Abstract
Background:
Attenuated cardioprotective effect of ischemic preconditioning (IPC) by reduced nitric oxide (NO) is a hallmark during diabetes mellitus (DM). Recently, we reported that the formation of caveolin–endothelial nitric oxide synthase (eNOS) complex decreases the release of NO, which is responsible for attenuation of IPC-induced cardioprotection in DM rat heart. Heme oxygenase-1 (HO-1) facilitates release of NO by disrupting caveolin–eNOS complex. The activity of HO-1 is decreased during DM. This study was designed to investigate the role of hemin (HO-1 inducer) in attenuated cardioprotective effect of IPC in isolated diabetic rat heart.
Methods:
DM was induced in male Wistar rat by single dose of streptozotocin. Cardioprotective effect was assessed in terms of myocardial infarct size and release of lactate dehydrogenase and creatine kinase in coronary effluent. The release of NO was estimated indirectly by measuring the release of nitrite in coronary effluent. Perfusion of sodium nitrite, a precursor of NO, was used as a positive control.
Result:
IPC-induced cardioprotection and increased release of nitrite were significantly attenuated in a diabetic rat as compared to a normal rat. Pretreatment with hemin and daidzein, a caveolin inhibitor, alone or in combination significantly restored the attenuated cardioprotection and increased the release of nitrite in diabetic rat heart. Zinc protoporphyrin, a HO-1 inhibitor, significantly abolished the observed cardioprotection and decreased the release of nitrite in hemin pretreated DM rat heart.
Conclusion:
Thus, it is suggested that hemin restores the attenuated cardioprotective effect in diabetic rat heart by increasing the activity of HO-1 and subsequently release of NO.
Introduction
Coronary heart diseases have been remarked as a major prevailing cause of morbidity and mortality worldwide. 1,2 Reduced supply of blood to myocardium leads to ischemia and early reperfusion is mandatory to salvage ischemic myocardium from infarction. 3 However, reperfusion after a sustained ischemic insult causes further injury of myocardium that is known as ischemia-reperfusion (I/R) injury. 4,5 Ischemic preconditioning (IPC) is one of the most promising strategies used to protect the myocardium from ischemic insults which comprises of brief sub-lethal bursts of ischemia followed by reperfusion before the subsequent ischemic insult, increases the tolerance against I/R injury. 6,7 IPC plays a major role in cardioprotection in various species including human beings through various pathways. 8 –14 The IPC-mediated cardioprotection has been reported to be attenuated during diabetes mellitus (DM), which is a major risk factor for ischemic heart disease. 15 –20
Caveolae is a pocket-like structure of plasma membrane that serves as organizing centers for cell signaling. 21,22 The signaling molecules like src-like kinases, tyrosine kinases, members of Ras–MAPK cascade, and endothelial nitric oxide synthase (eNOS) are generated within caveolae. 23,24 Caveolin, a caveolar membrane protein, is a well-known negative regulator of eNOS, which decreases the activity of eNOS by making caveolin–eNOS complex. 25 –28 It has been reported that during DM, expression of caveolin increases and IPC-induced release of nitric oxide (NO) in myocardium significantly decreases. 29 Recently, we have reported that the treatment of daidzein (DDZ), a phytoestrogen, increases the release of NO and restores the attenuated cardioprotective effect of IPC in isolated diabetic rat heart. 30
Heme oxygenase-1 (HO-1) is a rate-limiting enzyme in catabolism of heme into ferrous (Fe++) ion, carbon monoxide, and biliverdin, and it is present in the membrane of caveolae where it interacts with caveolin. 31,32 It has been documented that overexpression of HO-1 conversely regulates the expression of caveolin. 33 Moreover, HO-1 activates the eNOS by disrupting the caveolin–eNOS complex. 34 Decreased cardio-specific expression of HO-1 exacerbates, while upregulation of HO-1 produces cardioprotection against I/R injury. 34 –37 During DM, both expression and activity of HO-1 are noted to be reduced. 35,38,39 Therefore, the present study was designed to investigate the effect of hemin, an activator of HO-1, in attenuated cardioprotective effect of IPC in diabetic rat heart.
