Abstract
Crocin (CRO) and safranal (SAF) are bioactive constituents of saffron (dried stigma of Crocus sativus flower), an expensive spice with medicinal properties. Aqueous extract of saffron is known for its antigenotoxic effect against environmental genotoxins/carcinogens. However, there is need to identify saffron constituents responsible for this antigenotoxic effect. The aim of our investigation was to ascertain the role of CRO and SAF as inhibitors of in vivo genotoxic stress. For this purpose, Swiss albino mice were pretreated with CRO (50-mg/kg body weight (bw))/SAF (0.025- and 0.25-ml/kg bw) by gavage for 2 days. Thereafter, the pretreated mice were exposed to the genotoxic agents: (1) gamma radiation (GR; 2 Gy), (2) urethane (URE; 800 mg/kg) and (3) procarbazine (PCB; 60 mg/kg). In addition, CRO (50 mg/kg) was co-administered with the nitrosation reaction mixture of methylurea (MU; 300-mg/kg bw) + sodium nitrite (15 mg/kg) which can form N-nitroso-N-MU in the stomach. Genotoxic damage was measured by performing the bone marrow micronucleus test. Results obtained demonstrated significant reductions in the incidence of micronucleated polychromatic erythrocytes in the bone marrow of mice pretreated with CRO/SAF before exposure to the above DNA damaging agents, GR, URE and PCB. Co-administration of CRO with the nitrosation reaction mixture led to significant decrease in genotoxicity when compared to nitrosation reaction mixture alone. Histopathological studies revealed that these saffron constituents reduced testicular cell damage induced by the test genotoxins. The cell-free DNA-nicking assay using pBR322 DNA showed significant protective effects of CRO against hydroxyl radical-induced strand breaks.
Introduction
There is substantial historical evidence indicating that saffron, the dried stigma of Crocus sativus flower, has been used by humans for over 3000 years, owing to its unique medicinal properties and importance as an agent for flavouring and colouring food. 1 During the last two decades, a large number of in vitro and in vivo laboratory investigations have been carried out to elucidate the cellular and molecular mechanisms associated with pharmacophysiological effects of saffron. 2 –6 Some of these studies demonstrated protective effects of saffron against genotoxic stress and carcinogenesis. 7 –10 These findings are mainly from experiments using aqueous extract of saffron. 8 –10 This raises questions on the identity of bioactive saffron constituents responsible for the observed antigenotoxic and anticarcinogenic effects. Crocin (CRO), picrocrocin and safranal (SAF) are often considered as constituents of saffron responsible for its bioactivity. 11 CRO is a carotenoid pigment responsible for the bright red colour of C. sativus stigma. SAF is the main component of saffron essential oil, considered as responsible for its unique aroma. There are reports showing the protective effects of CRO and SAF against methyl methanesulfonate–induced DNA damage in mice organs using the comet assay. 12,13 Hence, we initiated the present study with the objective of assessing in mice the possible role of two bioactive constituents, CRO and SAF, as inhibitory agents against genotoxic stress.
Previous investigations on in vitro/in vivo antigenotoxic effects of saffron extract have focused on effects against chemical genotoxins. 8 –10 Therefore, in the present work, we included experiments to evaluate the impact of saffron constituents, CRO and SAF, on chromosomal damage induced by gamma radiation (GR), a physical DNA damaging agent. In addition, we used the genotoxic carcinogen urethane (URE), the chemotherapeutic drug procarbazine (PCB) and the nitrosation reaction mixture of methylurea (MU) + sodium nitrite (SN) (which forms N-nitroso-N-MU in the stomach) as genotoxins. URE is an environmental carcinogen known to induce tumours in different organs. 14,15 Humans are exposed to URE through ingestion of alcoholic beverages, yeast breads and use of cosmetics, pesticides and textiles. 16 PCB is a chemotherapeutic drug, effective for treatment of Hodgkin’s disease and tumours of the brain, lung and skin. 17 PCB is known to generate reactive oxygen species. 18 The genotoxic damage induced by GR, URE and PCB has been observed in bone marrow cells and germ cells of mice. 14,19 –22 In this study, modulation of genotoxic damage by CRO and SAF was evaluated by performing the bone marrow micronucleus test. Additional experimental work included histopathological studies to assess the effects of pretreatment with CRO and SAF on testicular damage induced by GR, URE and PCB. The cell-free DNA-nicking assay using pBR322 DNA was performed to assess the possible protective effects of CRO against hydroxyl radical–induced DNA damage.
