Abstract
Buprenorphine drug cartridge was made for autoinjector device for use in emergency and critical situations to reduce the morbidity and mortality. Water-filled cartridges were prepared and buprenorphine was injected aseptically in the cartridge, to make 0.05 and 0.10 mg/mL. Rats were injected intraperitoneally, buprenorphine (0.3 and 0.6 mg/kg), repeatedly with the autoinjector and compared with manual injection (7 days and 14 days) using various haematological and biochemical parameters. No significant change was observed in the body weight, organ to body weight ratio and haematological variables in any of the experimental groups compared with the control group. Except serum urea and aspartate aminotransferase, no significant change was observed in glucose, cholesterol, triglycerides, bilirubin, protein, albumin, creatinine, uric acid, alanine aminotransferase, gamma glutamyltransferase and alkaline phosphatase. The autoinjectors deliver the drugs with spray effect and force for faster absorption. In the present study, the autoinjector meant for intramuscular injection was injected intraperitoneally in rats, and the drug was delivered with force on the vital organs. No significant difference was observed in the autoinjector group compared to the manual group showing tolerability and safety of the buphrenorphine autoinjector. This study shows that buprenorphine autoinjector can be considered for further research work.
Introduction
Emerging catastrophes throughout the world have emphasized the importance in creating awareness and preparedness in primary medical care to reduce morbidity and mortality. Disasters, such as, earth quake, tsunami, flood, avalanche, explosion, accidents, building collapse, military operations and terrorism can precipitate mass casualty situations. 1,2 Efficient medical care during this emergency situation requires trained personnel with adequate medical equipments, which in reality is insufficient to protect the injured victims. 3 In many developing countries, death and disabilities have occurred due to road accidents and violence, resulting in large numbers visiting the emergency department. 4,5 The number of disasters, particularly accidents, have increased, and people killed and injured have increased in the recent years. 6 In these emergency situations, an antibacterial and an analgesic drug will be very much required as a first aid that can be used in the field with easiness, like an autoinjector.
Autoinjectors are self-injectable medical devices and available for intramuscular and subcutaneous administration. For managing several conditions, namely, nerve gas poisoning (atropine and pralidoxime), anaphylaxis (epinephrine), seizures (diazepam), opioid overdose (naloxone), multiple sclerosis (interferon β1a, peginterferon-α2a), hepatitis C (vasoactive intestinal peptide), alkylosis and rheumatoid arthritis (golimumab), autoinjectors are available. 7 Currently, sarilumab autoinjector is under clinical trial for rheumatoid arthritis. 8 In disasters like cyclones, about 80% of injuries occur due to scattering of building materials and about 15% of injured require self-treatment for wound care management. 9 During natural and man-made disasters, acute and chronic pain can be managed by an analgesic autoinjector and infection by an antibiotic autoinjector. The autoinjector delivers the drug by spray effect and increases the area of absorption, and the effect produced is nearly equal to intravenous route. 7 The interchangeable cartridge and the dose selection can make them more convenient during emergency situations. The concept of an analgesic autoinjector (buprenorphine) and an antibacterial autoinjector (amikacin) has been proposed. Amikacin autoinjector’s tolerability and acceptability were studied in rodents and nonrodents. 10,11 The latest introduction of methotrexate autoinjector for the treatment of rheumatoid arthritis has ensured the need for an analgesic autoinjector to manage these emergency situations. 12,13
Analgesics commonly used are mostly effective against nociceptive pain but less effective in neuropathic pain. Non-steroidal anti-inflammatory drugs are used in mild and moderate pain. Opioid analgesics are strong analgesics and are used in acute and chronic pain, including cancer pain. The main disadvantage of opioids is its physical dependence and reduction in immunity when they are used for chronic treatment. 14 Buprenorphine, an opioid analgesic, is preferred for the analgesic autoinjector, due to its long action, safety in pregnant women and ceiling effect on respiratory depression. 15,16 Studies have proved that there is no reduction in immunity from the prolonged use of buprenorphine in managing pain. 17 Buprenorphine do not cause withdrawal symptoms in opioid addicts. 18
Autoinjectors deliver the drug with a higher force than a conventional manual injection. Hence, the objective of this study is to compare the response of stress induced by repeated administration of buprenorphine intraperitoneally through autoinjector compared with the manual injection on haematological and biochemical parameters in rats.
Materials and methods
Animals
Randomly bred female Wistar rats (180–240 g; 5–6 months of age) from Centre for Laboratory and Animal Research, Saveetha University, were used for the study. They were housed in polypropylene cages, three per cage with sterilized paddy husk as the bedding material. They were provided with laboratory animal feed (VRK Nutritional Solutions, Sangli, Maharashtra, India) and filtered water ad libitum. The animals were maintained at a temperature of 25 ± 2°C and natural dark/light cycle. Care and maintenance of the animals were as per the approved guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (India). The study was initiated after the approval by Institutional Animal Ethical Committee of Saveetha University (SU/BRULAC/RD/001/2013).