Materials and methods
Animals
Male Wistar rats, weighing 180–250 g, housed in animal house and provided 12-h light and 12-h dark cycle. They were fed on a standard chow diet (Ashirwad Industries Ltd, Ropar, Punjab, India) and water ad libitum. The experimental protocol was approved by the Institutional Animal Ethics Committee (ISFCP/IPEC/CPCSEA/6/2010) in accordance with the national guidelines on the use of laboratory animals. All efforts were made to minimize animal suffering and to reduce the number of animals used.
Drugs and chemicals
DDZ (0.2 mg/kg, subcutaneously (s.c.); Enzo Life Sciences International, Inc., Farmingdale, New York, USA) was dissolved in 10% dimethyl sulfoxide (DMSO) and injected to the animals for 7 days after 3 weeks of streptozotocin administration. Hemin (4 mg/kg, intraperitoneally (i.p.); Hi Media Laboratories Pvt Ltd, Mumbai, Maharashtra, India) was dissolved in 0.2 M sodium hydroxide and was injected 18 h before isolation of heart. Zinc protoporphyrin (ZNPP; 50 µg/kg, i.p.; Enzo Life Sciences International, Inc.) was dissolved in DMSO and injected 6 h before hemin treatment. Triphenyltetrazolium chloride (TTC) stain, Tris-chloride buffer, sulfanilamide, phosphoric acid, and sodium nitrite were purchased from CDH Pvt Ltd, New Delhi, India. N-(1-Naphthyl) ethylenediamine dihydrochloride was purchased from Hi Media Laboratories Pvt Ltd. The lactate dehydrogenase (LDH) and creatine kinase (CK-MB) enzymatic estimation kits were purchased from Coral Clinical Systems, Salcete, Goa, India. All other reagents used in this study were of analytical grade and always freshly prepared before use.
Induction of experimental DM
DM in rat was induced by administration of single dose of streptozotocin (50 mg/kg, i.p.). 40 Serum level of glucose was estimated spectrophotometrically at 505 nm by glucose oxidase/pyruvate oxidase method 41 –43 using enzymatic kit (Coral Clinical Systems). Serum glucose level >200 mg/dL was considered to be hyperglycemic.
Isolated rat heart preparation
Rats were administered heparin to prevent the coagulation of blood, (500 IU/L, i.p) 20 min prior to sacrificing the animal by cervical dislocation. Heart was rapidly excised and immediately mounted on Langendorff’s apparatus. 44 Isolated heart was retrogradely perfused at constant pressure of 80 mmHg with Krebs–Henseleit (K-H) buffer (NaCl 118 mM, KCl 4.7 mM, CaCl2 2.5 mM, MgSO4·7H2O 1.2 mM, KH2PO4 1.2 mM, C6H12O6 11 mM), pH 7.4, and maintained at 37°C bubbled with 95% oxygen and 5% carbon dioxide. Flow rate was maintained at 7–9 mL/min using Hoffman’s screw. The surrounding temperature of heart was maintained by enclosing it in a double wall water jacket maintained at 37°C. IPC was produced by closing the inflow of K-H solution for 5 min followed by 5 min of reperfusion. Four such episodes were employed. Global ischemia was produced for 30 min followed by 120 min of reperfusion. Coronary effluent were collected to estimate lactate dehydrogenase (LDH), creatine kinase (CK-MB) and nitrite at the end of stablilization phase (basal) before ischemia, immediately (0 min), 5 min and 30 min after reperfusion. 45,46
Assessment of myocardial injury
The myocardial infarct size was measured using TTC staining method. The heart was removed from the Langendorff’s apparatus. Both the atria and root of aorta were excised and ventricles were kept overnight at −4°C temperature. Frozen ventricles were sliced into uniform sections of about 1–2 mm thickness. The slices were incubated in 1% w/v TTC stain at 37°C in 0.2 M Tris-chloride buffer for 30 min. The normal myocardium was stained brick red, while the infarcted portion remained unstained. Infarct size was measured by the volume method. 47 The level of LDH and CK-MB were measured to evaluate the occurrence of cardiac cell death. After the experiment, these levels in coronary effluent were estimated by using commercially available kits (Coral Clinical Systems, Goa, India). Values of LDH and CK-MB were expressed in international units per liter.
