Abstract
Objective:
To investigate the effects of in utero exposure to high-dose di-n-butyl phthalate (DBP) on testicular cell apoptosis in late embryonic and pubertal male rat offspring.
Methods:
Twenty pregnant Sprague-Dawley (SD) rats were divided into two groups. During gestation day (GD) 12 to GD 19, control group was given 1 ml day−1 of olive oil and experimental group was given DBP 500 mg kg−1 day−1 by gavage. On GD 19.5 and postnatal day (PND) 45, the testes were removed. Morphological analysis of the testes was observed by transmission electron microscopy and hematoxylin and eosin (H&E) staining. Testicular cell apoptosis was detected by terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL). The expression of Bcl-2, Bax, and p53 was presented by immunohistochemistry (IHC) and western blot. Data of the two groups was compared using independent samples t-test and Mann–Whitney test by SPSS 20.0.
Results:
H&E staining showed that spermatogenetic cells were significantly decreased in DBP exposed pubertal rat testis. The apoptosis index of testes in DBP-treated group was significantly lower on GD 19.5 but higher on PND 45 than that of the controls (p < 0.01). IHC and western blot revealed significantly increased expression of Bcl-2 in GD 19.5 rat testis and Bax and p53 in PND 45 rat testis after DBP exposure, compared with the control (p < 0.05).
Conclusion:
In utero exposure of high-dose DBP resulted in opposite effects on testicular cell apoptosis in late embryonic and pubertal rat offspring. The overexpression of Bcl-2, Bax, and p53 might be related to the occurrence of abnormal apoptosis and finally produce male infertility.
Introduction
Reproductive disorders of newborn (cryptorchidism and hypospadias) and young adult males (testicular germ cell cancer and infertility) are common and/or increasing in incidence. 1,2 In 2001, Skakkebaek et al. proposed testicular dysgenesis syndrome (TDS), a hypothesis to explain the etiology of a series of diseases such as testicular germ cell cancer, cryptorchidism, hypospadias, and low sperm counts. 1,2 Evidence suggests that this syndrome arises from abnormal development of Sertoli and Leydig cells either due to genetic defects or because of environmental factors. 2 –4 Many of the features described by the TDS hypothesis have been observed in rats developmentally exposed to phthalate acid esters (PAEs), a family of environmental endocrine disruptors. di-n-butyl phthalate (DBP) is one of many PAEs which are contained in a variety of consumer products, including plastic food wrap, insect repellents, skin emollients, perfumes, hair spray, and nail polish. Administration of the phthalate ester DBP to pregnant rats leads to a variety of male reproductive malformations, including underdeveloped or absent reproductive organs, hypospadias, decreased anogenital distance, and cryptorchidism, while the main effect to sexually mature male rat offspring was testicular dysfunction, including abnormal spermatogenesis, diminished sperm counts, and quality. 5 –7 These testicular findings in rats have been well defined and for this reason the rat may serve as a useful model for identifying cellular pathways of such findings in humans. 8
TDS originates in fetal life. 2 The animal model of hypospadias and cryptorchidism had been successfully established by gavaging rats in late pregnancy with DBP, and subsequently, the abnormal testis development was observed in these models. 9,10 The testicular dysgenesis could have numerous primary causes, such as secondary to hormonal or other malfunctions of the Leydig and/or Sertoli cells during male sexual differentiation, leading to increased risk of the reproductive disorders. The effects of phthalates on fetal Leydig cells involved the morphology, function, and cell mitosis, including the Leydig cell aggregation, a decrease in testosterone level, insulin-like growth factor 3 (insl3) expression, and the number of Leydig cells. 11 –14 DBP could also inhibit the maturation of Sertoli cells in the testis of rat offspring, and then later influence the spermatogenesis in adult testis. 4 The vimentin cytoskeleton in Sertoli cells could be changed by DBP, leading to the occurrence of multinucleated gonocyte (MNG) in testis, which might be closely related to testicular cancer. 15,16 Vimentin damage would also result in spermatogenic cell separation, which would develop apoptosis due to the lack of nutrition and support from Sertoli cells. 17 DBP differentially affects fetal germ cells in rats according to stage of gestation, including the effects on germ cell number and differentiation. 18 DBP exposure could induce MNG and aggregation of differentiated germ cells in human testis xenografts and in vivo in rats. Loss of Sertoli–germ cell membrane contact might be responsible for the latter. 19 Since the regulation of germ cells in the embryonic stage was poorly understood, the study of this mechanism provided a new direction for our study. Based on the mature animal model, we investigated the effects of DBP on the apoptosis of testicular germ cells in late embryonic and pubertal male rat offspring in order to study the molecular mechanisms of DBP-induced testicular dysgenesis. The aim was to identify the period of greatest vulnerability of fetal testis to disruption by phthalate exposure, hence the use of a single high dosage of DBP exposure, as this period may prove to be relevant to humans.
