Abstract
Glutathione-S-transferase (GST) and cytochrome P450 family 1 subfamily A polypeptide 1 (CYP1A1) metabolize and detoxify carcinogens, drugs, environmental pollutants, and reactive oxygen species. Changes of GST expression in tissues and gene mutations have been reported in association with many neoplastic skin diseases and dermatoses. Widely used artificial food coloring additives (AFCAs) also reported to effect primarily behavioral and cognitive function and cause neoplastic diseases and several inflammatory skin diseases. We aimed to identify the changes in expression of GSTs, CYP1A1, and vascular endothelial growth factor (VEGF) in rat skin which were maternally exposed AFCAs. A rat model was designed to evaluate the effects of maternal exposure of AFCAs on skin in rats. “No observable adverse effect levels” of commonly used AFCAs as a mixture were given to female rats before and during gestation. Immunohistochemical expression of GSTs, CYP1A1, and VEGF was evaluated in their offspring. CYP1A1, glutathione S-transferase pi (GSTP), glutathione S-transferase alpha (GSTA), glutathione S-transferase mu (GSTM), glutathione S-transferase theta (GSTT), and VEGF were expressed by epidermal keratinocytes, dermal fibroblasts, sebaceous glands, hair follicle, and subcutaneous striated muscle in the normal skin. CYP1A1, GSTA, and GSTT were expressed at all microanatomical sites of skin in varying degrees. The expressions of CYP1A1, GSTA, GSTT, and VEGF were decreased significantly, while GSTM expression on sebaceous gland and hair follicle was increased. Maternal exposure of AFCAs apparently effects expression of the CYP1A1, GSTs, and VEGF in the skin. This prominent change of expressions might play role in neoplastic and nonneoplastic skin diseases.
Introduction
Glutathione-S-transferase (GST)—which includes classes alpha (A), mu (M), pi (P), theta (T), and microsomal GST—metabolizes and detoxifies carcinogens, cancer chemotherapeutic agents, therapeutic drugs, insecticides, herbicides, environmental pollutants, and reactive compounds produced by reactive oxygen species (ROS). 1 GST also indirectly controls the levels of other antioxidant and drug-metabolizing enzymes. 2 Overexpression of GST in mammalian tumor cells has been associated with resistance to various anticancer agents and chemical carcinogens. GST negatively regulates inflammation-associated proteins and apoptosis-associated proteins. 3,4 GSTs and cytochrome P450 family 1 subfamily A polypeptide 1 (CYP1A1) play an important role in the elimination of oxidative stress, and they have a defined relationship with neoplastic and nonneoplastic skin diseases. 5,6 Changes to GST expression in tissues and gene mutations have been reported in many neoplastic skin diseases and dermatoses. 5 –22
Several skin diseases are believed to be associated with oxidative stress, including psoriasis, 7,8 urticaria, 9 polymorphic light eruption (PLE), 10 lichen planus, 11 dermatophytoses, 12 inflamed skin, 6 vitiligo, 13 solar keratoses, 14 malignant melanoma, 15,16 and basal cell carcinoma (BCC). 17 The effects of artificial food coloring additives (AFCAs) have been investigated previously, and in addition to their behavioral, cognitive, 18,19 and systemic effects, AFCAs are believed to be associated with several skin diseases, including psoriasis, atopic dermatitis, 20 urticaria, 21 erythema multiform, cutaneous vasculitis, and allergic contact dermatitis. 22 The effects of AFCAs on oxidative stress enzymes 23 –25 and the inhibition of endogenous antioxidant defense enzymes 25,26 in the skin have been identified. Amin et al. revealed that rats that consumed high and low doses of tartrazine or carmoisine exhibited a significant increase in serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase activities. 23 In this investigation, we evaluated changes in the immunohistochemical expression of GSTs, CYP1A1, and vascular endothelial growth factor (VEGF) in the skin of rats maternally exposed to AFCAs.
Materials and methods
The study was performed according to the recommendations for animal care and experimentation of the Pertinent European Communities Council Directive (86/609/EEC), and all the procedures were approved by the ethical committee at the experimental animal laboratory of the Süleyman Demirel University School of Medicine. Thirty adult female Wistar albino rats were used in the study. Rats were maintained ad libitum with water and food and housed separately on a 12-h light/12-h dark cycle at room temperature (23 ± 2°C). Rats were separated into control (n = 15) and study groups (n = 15). In the study group, each rat was housed in a separate mesh cage. AFCA mixtures were given from 1 week before pregnancy to birth. After 1 week from starting to give the food dye, a male rat was left in each cage for copulation to produce a pregnancy over the course of 1 week. The first day that male rats were left in the cages was accepted as the first day of pregnancy. Offspring were left with their mothers for 1 month and then they were separated into two groups (study and control) of 20 rats each. One male and one female offspring was randomly selected from each mother. Finally, the following four groups were obtained: female controls (n = 10), male controls (n = 10), AFCA females (n = 10), and AFCA males (n = 10). Rats were housed in the laboratory until they reached maturity at 3 months for assessment of the effects of maternal exposure to food dyes.