Nitrite estimation
Nitrite is a stable nitrogen intermediate produced from the degradation of NO, which can be measured easily and can be used to infer levels of NO production. 48 The nitrite release in coronary effluent was measured. Griess reagent 0.5 mL (1:1 solution of 1% sulfanilamide in 5% phosphoric acid and 0.1% N-(1-Naphthyl) ethylenediamine dihydrochloride in water) was added to 0.5 mL of coronary effluent. The optical density at 550 nm was measured using spectrophotometer (UV-1700 Spectrophotometer; Shimadzu, Japan). Nitrite concentration was calculated by comparing with the spectrophotometer reading of standard solution of sodium nitrite prepared in K-H buffer. 30,45,49
Experimental design
The present study was conducted on 12 groups, and each group comprised of six rats. The diagrammatic representation of experimental protocol is shown in Figure 1.

Diagrammatic representation of experimental protocol. S: stabilization; P: perfusion; I: ischemia; R: reperfusion; K-H: Krebs–Hensleit; SN: sodium nitrite.
Group 1(sham control; n = 6): Isolated rat heart preparation was stabilized for 10 min and then perfused continuously with K-H buffer solution for 190 min without subjecting them to global ischemia.
Group 2 (DDZ per se; n = 6): Isolated rat heart preparation from DDZ (0.2 mg/kg/day, s.c. in DMSO, 1 week) treated normal rat was allowed to stabilize for 10 min and then perfused continuously with K-H buffer solution for 190 min without subjecting them to global ischemia.
Group 3 (hemin per se; n = 6): Isolated rat heart preparation from hemin (4 mg/kg, i.p., 18 h before the isolation of heart) treated normal rat was allowed to stabilize for 10 min and then perfused continuously with K-H buffer solution for 190 min without subjecting them to global ischemia.
Group 4 (I/R control; n = 6): Isolated rat heart preparation was allowed to stabilize for 10 min and then subjected to 30 min global ischemia followed by 120 min of reperfusion.
Group 5 (IPC control; n = 6): Isolated rat heart preparation was allowed to stabilize for 10 min and subjected to four cycles of IPC, each cycle comprised of 5 min global ischemia followed by 5 min reperfusion with K-H solution. Then, the preparation was subjected to 30-min global ischemia followed by 120 min of reperfusion.
Group 6 (IPC in sodium nitrite (10 µmol/L) perfused normal rat heart; n = 6): Isolated rat heart preparation from normal rat was allowed to stabilize for 10 min and perfused with sodium nitrite (10 µmol/L) for 30 min and subjected to four cycles of IPC as described earlier in group 5.
Group 7 (IPC in diabetic rat heart; n = 6): Isolated heart preparation from diabetic rat was allowed to stabilize for 10 min and subjected to four cycles of IPC as described earlier in group 5.
Group 8 (IPC in sodium nitrite (10 µmol/L) perfused diabetic rat heart; n = 6): Isolated rat heart preparation from diabetic rat was allowed to stabilize for 10 min and perfused with sodium nitrite (10 µmol/L) for 30 min and then subjected to four cycles of IPC as described earlier in group 5.
Group 9 (IPC in DDZ (0.2 mg/kg/s.c.) pretreated diabetic rat heart; n = 6): Isolated rat heart preparation from DDZ (0.2 mg/kg/day, s.c. in DMSO, 1 week) treated diabetic rat was allowed to stabilize for 10 min and then subjected to four cycles of IPC as described earlier in group 5.
Group 10 (IPC in hemin (4 mg/kg/i.p.) pretreated diabetic rat heart; n = 6): Isolated rat heart preparation from hemin (4 mg/kg/i.p.) treated diabetic rat was allowed to stabilize for 10 min and then subjected to four cycles of IPC as described earlier in group 5.
Group 11 (IPC in DDZ (0.2 mg/kg/s.c.) and hemin (4 mg/kg/i.p.) pretreated diabetic rat heart; n = 6): Isolated rat heart preparation from DDZ (0.2 mg/kg/day, s.c. in DMSO, 1 week) and hemin (4 mg/kg/i.p., 18 h before isolation of heart) treated diabetic rat was allowed to stabilize for 10 min and then subjected to four cycles of IPC as described earlier in group 5.
