Abstract
Conventional chemotherapy is the most valid method to cope with cancer; however, it has serious drawbacks such as decrease in production of blood cells or inflammation of the lining of the digestive tract. These side effects occur since generally the drugs used in chemotherapy are distributed evenly within the body of the patient and cannot distinguish the cancer cells from the healthy ones. In this study, folic acid (FA)-conjugated, polyethylene-coated magnetic nanoparticles (FA-MNPs), and doxorubicin (Dox)-loaded formulation (Dox-FA-MNPs) were prepared. The cytotoxicity of these nanoparticles on HeLa and Dox-resistant HeLa cells was investigated. Magnetic nanoparticles (MNPs), polyethylene glycol (PEG)-coated MNPs (PEG-MNPs), and FA-MNPs were successfully synthesized and characterized by several methods. Dox loading of FA-MNPs and release profile of Dox from the nanoparticles were studied. Cytotoxic effects of FA-MNPs and Dox-FA-MNPs on HeLa cells were analyzed. MNPs, PEG-MNPs, and FA-MNPs all had small sizes and supermagnetic behavior. High amounts of Dox could be loded onto the nanoparticles (290 μgmL−1). In 24 h, 15.7% of Dox was released from the Dox-FA-MNPs. The release was increased in acidic conditions (pH 4.1). Internalization studies showed that FA-MNPs and Dox-FA-MNPs were taken up efficiently by HeLa cells. The investigation of cytotoxicity of the particles indicated that 38–500 μgmL−1 Dox-FA-MNPs significantly decreased the proliferation of HeLa cells compared to FA-MNPs. Due to their size, magnetic properties, internalization, drug release, and cytotoxicity characteristics, the MNPs prepared in this study may have potential application as a drug delivery system in cancer chemotherapy.
Introduction
Drug delivery systems (DDSs) enhance the efficiency and safety of a drug by determining how long, how fast, and where drug release occurs in the body. 1 Chemotherapeutic drugs are generally spread out evenly within the body; as a result, they have effect not only on cancer cells but also on healthy cells. Due to this even distribution, cancerous tissue gets limiting dose of the drugs. Increase in the dose of the drug to kill more cancer cells causes excessive toxicity in the body. 2 Therefore, DDSs have been designed to increase influence of chemotherapy and decrease drug toxicity by not only providing increment of drug accumulation in tumor site but also lowering quantity of drug distributed to healthy parts of body. 3 The most important feature of DDSs is that drugs are released in a controlled manner. Until now, many new DDSs have been developed such as capsules, polymers, liposomes, microparticles, and nanoparticles. These systems must have certain required properties which include biocompatibility, biodegradability, and a targeted biodistribution at desired regions supplying the therapeutic agents for a long period of time. 4
Nano-DDS offer some advantages in directing and release of large amounts of therapeutic compounds in specific regions. Many different types of these nanostructures such as simple metal core or complex lipid–polymer constructions become functional in many ways to work as drug carriers for diversity of situations. 5 Although they have different structures, all magnetic nanoparticles (MNPs) have to own some fundamental properties to function as DDS. Moreover, these particles need to have enough magnetic properties to accumulate at the desired regions in the presence of a magnetic field. The architecture of MNPs needs to be compatible enough to apply onto living cells or organisms. In the structure of MNPs, a magnetic core is found, composed of magnetic iron oxide (Fe3O4) or maghemite. Synthetic polymers protect MNPs and make them biologically functional by coating the magnetic core. Drugs or gene vectors are sometimes attached onto the MNPs with specific organic linkers. 6 Polyethylene glycol (PEG) is a good choice for the polymeric component of the drug carrier systems because it is biocompatible, least toxic and antigenic, and highly soluble. Previously, numerous agents and biological materials, like proteins and enzymes, have been linked to PEG and it was shown that they can stay biologically active. 7
Folic acid (FA) targets to the folate receptor which is on the cell surface. The folate receptor is overexpressed by many different cancer types like endometrial, ovarian, cervical, breast, lung, kidney, colorectal, and brain tumors, while healthy cells have low expression of the receptor. 8 Therefore, FA is a useful targeting molecule for applications of targeted drug delivery in cancer.