Methods
Reagents
DBP (99.8%, AR, 1.04 g ml−1, CAS#84-74-2) and hematoxylin were acquired from Sigma Chemical (St Louis, Missouri, USA). TUNEL In Situ Apotosis Detection Kit (11684817910) was acquired from Roche (Basel, Switzerland). Rabbit polyclonal to Bcl-2 antibody (441201) and rabbit polyclonal to Bax antibody (CA06131) were acquired from Bioworld Technology (Minion, USA). Rabbit polyclonal to p53 antibody (#F2513) was acquired from Santa Cruz Biotechnology (Heidelberg, Germany). Sheep anti-rabbit immunoglobulin G (IgG; GR26562-1), sheep anti-mouse IgG (GR193588 -1), rabbit anti-sheep IgG (GR18798-25), and β-actin were acquired from Abcam (Cambridge, UK). DAB Color Kit was acquired from Beijing ZhongShan Biotechnology Co., China; Microscope from BX43, Olympus, Japan; and transmission electron microscopy (TEM) Tecnai G2 Spirit BioTwin, FEI Company, Hillsboro, USA.
Samples
Twenty-eight-week-old female Sprague–Dawley rats were kept in the laboratory animal center of Nanjing Medical University under controlled temperature (20−22°C), light (12/12-h light dark cycle), and humidity (50–70%) and received sterile water and a soy-free breeding diet in accordance with the Guide for the Care and Use of Laboratory Animals. Both of the female and male rats used for insemination were from the laboratory animal center of Nanjing Medical University. The day when sperm was found in the vagina of the matched female was considered to be gestation day 0 (GD 0). Dams from GD 12 to GD 19 were daily treated by gavage with 1 ml olive oil (group A, n = 10) or 500 mg kg−1 DBP dissolved in olive oil (group B, n = 10). Body weights were recorded daily before dosing. Food consumption was monitored biweekly throughout the dosing period. Six dams in each dose group were killed by carbon dioxide asphyxiation at GD 19.5 within 24 h of last dose, when there was a rapid increase to the peak of testosterone production. 20 According to the study of Thompson et al., steroidogenesis-related protein levels remained low 24 h after withdrawal of DBP treatment but increased 48 h after cessation of DBP exposure. 21 Upon killing, live fetuses were immediately removed and sexed by measuring anogenital distance under a dissecting microscope with a micrometer lens (accuracy 0.05 mm). Testes were isolated from male fetuses. Another four dams in each group were raised to delivery. On postnatal day (PND) 45, which was considered to be in the sexual developmental period of rat as well as the pubertal stage of human, the male pups were checked for occurrence of cryptorchidism and hypospadias by visual inspection and palpation. Testes were isolated from male offspring. The specimens isolated from the same pregnant rat were pooled and immediately snap-frozen for protein extraction. The others were fixed in glutaraldehyde for immunocytochemical analysis and electron microscopic observation or in Bouin’s liquid for hematoxylin and eosin (H&E) staining.
Transmission electron microscopic observation
For TEM, the testes isolated from male rat offspring were cut to less than 1 mm 3 , perfused with 5% glutaraldehyde at 4°C for 2 h, rinsed in phosphate-buffered solution (PBS), and post-fixed in 1% osmium tetroxide (OsO4) at 4°C for 2 h. The samples were then dehydrated in a graded series of acetone, infiltrated in propylene oxide, and embedded in Araldite M (Sigma-Aldrich, Taufkirchen, Germany). Ultrathin sections were cut and stained with uranyl acetate and lead citrate. Tissue sections were examined using a Tecnai G2 transmission electron microscope (FEI Company, Hillsboro, USA) at 120 kV. Evaluation was limited to characterization of subtle lesions, abnormal cells, and apoptosis.
Pathological examination
The testes samples of pubertal rats were embedded in paraffin. Sections were cut at about 4 μm in thickness, dried in a constant temperature box at 45°C, deparaffinization was performed with xylene, and then stained with H&E. Tissue sections were examined using optical microscope, to observe the development of germ cells at all levels.
Apoptosis detection
The testis tissue samples were stained using the TUNEL method. Apoptotic cells were counted under a microscope at a magnification of 200×. Using the TUNEL technique, all cut sections were preserved with lysine for three nights at 37°C and then treated for one night at 65°C in an incubator. Deparaffinization was performed by processing the cut sections three times with xylene (15 min; first xylene in the incubator, and the others at room temperature). The tissue sections were then rehydrated by flushing with a series of alcohol solutions of decreasing degree (absolute, 95%, and 80%); the samples were then stored in distilled water for 5 min. After absorbing water from the edge of each section, proteinase K (Tiangen, China) was applied for 15 min at 37°C. The sections were then washed thrice with PBS for a period of 3 min each. After drying the cross sections, 3% H2O2 was applied for 5 min to inhibit tissue endogenous peroxidase, and the sections were then washed thrice with PBS for 3 min each. The cross-section slices were then dried, and an equilibration buffer was applied for 10 min at room temperature. A total of 55 μl of the enzyme terminal deoxynucleotidyl transferase was then applied to each cross section. The coverslips of each cross section were incubated for 1 h at 37°C. After removal from the incubator, stop/wash buffer was then applied to the sections for 10 min at room temperature. The sections were then washed three times for 3 min each with PBS at 37°C, dried, and incubated with anti-streptavidin peroxidase at 37°C for 30 min. The sections were then washed with PBS three times for 3 min each to determine the visibility of the TUNEL reaction, and the samples were then stained with diaminobenzidine (DAB). After washing with distilled water, counterstaining was performed using hematoxylin. The sections were then washed three times with distilled water. After three times searing processing with xylene for 20 min, closure was performed with neutral resin. According to the distribution of positive testicular cells, six positive views of each paraffin-embedded sample from each group were selected under the microscope to count the number of positive cells as apoptotic index.