Group 12 (IPC in ZNPP (50 µg/kg/i.p.) and hemin (4 mg/kg/i.p.) pretreated diabetic rat heart; n = 6): Isolated rat heart preparation from hemin (4 mg/kg/i.p., 18 h before isolation of heart) and ZNPP (50 µg/kg/i.p., 6 h before hemin treatment) treated diabetic rat was allowed to stabilize for 10 min and then subjected to four cycles of IPC as described earlier in group 5.
Statistical analysis
All values were expressed as mean ± standard deviation (SD). Statistical analysis was performed using GraphPad Prism Software (version 5.0). The data obtained from the various groups were statistically analyzed using Student’s t-test, one-way analysis of variance (ANOVA), and two-way ANOVA followed by Tukey’s multiple comparison test. A p value of <0.05 was considered to be statistically significant.
Results
Effect of streptozotocin on serum glucose
The administration of single dose of streptozotocin (50 mg/kg, i.p.) significantly increased the blood glucose level as compared to basal value (Figure 2).

Effect of streptozotocin administration on serum glucose level in rat. Values are expressed as mean ± SD. *p < 0.05 as compared to normal control animal. SD: standard deviation.
Effect of IPC and pharmacological interventions on myocardial infarct size
Global ischemia for 30 min followed by 120 min of reperfusion significantly increased the myocardial infarct size as compared to sham control. Four episodes of IPC significantly decreased I/R-induced increase in myocardial infarct size. However, IPC failed to decrease I/R-induced infarct size in diabetic rat heart. Moreover, IPC-induced decrease in myocardial infarct size was significantly restored in sodium nitrite–perfused diabetic rat heart. Similar results were obtained in DDZ, hemin, and DDZ with the hemin-treated group. Administration of ZnPP significantly blocked the IPC-induced decrease in the infarct size in hemin-treated diabetic rat (Figure 3).

Effect of pharmacological interventions on myocardial infarct size. Values are expressed as mean ± SD. *p < 0.05 versus Sham control; # p < 0.05 versus I/R control; α p < 0.05 versus IPC control; β p < 0.05 versus IPC in diabetic rat heart; γ p < 0.05 versus IPC in hemin-treated diabetic rat heart. SD: standard deviation; I/R: ischemia–reperfusion; IPC: ischemic preconditioning.
Effect of IPC and pharmacological interventions on myocardial release of LDH and CK-MB in coronary effluent
Global ischemia for 30 min followed by 120 min of reperfusion significantly increased the release of LDH and CK-MB in isolated normal rat heart as compared to sham control. Four episodes of IPC significantly decreased I/R-induced increase in the release of LDH and CK-MB. However, IPC failed to decrease I/R-induced release of LDH and CK-MB in isolated diabetic rat heart. Moreover, IPC-induced decrease in the release of LDH and CK-MB was significantly restored in sodium nitrite–perfused diabetic rat heart. Similar results were noted in DDZ, hemin, and DDZ with the hemin-treated group. Administration of ZnPP significantly attenuated the IPC-induced decrease in the release of LDH and CK-MB in hemin-treated diabetic rat (Figures 4 and 5).