FA is a non-immunogenic and inexpensive molecule and also it can retain its stability over a large scale of temperatures and wide range of pH values. Conjugation of FA to drug carrier does not affect the ability of FA to interact with the folate receptor. Binding of FA to its receptor causes endocytic internalization of the carrier. Drop of the pH to 5 in endosomes triggers dissociation of the folate from its receptor and causes the release of the drug. 9
The aim of this study is to synthesize and characterize PEG-coated, FA-conjugated, doxorubicin (Dox)-loaded MNPs; investigating their drug release properties, internalization, and antiproliferative effects on HeLa cells as a potential targetable DDS in cervical cancer treatment.
Material and methods
Materials
Oleylamine (OM; ≥70%), oleic acid (OA; 90%), and iron(III) acetylacetonate (Fe(acac)3; 97%) are purchased from Sigma-Aldrich (Germany). Roswell Park Memorial Institute (RPMI) 1640 Medium, fetal bovine serum (FBS), trypsin–ethylenediaminetetraacetic acid (EDTA), phosphate buffered saline (PBS), and gentamycin were purchased from Sigma-Aldrich Chemie GmbH. XTT cell proliferation assay kit (2,3-Bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide salt (XTT)) was supplied by Biological Industries, Israel Beit Haemek Ltd.
Synthesis of OA- and OM-coated magnetic iron oxide (Fe3O4) nanoparticles
In this study, Fe3O4 MNPs were prepared by the thermal decomposition of Fe(acac)3 in OA and OM solution at 300°C. 10 Fe(acac)3 was dissolved in the mixture of OA and OM and then mechanically stirred under a flow of nitrogen. The solution was dehydrated at 120°C for 1 h and then quickly heated to 300°C and kept at this temperature for 1 h. Heat source was removed to cool the black solution. Ethanol was added to the black solution and MNPs were precipitated by magnetic force. The nanoparticles were washed several times with ethanol. The synthesized OA- and OM-coated magnetic iron oxide (OL-Fe3O4) nanoparticles which have OA and OM on their surface were labeled as MNPs.
Synthesis of PEG-coated MNPs
MNP has hydrophobic property due to the hydrophobic surfactants (OA and OM) on the surface of Fe3O4 particles. To be biocompatible, soluble in hydrophilic environment, and able to carry drug, the MNPs should be coated with an appropriate polymer or dendrimer. In this research, OL-Fe3O4 MNPs were mixed with PEG in distilled water (dH2O) and the solution was stirred overnight at room temperature. Then, PEG-coated MNP (PEG-MNP) was precipitated by magnet. The nanoparticles were washed with dH2O until clear supernatant was observed. 11
FA modification of PEG-MNP
PEG-MNP was modifed with activated FA. 12 FA is dissolved in dimethyl sulfoxide. Dicyclohexyl carbodiimide is added to the solution and the solution is stirred for 2 h under a nitrogen atmosphere. Then, PEG-MNP is added and continuously stirred for 2 h under a nitrogen atmosphere. Finally, folic acid-conjugated, polyethylene-coated magnetic nanoparticles (FA-MNP) was washed twice with dH2O and stored in dH2O at room temperature.
Characterization of the nanoparticles
Crystal structures of synthesized MNPs were analyzed by X-ray powder diffraction (XRD). The chemical groups and chemical interactions involved in synthesized MNPs were identified using the Fourier transform infrared spectroscopy (FTIR). The sizes and morphological properties were observed through transmission electron microscopy (TEM) images. The percentage of PEG on the surface of the nanoparticles has been determined by thermal gravimetric analysis (TGA). Magnetic properties of MNPs were determined through vibrating sample magnetometer (VSM) analyses.
Drug loading
Different amounts of Dox (23–1500 μg) were added to the Eppendorf tubes containing 1.5 mg FA-MNP. The tubes were placed on the rotary shaker (Biosan Multi RS-60 Rotator, Latvia) and shaked at room temperature for 24 h. Dox-loaded FA-MNP was precipitated using a magnet. Then, supernatant was collected and absorption of the supernatant was measured to determine the remained Dox concentration. The loaded amount of Dox was calculated from the difference between the initial amount of Dox added to the tubes and the amount of Dox remaining in the supernatant.
Drug release studies
Dox-loaded FA-MNPs are washed with dH2O and acetate buffer was added at pH 4.1, 5.1, and 7.4 in separate Eppendorf tubes. Then, it is placed in the rotator shaker. After 3 h, particles are precipitated by magnet and supernatant is taken to a new tube for measurement of absorbance. After that, new buffer solutions were added to the Eppendorf tubes and rotated. This process was repeated several times for 72 h. Finally, concentration of released drug is calculated from its absorbance at 480 nm measured by a ultraviolet (UV) spectrophotometer (Multiskan GO; Thermo Scientific, Finland) via the slope of standard curve.
