (TaKaRa Co., Shanghai, China). RT-PCR was performed with the following program: initial denaturation at 95°C for 30 s, followed by 40 cycles of 95°C for 5 s, and 60°C for 30 s. Each lung tissue RNA sample was measured with three repeats, and triple reactions in one repeat. Pfaffl method 15 was used to calculate the relative expression levels of mRNA.
Oligonucleotide primer sequences and theoretical amplification length.

TGF-β1: transforming growth factor β1; Mmp9: metalloproteinase 9; TIMP-1: tissue inhibitor of metalloproteinase-1.
Measurement of hydroxyproline content in lung tissues
The collagen content of the lung tissue was determined by analysis of hydroxyproline. According to the detection kit instruction (Nanjing Jiancheng Bioengineering Institute, China), detection samples of lung tissues were prepared, and absorbance was measured at 550 nm to determine the hydroxyproline content. Data are expressed as micrograms of hydroxyproline per milligrams of lung tissue.
Measurement of collagen in lung tissues
Approximately, 100 mg lung tissues were homogenized in 1.0 ml radioimmunoprecipitation assay buffer containing 10 μl phenylmethylsulfonyl fluoride and centrifuged for 40 min at 13,000 r/min at 4°C. The supernatant was collected and protein concentrations were measured using the bicinchoninic acid protein assay kit (Beyotime Institute of Biotechnology, Jiangsu, China). For gel electrophoresis, equal quantities of protein (50 μg) were suspended in sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) buffer. The PCR instrument was used to denature protein at 99°C for 5 min. Collagen type I and β-actin were separated by 10% SDS-PAGE and collagen type III was separated by 8% SDS-PAGE, respectively, and then transferred to the polyvinylidene difluoride membranes (Millipore, Billerica, Massachusetts, USA). Membranes were then blocked overnight at 4°Cwith 5% nonfat milk in Tris-buffered saline (TBS) and then rinsed with TBS–Tween 20 (TBST) buffer for 10 min with three repeats. The membranes were incubated with goat anti-rat collagen type I, collagen type III, and β-actin antibodies at a dilution of 1/250, respectively. Blots were washed in TBST with three repeats and incubated in HRP-conjugated rabbit anti-goat secondary antibody at a dilution of 1/5000. For immunodetection, membranes were incubated with enhanced chemiluminescence solution according to the manufacturer’s instructions (Applygen, Beijing, China) and exposed in Molecular Imager ChemiDoc XRS System (Bio-Rad, Hercules, California, USA). The relative expression levels of collagen type I and III were analyzed by the band intensities of collagen type I/β-actin and collagen type III/β-actin, respectively.
Histological analysis
The middle lobe of the right lung fixed in 10% neutral-buffered formalin was processed through graded alcohol, xylene, and paraffin using an automatic tissue processor and blocked in paraffin. Sections of 4 μm thickness were obtained and stained by hematoxylin and eosin (H&E). In addition, the upper lobe of the left lung was kept in Bouin’s solution for Masson’s trichrome staining to assess collagen deposition. One pathologist blind to the study groups performed all the histological examinations. The presence of inflammation and fibrosis was evaluated in the samples.
Statistical analysis
Data were entered into a database and analyzed using SPSS 19.0 software and are expressed as the mean and standard errors. Statistically significant differences between groups were determined by one-way analysis of variance. The significance was accepted when p < 0.05.
Results
Analysis of TGF-β1, Mmp9, and TIMP-1 mRNA expressions
In the lung sample analyses, the threshold cycle value of each sample was determined with the same threshold, and the expression analysis of each sample was calculated using Pfaffl method. As shown in Figure 1, compared with the control group, the lung mRNA expression of TGF-β1, Mmp9, and TIMP-1 increased significantly in the PQ group ( p < 0.05), and the lung mRNA expression of Mmp9 and TIMP-1 in the losartan 7d group was still higher than in the control group ( p < 0.05). In contrast, treatment with losartan significantly decreased TGF-β1 and TIMP-1 mRNA expression levels in losartan 7d and 14d groups, but Mmp9 mRNA expression only had a statistical significance in losartan 14d group compared with the PQ group ( p < 0.05).

Effect of treatment with losartan on PQ-induced changes in TGF-β1, Mmp9, and TIMP-1 mRNA expression in lung tissue. Data are expressed as the mean and standard error. n = 8. (a) p < 0.05, when compared with control group and (b) p < 0.05, when compared with PQ group. PQ: paraquat; TGF-β1: transforming growth factor β1; Mmp9: matrix metallopeptidase 9; TIMP-1: tissue inhibitor of metalloproteinase-1; mRNA: messenger RNA.