Materials and methods, Chemicals
CRO, SAF, URE and newborn calf serum were purchased from Sigma-Aldrich Pvt. Ltd (New Delhi, India). PCB was a gift from Hoffmann-La Roche, (Basel, Switzerland). MU and SN were obtained from Merck (New Delhi, India). All the other reagents and solvents used for the experiments were of analytic grade from Qualigens Chemicals Pvt. Ltd, New Delhi, India.
DNA nicking assay
DNA nicking assay was performed using supercoiled pBR322 DNA (Bangalore Genei Ltd, Bangalore, India) according to the method of Lee et al. 23 with minor modifications.
Animals
All the in vivo experiments were carried out with 10–12-weeks old male Swiss albino mice weighing 25–30 g. These animals were bred in the university animal house and maintained at 25 ± 2°C on the standard mouse diet and water ad libitum in accordance with CPCSEA, India, guidelines. Approval for this work was obtained from the University Animal Ethical Committee (IAEC-JNU 15/2014).
Pretreatment
CRO (50 mg) and SAF (0.025 and 0.25 ml) were diluted in double distilled water and liquid paraffin (LP), respectively, and administered to the experimental animals by gavage (10-ml/kg bw for CRO) and (5-ml/kg bw for SAF) for two consecutive days. From the preliminary work, we observed that the efficacy of this pretreatment schedule for antigenotoxicity was not significantly different when compared to those with administration of saffron constituents for 4 or 5 days. Control animals received the same volume of distilled water or LP. Each control/treatment group consisted of five mice. The test doses of CRO and SAF were decided based on the preliminary experiments and the information from publications by Hosseinzadeh and Sadeghnia 12 and Hosseinzadeh et al. 13 on antigenotoxicity of these saffron constituents against methyl methanesulfonate.
Irradiation
The experimental animals were whole body irradiated at room temperature in a gamma chamber (Co60 source, 240 TBq, Model 4000A) obtained from Bhabha Atomic Research Centre, Mumbai, India. The mice were restrained in a well-ventilated acrylic cylinder and placed in the gamma chamber. The dose rate was 1 Gy/28 s. The animals received CRO/SAF 24 h and 30 min before irradiation.
Genotoxin treatment
PCB (60-mg/kg body weight (bw)) and URE (800-mg/kg bw) were diluted in distilled water and injected intraperitoneally 30 min after pretreatment with CRO (50-mg/kg bw)/SAF (0.025-ml or 0.25-ml/kg bw) on the second day. The test doses of PCB and URE were decided on the basis of our previous studies. 24,25
Treatment of animals with CRO and nitrosation reaction mixture
As described by Abraham and Khandelwal, 26 the nitrosation reaction mixture consisting of MU (300-mg/kg bw) + SN (15-mg/kg bw) was used for the experiment. The four groups of experimental animals received by gavage (10-ml/kg bw) are as follows: (a) distilled water (10-ml/kg bw, negative control), (b) CRO (50-mg/kg bw in distilled water), (c) MU (300-mg/kg bw) + SN (15-mg/kg bw) in distilled water and (d) MU (300-mg/kg bw) + SN (15-mg/kg bw) and CRO (50-mg/kg bw) in distilled water.