Internalization of nanoparticles
Internalization of nanoparticles by HeLa cells was investigated by Prussian blue staining (Sigma-Aldrich) and by fluorescent microscopy (Leica DM 6000 FS, Germany).
Iron oxide nanoparticles were stained by Prussian blue staining method. 13 The nanoparticles that were internalized by HeLa cells were detected by this technique. This method provides two solutions: working iron solution and working pararosaline solution. To prepare working iron stain solution, potassium ferrocynanide solution and hydrochloric acid (1:1 v/v) are mixed in a falcon tube. For preparation of working pararosaline solution, pararosaline is added to dH2O to be 2% (v/v) in a falcon tube. Acid ferrocyanide in working iron stain solution reacts with iron producing blue or dark blue color in case of heavy deposit of iron, while working pararosaline solution stains cell producing red color in nucleus and pink color in cytoplasm.
Procedure of Prussian blue staining is modified. Firstly, cover slides were placed inside the wells of six-well plate. About 200,000 cells were seeded to each well. Old medium was removed after 24 h and cells were washed with PBS solution. Two millilitre of fresh media containing different concentrations of Fe3O4 particles were added to the wells. Cells were incubated at 37°C and 5% carbon dioxide (CO2) for 7 h. Then, cells were washed five times with PBS to get rid of MNPs that have not been internalized by cells. Working pararosaline solution is added to wells containing cells and incubated for 10 min. The cells are then washed with dH2O. Working pararosaline solution is added to the flask containing cells and incubated for 5 min. The cells are washed with dH2O and left to dry. Finally, the cells are observed under light microscopy and photographed.
The second method used for determination of the nanoparticle internalization is based on fluorescent property of Dox. When Dox is loaded onto FA-MNPs, they can be detected by fluorescent microscopy. Firstly, about 200,000 cells were seeded to each well of a six-well plate. The medium was removed after 24 h and cells were washed with PBS solution. Then, 2 mL fresh media containing different concentrations of Dox-loaded MNPs were added to the wells. Cells were incubated at 37°C and 5% CO2 for 7 h. Then, cells were washed five times with PBS to get rid of the MNPs that have not been internalized by cells. The cells are observed under fluorescent microscope and photographed.
Cytotoxicity of MNPs
HeLa cells were used for cytotoxicity studies. Cells were grown in 75 T culture flasks (Corning, Product #430725U) in RPMI-1640 culture medium supplemented with 10% FBS and 1% gentamycin solution at 37°C under 5% CO2. The cells were subcultured two or three times per week with 0.25% trypsin–EDTA.
Antiproliferative effects of MNPs on HeLa were evaluated by means of the Cell Proliferation Kit (Biological Industries, Cromwell, Connecticut, USA) according to manufacturer’s instructions. Assay was a colorimetric test based on the reduction of tetrazolium salt, XTT, to colored formazan products by mitochondria of live cells. In brief, cells were seeded to 96-well microtiter plates (Greiner, Austria) at a concentration of 1.0 × 104cells/well and incubated for 72 h in medium containing horizontal dilutions of nanoparticles. In each plate, assay was performed with a column of blank medium control and that of cell control. Then, XTT reagent was added and the optical density was measured at 492 nm with 96-well plate reader (Spectromax 340 Molecular Devices, Sunnyvale, California, USA).
Results
The preparation of MNPs
Coprecipitation and thermal decomposition are the most widely used methods for synthesis of Fe3O4 MNPs. The simplest and more preferred method to synthesize MNPs is the coprecipitation technique. However, thermal decomposition method provides control on size and morphology of the nanoparticles. MNPs are synthesized in small size and narrow size range via thermal decomposition method. 14
OA and OM make the synthesized MNPs hydrophobic on the surface. They are produced by decomposition of Fe(acac)3 in mixture of these two surfactants at high temperature. Then, the synthesized MNPs are coated with PEG monooleate by hydrophobic interaction between hydrophobic surfactants on MNPs and oleate part of PEG monooleate in dH2O which is a hydrophilic solvent.