Hydroxyproline content in lung tissue
As shown in Figure 2, the hydroxyproline content was up to 0.72 ± 0.13 μg/mg in PQ group, which was significantly higher than the control group (0.53 ± 0.10 μg/mg; p < 0.05). When treated with losartan for 7 days, the hydroxyproline content dropped to 0.64 ± 0.18 μg/mg, but there was no statistical significance compared with PQ group ( p > 0.05). After treatment with losartan for 14 days, the hydroxyproline content significantly decreased to only 0.55 ± 0.13 μg/mg ( p < 0.05).

Effect of treatment with losartan on PQ-induced changes in hydroxyproline content in lung tissue. Data are expressed as the mean and standard error. n = 8. (a) p < 0.05, when compared with control group and (b) p < 0.05, when compared with PQ group. PQ: paraquat.
Western blot analysis of collagen type I and III
Compared with the control group, PQ administration significantly increased the levels of collagen type I and III contents in the lung tissue of the PQ group ( p < 0.05). On the other hand, the treatment of losartan gradually decreased the levels of collagen type I and III in the lung tissue of the losartan 7d and 14d groups (Figure 3(a) and (b)).
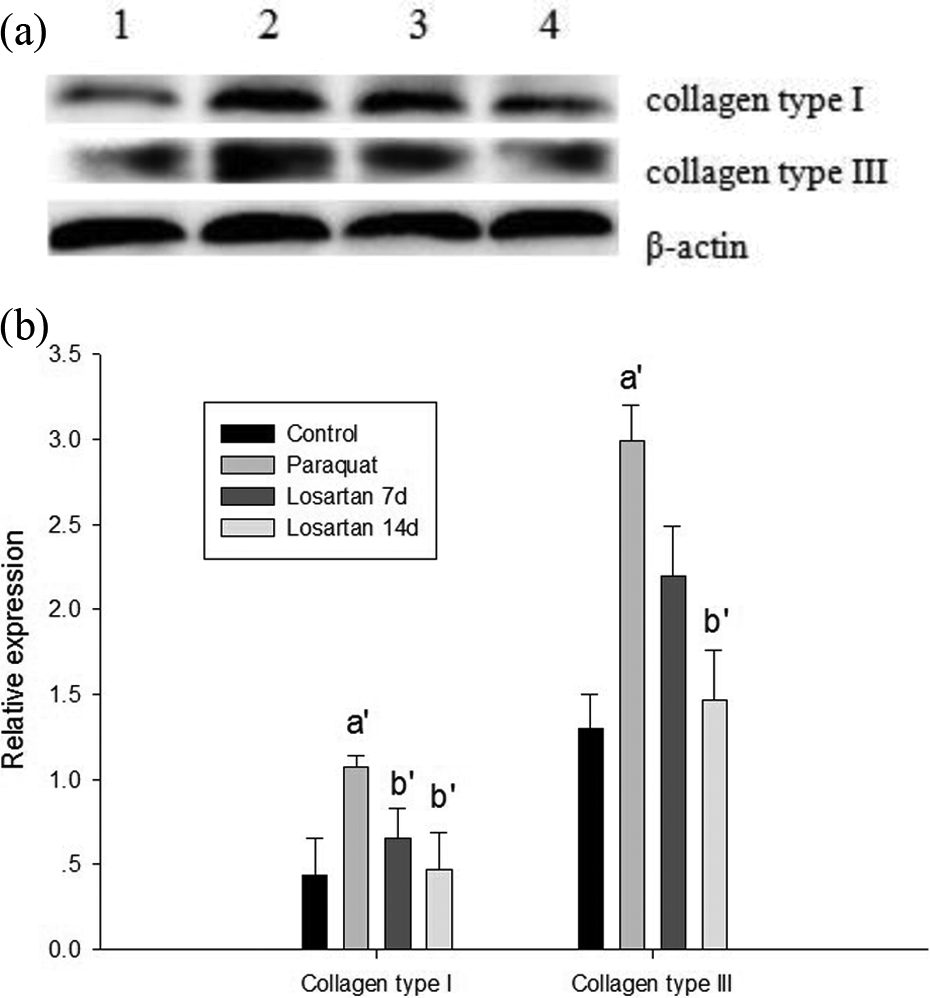
Western blot analysis of collagen type I and III in the lung tissue of rats. (a) Lane 1 represents control group; lane 2 represents PQ group; lane 3 represents PQ + losartan 7d group; and lane 4 represents PQ + losartan 14d group. (b) Densitometric analysis showed a significant increase in collagen type I and III in the lung tissue in PQ group. In contrast, the groups treated with losartan for 7 and 14 days showed a gradual decrease in two proteins compared with PQ group. The mean and standard error of three independent experiments were indicated; (a′) p < 0.05, when compared with control group and (b′) p < 0.05, when compared with PQ group. PQ: paraquat.