Micronucleus test
The micronucleus test for evaluating genotoxic damage was carried out as described in our recent publications. 25,27 The experimental animals were sacrificed by cervical dislocation 24 h after treatment, and bone marrow cells from both the femurs were flushed into foetal calf serum in a 1-ml centrifuge tube. Special care was taken to collect the cells in the form of a fine suspension. Thereafter, the cell suspension was centrifuged at 2000 rpm for 5 min, and the supernatant was discarded. The pellet was used for preparing slides which were air-dried and stained with May–Grünwald and Giemsa stain. Five mice were used for each experimental point, and 2000 PCEs/animal were scored from a single slide to assess the incidence of micronucleated polychromatic erythrocytes (MnPCEs).
Histopathological analysis of mouse testis
Testes from control/treated mice groups were removed and fixed in Bouin’s solution for 24 h, then rinsed in distilled water and preserved in 70% ethanol. The samples were dehydrated in ascending grades of ethanol, cleared in xylene and embedded in paraffin for sectioning. Sections of 5-µm thickness were mounted on glass slides and deparaffinized using descending grades of alcohol before staining with haematoxylin and eosin for histological analysis. These tissue sections were processed and stained according to Bancroft and Gamble. 28
Statistical analysis
Data is expressed as mean ± standard deviation. Student’s t-test was performed using Graph pad software, 2016 version for comparing two groups for genotoxic damage. Results were considered statistically significant at p < 0.05.
Results
From the data presented in Figures 1 to 4, it is evident that oral administration of either CRO or SAF alone has not significantly increased the frequencies of MnPCEs when compared to that in the negative controls. Figure 1 illustrates the protective effects of CRO against the genotoxic damage induced by PCB and URE. Significant reductions were observed in the frequencies of MnPCEs induced by PCB (60-mg/kg bw) and URE (800-mg/kg bw) following pretreatment with CRO (50-mg/kg bw) for 2 days. The data presented in Figure 2 shows the protective effect of CRO (50-mg/kg bw) pretreatment on the incidence of MnPCEs induced by exposure to GR (2 Gy). Figure 3 shows the antigenotoxic effect of SAF against PCB and URE. The frequencies of MnPCEs registered significant reductions when the animals received SAF (0.025 and 0.25 ml/kg against PCB and URE, respectively) before the genotoxin treatment. As shown in Figure 4, pretreatment with SAF (0.25 ml/kg) exerted a significant protection against the induction of MnPCEs by exposure to GR (2 Gy). The inhibitory effect of CRO (50-mg/kg bw) against the genotoxic damage induced by endogenous nitrosation has been shown in Figure 5. Co-administration of CRO (50 mg/kg) with the nitrosation reaction mixture (MU (300 mg/kg) + SN (15 mg/kg)) showed a significant reduction in the incidence of MnPCEs when compared to that with the nitrosation reaction mixture alone.

Inhibitory effects of CRO (50-mg/kg bw) on MnPCEs induced by PCB (60-mg/kg bw) and URE (800-mg/kg bw). Values are mean ± standard deviation from five mice. *Significantly different from the genotoxin alone group (p < 0.05). bw: body weight; CRO: crocin; MnPCE: micronucleated polychromatic erythrocyte; PCB: procarbazine; URE: urethane.