Immunohistochemical staining
Sections of 4-mm-thick wax-embedded tissue were cut and placed on 3% aminopropylethoxysilane-coated glass slides. These sections were then de-waxed and rehydrated through descending grades of alcohol to distilled water. Immunohistochemistry (IHC) was performed on the sections as previously described. 22 Briefly, sections were incubated in 1% hydrogen peroxide, washed in PBS, and nonspecific protein binding was blocked with rabbit serum (Beijing ZhongShan Biotechnology Co.). Sections were then incubated overnight in a humid chamber at 4°C with affinity purified goat antibodies, specific to Bcl-2, Bax or p53, at 1:100 antibody dilution. Following three PBS washes, sections were incubated with horseradish peroxidase (HRP)-conjugated anti-goat secondary antibody, at 1:200 antibody dilution. Immunoreactive sites were visualized as brown in color with DAB and mounted for bright field or differential interference contrast microscopy. Negative controls were incubated with dilution lacking primary antibody and were otherwise subject to all the immunohistochemical procedures. The staining results were analyzed using semi-quantitative method. Staining intensity score was calculated by staining intensity of most cells minus background coloration (score 0 for no obvious coloring, score 1 for light yellow, score 2 for deep yellow, and score 3 for brown). Six different horizons (200×) of each paraffin selection were observed, and the number of positive cells of 100 cells in each field were counted to calculate the percentage of positive cells (score 0 for 0–5%, score 1 for 6–25%, score 2 for 26–50%, score 3 for 51–75%, and score 4 for 76–100%). A specific staining index, expressed as total immune staining score was used, and was calculated by staining intensity times the fraction of positive cells, giving a maximal score of 12 points. The staining was graded independently by two blinded investigators. The interobserver agreement on staining index was 85%; slides with incongruent gradings were scrutinized again and the results weighted together.
Western blot
Testes isolated from offspring of the same pregnant rat were pooled to obtain a sufficient sample for analysis. Protein extracts were prepared with the treatment of tissues in lysis buffer (7 M urea, 2 M thiourea, 4% (w/v) CHAPS, 2% (v/v) IPG buffer, pH 3∼10, 2% (w/v) DTT) in the presence of protease inhibitor—1 mM phenylmethylsulfonyl fluoride. The mixture was homogenized (Ultra Turrax, IKA, Staufenim Breisgau, Germany) at 11,000 p.m. × 2 s × 12 on ice and then left at 4°C for 1 h. The mixture was centrifuged at 40,000 × g for 1 h at 4°C, and the supernatant saved and stored at −70°C. The protein concentration was estimated by the method of Bradford 23 using BSA as the standard. Samples containing 50 μg of protein from control and DBP-treated rat testes were electrophoresed on a 12% SDS polyacrylamide gel and transferred to a nitrocellulose filter (Amersham Life Science, Buckinghamshire, UK). The filters were blocked in PBS containing 5% nonfat milk powder for 1 h and then incubated overnight with a 1:1000-diluted anti-rat Bcl-2, Bax, or p53 goat polyclonal antibody in PBS containing 5% nonfat milk powder. They were washed three times (10 min each) with PBS. The filters were then incubated for 1 h with HRP-conjugated anti-goat IgG (Beijing ZhongShan Biotechnology Co.). At the same time, a 1:1000-diluted anti-beta actin antibody was used to verify the amount and integrity of the proteins. Specific proteins were detected by an ECL kit (Amersham Biosciences, Buckinghamshire, UK) and AlphaImager (FluorChem5500; Alpha Innotech, San Leandro, California, USA). The protein expression level was analyzed by AlphaEaseFC software (Alpha Innotech).
Statistical analysis
The data of experimental results were described by mean and standard deviation. Independent samples t-test and Mann–Whitney U-test were performed to determine the significant differences between the two groups by ImageMaster and SPSS statistics 20.0 (IBM, New York, New York, USA). The p values less than 0.05 were considered statistically significant.
Results
Reproductive malformations of male rat offspring
In the male rat offspring dissected at GD 19.5 and PND 45, the position of testes in DBP exposed group was found to be higher than in control group (Figure 1). The incidence of hypospadias and cryptorchidism at PND 45 in DBP-exposed group was 37.5% and 65.6%, respectively, and the incidence of simultaneously occurring hypospadias and cryptorchidism was 31.3%. No male rat with hypospadias or cryptorchidism was found in control group (Table 1).