The AFCA mixture and the doses we used in the study are shown in Table 1. The doses used in the study were the “no observable adverse effect levels” of these AFCAs, which were obtained from monographs of the Joint Expert Committee on Food Additives. 36 AFCA mixtures were prepared weekly and stored at 4°C. Each color additive was weighed separately and dissolved in water. Rats were given 1 ml of mixture per 100 g of body weight once daily by gavage. The control group was synchronously given the same volume of tap water.
Mixture and the doses of AFCAs.

AFCA: artificial food coloring additive.
Rats were anesthetized and killed with an intraperitoneal (ip) injection of 10% ketamine (80 mg/kg ip; Brema-Ketamin, 10%) and 2% xylazine (10 mg/kg ip). Samples of dorsal skin tissue at least 2 cm long were collected and fixed in 10% buffered formaldehyde. After standard tissue processing, tissues were embedded in paraffin blocks, sliced to 4–6 µm in thickness, and stained with hematoxylin–eosin. During immunohistochemical staining, endogenous peroxidase activity was blocked by incubating the sections in 1% hydrogen peroxide (v/v) in methanol for 10 min at room temperature. The sections were subsequently washed in distilled water for 5 min, and antigen retrieval was performed for 3 min using a 0.01 M citrate buffer (pH 6.0) in a domestic pressure cooker. The sections were transferred into 0.05 M Tris-HCl (pH 7.6) containing 0.15 M sodium chloride Tris-buffered saline. After washing in water, the sections were incubated at room temperature for 10 min with super block (SHP125; ScyTek Laboratories, Logan, Utah, USA) to block nonspecific background staining. The following antibodies were included in the analysis: anti-CYP1A1 (H-70 polyclonal, Santa Cruz Biotechnology, Dallas, Texas, USA; 1:100 dilution), anti-GSTT (GSTT1/glutathione S-transferase theta 1; clone polyclonal, Bioss Inc., Woburn, Massachusetts, USA; 1:350 dilution), anti-GSTM (glutathione S-transferase mu 1/GSTM1 antibody; clone 1H4F2, Novus Biologicals, Littleton, Colorado, USA; 1:250 dilution), anti-GSTA (GSTA1/glutathione S-transferase alpha; clone polyclonal, Bioss Inc.; 1:100 dilution), anti-GSTP (glutathione S-transferase P 1(GSTP1; clone polyclonal, Boster Biological, Pleasanton, California, USA; 1:300 dilution), and anti-VEGF (polyclonal, Novus Biologicals; 1:250 dilution).
The immunohistochemically stained sections were evaluated and scored in double-blinded fashion. Intensity of staining on the epidermal keratinocytes, dermal fibroblasts, hair follicles, sebaceous glands, peripheral nerve cells, vascular endothelia, vascular walls (smooth muscle), subcutaneous striated muscle, and adipocytes was scored semiquantitatively according to the following degrees of staining: 0 (none), 1 (mild), and 2 (marked). All statistical assessments were performed by SPSS v.21 for Windows. Pearson’s χ 2 test and Fischer’s exact test were used to compare the changes between tissues in the enzyme groups. The statistical significance level was established at p < 0.05, and the confidence interval was 95%.
Results
In the control group, all markers were expressed by epidermal keratinocytes, dermal fibroblasts, sebaceous glands, hair follicles, and subcutaneous striated muscle. CYP1A1, GSTA, and GSTT were expressed at all microanatomical sites of the skin in varying degrees.
GSTP was expressed prominently in epidermal keratinocytes, sebaceous glands, and striated muscle and expressed mildly in dermal fibroblasts and hair follicles. GSTM was markedly expressed in epidermal keratinocytes, sebaceous glands, hair follicles, dermal fibroblasts, and striated muscle, whereas it was mildly expressed in peripheral nerve cells, vascular endothelia, and vascular smooth muscle. GSTP expression in the AFCA group did not vary significantly according to microanatomical site.