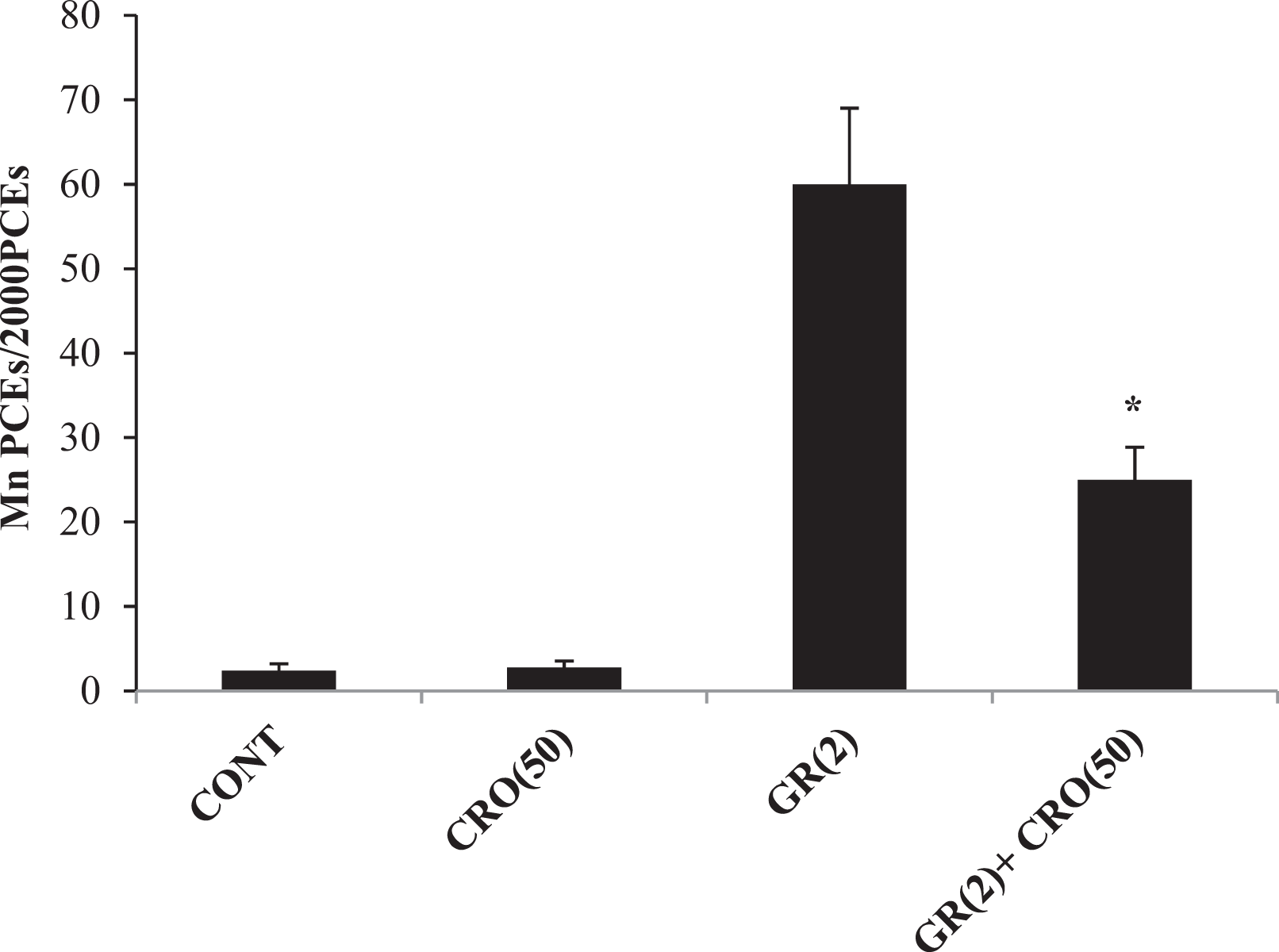
Inhibitory effects of CRO (50-mg/kg bw) on the induction of MnPCEs by GR (2 Gy). Values are mean ± standard deviation from five mice. *Significantly different from the GR (2 Gy) group (p < 0.05). bw: body weight; CRO: crocin; MnPCE: micronucleated polychromatic erythrocyte; GR: gamma radiation.

Inhibitory effects of SAF (0.025- or 0.25-ml/kg bw) on MnPCEs induced by PCB (60-mg/kg bw) and URE (800-mg/kg bw).Values are mean ± standard deviation from five mice. *Significantly different when compared to the groups treated with the genotoxin (PCB or URE) alone (p < 0.05). bw: body weight; SAF: safranal; MnPCE: micronucleated polychromatic erythrocyte; PCB: procarbazine; URE: urethane.

Inhibitory effect of SAF (0.25-ml/kg bw) on MnPCEs induced by GR (2 Gy). Values are mean ± standard deviation from five mice.*Significantly different when compared with the GR (2 Gy) alone group (p < 0.05). bw: body weight; SAF: safranal; MnPCE: micronucleated polychromatic erythrocyte; GR: gamma radiation.