Gross anatomical view of reproductive malformations of pubertal male rat offspring. (a) Normal penis and testes in scrotum in control group. (b) Hypospadiac penis and undescended testes in DBP-exposed group. DBP: di-n-butyl phthalate.
Reproductive malformations of male rat offspring observed at PND45 after prenatally (GD 12 to GD 19) exposure to DBP.

PND: postnatal day; GD: gestation day; DBP: di-n-butyl phthalate.
aSignificantly different from control (p < 0.05).
Morphological changes of testis
Transmission electron microscope
At GD 19.5, more cell vacuolization, mitochondrial crest breakage, and vacuole degeneration were observed in DBP-exposed embryonic rat testicular cytoplasm than in control group. MNG occurred in the embryonic rat testis after DBP exposure. At PND 45, more germ cell and Sertoli cell apoptosis as well as cell vacuolization were found in DBP-exposed pubertal male rats.
H&E staining of pubertal male rat testis
Spermatogonia, primary spermatocytes, secondary spermatocytes, spermatids, and spermatozoa (the sperm) were observed in the convoluted seminiferous tubules of PND 45 rat testis in control group (Figure 2(a)). While in DBP-exposed group, mainly Sertoli cells manifesting vacuolization changes existed in the convoluted seminiferous tubules. Germ cells were significantly reduced at all levels, only spermatogonia were visible, with no sperm (Figure 2(b)). No significant difference of Leydig cells was observed between the two groups.
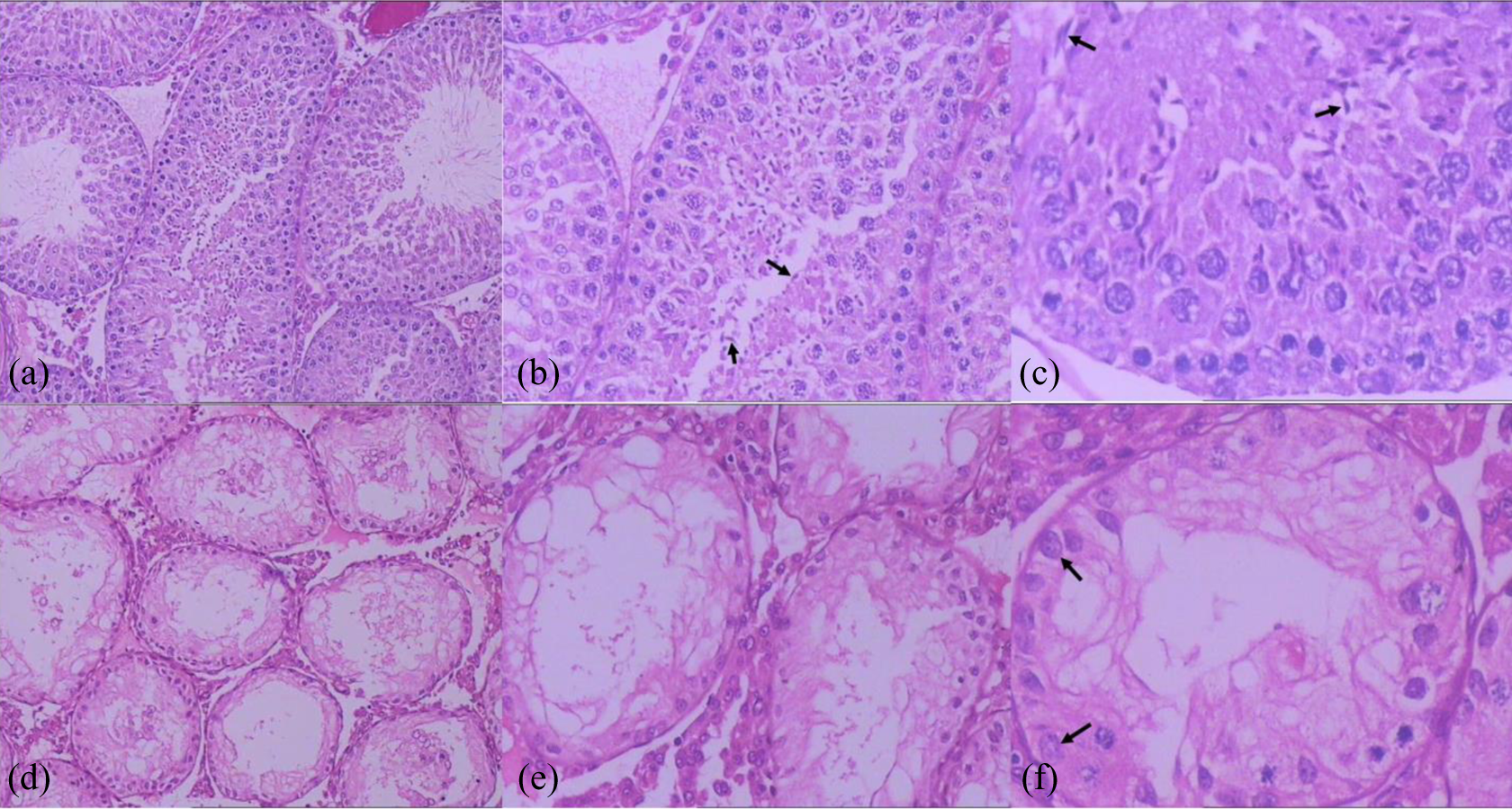
H&E staining of the testis tissue of the pubertal (PND 45) male rat offspring in the control and DBP-exposed groups. (a) Control group (×100); (b) control group (×200); (c) control group (×400), the arrows indicate sperm. (d) DBP-treated group (×100); (e) DBP-treated group (×200); (f) DBP-treated group (×400), the arrows indicate spermatogonia. In DBP-treated group, germ cells of all levels were significantly reduced, and no mature sperm was observed. DBP: di-n-butyl phthalate; H&E : hematoxylin and eosin.