There was a significant difference in CYP1A1 expression between the AFCA group and the control group in regards to the sebaceous glands (Fisher’s exact test, p = 0.003) and striated muscle (Pearson’s χ 2 test, χ 2 = 9.46; p = 0.009; Figure 1). In the control group, 100% of sebaceous glands stained mildly, whereas in the AFCAs group, 60% stained mildly and 40% stained markedly. Striated muscle stained 95% mildly and 55% mildly in the control and study groups, respectively (Table 2).

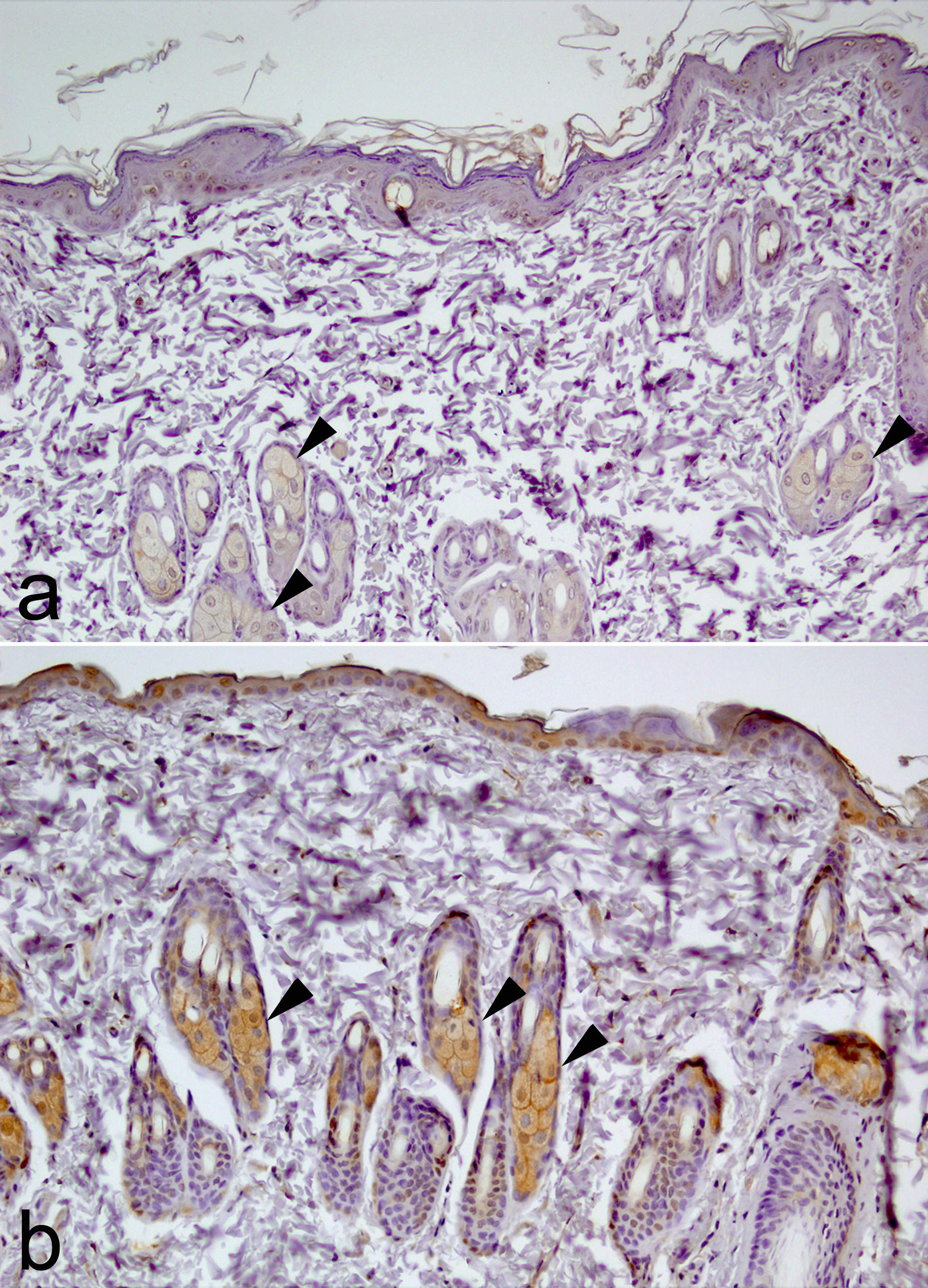
Immunohistochemical expression of CYP1A1 in rat skin. (a) Mildly stained sebaceous glands in the control group and (b) markedly stained sebaceous glands in the AFCA group (CYP1A1 immunohistochemical staining, original magnification ×100). CYP1A1: cytochrome P450 family 1 subfamily A polypeptide 1; AFCA: artificial food coloring additive.
p Value of enzymes scores in subanatomical sites of skin.