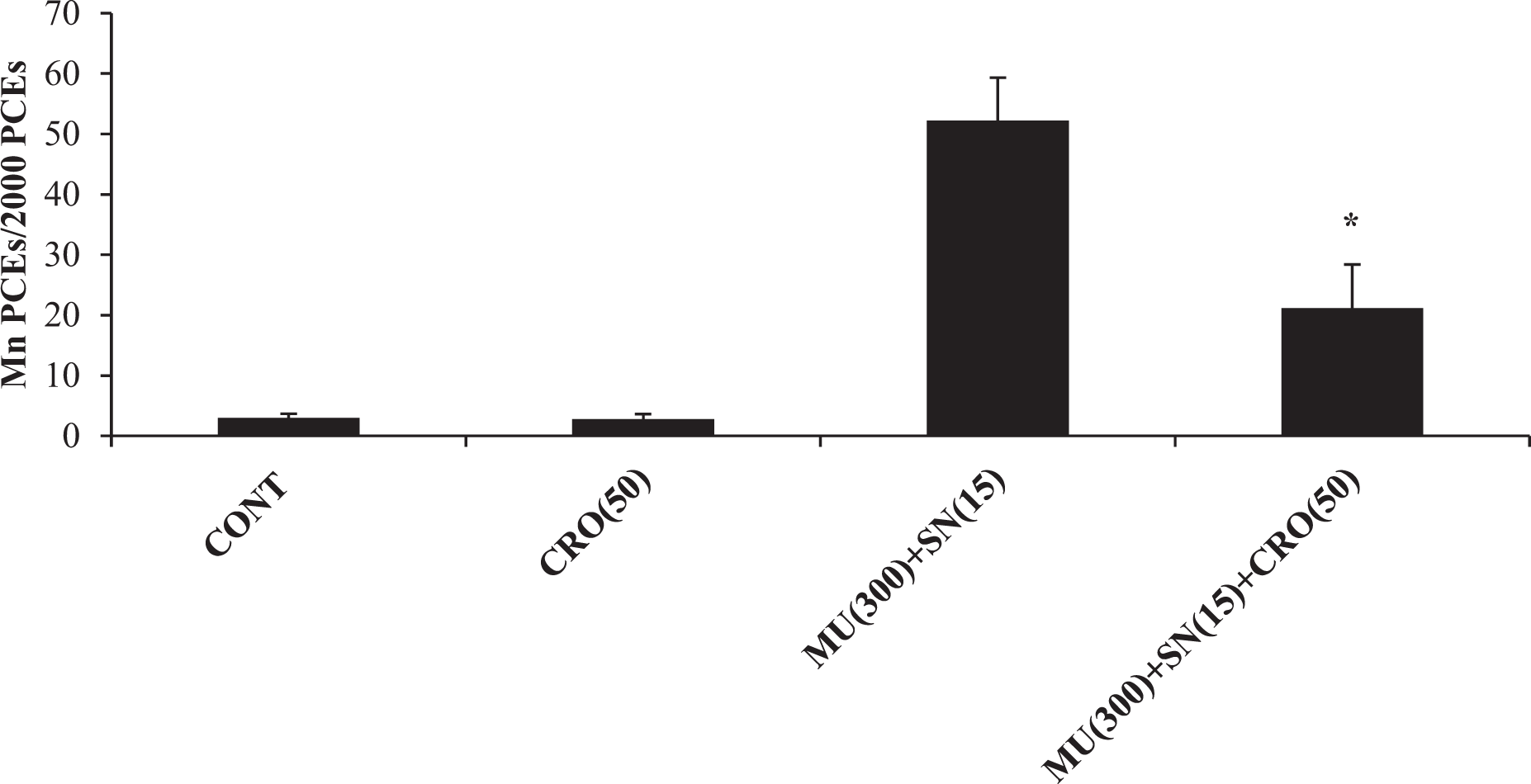
Endogenous nitrosation-induced micronucleus formation and its reduction by co-treatment with CRO (50-mg/kg bw). Values are mean ± standard deviation from five mice. *Significantly different when compared with MU (300-mg/kg bw) + SN (15-mg/kg bw) treated group (p < 0.05). bw: body weight; CRO: crocin; MU: methylurea; SN: sodium nitrite.