Characterization of MNPs
TEM of MNPs
Size and morphology of synthesized OL-Fe3O4 MNPs have been observed by TEM (FEI/Tecnai G2 F30, Hillsboro, Oregon, USA). Obtained images (Figure 1(a) and (b)) showed that MNPs are almost spherical and have more uniform size distribution. The average diameter of MNPs is approximately 10 nm. Images of MNPs in two solvents (ethanol and hexane) are taken by TEM. Ethanol can dissolve both hydrophobic and hydrophilic substances because it is a versatile solvent, while hexane dissolves only hydrophobic materials due to its hydrophobic property. As a result, MNPs are more dispersed in hexane than in ethanol because MNPs are hydrophobic due to the presence of OA and OM on their surface.

TEM images of MNPs: (a) in ethanol, (b) in hexane. TEM: transmission electron microscopy; MNP: magnetic nanoparticle.
X-ray diffraction
The crystal structure of sythesized iron oxide (OL-Fe3O4) nanoparticles was determined by XRD. Diffraction peaks at 2θ values of 30.25, 35.41, 43.41, 57.30, 62.70, and 74.49 (degrees) are corresponding to specific diffractive plane indexes (220), (311), (400), (422), (511), (440), and (533), respectively (Figure 2). XRD patterns of OL-Fe3O4 were examined by comparing the peaks of standard magnetic iron oxide in Joint Committee on Powder Diffraction Standards (JCPDS) file (PDF no: 01-075-1609). All peaks are characteristic peaks of the magnetic iron oxide (Fe3O4) crystals that have an inverse cubic spinel structure. XRD results revealed the presence of the Fe3O4 crystals in the synthesized nanoparticles OL-Fe3O4. The peaks shown in the XRD pattern of the prepared sample are sharp and intense, indicating crystallinity of the sample. The crystallinity of nanocrystals had a significant effect on magnetic properties of nanocrystals. 15
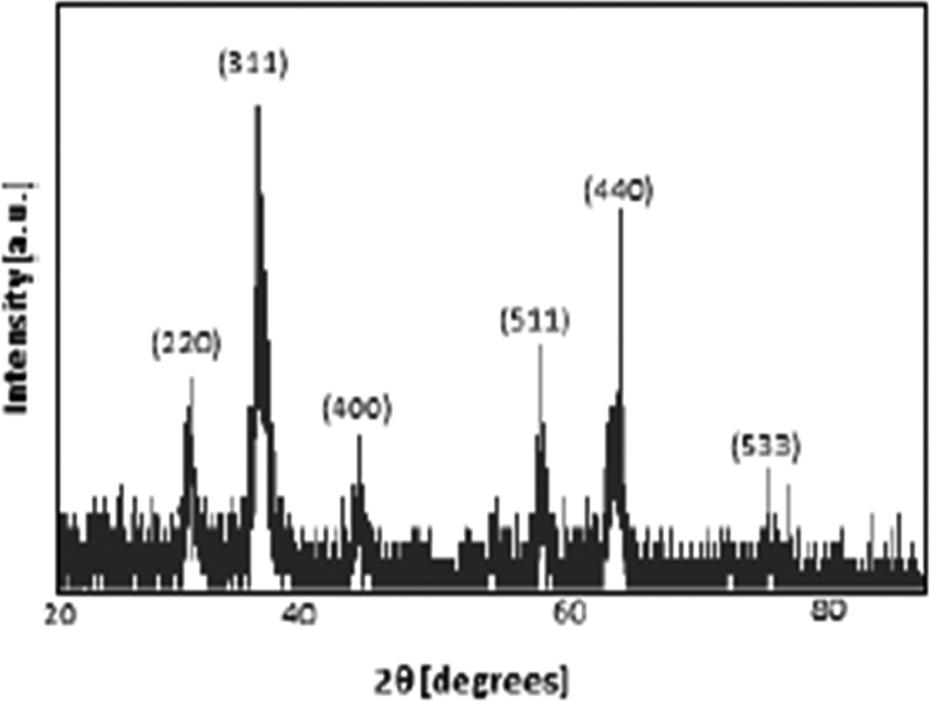
XRD patterns of synthesized iron oxide (OL-Fe3O4) nanoparticles. XRD: X-ray powder diffraction; OL-Fe3O4: OA- and OM-coated magnetic iron oxide.