Drug cartridge preparation
Sterile water-filled glass cartridges with 23 gauge needle were prepared by M/s Neon Laboratories Ltd (Mumbai, Maharashtra, India). Each cartridge contained 2.3–2.4 mL of sterile water for injection and upon firing by autoinjector without restrictor, 2.1–2.2 mL of the contents would be delivered. With the restrictor, about 1.5 mL would be delivered. 19 Two concentrations of buprenorphine cartridges were prepared using the sterile water-filled cartridges. Buprenorphine (as buprenorphine hydrochloride) 0.3 mg/mL (2 mL vial) was procured from M/s Neon Laboratories Ltd. About 0.4 mL or 0.8 mL of sterile water was removed from the water-filled cartridges under a laminar flow and 0.4 mL or 0.8 mL of buprenorphine was injected back, making the drug cartridges with 0.05 mg/mL and 0.10 mg/mL of buprenorphine, respectively. The autoinjectors were supplied by M/s Sigma Engineering (Hyderabad, Telangana, India).
Quality control parameters
A battery of quality control parameters, namely, sterility test of the prepared drug cartridge, injection time, injection volume, ejected needle lengths, corrosion test of the needle, low pressure test and ruggedness test were carried out.
Autoinjector usage in rats
The rats were held gently on the operating table in the supine position exposing the abdomen. The safety mechanism of the autoinjector was turned clockwise to make the autoinjector ready for use. The autoinjector was used with the restrictor to deliver a partial dose with partial needle ejection. The autoinjector was positioned firmly on the abdomen of the rat. The trigger mechanism was pressed and kept in position for about 10 s for the efficient ejection of the drug. The autoinjector was gently removed and the rat was freed. The drug cartridge was weighed before and after the drug delivery for the calculation of the injected volume and dose. The ejected needle length was also measured using a Vernier calliper (Figure 1).

Water-filled fresh cartridges, autoinjector used for the experiments and partially ejected drug cartridges.
Manual injection
Two concentrations of buprenorphine, 0.05 mg/mL and 0.10 mg/mL in sterile water, were prepared. For each rat, 1.2 mL was injected intraperitoneally for 7 days and 14 days consecutively.
Experimental groups
The following are the experimental groups: Group 1: Control (C) Group 2: Buprenorphine (0.05 mg/mL), injection by autoinjector for seven consecutive days (A 0.05–7 days) Group 3: Buprenorphine (0.05 mg/mL), injection by manual for seven consecutive days (M 0.05–7 days) Group 4: Buprenorphine (0.10 mg/mL), injection by autoinjector for seven consecutive days (A 0.10–7 days) Group 5: Buprenorphine (0.10 mg/mL), injection by manual for seven consecutive days (M 0.10–7 days) Group 6: Buprenorphine (0.05 mg/mL), injection by autoinjector for 14 consecutive days (A 0.05–14 days) Group 7: Buprenorphine (0.05 mg/mL), injection by manual for 14 consecutive days (M 0.05–14 days)
Each group consisted of six rats.
Sample collection
The rats were weighed daily and food and water intake was recorded. The gross behaviour of the animals was observed, including abdominal writhing due to pain. Twenty-four hour after the last injection (8th or 15th day), the rats were anaesthetized with isoflurane, USP (Raman and Weil Pvt Ltd, Mumbai, Maharashtra, India). Blood was withdrawn from the orbital plexuses and collected in two vials with anticoagulant (tri-potassium ethylenediaminetetraacetic acid) and without anticoagulant for haematological and biochemical estimations. The rats were euthanized with an overdose of isoflurane and vital organs, such as, lung, liver, spleen, kidney and heart were removed. The organs were cleaned with saline, weighed and preserved in 10% formalin for further histological studies.
Haematological parameters
Various haematological parameters, namely, haemoglobulin (Hb), packed cell volume (PCV), red blood cell (RBC) count, white blood cell (WBC) count and platelet count were estimated using the whole blood by Beckman Coulter cell counter (Beckman Coulter, Chennai, Tamil Nadu, India) as per the instrument guidelines. All tests were done in duplicate.
Biochemical parameters
Serum was separated from the clotted blood and various biochemical parameters were estimated, namely, glucose, cholesterol, triglycerides, bilirubin, protein, albumin, aspartate amino transferase (AST), alanine amino transferase (ALT), gamma glutamyl transferase (GGT), alkaline phosphatase (ALP), urea, creatinine and uric acid using Roche Modulator EVO 9000 autoanalyser (Germany) as per the instrument procedure. All tests were done in duplicate.
Organ to body weight ratio
The organ to body weight ratio of all the vital organs was determined by calculating organ weight divided by body weight and multiplied by 100.
Statistical analyses
All the parameters were analysed using one-way analysis of variance and compared with control using Dunnett’s test. A probability of 0.05 and less was taken as statistically significant. The analysis and plotting of graphs were carried out using SigmaPlot 12 (Systat software Inc., San Jose, California, USA).
Results
The buprenorphine drug cartridge for the autoinjector complied with all the quality control parameters. Sterility test carried out on randomly selected drug cartridges did not show any growth in the agar plate test.
All the animals injected with buprenorphine through autoinjector or through manual injection did not show any gross abnormality in their behaviour. There was no mortality in any of the groups. When the animals were autopsied after euthanasia, no sign of bleeding was observed in the abdomen. The food and water consumption of all the groups did not show any difference apparently.