Photomicrographs showing cross sections of the testes from control, CRO and SAF pretreated mice exhibited testicular tissue with densely packed intact seminiferous tubules without any damage (Figure 6(a) to (c)). In comparison, mice exposed to GR, URE and PCB (Figure 6(d), (g) and (j)) show severe damage to different germ cell stages. This damage to the testicular cells is reduced considerably when the animals are pretreated with CRO/SAF. The ameliorative effect was more with CRO pretreatment (Figure 6(e), (h) and (k)). A reduction in damage to different germ cell stages was observed following pretreatment with SAF before exposure to GR, URE and PCB (Figure 6(f), (i) and (l)).
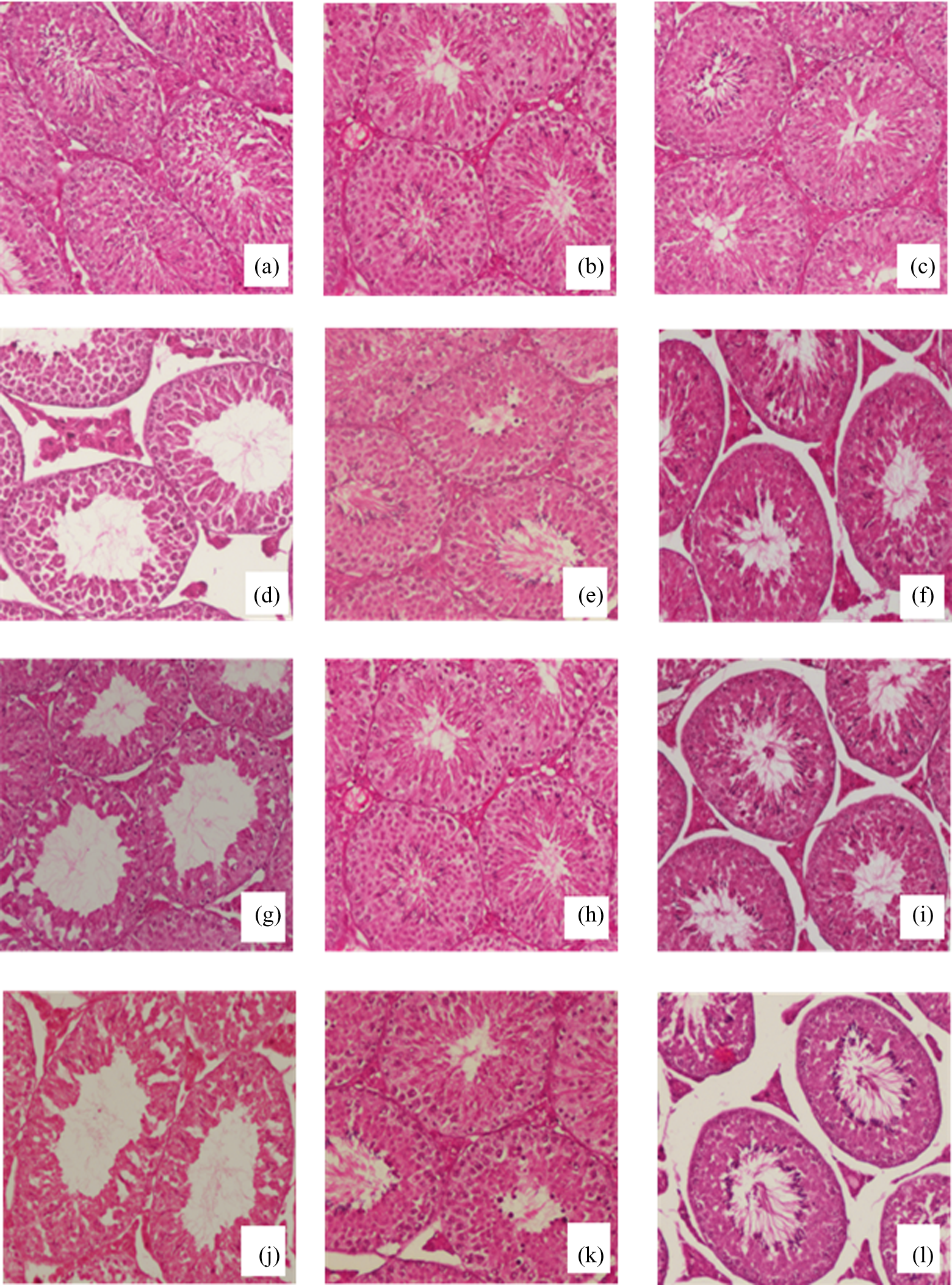
Photomicrographs of Haematoxylin and eosin stained sections of mice testes (×10) showing seminiferous tubules: (a) CONT, (b) CRO (50-mg/kg bw), (c) SAF (0.25-ml/kg bw), (d) GR (2 Gy), (e) GR (2 Gy) + CRO (50-mg/kg bw), (f) GR (2 Gy) + SAF (0.025-ml/kg bw), (g) URE (800-mg/kg bw), (h) URE (800-mg/kg bw) + CRO (50-mg/kg bw), (i) URE (800-mg/kg bw) + SAF (0.25-ml/kg bw), (j) PCB (60-mg/kg bw), (k) PCB (60-mg/kg