FTIR of MNPs
In order to confirm the chemical composition of synthesized nanoparticles, FTIR analysis was obtained. The peak located at the 583 cm−1 region, characteristic for the Fe–O group MNPs’ spectra, confirms that the products contain magnetic iron oxide (Fe3O4). All characteristic peaks of OL-Fe3O4 are shown in Figure 3. The band at 580 cm−1 corresponded to the vibration of the Fe–O bonds in the crystalline lattice of Fe3O4. 16 The bands at 2852 and 2922 cm−1 were attributed to the asymmetric methylene (CH2) stretch and the symmetric CH2 stretch in OA, respectively. 17 The band at 1409 cm−1 corresponded to the methyl group umbrella mode in OA. The bands at 1427 and 1523 cm−1 were attributed to the asymmetric (COO) carboxyl and symmetric (COO) stretch vibration band. 18

FTIR spectrum of MNPs. FTIR: Fourier transform infrared spectroscopy; MNP: magnetic nanoparticle.
TGA of MNPs
The percentages of Fe3O4 and OL (OA and OM) in the nanoparticles were measured by thermogravimetric analyzer. The TGA analysis of MNPs (OL-Fe3O4) provides qualitative and quantitative information about the nanoparticle composition. The TGA analysis of MNPs has two weight loss phases (Figure 4). The first weight loss (about 2%) belongs to the decomposition of ethanol absorbed by OL molecules on the surface of Fe3O4 particles. In the second phase of weight loss which is almost 10% of total mass, OL molecules were decomposed by increasing the temperature. TGA result also suggests that MNPs have OL molecules on their surface.

TGA of MNPs. TGA: thermal gravimetric analysis; MNP: magnetic nanoparticle.
VSM of MNPs
Magnetic hysteresis curve (Figure 5) was obtained by VSM (Cryogenic Limited PPMS, UK). The applied magnetic field was changed and magnetization properties of synthesized OL-Fe3O4 nanoparticles were measured at 37°C. Remanence and coercivity were not observed in the hysteresis curve. This phenomenon proved that all nanoparticles synthesized in this study were superparamagnetic. Supermagnetic propety of MNPs is important for anticancer drug delivery. After targeting MNPs in a tumor region by an external magnetic field, magnetic agglomeration of the nanoparticles must be avoided in the absence of magnetic field. 19 The saturated magnetization of MNPs is 55 emug−1.
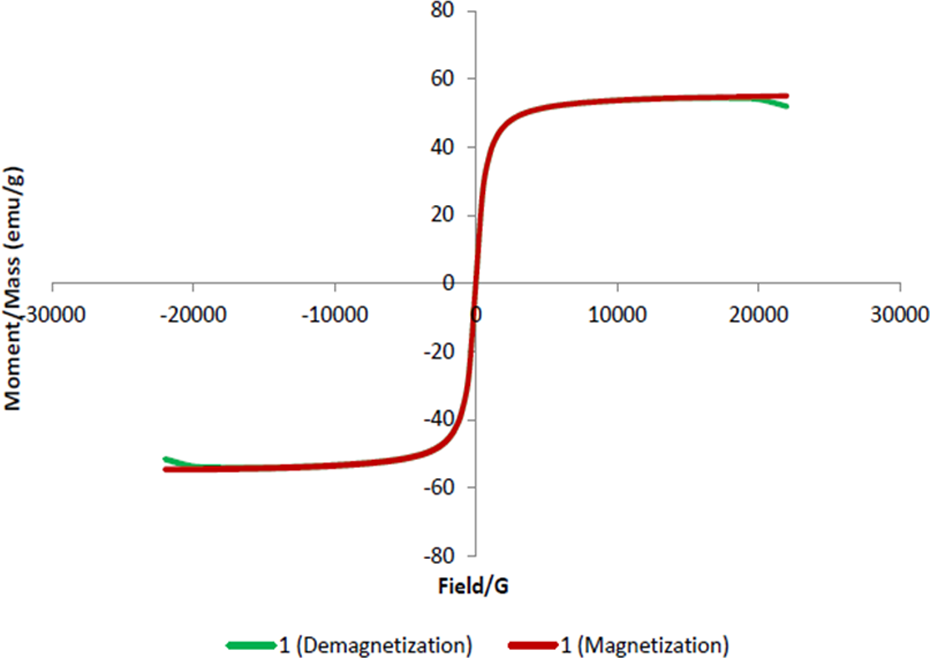
VSM results show magnetization and demagnetization curves of MNPs (55 emug−1) at 37°C. VSM: vibrating sample magnetometer; MNP: magnetic nanoparticle.