Testicular cell apoptosis detected by TUNEL
Less testicular cell apoptosis was observed in DBP-exposed group than that in control group at GD 19.5 (apoptotic index 1.00 ± 0.00 vs. 3.38 ± 1.15; t = 8.279, p = 0.000; Z = −2.747, p = 0.006). The apoptotic cells which occurred in control group were mainly found in germ cells. Conversely, apoptosis observed in the testes of DBP-exposed group was significantly higher than that in control group at PND 45 (apoptotic index 13.89 ± 4.60 vs. 3.74 ± 1.15; t = −17.213, p = 0.000; Z = −6.768, p = 0.000). These apoptotic cells mainly appeared in germ cells and secondly in Sertoli cells (Figures 3 and 4). In the control group, similar apoptotic index was obtained in GD 19.5 and PND 45 testicular specimens.
Apoptotic indexes of the testicular cells of male rat offspring in the control and DBP-treated groups at GD 19.5 and PND 45 (TUNEL ×200). (a) control group at GD 19.5; (b) DBP-treated group at GD 19.5; (c) control group at PND 45; (d) DBP-treated group at PND 45. The arrows indicate apoptotic cells. Scale bar, 50 μm. DBP: di-n-butyl phthalate; PND: postnatal day; GD: gestation day.

Error bars: The comparison of apoptotic indexes between the control and DBP-treated groups at GD 19.5 and PND 45. Values are mean ± SEM representing six animals in the control group and six animals in the DBP-treated group. *p < 0.01 versus control. DBP: di-n-butyl phthalate; PND: postnatal day; GD: gestation day; SEM: standard error of mean.
Expression of apoptosis regulatory proteins
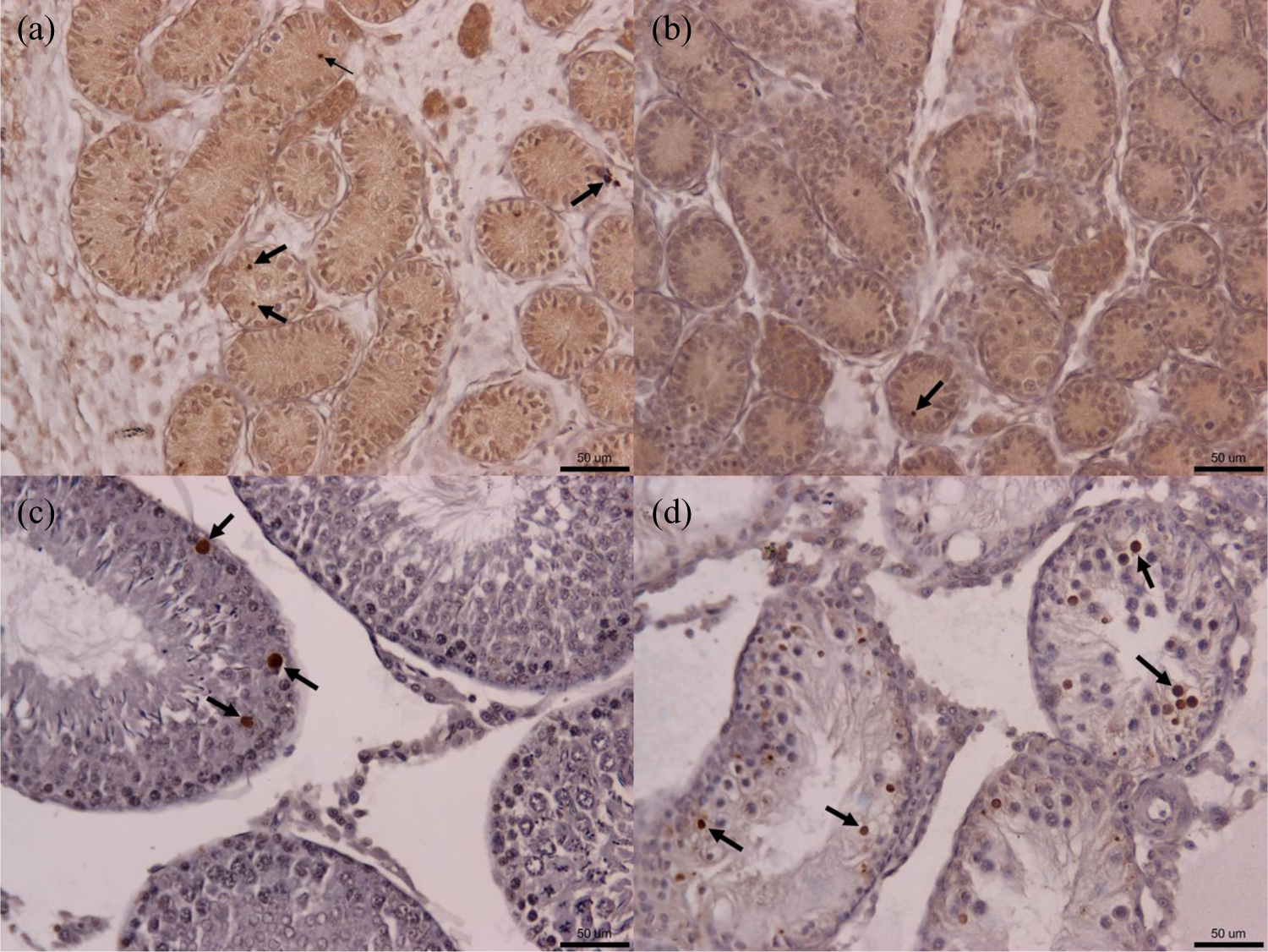
To analyze the expression of apoptosis regulatory proteins Bcl-2, Bax, and p53 in rat testis, immunohistochemical studies were performed. The results of the GD 19.5-old rat testis tissue sections showed a stronger staining of Bcl-2, which was mainly expressed in cytoplasm of germ cell and Leydig cell, in DBP-exposed group (Figures 5 and 7). Relatively faint positive signal was detectable in control group. The PND 45-old rat testis tissue sections showed a significantly enhanced staining of Bax and p53, mainly expressed in nucleus and cytoplasm of germ cell, respectively, in the DBP-exposed group (Figures 6 and 7). The results of western blot confirmed the IHC findings (Figures 8 and 9).

