Abstract
Nano nickel oxide (NiO), widely used in industry, has recently been discovered to have pulmonary toxicity. However, no subchronic exposure studies about nano NiO-induced pulmonary fibrosis have been reported. The objective of this study was to investigate pulmonary fibrosis induced by nano NiO and its potential mechanism in rats. Male Wistar rats (n = 40, 200–240 g) were randomized into control group, nano NiO groups (0.015, 0.06, and 0.24 mg/kg), and micro NiO group (0.024 mg/kg). All rats were killed to collect lung tissue after intratracheal instillation of NiO particles twice a week for 6 weeks. To identify pulmonary fibrosis, Masson trichrome staining, hydroxyproline content, and collagen protein expression were performed. The results showed widespread lung fibrotic injury in histological examination and increased content of hydroxyproline, collagen types I and III in rat lung tissue exposed to nano NiO. To explore the potential pulmonary fibrosis mechanism, transforming growth factor beta 1 (TGF-β1) content was measured by enzyme-linked immunosorbent assay, and the messenger RNA expression of key indicators was detected by reverse transcription-quantitative polymerase chain reaction (RT-qPCR). The TGF-β1 content was increased in nano NiO exposure groups, as well as the upregulated gene expression of TGF-β1, Smad2, Smad4, matrix metalloproteinase, and tissue inhibitor of metalloproteinase. The findings indicated that nano NiO could induce pulmonary fibrosis, which may be related to TGF-β1 activation.
Introduction
Along with the progress of nanotechnology, nano nickel oxide (NiO) has been widely produced and applied for its unique physicochemical properties to meet novel functionalities. 1 Since nano NiO exists greatly in exhaust emission, welding fume, and mineral exploitation as inhalable particles, lungs become the target organs to burden risk in case without protection. 2,3 Compared to 1 μm NiO particles, 100 nm NiO induced remarkable injury and oxidative stress in the rat lung for a single instillation. 4 The distinct toxicity indicated that it is improper to infer the health hazard of nano NiO from micro NiO. It is imperative to assess the pulmonary toxicity induced by nano NiO to promote the development of nanoscience and nanotechnology.
As an insoluble metal oxide in aqueous phase, nano NiO induced apoptosis in human lung epithelial cell line (H460) via cellular intake and subsequent mobilization of nickel ions. 5 An in vitro study discovered that nano NiO increased lipid peroxidation level in human carcinoma cells (A549), resulting in the activation of antioxidative system including glutathione and heme oxygenase-1. 6 In nano NiO-treated human monocyte cells (THP-1), the gene expression levels of inflammatory cytokines such as interleukins (IL-1β, IL-6, IL-8) and tumor necrosis factors-α were upregulated. 7 After nano NiO acute exposure in rats, the polymorphonuclear neutrophils and cytokine-induced neutrophil chemoattractants in bronchoalveolar lavage fluid were increased. 8,9 In addition, pathological images showed foamy macrophages infiltrated in the alveolar cavities as well as hypertrophy and hyperplasia in alveolar epithelial cells after a single intratracheal instillation of nano NiO in rats. 10 Existing in vivo and in vitro studies have indicated that acute exposure to nano NiO induced pulmonary toxicity such as oxidative stress and inflammatory response, while there is little evidence for subsequent pulmonary fibrosis. 11 On the other hand, nano NiO is an industrial contaminant, which needs a subchronic or chronic exposure animal model to assess its occupational exposure risk. So, we performed intratracheal instillation of nano NiO twice a week for 6 weeks in rats, exploring the potential pulmonary toxicity and its mechanism.