Characterization of PEG-MNP
TEM characterization of PEG-MNP
The synthesized MNPs are coated with PEG monooleate. Size and morphology of PEG-MNPs were examined by TEM. TEM results (Figure 6(a) and (b)) demonstrated that PEG-MNPs had almost spherical and more uniform size distribution. The average diameter of MNPs was approximately 15 nm. After coating MNPs with PEG, the size of the particles increased when the TEM images of MNPs and PEG-MNPs were compared. This indicated that PEG coated the MNPs and increased the size of the particles.

TEM images of PEG-MNPs. TEM: transmission electron microscopy; PEG: polyethylene glycol; MNP: magnetic nanoparticle.
FTIR of PEG-MNP
The chemical groups and chemical interactions involved in PEG-MNPs were analyzed using the FTIR method. The FTIR analyses of PEG-MNP were performed to confirm that MNPs were coated with PEG monooleate. The peaks at 946 cm−1 and 1109 cm−1 indicated the stretching vibration of functional CH2 group and the stretching vibration of functional group C–O of PEG, respectively, while the absorption band observed at 1735 cm−1 was caused by C=O carbonyl group of PEG (Figure 7). Oleate group of PEG monooleate formed hydrophobic interaction with OA on the surface of MNPs. Thus, this interaction formed interpenetration layer and PEG formed secondary layer by coating MNPs. The intense peak at 1705 cm−1 was due to the C=O stretching of oleate part of PEG monooleate. This peak was at 1710 cm−1 for second layer of OA on MNPs by Kun et al. 18 The FTIR result showed that MNPs were successfully coated with PEG.

FTIR spectrum of PEG-MNPs. FTIR: Fourier transform infrared spectroscopy; PEG: polyethylene glycol; MNP: magnetic nanoparticle.
TGA of PEG-MNPs
TGA was used to show existence of PEG on the surface of particles and to determine amount of PEG by decomposition of particles. TGA analysis revealed that MNPs had 12% weight loss due to decomposition of OA and OM, while PEG-MNPs had 44% weight loss (Figure 8). The difference of 32% showed that PEG layer exists on the surface of the nanoparticles.

TGA of PEG-MNPs. TGA: thermal gravimetric analysis; PEG: polyethylene glycol; MNP: magnetic nanoparticle.
Characterization of FA-conjugated MNPs
FTIR of FA-MNPs
The FTIR result of FA-MNP showed the characteristic absorption peaks at 1433 and 1606 cm−1 (Figure 9). The characteristic band of 1433 cm−1 wavelength was belonging to the phenyl ring of FA. 1606 cm−1 band indicated amine bending vibration of FA. 11 These results indicated the modification of PEG-MNPs with FA.

FTIR spectrum of FA-MNPs. FTIR: Fourier transform infrared spectroscopy; MNP: magnetic nanoparticle; FA: folic acid.
Drug loading
Different amounts of Dox were mixed with 1.5 mg FA-MNPs in 1.5 mL Tris buffer having pH 7 by rotary shaker at room temperature for 24 h. Increase in the total drug concentration in the mixture increased the amount of the Dox loaded to FA-MNPs (Table 1). At the highest Dox concentration (1724 μM), 439 μg Dox was loaded to 1.5 mg FA-MNPs, which was 29% of total Dox amount. Thus, 290 μgmL−1 Dox was loaded to 1 mgmL−1 nanoparticles (Figure 10).
Concentration of Dox loaded to FA-MNPs.

Dox: doxorubicin; SEM: standard error of the mean; FA: folic acid; MNP: magnetic nanoparticle.

Percentages of Dox loaded to FA-MNPs in different concentrations of Dox. Dox: doxorubicin; FA: folic acid.
Drug release
The release profiles of Dox from dox-loaded FA-MNPs (Dox-FA-MNPs) in acetate buffer with pH 4.1, 5.1, and 7.4 at 37°C were measured at 480 nm in 3-h intervals by UV spectrophotometer. Drug release was investigated at acidic pH to mimic the endosomal condition. Moreover, the release temperature was 37°C which is equal to the body temperature and also incubation temperature of cells. According to the results, at low pH 4.1, drug release from nanoparticles occurred faster and higher than at higher pH values (Figure 11). Acidity of environment affected the release of Dox in vitro. In more acidic condition, the release rate of Dox was higher. Percentages of drug releases from Dox-FA-MNPs were 15.7%, 14.3%, and 10.0% in acetate buffers with pH 4.1, 5.1, and 7.4, respectively, for 72 h. 19

Released Dox concentration from Dox-FA-MNPs in acetate buffers with different pH values. Dox: doxorubicin; MNP: magnetic nanoparticle; FA: folic acid.