Buprenorphine was administered using 0.05 mg/mL and 0.10 mg/mL cartridges through the autoinjector for 7 days and 14 days repeatedly. The volume of drug delivered (mean ± standard error (SE)) was 1.34 ± 0.05 in 0.05 mg/mL cartridge (7-day group), 1.28 ± 0.03 in 0.10 mg/mL cartridge (7-day group) and 1.18 ± 0.05 in 0.05 mg/mL cartridge (14-day group). The dose of buprenorphine administered (mean ± SE) through the autoinjector was 0.324 ± 0.022 mg/kg in 0.05 mg/mL (7-day group), 0.678 ± 0.040 mg/kg in 0.10 mg/mL cartridge (7-day group) and 0.380 ± 0.019 mg/kg in 0.05 mg/mL cartridge (14-day group). The effective needle length in all the groups ranged from 1.4 mm to 1.5 mm. The dose of buprenorphine administered (mean ± SE) by the manual injection was 0.323 ± 0.014 mg/kg in 0.05 mg/mL (7-day group), 0.649 ± 0.015 mg/kg in 0.10 mg/mL (7-day group) and 0.350 ± 0.010 mg/kg in 0.05 mg/mL (14-day group).
The body weight changes of the control and experimental groups are given in Table 1. There was no significant change in the body weight of the experimental groups when compared to the control group. Table 2 shows the organ to body weight ratio of the vital organs in the control and the experimental groups. Statistically no significant difference was observed between the control and the experimental groups. Table 3 lists the results of various haematological parameters, namely, Hb, PCV, RBC, WBC and platelets. There was no significant change in any of the haematological parameters among the experimental groups (7 days and 14 days) when compared with the control group.
Body weight changes (percent).a
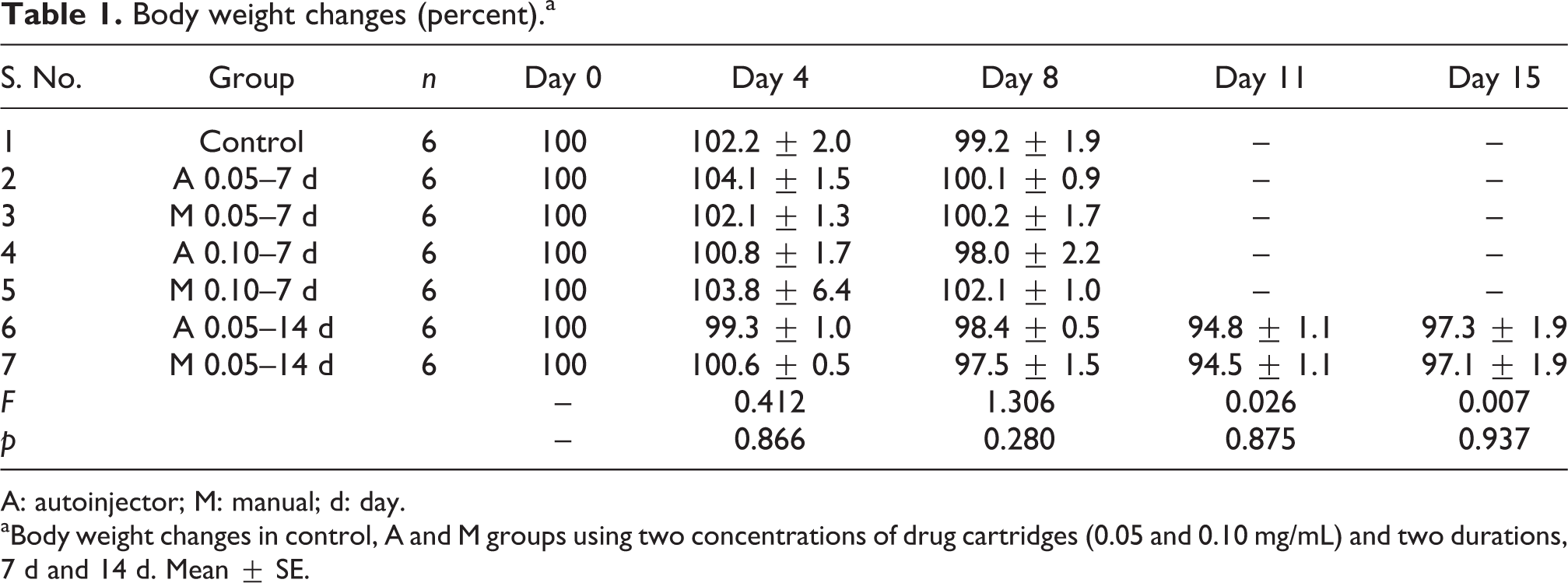
A: autoinjector; M: manual; d: day.
aBody weight changes in control, A and M groups using two concentrations of drug cartridges (0.05 and 0.10 mg/mL) and two durations, 7 d and 14 d. Mean ± SE.