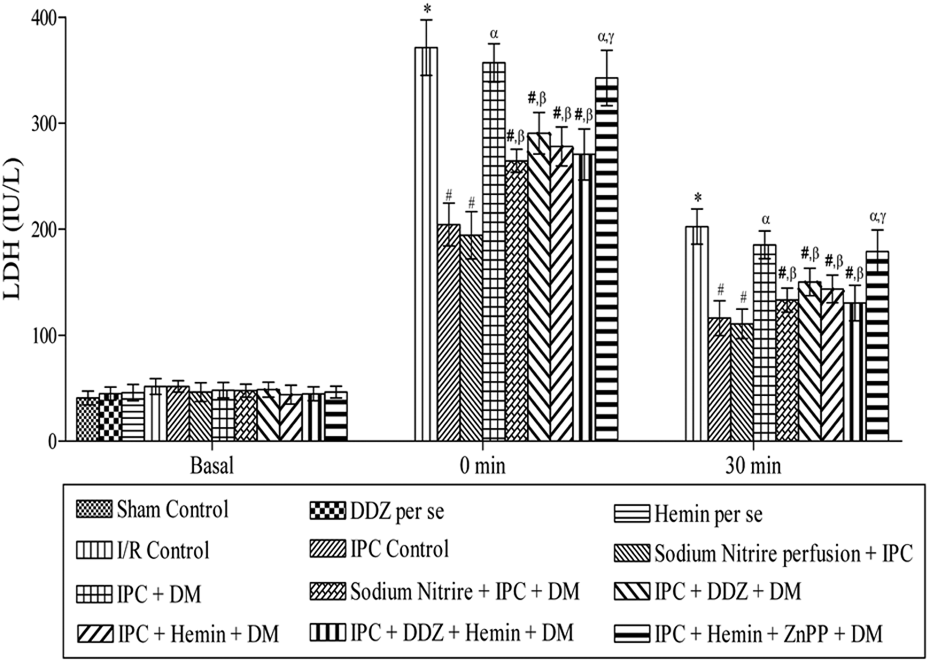
Effect of pharmacological interventions on myocardial release of lactate dehydrogenase. Values are expressed as mean ± SD. *p < 0.05 versus Sham control and basal value; # p < 0.05 versus I/R control; α p < 0.05 versus IPC control; β p < 0.05 versus IPC in diabetic rat heart; γ p < 0.05 versus IPC in hemin-treated diabetic rat heart. SD: standard deviation; I/R: ischemia–reperfusion; IPC: ischemic preconditioning.

Effect of pharmacological interventions on myocardial release of creatine kinase (CK-MB). Values are expressed as mean ± SD. *p < 0.05 versus Sham control and basal value; # p < 0.05 versus I/R control; α p < 0.05 versus IPC control; β p < 0.05 versus IPC in diabetic rat heart; γ p < 0.05 versus IPC in hemin-treated diabetic rat heart. SD: standard deviation; I/R: ischemia–reperfusion; IPC: ischemic preconditioning; CK-MB: creatine kinase.
Effect of IPC and pharmacological interventions on nitrite release in coronary effluent
Global ischemia for 30 min followed by 120 min of reperfusion markedly decreased the nitrite release from isolated rat heart. Four episodes of IPC significantly increased the nitrite release in normal rat heart. However, IPC failed to increase the nitrite release in isolated diabetic rat heart. Pretreatment with DDZ and hemin, alone or in combination, restored the IPC-induced increase in release of nitrite. Furthermore, administration of ZnPP significantly abolished the IPC-induced increase in the release of nitrite from isolated heart of hemin-treated diabetic rat (Figure 6).

Effect of pharmacological interventions on myocardial release of nitrite. Values are expressed as mean ± SD. *p < 0.05 vs. Sham control and basal value; # p < 0.05 versus I/R control; α p < 0.05 versus IPC control; β p < 0.05 versus IPC in diabetic rat heart; γ p < 0.05 versus IPC in hemin-treated diabetic rat heart. SD: standard deviation; I/R: ischemia–reperfusion; IPC: ischemic preconditioning.
Discussion
In our study, four episodes of 5 min of global ischemia and 5 min of reperfusion produced cardioprotection against I/R-induced injury noted in terms of decrease in infarct size and decrease in the release of LDH and CK-MB in isolated normal rat heart. These results are in agreement with earlier published work. 50,51 However, this observed cardioprotective effect of IPC significantly attenuated in heart isolated from diabetic rat. These results are in accordance with other published studies from our laboratory 30,43,49 and from other laboratories too. 15,17,18
IPC produces cardioprotection against I/R-induced injury by increasing the release of NO from myocardium. 13,28 In our study, IPC significantly increased the release of nitrite (measured in coronary effluent), a marker of NO as compared to I/R control group. We have reported that perfusion of sodium nitrite (NO donor) restores the cardioprotection in isolated heart from diabetic rat subjected to global ischemia. 30 In the present study, IPC in sodium nitrite–perfused diabetic rat heart produced significant cardioprotection noted in terms of decrease in infarct size and release of LDH and CK-MB. It is probable that the attenuation of cardioprotective effect of IPC in diabetic rat is due to reduced level of NO.