Histopathological evaluation
Light microscopic histopathological evaluation of lung tissue was performed for the four different experimental groups. Figure 4 shows H&E and Masson’s trichrome staining for lung injury and collagen deposition of lung tissue. H&E staining showed that the structure of lungs in the control group was normal under a light microscope, and no lesion was evident. The PQ group showed extensive alveolar wall thickness, and the alveolar space turned into small or closure considerably. Besides, there was a large amount of inflammatory cell infiltration in some areas. In addition, the small vascular wall in lung interstitium became thick and the lumen turned into stenosis. Compared with the PQ group, after 7 days’ intervention of losartan, the lung injury was improved but the thickening of small vascular wall was still not improved. After 14 days’ intervention of losartan, we could find that most of lung tissue became normal and that the interstitial small blood vessels were slightly thickened.

Effect of losartan on histopathological changes of lung tissue induced by PQ. (a–d) represent H&E staining for lung injury (H&E ×200). (e–h) represent Masson’s trichrome staining for collagen deposition of lung tissue (Masson ×400). (a and e) Lung tissue of control group was normal. (b and f) Lung tissue of PQ group showed the small vascular wall and extensive alveolar wall thickness and widespread inflammatory cell infiltration in the alveolar space and septum in H&E staining group and a large amount of collagen deposition in interstitial tissue in Masson staining group. (c and g) Lung tissue of PQ + losartan 7d group showed less inflammatory cell infiltration and collagen deposition in interstitial tissue, but the thickening of small vascular wall was still not improved. (d and h) Lung tissue of PQ + losartan 14d group showed that most of the lung tissue became normal and the interstitial small blood vessels were slightly thickened, and inflammatory cell infiltration and collagen deposition in interstitial tissue decreased further. Parts of picture (b–d) were amplified to observe inflammatory cells (×400). → and ▴ point at interstitial small blood vessels and inflammatory cells, respectively. PQ: paraquat; H&E: hematoxylin and eosin.
Masson’s trichrome staining differentiated collagen from smooth muscle and elastin and better visualized the extent of fibrosis. After Masson’s trichrome staining, collagen in the pulmonary interstitial was presented as a dense bluish-tinged material, as shown in Figure 4. More collagen deposition was found in PQ-exposed rats than those in control group. In contrast, losartan-treated rats for 7 and 14 days exhibited significantly less collagen deposition compared with PQ group.
Discussion
Pulmonary fibrosis is a frequent response to different injuries of the lung. Although there are numerous inducing factors, the final phases of fibrosis are the proliferation and progressive accumulation of connective tissue that replaces normal functional parenchyma. 16 In particular, PQ can cause severe lung injury and fibrosis in poisoning humans and experimental animals, characterized by alveolitis, pulmonary edema, and infiltration of inflammatory cells, followed by a final fibrotic stage where fibroblasts proliferate, collagen is deposited, and massive pulmonary fibrosis occurs. 3 Our findings showed a fibrogenetic effect of PQ in lung tissue of the rats (Figure 4(b) and (f)) and a significant increase in hydroxyproline content after PQ intoxication. The results suggested that the PQ poisoning model was successful.