Organ to body weight ratio.a
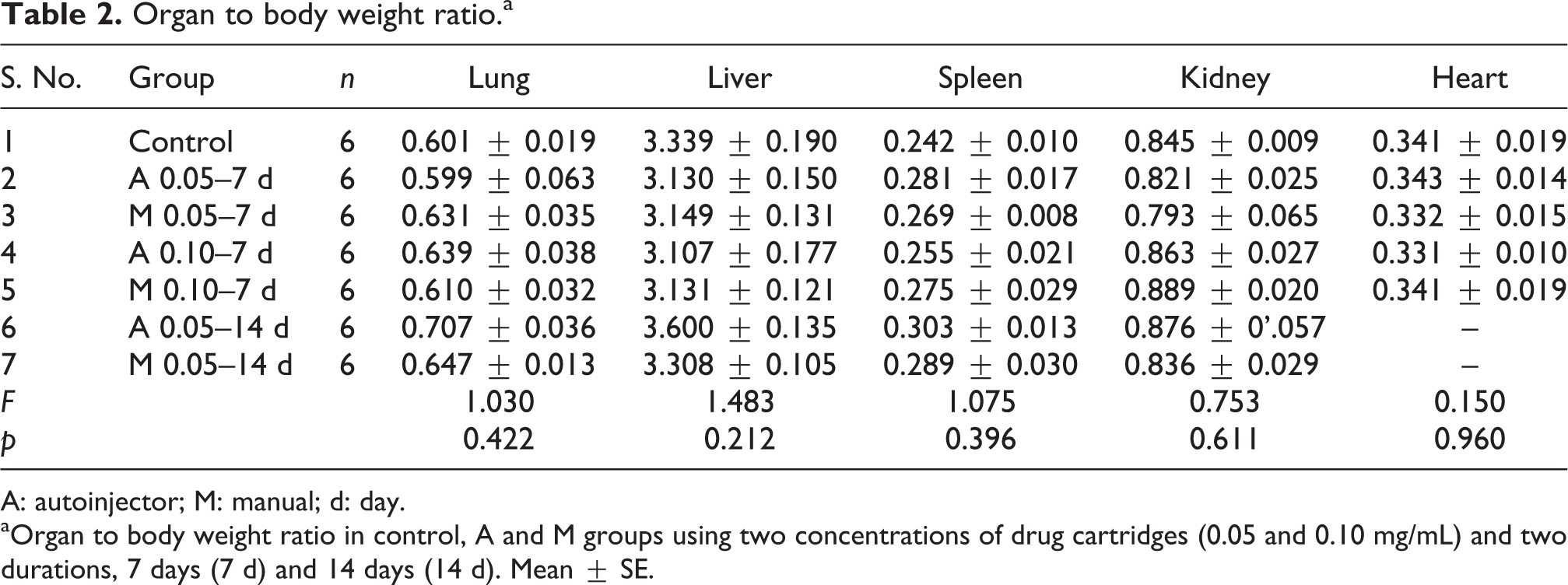
A: autoinjector; M: manual; d: day.
aOrgan to body weight ratio in control, A and M groups using two concentrations of drug cartridges (0.05 and 0.10 mg/mL) and two durations, 7 days (7 d) and 14 days (14 d). Mean ± SE.
Haematological variables.a
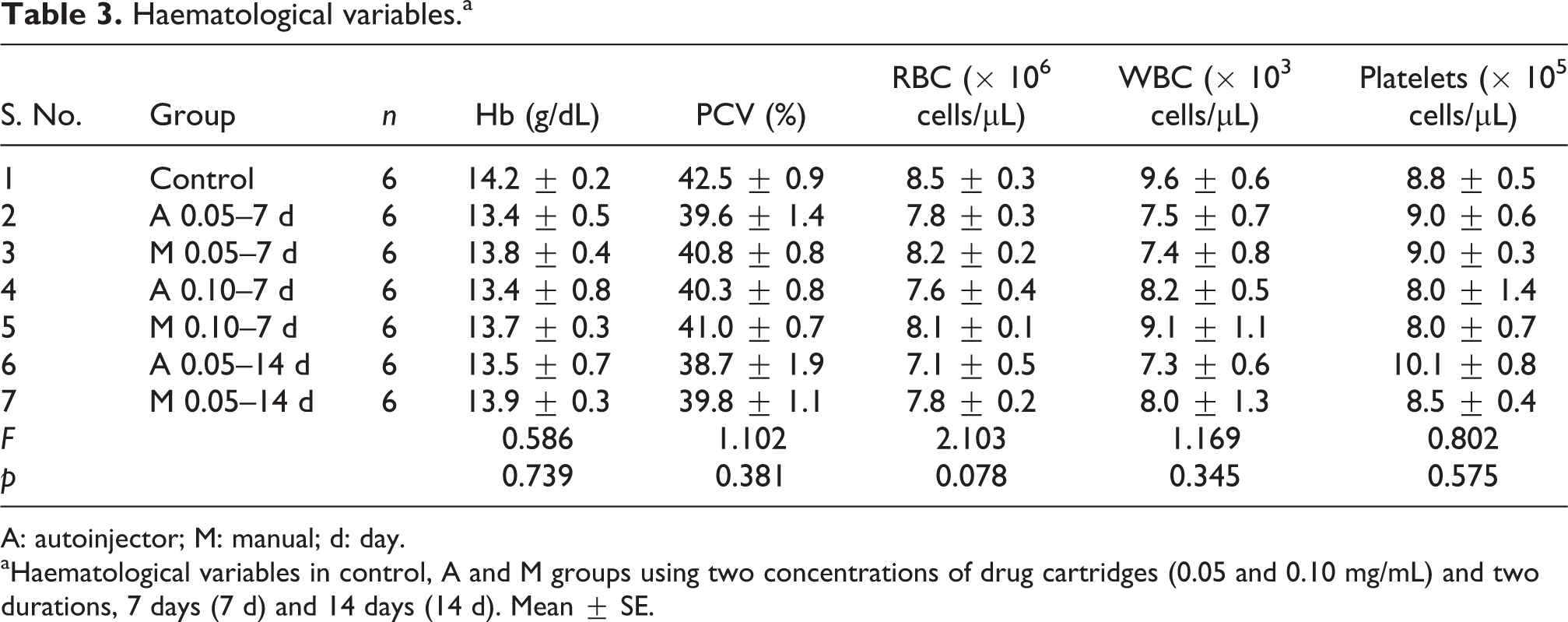
A: autoinjector; M: manual; d: day.
aHaematological variables in control, A and M groups using two concentrations of drug cartridges (0.05 and 0.10 mg/mL) and two durations, 7 days (7 d) and 14 days (14 d). Mean ± SE.