bw) + CRO (50-mg/kg bw) and (l) PCB (60-mg/kg bw) + SAF (0.025-ml/kg bw). bw: body weight; CONT: control; CRO: crocin; SAF: safranal; GR: gamma radiation; URE: urethane; PCB: procarbazine.
Figure 7(a) shows that the presence of CRO (0.062–1 µg/µl) along with plasmid pBR 322 DNA during Fenton’s reaction protected DNA from hydroxyl radical-induced strand break in a concentration dependent manner. Maximum protection of 93% was observed with a concentration of 1 µg/µl (Figure 7(b)).

Protective effects of CRO on hydroxyl radical (OH•−)-induced pBR322 DNA strand breaks. (a) Lane 1: plasmid DNA (0.5 µg/µl), Lane 2: plasmid DNA (0.5 µg/µl) + Quercetin (125 mM), Lane 3: FR, Lane 4: DNA (0.5 µg/µl) + CRO (1 µg/µl) + FR, Lane 5: DNA (0.5 µg/µl) + CRO (0.5 µg/µl) + FR, Lane 6: DNA (0.5 µg/µl) + CRO (0.25 µg/µl) + FR, Lane 7: DNA (0.5 µg/µl) + CRO (0.125 µg/µl) + FR and Lane 8: DNA (0.5 µg/µl) + CRO (0.062 µg/µl) + FR. (b) Total protective percentage of CRO (0.062–1 µg/µl) on hydroxyl radical (OH•−)-induced plasmid DNA strand breakage. Data are presented as mean ± standard deviation of three experiments. FR: Fenton’s reagent; CRO: crocin.