Caveolae are originated by enfolding of plasma membrane domains and act as a centers of signal transduction. 1,52 Caveolins are proteins that form the structure of caveolar membrane and act as signalosomes for other molecules including eNOS. 53 –56 Increased expression of caveolin decreases the activity of eNOS by making caveolin–eNOS complex resulting in decreased bioavailability of NO. 25 –27 Upregulation of caveolin is noted to decrease the myocardial release of NO in diabetic rat heart. 57,58 In our study, 1-week treatment of diabetic rat with DDZ, a caveolin inhibitor, 59,60 significantly restored the attenuated cardioprotective effect of IPC and also increased the release of nitrite as compared to the diabetic control group. This result is in accordance of our previous study 30 and supported by other studies, that is, administration of DDZ increases bioavailability of NO by inhibiting the caveolin–eNOS complex. 60 Since DDZ is a phytoestrogen, we have selected male animals to overcome the effect of body estrogen.
HO-1 is localized in the caveolae of the plasma membrane where it interacts with caveolin. 32 It has been reported that a decrease in the cardio-specific expression of HO-1 exacerbates, 61 while an upregulation of HO-1 produces cardioprotection against I/R injury. 37,62,63 It is well documented that HO-1 facilitates release of NO by disrupting the complex of caveolin and eNOS. 33,34,37 But the expression of HO-1 is diminished in diabetic myocardium. 35,38,39 In our study, pretreatment with hemin, a HO-1 inducer, 64 significantly increased the IPC-induced release of NO and restored the attenuated cardioprotection in diabetic rat heart. Administration of ZnPP, an inhibitor of HO-1, significantly blocked this observed cardioprotection and decreased the release of nitrite in isolated diabetic rat heart. As expected treatment of hemin, a specific inducer of HO-1 increased the release of nitrite in diabetic rat heart. Our finding is supported by studies of other laboratories, that is, activation of HO-1 increases the release of NO by disruption of caveolin–eNOS complex. 32 –34,37
Furthermore, the restoration of the attenuated cardioprotective effect of IPC in diabetic rat heart by treatment of DDZ and hemin in combination was not greater than when both the drugs were administered alone. This supports that treatment with hemin facilitates the release of NO by acting on the same platform, that is, caveolin–eNOS complex. 33,34
Our data indicate that HO-1 with IPC protects the myocardium from diabetes-induced myocardial injury. So, our findings may lead to novel strategy for preventing cardiac injury due to ischemia and reperfusion in diabetic patients, which could be a beneficial adjunctive for myocardial protection during open-heart surgery.
Conclusion
On the basis of aforementioned discussion, it is assumed that activation of HO-1 by a specific activator, that is, hemin, restored the attenuated cardioprotective effect of IPC in diabetic rat heart by disrupting the caveolin–eNOS complex and thereby enhancing the release of NO. Further, pretreatment with ZnPP, a specific HO-1 inhibitor, significantly blocked the restoration of cardioprotective effect of IPC in hemin-pretreated diabetic rat heart. Therefore, it may be concluded that attenuation of cardioprotective effect of IPC in diabetic rat heart is due to reduced activity of HO-1 and subsequently decreased activity of eNOS.
Limitations of the present study
Ideally, in this study, the proposed HO-1 level should have been assessed by Western blotting, which could have supported the findings of the present study.
Footnotes
Acknowledgment
We are grateful to our teacher Late Prof. Manjeet Singh. Prof. P.L. Sharma (Supervisor) and Dr. H.N. Yadav (Co-supervisor) developed the concept and designed experiment. Ishan Gupta performed experiment on Langendorff’s apparatus in I.S.F. College of Pharmacy, Moga, Punjab, India. Prof. P.L. Sharma, Dr. H.N. Yadav and Ishan Gupta carried out analysis of infarct size and data of other parameters (LDH, CK-MB, and Nitrite). Ishan Gupta, Dr. H.N. Yadav, Niraj K. Singh and Ahsas Goyal prepared the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