The effect of autoinjector and manual injection was compared with control after buprenorphine administration for serum parameters such as glucose, cholesterol, triglycerides and bilirubin (Figure 2). No significant change was observed in the above parameters in the experimental groups when compared with the control group. Figure 3 shows the effect on total protein and albumin levels. Compared with the control group, no significant difference was observed in the total protein and albumin levels in the experimental groups.
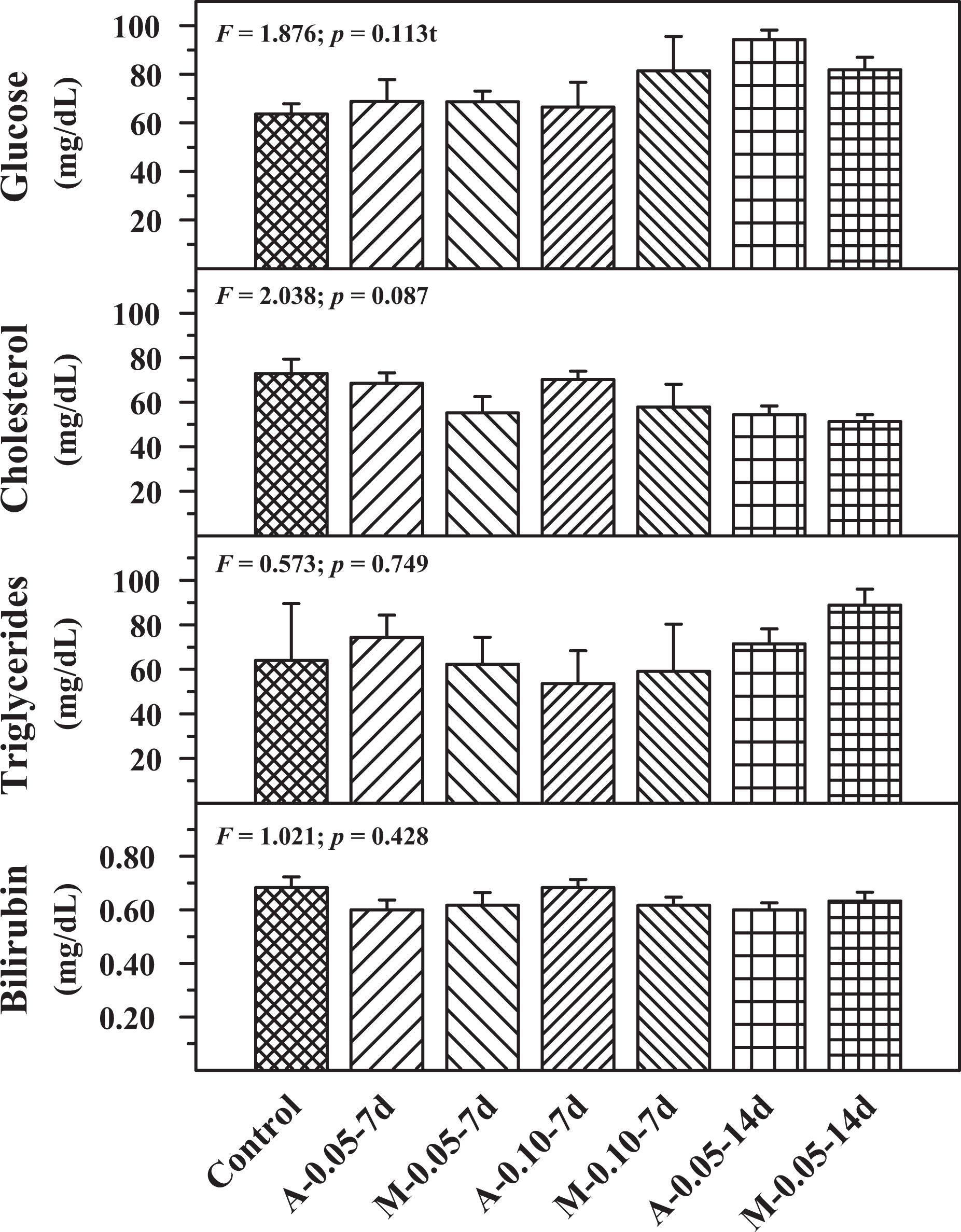
Serum glucose, cholesterol, triglycerides and bilirubin levels in control, A and M groups using two concentrations of drug cartridges (0.05 and 0.10 mg/mL) and two durations, 7 d and 14 d. Mean ± SE (n = 6). A: autoinjector; M: manual; d: day.