Discussion
Saffron has remained for several centuries as an important ingredient of folk medicine used to treat a wide range of ailments in humans. 2,3 Currently, laboratory investigations are unravelling the possible mechanisms responsible for the curative properties of saffron. 4 –10 Along with this, attempts are made to identify saffron constituents endowed with a wide spectrum of bioactivity. As stated in the Introduction, the main focus of our present work was to assess the role of CRO and SAF as antigenotoxic agents. The results we obtained from the mouse bone marrow micronucleus test demonstrated the importance of these two constituents as protective agents against physical (GR) and chemical (URE, PCB and endogenous nitrosation) DNA damaging agents in bone marrow cells of mice. The induction of micronuclei is considered as an important biomarker for assessing the genotoxic damage induced by environmental mutagens and carcinogens. 29 These findings have strengthened previous reports on the in vivo antigenotoxic effects of aqueous saffron extract against URE and PCB. Furthermore, the present results suggest that CRO and SAF are two of the main bioactive saffron constituents with antigenotoxic activity. Recent reports based on studies with cancer cells and animal models have highlighted the importance of CRO and SAF for cancer therapy and prevention. 30 –36 CRO exerted protective effects against N-nitroso-N-MU-induced breast cancer and doxorubicin-induced cardiotoxicity in rats. 32,33 Therapy with GR in combination with CRO increased apoptosis in MCF-7 cell line. 34 Combination of CRO with cisplatin increased the chemosensivity in lung cancer cells. 35 SAF showed neuroprotective effects in a rat model of spinal cord injury. 36
GR, URE and PCB are well-established agents for inducing chromosomal damage and mutations in the mouse germ cells. 14,16,19 –22 From the present histopathological analysis, there are clear indications that the saffron components CRO and SAF can ameliorate the testicular damage induced by these DNA-damaging agents. Further detailed investigations are needed to obtain information on the protective effects of CRO and SAF against germ cell damage induced by environmental genotoxins.
To date, all the reports on role of saffron or its constituents as protective agents against DNA damage have come mainly from studies with well-known genotoxic chemicals. Hence, we included experiments to answer the question whether or not intake of saffron constituents CRO and SAF can have an impact on the genotoxic damage induced by GR, a physical DNA damaging agent. Our present results have demonstrated for the first time the efficacy CRO and SAF as protective agents against GR-induced in vivo chromosomal damage.
N-nitroso compounds, which are cytotoxic, genotoxic and carcinogenic, can be formed in the stomach when nitrite and secondary amines or N-substituted amides react under acidic conditions. 37,38 In the present study, we used a model nitrosation reaction mixture consisting of MU and SN which can react to form N-nitroso-N-MU, a highly potent genotoxic and carcinogenic agent. There are many phytochemicals present in common fruits and vegetables, which inhibit endogenous nitrosation leading to the formation of carcinogenic compounds. Ascorbic acid and many polyphenols are efficient antinitrosating agents. 26 Our present study has furnished evidence for the role of CRO as an inhibitor of endogenous nitrosation leading to a reduction in genotoxic damage.
Analysis of the possible mechanisms responsible for the antigenotoxic/anticarcinogenic effects of saffron and its constituents furnished evidence which suggests that the antioxidant and free radical scavenging activity as one of the main mechanisms. Our present results from the cell-free DNA-nicking assay clearly demonstrated the free radical scavenging property of CRO.
The absorption and metabolism of CRO and SAF following administration by gavage are important factors which can determine their activity as antigenotoxic agents. After oral intake, CRO is rapidly absorbed into the blood circulation. CROs are hydrolyzed to crocetin before/during intestinal absorption. 39 Thereafter, the absorbed crocetin gets metabolized to mono- and diglucuronide conjugates. In the mouse plasma, crocetin–glucuronides are retained for 8 h after the administration of crocetin and CRO. 39 SAF is known to have antioxidant properties, and it protects DNA from harmful damage via external binding or intercalative modes. 40,41
Overall, the information on in vivo antigenotoxic effects of CRO and/or SAF against GR, URE, PCB, endogenous nitrosation (from the present study) and methyl methanesulfonate 12,13 indicate that these phytochemicals can play an important role in reducing genotoxic damage induced by genotoxins with diverse mechanisms of action. However, there is need for further in vivo investigations to assess the efficacy of low dose intake of saffron and its constituents for chemoprevention of genotoxic stress induced by a wider range of environmental toxicants.
In conclusion, the findings from our study have demonstrated the efficacy of CRO and SAF as constituents of saffron which can reduce genotoxic stress induced by well-established DNA damaging agents.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by University Grants Commission (NETWORKING) and the Department of Science and Technology (PURSE).