Expressions of Bcl-2, Bax, and p53 in the testis tissue (brown) of the GD 19.5 male rat offspring in the control and DBP-treated groups (IHC × 200). The GD 19.5-old rat testis tissue sections showed a stronger staining of Bcl-2 in DBP-treated group. Bcl-2 was mainly expressed in the cytoplasm of germ cells and Leydig cells, and it was less expressed in the cytoplasm of Sertoli cells. The arrows show positive staining. Arrowheads indicate MNG. Scale bar, 50 μm. GD: gestation day; DBP: di-n-butyl phthalate; IHC: immunohistochemistry; MNG: multinucleated gonocyte.

Expressions of Bcl-2, Bax, and p53 in the testis tissue (brown) of PND 45 male rat offspring in the control and DBP-treated groups (IHC × 200). The PND 45-old rat testis tissue sections showed a significantly enhanced staining of Bax and p53 in DBP-treated group. Bax was mainly expressed in the cytoplasm of germ cells, and it was weakly or even not expressed in Leydig cells and Sertoli cells. p53 was mainly expressed in the nucleus of germ cells and less expressed in the cytoplasm of Leydig cells and germ cells. The arrows show positive staining. Scale bar, 50 μm. PND: postnatal day; GD: gestation day; DBP: di-n-butyl phthalate.

Error bars: The differential expression of Bcl-2, Bax, and p53 between the control and DBP-treated groups at GD 19.5 and PND 45. Values are mean ± SEM representing six animals in the control group and six animals in the DBP-treated group. *p < 0.05 versus control. PND: postnatal day; GD: gestation day; DBP: di-n-butyl phthalate; SEM: standard error of mean.

Expression of Bcl-2, Bax, and p53 in the testis tissue of GD 19.5 and PND 45 male rat offspring in the control and DBP-treated groups by western blot. β-actin was used as an internal control. PND: postnatal day; GD: gestation day; DBP: di-n-butyl phthalate.