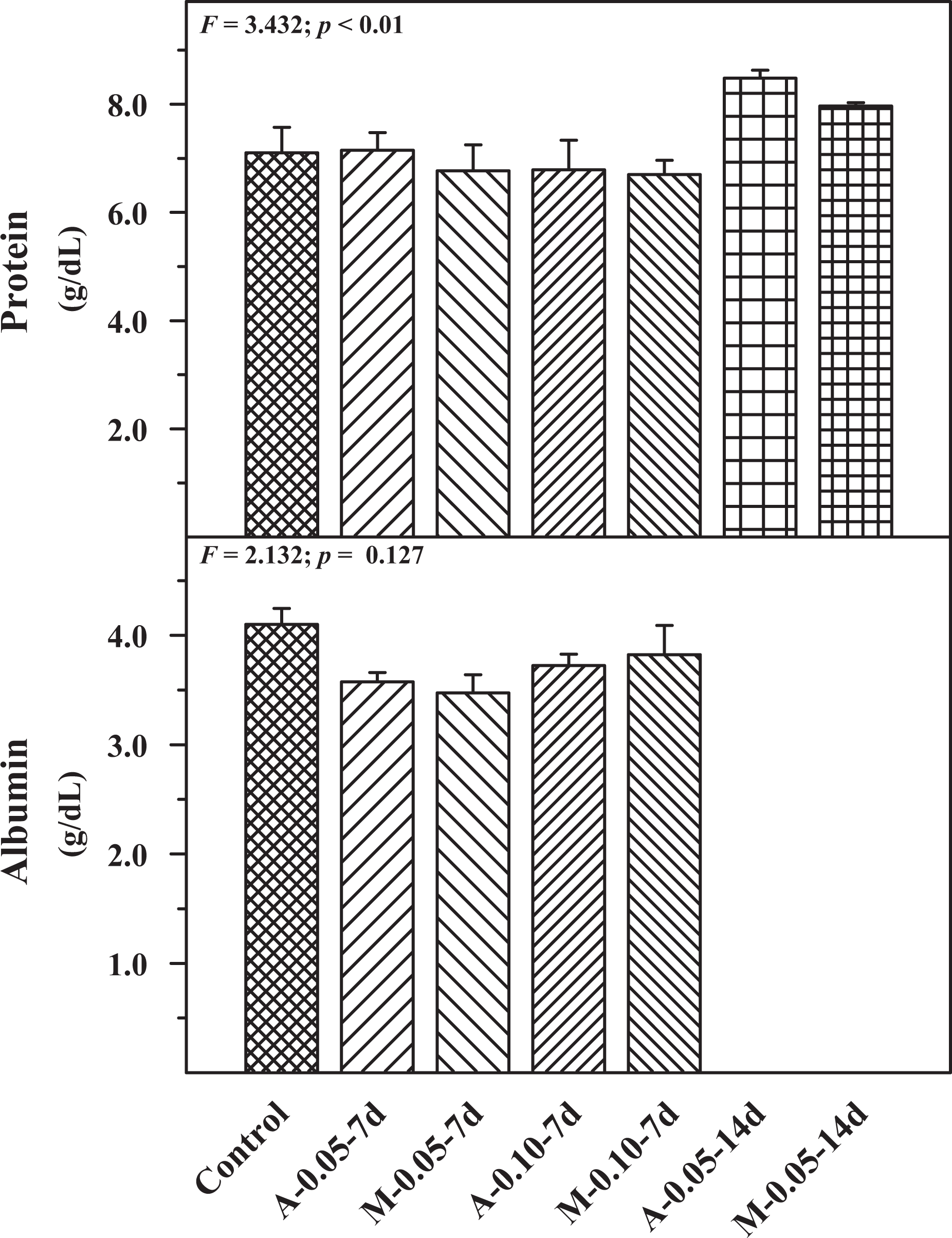
Serum protein and albumin levels in control, A and M groups using two concentrations of drug cartridges (0.05 and 0.10 mg/mL) and two durations, 7 days (7 d) and 14 days (14 d). Mean ± SE (n = 6). A: autoinjector; M: manual.
Figure 4 shows the effect of buprenorphine administration through autoinjector or manual injection on serum urea, creatinine and uric acid. Creatinine and uric acid did not show any significant difference in the experimental groups when compared to the control group. Serum urea showed a significant decrease when compared to the control group in 0.05 mg/mL, 14-day repeated administration both in autoinjector and manual injection groups. The effect on AST, ALT, GGT and ALP in experimental and control groups is shown in Figure 5. ALT, GGT and ALP did not show any statistically significant difference in the experimental groups when compared with the control group. AST level showed a significant increase in 0.10 mg/mL autoinjector cartridge, 7-day administration, and 0.05 mg/mL autoinjector cartridge, 14-day administration, when compared to the control.