CYP1A1: cytochrome P450 family 1 subfamily A polypeptide 1; GSTA: glutathione S-transferase alpha; GSTM: glutathione S-transferase mu; GSTP: glutathione S-transferase pi; GSTT: glutathione S-transferase theta; VEGF: vascular endothelial growth factor.
aNo statistics was computed because values were constant.
bFisher’s exact test.
Bold values indicate statistical significance and closeness to statistical significance.
GSTA expression in dermal fibroblasts in the study group was significantly lower than in the control group (Fisher’s exact test, p = 0.008). In the study group, there was no staining, whereas 35% of the control group was stained mildly. There were no statistically significant differences between the control and AFCA groups in terms of other histological components (p > 0.05).
GSTM expression in sebaceous glands was significantly higher in the study group (Pearson’s χ 2 test, χ 2 = 7.06; p = 0.029; Figure 2). In the control group, 70% of sebaceous glands displayed mild GSTM expression, whereas in the study group, 100% displayed mild expression. Mild GSTM expression in hair follicles was 25% and 55% in the control and AFCA groups, respectively (Pearson’s χ 2 test, χ 2 = 3.73; p = 0.053). There were no statistically significant differences between the two groups in terms of GSTP expression.
Immunohistochemical expression of GSTM in the control group (a) and the study group (b). Mild expression in sebaceous glands was observed in both groups (70% in the control group and 100% in the study group; GSTM immunohistochemical staining, original magnification ×100). GSTM: glutathione S-transferase mu.
GSTT expression in vascular endothelia was significantly different between groups (Fisher’s exact test, p = 0.008). In the control group, 35% of endothelia exhibited mild GSTT staining, whereas there was no GSTT staining in the AFCA group.
Expression of VEGF in vascular endothelia (Pearson’s χ 2 test, χ 2 = 16.46; p < 0.0001) and epidermal keratinocytes (Pearson’s χ 2 test, χ 2 = 8.64; p = 0.003) was significantly lower in the AFCA group.
Discussion
GST genes play a role in the detoxification of carcinogens and mutagens, environmental pollutants, therapeutic drugs, and reactive compounds produced by ROS, and GSTP plays a major role in carcinogenesis that is distinct from its role in detoxification. 1,2,5,14,37,38 GST-π has been immunohistochemically detected in human tumor cell lines and in normal human tissues. In the epidermis, sebaceous glands and sweat glands express GST-π regardless of body region. GST-π antibody expression has been found to be very strong in the stratum basale and weaker in the superficial layers of the skin. 37 In the dermis, sebaceous and sweat glands also tend to stain strongly. 37,39 Raza et al. demonstrated the existence of multiple forms of GST (alpha, mu, and pi classes) in human and rodent skin. Among these, the pi class is the predominant isozyme. GST and cytochrome P-450 are predominantly localized in sebaceous glands and in the outer root sheath of the hair follicles. 39 In our control group, CYP1A1 was predominantly expressed in sebaceous glands and in the hair follicles, as Raza et al. reported. In addition to sebaceous glands and hair follicles, epidermal keratinocytes, subcutaneous striated muscle, and dermal fibroblasts also expressed CYP1A1.
The role of GST M1, T1, P1, and Z1 gene polymorphisms in susceptibility to solar keratoses has also been examined, and a significant association between GSTM1, the GSTT1 null genotype, and solar keratoses has been detected. 14,40 GST activity has been found to be significantly decreased in malignant melanoma. 15 Ibarrola-Villava et al. proposed GSTP1 as a malignant melanoma susceptibility gene, 16 and Yang et al. showed that GSTP1 variants were inversely associated with BCC rates in naevoid BCC syndrome. 17 Decreased levels of GST have been reported in induced skin inflammation. 6 Overexpression of GSTP1 has been observed above the middle layer of the epidermis in psoriatic skin lesions. Increased GSTP1 may play a role in reduction of ROS stress in cells, thereby preventing ROS-induced DNA damage or cell death. 7 In addition, microsomal glutathione S-transferase 2 mutations were observed in a Chinese family affected by psoriasis vulgaris. 8 Millard et al. obtained experimental data supporting the role of ROS in the pathogenesis of PLE, and the family of GST enzymes plays a critical physiological role in cellular protection against this oxidative damage. The GSTP1 allele has been associated with PLE and plays a role in ROS production in the pathogenesis of PLE. 10 However, Zirbs et al. did not find any differences in the prevalence of GST (genotypes GSTM1, GSTT1, and GSTP1) polymorphisms between patients with PLE and healthy controls. 41 Increased nitric oxide levels in patients with lichen planus 11 and vitiligo 13 have been reported, resulting in production of ROS, and it has been suggested that oxidative stress may also lead to increased production of ROS, thus resulting in increased lipid peroxidation. Unlike the study by Ryu et al., no significant difference was observed in GSTP expression between the study group and controls in this study. Marked changes of other subtypes of GST expression in rats exposed to AFCAs suggest that AFCAs provoke a predisposition for solar keratosis, BCC, malignant melanoma, and dermatoses.