Cell culture studies
Internalization of nanoparticles
FA-MNPs internalized by HeLa cells were investigated by Prussian blue staining and Dox-FA-MNPs uptaken by HeLa were detected by fluorescent microscopy using fluorescent property of Dox.
Detection of FA-MNPs in HeLa cells by Prussian blue staining
Prussian blue staining method stains cellular components and iron core of particles in different colors. By this technique, cell and cell components are stained purple, while nanoparticles are blue or dark blue in order to observe internalization of the nanoparticles. Different concentrations of FA-MNPs were given to HeLa cells and incubated at 37°C for 7 h. Then, the cells were stained by Prussian blue staining method. Increasing concentration of the nanoparticles increased the amount of the particles in the cells (Figure 12). Non-treated HeLa cells were seen in only pink color, while FA-MNPs-treated HeLa cells were colored as both pink and blue. This result showed that FA-MNPs were internalized by the cells. Also, internalized nanoparticles in low dose were localized together in cells, while in highest dosage, they covered all around the cytoplasm.

Images of HeLa cells treated with (a) 0 μgmL−1, (b) 25 μgmL−1, (c) 50 μgmL−1, (d) 100 μgmL−1, (e) 150 μgmL−1, and (f) 200 μgmL−1 FA-MNPs. (The photographs of the cells were taken under light microscope. Magnification is ×10.) MNP: magnetic nanoparticle; FA: folic acid.
Detection of Dox-FA-MNPs in HeLa cells by fluorescent microscopy
Other detection method for internalized nanoparticles was using the fluorescent property of Dox. As Dox was loaded to FA-MNPs, they gained its fluorescent property. HeLa cells were treated with Dox and different concentrations of Dox-FA-MNPs and incubated at 37°C for 7 h. In order to prevent the background fluorescence from non-internalized Dox-FA-MNPs, HeLa cells were extensively washed with PBS. Only Dox-treated HeLa cells had fluorescence in their nucleus because Dox targets nucleus, while due to endosomal internalization of the particles, there was intense fluorescent emission in cytoplasm of Dox-FA-MNP-treated HeLa cells (Figure 13).

Images of HeLa cells treated with (a) Dox, (b) 50 μgmL−1, (c) 150 μgmL−1, and (d) 200 μgmL−1 of Dox-FA-MNPs. (The photographs of the cells were taken under fluorescent microscope. Magnification is ×20.) Dox: doxorubicin; MNP: magnetic nanoparticle; FA: folic acid.
Antiproliferative activity of nanoparticles
The antiproliferative effects of drug-loaded MNPs (Dox-FA-MNP) and non-loaded MNPs (FA-MNPs) were determined by XTT cell proliferation assay. For this, HeLa cells were treated with different doses (38–500 μgmL−1) of FA-MNPs and Dox-FA-MNPs, and then incubated for 72 h. Dose-dependent effects of FA-MNPs and Dox-FA-MNPs on HeLa cells were analyzed. The result demonstrated that FA-MNPs did not have cytotoxicity on HeLa cells, while those of Dox-FA-MNPs caused significant decreases on cell proliferation of HeLa cells (Figure 14).

Antiproliferative effects of FA-MNPs and Dox-FA-MNPs on HeLa cell line. Results were obtained from three independent experiments, represented as mean ± SEM. #p < 0.05. MNP: magnetic nanoparticle; Dox: doxorubicin; SEM: standard error of the mean; FA: folic acid.