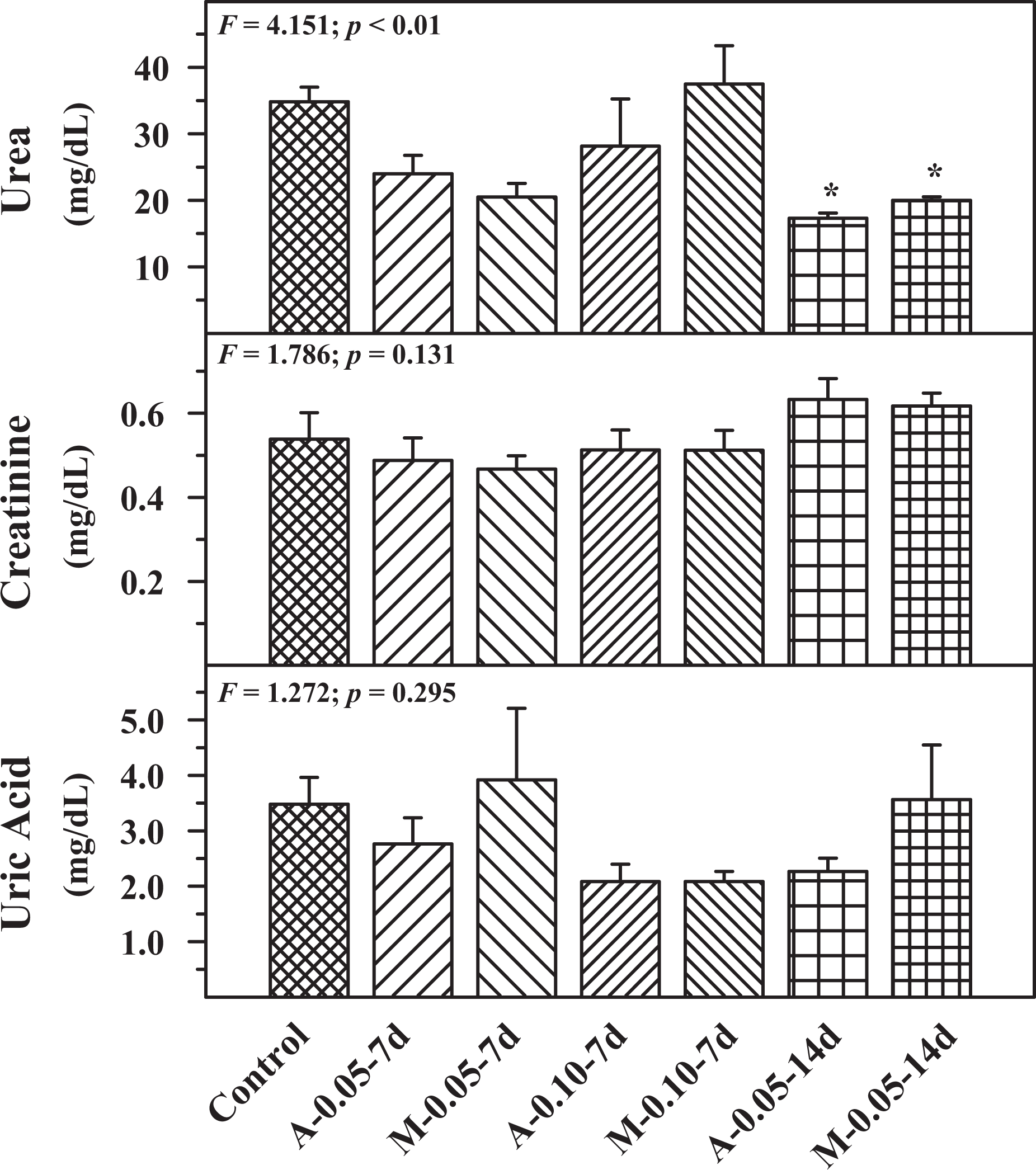
Serum urea, creatinine and uric acid levels in control, A and M groups using two concentrations of drug cartridges (0.05 and 0.10 mg/mL) and two durations, 7 days (7 d) and 14 days (14 d). Mean ± SE (n = 6). *Statistically significant from control group. A: autoinjector; M: manual.
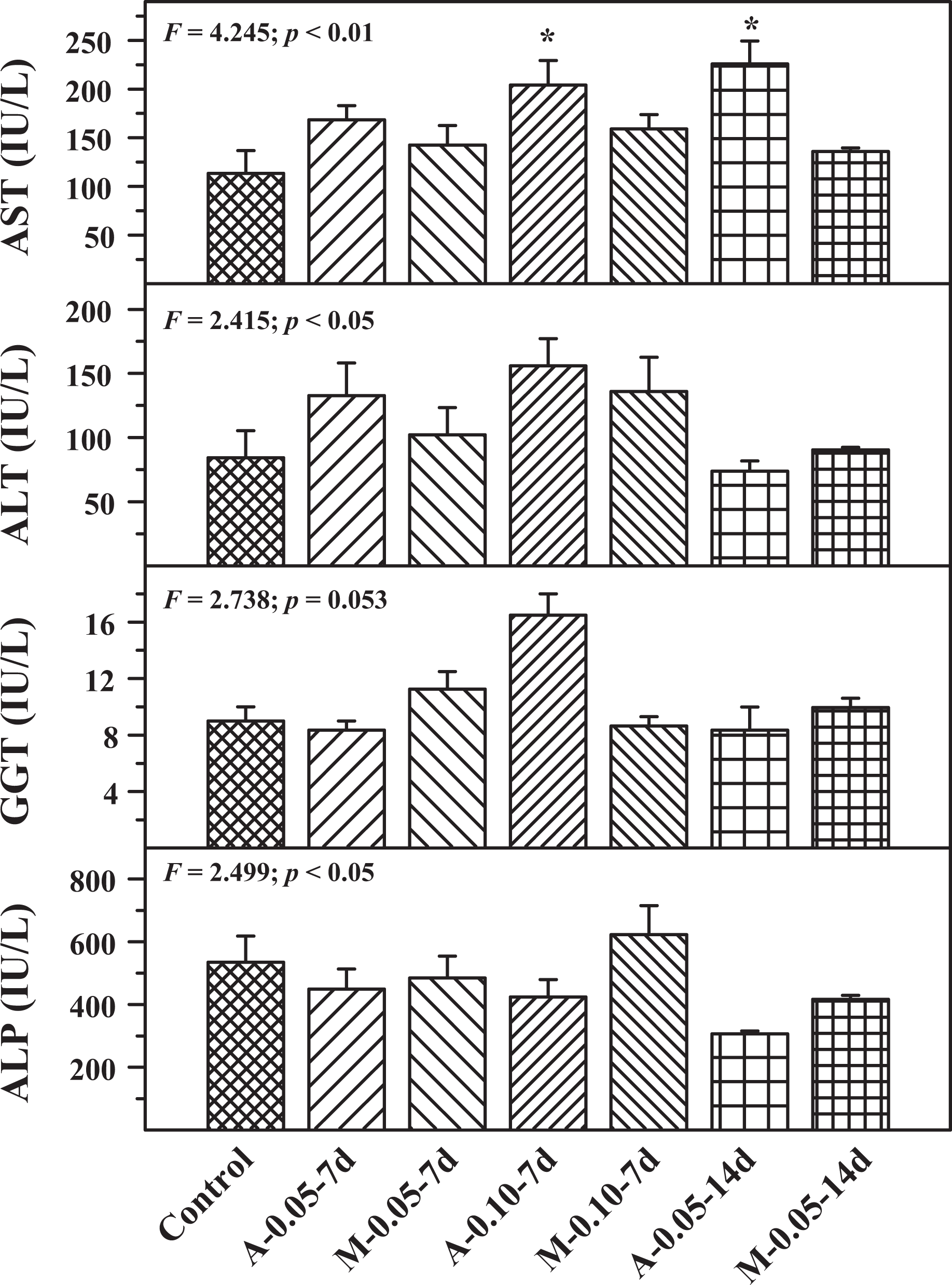
Serum aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma glutamyltransferase (GGT) and alkaline phosphatase (ALP) levels in control, A and M groups using two concentrations of drug cartridges (0.05 and 0.10 mg/mL) and two durations, 7 days (7 d) and 14 days (14 d). Mean ± SE (n = 6). *Statistically significant from control group. A: autoinjector; M: manual.
Discussion
Providing appropriate medical assistance during emergency situation would reduce the morbidity and mortality. Man-made disasters like the attack on the World Trade Centre in the United States or the natural disasters like the tsunami have resulted in huge loss of life and serious injuries. 20,21 Managing the mass casualties by providing immediate and trained medical assistance would save the victims by reducing their pain and spread of infection. With this intention, autoinjectors were developed for safe and easy administration of antibiotics and analgesics. 7 The present autoinjector is reusable with interchangeable cartridges. In injuries, intramuscular injection would be appropriate for faster absorption of the drugs as there would be hypotension due to blood loss. 22 The present autoinjector can deliver two doses (full for adults and partial for children) and can be used over the cloth in an emergency.
Earlier, the developed buprenorphine drug cartridges were subjected to quality control measures, to sterility test and for the assessment of analgesic effect in rats. 23 The cartridges successfully passed the quality control tests and the sterility test and showed significant analgesic effect when compared with manual injection. The buprenorphine autoinjector is designed for intramuscular administration. The present study was carried out to establish the safety and acceptability of the developed analgesic autoinjector in rats. Hence, various haematological and biochemical variables were estimated by administering the drug intraperitoneally in rats. An intraperitoneal injection with force using autoinjector can create stress in rats that can influence on the endocrine, haematopoietic and immune systems. 