High and moderate doses of tartrazine are thought to promote lipid peroxidation products and ROS, inhibiting endogenous antioxidant defense enzymes. 26 Orally administered tartrazine causes depression of the antioxidant enzyme system, resulting in an imbalance of the prooxidant and antioxidant system in the testis of rats. 25 Amin et al. revealed that rats given high and low doses of tartrazine or carmoisine exhibited a significant increase in serum ALT, AST, alkaline phosphatase activity, and creatinine and urea concentration. Rats given high doses of tartrazine showed a significant decrease in liver glutathione content. 23 Effects become more harmful at higher doses because tartrazine induces oxidative stress by the formation of free radicals. 23 Tartrazine has also been found to significantly decrease intracellular glutathione, which may weaken cellular defense, thereby opening the door for a variety of oxidative stress-related disorders, including cell death. 24
An increasing number of studies have reported toxic 22 and behavioral 18,19 effects of the AFCAs used in food products. AFCAs have long been thought to adversely affect learning and behavior in children. Madsen et al. reported intolerance to food additives (Ponceau 4R, azorubine, tartrazine, and sunset yellow) with low and high doses among children with various atopic symptoms, such as urticaria, atopic dermatitis, colic, asthma, and rhinitis. 21
AFCAs are referred to as “haptens” because of their low molecular weight. Sensitization to the hapten carrier results in activation of T cells and release of granules and cytokines by mast cells, which trigger typical allergy symptoms and increased Immunoglobulin E (IgE) levels. 20 Studies on the impact of tartrazine on GSTs have consistently reported that maternal exposure to AFCAs significantly decreases CYP1A1 and GSTs in the skin, and this supports the possibility that AFCAs play a role in the development of skin tumors and dermatoses.
VEGF induces increased microvascular permeability, and it is a selective endothelial cell mitogen that is suspected to play an important role in promoting angiogenesis by stimulating capillary proliferation, migration, and permeability and by regulating the growth, differentiation, and development of a variety of tissues and malignancies. 42,43 In normal skin, VEGF is expressed by keratinocytes, 44 hair follicles, sebaceous glands, and eccrine sweat glands. 43 In addition, excess VEGF production has been reported in skin affected by psoriasis and cutaneous lichen planus, 45 which are commonly associated with prominent permeability barrier abnormalities. VEGF receptors are also strongly expressed in nonlesional, perilesional, and lesional psoriatic keratinocytes in all epidermal strata in vivo. 45 –47 However, no correlations have been found between VEGF and chronic urticaria. 48 Angiogenesis and lymphangiogenesis are tightly connected to the pathogenesis of psoriasis and cutaneous lichen planus. 45 VEGF gene polymorphisms and elevated VEGF serum levels have been found in BCC patients. Positive associations between VEGF serum levels and tumor size and site have also been identified, which may indicate a role of VEGF in tumor aggressiveness. 42 Bao et al. reported accelerating hair regrowth in skin after application of VEGF. 49 Marked decrease of expression of VEGF in epidermal keratinocytes and the vascular endothelium clearly plays a role in retarded wound healing and may affect the progression of chronic disease.
Conclusion
In this study, we have found that maternal exposure to AFCAs apparently affects expression of CYP1A1, GSTs, and VEGF in the skin. The expressions of CYP1A1 in sebaceous glands and subcutaneous striated muscle, GSTA in dermal fibroblasts, GSTT in vascular endothelia, and VEGF in epidermal keratinocytes and vascular endothelia were all decreased significantly following maternal exposure to AFCAs. To the best of our knowledge, the role of AFCAs in the expression of GSTs, CYP1A1, and VEGF in the skin has not been previously investigated. Therefore, our study highlights that significant changes in the expression of these substances play a role in neoplastic and nonneoplastic skin diseases. However, further investigations should be performed to evaluate the effects of exposure to individual color additives on rat skin.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