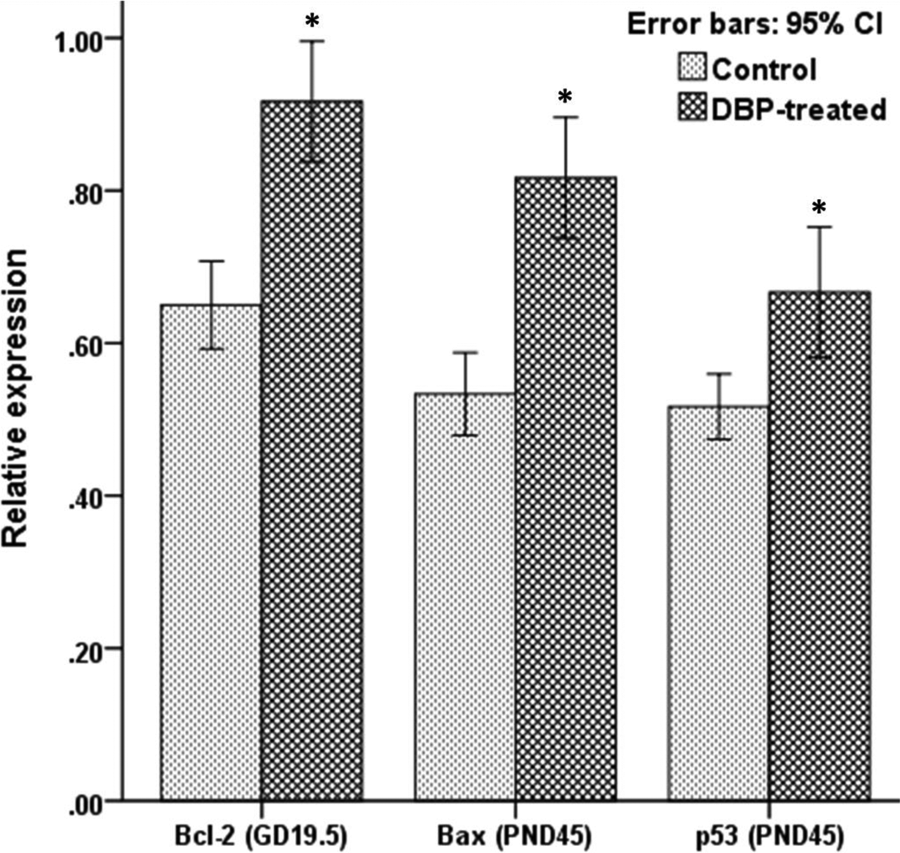
Error bars: The differential expression of Bcl-2, Bax, and p53 between the control and DBP-treated groups at GD 19.5 and PND 45 by western blot. Values are mean ± SEM for pups from six pregnant rats per group. *p < 0.05 versus control. PND: postnatal day; GD: gestation day; DBP: di-n-butyl phthalate; SEM: standard error of mean.
Discussion
Since the concept of TDS was proposed in 2001, people have had a new understanding of male infertility. Although there seems to be no connection between these disorders, they should be considered as different clinical manifestations of a syndrome, which share many risk factors as well as being risk factors for each other. 24 Pregnant rats exposed to DBP leaded to a variety of male reproductive malformations similar to the performances of TDS, and many repeated animal experiments also confirmed that DBP was the most thorough and representative testicular toxic substance studied. Using this animal model, we could prove the hypothesis of TDS and study its pathogenesis. In addition to testicular cancer, all of the TDS phenotypes could be induced by administration of PAEs to rats during late pregnancy, which could lead to significant reduction of the expression of key enzymes involved in testosterone synthesis and decrease in the testosterone level in vivo in the embryonic rat offspring, followed by a decline in fertility after sexual maturity. 12,25 –28 The infertility might be developed as a result of decreased testosterone secreted by Leydig cells or the direct damage of germ cells and Sertoli cells.
Apoptosis is a process of cell death triggered by the pre-stored procedure of death in vivo and in vitro, which is in turn regulated by related genes. In order to induce the active suicide behavior of cells, the transcription and expression of some intracellular genes can be activated by extracellular factors. Normal testicular cell apoptosis was considered to be the essential process of testicular development, and germ cell apoptosis was an important way to remove the damaged germ cells and regulate the balance of cell proliferation and differentiation. During the critical period of testicular development, the inhibition of apoptosis would affect the maturation of spermatogonia, however, increased apoptosis might lead to testicular spermatogenic dysfunction and infertility. 29 Shultz et al. 28 reported that the expression of cell survival-related factors TRPM-2 and Bcl-2 was increased in embryonic testis after maternal exposure to DBP. As a proto-oncogene, the high expression of Bcl-2 could inhibit apoptosis induced by a variety of pro-apoptotic factors, such as X-ray and chemical drugs. In our study, using the methods of TEM observation and apoptosis detection by TUNEL, we found that in utero exposure of DBP significantly reduced the apoptosis of germ cell in embryonic rat testes but increased the apoptosis of germ cell and Sertoli cell in pubertal rat testes. No abnormal apoptosis of Leydig cell was observed. The results of IHC and western blot showed increased expression of Bcl-2 in the DBP exposed rat testes at GD 19.5, which indicated that DBP inhibited the apoptosis of germ cell in embryonic rat testis by upregulating the expression of anti-apoptotic factors. On the other hand, enhanced expression of Bax and p53 in PND 45 rat testes following maternal exposure of DBP revealed that upregulated pro-apoptotic factors might play an important role in facilitating germ cell apoptosis in pubertal rat testis. During late embryonic rat testis, the decreased apoptosis of germ cell might inhibit the maturation of spermatogonia, which would affect the development of spermatocytes and spermatids, leading to spermatogenic dysfunction. The increased apoptosis of Sertoli cell, which played an important role in supporting nutrition to germ cell and regulating germ cell cycle, would affect germ cell development in the pubertal stage. Meanwhile, the increased apoptosis of germ cell would directly lead to spermatogenic dysfunction. Just as we observed in this study by H&E staining, germ cells at all levels, except for spermatogonia, were significantly reduced and no sperm was displayed in DBP-exposed pubertal rat testes.