Discussion
The main purpose of this study was to synthesize FA-modified PEG-coated MNPs for Dox delivery. MNPs are important candidate for drug targeting due to their magnetic property. This propety can be utilized to accumulate the particles around the tumor site by an external magnetic field. 20 The MNPs can be used to carry anticancer drug to tumor site. By this way, systemic side effects of the drug could be eliminated or minimized. VSM result indcated the superparamagnetic feature of the systhesized particles (Figure 5). In other word, the particles had magnetic behaviour only under a magnetic field. OL-Fe3O4 nanoparticles were coated with PEG polymer because PEG molecules improve the biocompatibility and circulation of the particles in blood. 21 The desiresd nanoparticle size must be between 5.5 nm and 200 nm to inhibit rapid renal removal from blood and endocytosis by phagocytic cells. 22 The size of PEG-MNPs obtained in this study was approximately 15 nm determined by TEM. In addition to magnetic targetable characteristics of the nanoparticles, FA was conjugated to PEG-MNPs as a chemical targeting agent. Numerous cancer types overexpress folate receptor, including ovarian carcinomas, endometrial carcinomas, renal cell carcinomas, lung adenocarcinomas, mesotheliomas, and some breast cancers. 23
In this study, the MNPs synhesized by thermal decomposition method were small, spherical, and had narrow size distribution. Also, due to the method of the synthesis, the MNPs had hydrophobic surfactants (OA and OM) on their surfaces. Therefore, an additional step for coating the nanoparticles with OA or other surfactants was skipped. 11
The drug loading capacity of FA-MNPs was 29%. The amount of Dox was 290 μgmL−1 loaded to 1 mgmL−1 nanoparticles. In another study, similar FA-MNPs had nearly 50% Dox loading capacity 12 which was higher than that found in this study. There are some factors that affect the loading capacity of particles. First, the molecular weight of PEG could have role on loading capacity. If the polymeric chains that coat the particles are long, the particles could have higher drug loading capacity. Also, pH of environment and buffer type used during drug loading could affect the amount of drug loaded to the particles. Furthermore, the amount of the polymer coating the particles affects their drug adsorption. The carriers with 0.5%, 1%, 2%, and 5% polyvinyl alcohol have different drug loading capacities: 35 μg, 41 μg, 47 μg, and 58 μg of Dox per milligram of carrier, respectively. 24
pH has a remarkable effect on the release of Dox from the nanoparticles. Drug release profiles from Dox-FA-MNPs were affected by pH value of buffer. Drug releases were 15.7%, 14.3%, and 10.0% in acetate buffers with pH 4.1, 5.1, and 7.4, respectively, in 72 h. As a result, pH-responsive drug carriers provide selective drug release at acidic intracellular vesicles such as endosomes and lysosomes in targeted tumor cells. 25 Dox, which was loaded to FA-MNPs, could be successfully delivered to the cell’s late endosome near to the nucleus where Dox shows its anticancer activity.
In the study by Kim et al., pH-sensitive mixed-micelle system conjugated with FA was prepared in order to overcome multidrug resistance in cancers. The micelles were composed of poly(histidine (His)-co-phenylalanine (Phe))-b-PEG and poly(L-lactic acid) (PLLA)-b-PEG-folate. 26 The release of Dox was found as pH dependent and higher release was observed at lower pH values as compatible with our results. In 24 h, less than 7% of Dox was released from Dox-PLGA nanoparticles and it is suggested that in longer periods of time, the release of the remaining Dox in particles could be achieved. 27
The cellular internalization of FA-MNPs by HeLa cells was determined by Prussian blue staining, while Dox-FA-MNPs internalized by the cells were determined via fluorescent microscopy using fluorescent feature of Dox. Both results confirmed that the nanoparticles were highly uptaken by HeLa cells (Figures 12 and 13). The nanoparticles were accumulated as clusters in the cytoplasm. The internalization of nanoparticles by the cells started as an interaction (adsorbing) followed by vesicle formation and then the nanoparticle-containing vesicles are taken up by endocytosis. 28 The nanoparticles were generally located near nucleus.
The nanoparticles get attached to cell membrane because bare iron oxide nanoparticles (free MNPs) were not taken up by the cells due to their negative surface charge coming from the abundant OH− ions. After that, positively charged Dox-FA-MNPs will be easily attached to negatively charged cell membrane, which will result in an increased rate of cellular internalization. 29 –31
The cytotoxicities of FA-MNPs and Dox-FA-MNPs on HeLa cells were compared to determine the cell death. Results indicated that Dox-FA-MNPs at higher doses inhibited the cell proliferation more than 70%. FA-MNPs were non-cytotoxic by themselves. It is clear that Dox released from Dox-FA-MNPs effectively killed HeLa cells.
As a result, Dox-loaded FA-conjugated PEG-coated MNPs are effective to kill cancer cells and may decrease the side effects of Dox.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by METU Research Fund (grant ID: BAP-07-02-2012-101).