24 Haematological and biochemical changes have been reported following chemical toxicity, internal bleeding, ischaemia–reperfusion injury and intra-abdominal hypertension. 25 –28 No significant effect was observed in the autoinjector group using two doses (0.3 mg/kg and 0.6 mg/kg using 0.05 mg/mL and 0.10 mg/mL cartridges) and two durations, 7 and 14 days. Damage of RBCs may produce an increase in bilirubin level and no change was observed in this study, showing that autoinjector and manual injection did not cause any abdominal injury. No significant change in albumin, protein, glucose, cholesterol and triglycerides was seen in all the experimental groups supporting that the forceful injection through the autoinjector did not cause any injury.
Biomarkers of liver injury, such as, AST, ALT, GGT and ALP did not alter, except an increase in AST that was observed in the autoinjector group. Liver injury is known to increase the levels of liver enzymes. 29 The effective needle length in the autoinjector was about 15 mm and used vertically in the abdomen of the rat. The spray effect of the drug and the forceful delivery did not cause any kidney damage as evidenced by no effect on serum urea, creatinine and uric acid levels. Kidney damage may occur with increased levels of these biochemical markers. 30
The developed analgesic autoinjector with two different doses, 0.3 mg/kg and 0.6 mg/kg using 0.05 mg/mL and 0.10 mg/mL cartridges with repeated administration for 7 and 14 days through intraperitoneal injection in rats proved the safety of the device. Technical performance, usability, tolerability and ease of administration in humans are under clinical trial for sarilumab and methotrixate autoinjectors. 8,13 Buprenorphine can be safely used in cancer pain and also in the aged people with renal and hepatic problems. 31 –33 The autoinjector can be used in children as well as in farm and pet animals also. It is well suited for mass casualty management and gives scope for further research.
Footnotes
Acknowledgement
The authors thank M/s Neon Laboratories for providing water-filled cartridges.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