P53 is a DNA-binding protein encoded by wild-type p53 gene, which has the function of inducing apoptosis. It can activate DNA repair proteins when DNA has sustained damage. If DNA damage proves to be irreparable, it will initiate apoptosis and kill the cells that may evolve into cancer, in a rudimentary way. Germ cell apoptosis induced by p53 in DBP-exposed male rat offspring might also have a role in inhibiting the occurrence of testicular cancer, which was one of the phenotypes of TDS. Similar morphology and common biochemical features of carcinoma in situ and gonocytes, 30 as well as the correlation between presence of MNG in cryptorchid testes in boys and development of testicular cancer later in life, 16 led to the hypothesis that aneuploid embryonic germ cells are the precursors of germ cell tumorigenesis. 30,31 In DBP exposed rats, polyploid MNG persisted 5 days after birth and migrated to the basal lamina, where they might become progenitors of genetically altered spermatogonia. Previous data demonstrated that occurrence of MNG significantly increased following in utero exposure to the high dose of DBP. 15 Nevertheless, testicular germ cell cancer has not yet been reported in rats exposed in utero to various doses of DBP. Gestational exposure to DBP in p53-null mice induced greater numbers of MNG than in p53-heterozygous or wild-type mice, and histologic examination of adult mice exposed in utero to DBP revealed persistence of abnormal germ cells only in DBP-treated p53-null mice. 32 This model identified a role for p53 in the apoptosis of DBP-induced abnormal germ cells, for example, MNG, that could lead to germ cell cancer.
Cytoplasmic vacuolization observed in DBP-exposed rat testes might be another phenomenon of cell death, which was called non-apoptotic programmed cell death. 33,34 This alternative form of programmed cell death appeared during development and was lack of caspase activation or response to caspase inhibitors. Vacuolization derived from endoplasmic reticulum and mitochondrion, and lack of apoptotic body formation, was considered to be its features. Coexpression of Bak and Bcl-XL could provoke cytoplasmic vacuolization. 35 Currently, it is difficult to determine whether the abnormal expression of Bcl-2 family and p53 in the testis of male rat offspring following in utero exposure to DBP has participated in the process of cell vacuolization or not.
Although we found that DBP exposure during pregnancy leaded to different effects on the apoptosis of testicular germ cells in late embryonic and adolescent offspring, it did not mean that all PAEs would cause the similar effects. An apoptosis assessment about early testicular effects in rats perinatally exposed to di(2-ethylhexyl) phthalate (DEHP) in combination with di(2-ethylhexyl) adipate (DEHA) revealed that a few animals had a high number of TUNEL positive apoptotic cells in degenerating seminiferous tubules at PND 22 and 190, whereas most exposed animals had low levels of germ cell apoptosis at GD 21, PND 22, or PND 26. 36 While it was considered that apoptosis did not appear to be an important element of the testicular effects of perinatal exposure to DEHP or to DEHP in combination with DEHA in fetal testes, apoptosis was involved in the degeneration of seminiferous tubules in prepubertal and adult testes.
The present study suggested that in utero exposure of high-dose DBP resulted in opposite effects on testicular cell apoptosis in late embryonic and pubertal male rat offspring (Figure 10). DBP exposure during pregnancy leaded to overexpression of anti-apoptotic protein Bcl-2 in late embryonic rat testis and inhibited the apoptosis of germ cell, which could influence the maturation of spermatogonia. Due to the persistent effect of maternal exposure to DBP on postnatal male rat offspring, the expression of pro-apoptotic proteins Bax and p53 was upregulated in the testis of pubertal male rat offspring, resulting in increased apoptosis of germ cell and Sertoli cell. The excessive apoptosis of germ cell could cause spermatogenic dysfunction, and the same impact on Sertoli cell could affect the development of germ cell. Since either spermatogonial maturation or germ cell development was interfered with, and spermatogenic dysfunction occurred, these induced disorders could cause male infertility. Searching for small RNAs which function in regulating the aberrant expression of these apoptosis-related proteins, and studying its molecular mechanisms, has been our subsequent ongoing research.

Flow diagram of the effects of in utero DBP exposure on testicular cell apoptosis in the testis of late embryonic and pubertal male rat offspring, according to the current study. The sign “×” indicates inhibition or blocking. DBP: di-n-butyl phthalate.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This study was supported by Science and Technology Development Program in Nanjing City (201303028).