To date, the pathogenesis of pulmonary fibrosis has not been precisely explained, but previous studies have shown that inflammation response is a major component in the pathogenesis of interstitial lung fibrosis that is induced in part by endogenous and migrating inflammatory cells. These cells together with lung epithelial and endothelial cells can produce abundant inflammatory cytokines, chemokines, or growth factors (such as tumor necrosis factor-α, monocyte chemotactic protein 1, TGF-β1, Mmp2, and TIMP-1), which may amplify or attenuate inflammatory reactions and involve in the conversion of fibroblast into myofibroblasts and enhancement of collagen accumulation. 17,18 In addition, some other studies also documented that Ang II is an inducer of procollagen production in normal fibroblasts, 19 which is produced primarily in the lung and serves as an inducer of TGF-β1-mediated fibrosis. When combined with AT1Rs, Ang II can stimulate collagen synthesis in many tissues. Studies verified that Ang II can affect human lung fibroblasts and promote extracellular matrix (ECM) synthesis. 20,21 Type I collagen is a principal matrix protein in the lung interstitium. Excessive collagen type I, mainly synthesized by activated fibroblasts, has been largely involved in the pathogenesis of fibrosis. This profibrotic effect of Ang II has been observed in the heart and kidney, where its effects have been attenuated by Ang II blockade. 22,23 Furthermore, excessive deposition of ECM in the lung interstitium results in respiratory failure associated with inflammation and neutrophil recruitment and the turnover of the ECM is partially regulated by proteases such as Mmp2 and TIMP-1. 24 The Mmp family and the endogenous regulators of Mmp activity (tissue inhibitors of Mmps and TIMPs) certainly play a crucial role in pathological conditions by degrading matrix proteins and connective tissues. 25 Lemjabbar et al. 26 reported that Mmp9 expression was increased in cultured alveolar macrophages and in bronchoalveolar lavage fluid from patients suffering from idiopathic pulmonary fibrosis. Other investigators reported that Mmp9 could be linked to inflammation-induced tissue remodeling, while Corbel et al. 24 observed a significant increase in Mmp9 activity in bronchoalveolar lavage fluid from lipopolysaccharide-exposed mice concomitantly with collagen deposition, and Mmps appear to have an important role in causing damage to lung ECM and to be associated with the development of pulmonary fibrosis. Furthermore, TIMPs, and particularly TIMP-1 induction could lead to a “noncollagenolytic microenvironment,” building adequate conditions for further ECM deposition to occur. 27 In humans, increased levels of TIMP protein and RNA are observed in the lungs of patients with idiopathic pulmonary fibrosis, and TIMP expression there exceeds that of Mmp. 28 Previous studies have demonstrated that the mRNA expression of Mmp9, TIMP-1, and collagen type I and III were significantly increased after PQ intoxication. 18,29 In the present study, we found that the mRNA expression of TGF-β1, Mmp9, and TIMP-1 and the expression of collagen type I and III increased significantly after PQ poisoning, which revealed that serious lung injury and fibrosis occurred when rats were poisoned with PQ and which was consistent with previous studies.
Losartan, an AT1R antagonist, can attenuate pulmonary fibrosis, which has been verified in many studies. Losartan at a dose of 10 mg/kg effectively prevented collagen gene activation and ameliorated the degree of renal vascular fibrosis without altering the systolic blood pressure in the transgenic mice. 30,31 Besides, the same dose of losartan was verified to have therapeutic strategy for preventing hyperoxia-induced lung fibrosis in newborn rats. 14 The potential actions by which AT1R antagonists could act on the lung include decreased vascular tone and permeability and changed fibroblast activity. 32 Thus, it is possible that the antifibrotic effect of losartan might be associated with lowering the systemic blood pressure. However, blood pressure was not measured in the present study, and other data do not exclude that possibility. As a potent AT1R antagonist, losartan is able to alleviate fibrosis by reducing the expression of TGF-β1 and inhibition of binding of Ang II to the AT1R in bleomycin-induced lung fibrosis in mice. In addition, collagen deposition was also inhibited, which has been demonstrated in previous animal studies. 33,34
The successful establishment of the PQ poisoning model by intragastric administration of PQ (Figures 4(b) and (f)) made it possible for us to evaluate the effect of losartan against PQ-induced pulmonary fibrosis in rats. The histopathological photographs and decrease of hydroxyproline contents (Figures 1 and 4) in the present study verified that the treatment of losartan could effectively attenuate PQ-induced lung injury and fibrosis. In addition, intragastric treatment with losartan for 7 and 14 days resulted in a significant decrease of TGF-β1 levels in lung tissue. The results suggested that the mechanisms of the protective effect of losartan on PQ-induced lung fibrosis might be related to its inhibition of TGF-β1 mRNA expression. Furthermore, the mRNA expressions of Mmp9 and TIMP-1 and relative expressions of collagen I and III decreased significantly (Figures 1 and 3). The results of the present study were consistent with those of previous studies regarding acute lung injury with other chemical reagents exposure. 14,35 We suggest that losartan might play an important role in preventing the development of pulmonary fibrosis through alleviating inflammatory reaction and inhibiting collagen synthesis, which all verified the hypothesis that an AT1R antagonist, losartan, would attenuate PQ-induced lung fibrosis in rats.
In conclusion, we have demonstrated a possible intervention effect of losartan in PQ-induced pulmonary fibrosis. It may be related to the inhibition of the mRNA expression of TGF-β1 and collagen synthesis by losartan. The present study indicates that losartan might be a new therapeutic agent for the prevention of pulmonary fibrosis induced by PQ intoxication. However, the exact mechanism of losartan’s protective effect against PQ-induced pulmonary fibrosis should be verified with further animal and human studies.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research was financially supported by the Foundation of Chengguan District in Lanzhou City, Gansu, China (grant number 2012-1-4).