Collagen deposition of two regions (alveolar duct and pleura) by the point counting method found that the 27 nm NiO group had a tendency to increase after a single intratracheal instillation in rats. 10 Morimoto et al. 12 reported that nano NiO inhaled for 4 weeks (6 h/d) could upregulate gene expression of collagen type I in rat lung tissue. If exposed to extended period, fibrosis may have been observed among the nano NiO-instilled groups throughout our study. To date, the mechanisms of pulmonary fibrosis induced by nano NiO are still unclear, but a great deal of research studies have suggested that transforming growth factor beta 1 (TGF-β1) plays a critical role in paraquat or carbon nanotube-induced fibrosis. 13,14 TGF-β1 ligand binding result in the formation of a complex consisting of Smad2 and Smad3, phosphorylated complex combine Smad4 and transfer into the nucleus to regulate target gene which is related to the secretion of extracellular matrix. In carbon nanotube or bleomycin-induced pulmonary fibrosis, TGF-β1, Smad2 and Smad4 gene, and protein expression were upregulated in vivo or in vitro. 15,16 Following the activation of TGF-β1, Smad2, and Smad4, myofibroblast can secrete excessive extracellular matrix components under the regulation of matrix metalloproteinases (MMPs) and tissue inhibitor of metalloproteinases (TIMPs). 17 MMP-9 and TIMP-1 gene expression were elevated in human myelomonocytic cells (U937) treated with nano nickel for 48 h. 18 Furthermore, connective tissue growth factor (CTGF) promoted fibroblasts migration and proliferation, and increased in fibrogenic process. 19
In this study, we hypothesized intratracheal instillation of nano NiO twice a week for 6 weeks could induce pulmonary fibrosis in rats, thus histopathological examination, hydroxyproline content, and collagen protein expression in lung tissue were used to verify the hypothesis. To explore the potential mechanism of pulmonary fibrosis, we quantified TGF-β1 content and messenger RNA (mRNA) expression of critical indicators including TGF-β1, Smad2, Smad4, MMP-9, TIMP-1, and CTGF in lung tissue.
Materials and methods
NiO particles characterization and samples preparation
Nano NiO and micro NiO were purchased from ST-Nano Science and Technology Co., Ltd. (Shanghai, China). Particle size of NiO was measured using a scanning electron microscope (Jeol, Jsm-7610 F, Japan). The purities of NiO particles were detected by inductively coupled plasma mass spectrometry (Agilent, 7500i, Santa Clara, California, USA). Specific surface area of NiO particles was detected by Brunauer–Emmett–Teller method (Beishide, 3H-2000PS2, China), and the crystal structure of NiO were characterized using an X-ray diffractometer (Shimadzu, XRF-1800, Japan).
NiO particles were sterilized at 120°C for 30 min in a high-pressure steam autoclave (Panasonic, MLS-3751L, Japan) before each intratracheal instillation. Nano NiO particles were dispersed into 0.9% normal saline for gradient concentration at 0.015, 0.06, and 0.24 mg/mL, and a single concentration of micro NiO at 0.24 mg/mL. Suspensions were ultrasonicated using an ultrasonic homogenizer at output of 750 W for 30 min (Cole-Parmer, CP750, Vernon Hills, Illinois, USA), and vortically oscillated for 5 min before intratracheal instillation.
Animals and intratracheal instillation
A total of 40 adult male Wistar rats (200–240 g) were obtained from the Experimental Animal Center of Lanzhou University (Lanzhou, China). All rats were housed in laboratory animal room with 20 ± 2°C temperature, 60% relative humidity, 12-h light/12-h dark cycle, and supplied with commercial diet and tap water ad libitum. Animals received intratracheal instillation after 1 week acclimation period. All procedures involving animals were conducted under an institution criteria approved by the animal care and use committee of Lanzhou University.
Nishi et al. 20 reported the number of polymorphonuclear neutrophils in bronchoalveolar lavage fluid was increased after a single intratracheal instillation of 0.66 mg/kg nano NiO (20 nm) in rats. In this study, we chose 0.06 mg/kg for 12 times as the middle dosage, which was approached to 0.66 mg/kg nano NiO. We randomized 40 adult male Wistar rats into five groups for intratracheal instillation based on body weight as (i) control group, (ii) 0.015 mg/kg nano NiO group, (iii) 0.06 mg/kg nano NiO group, (iv) 0.24 mg/kg nano NiO group, and (v) 0.24 mg/kg micro NiO group. Rats were anesthetized with inhaled diethyl ether, then intratracheally instilled with 0.9% normal saline and gradient concentrations NiO suspensions at the volume of 1 mL/kg twice a week for 6 consecutive weeks. All animals were killed by exsanguination via heart after intoxication, and the lung tissues were immediately dissected and stored at −80°C for further analysis.
Histological examination
To identify whether nano NiO could induce pulmonary fibrosis in five rats of each group, the posterior lobe of right lung was fixed in Bouin’s solution, then stored at 4°C for 24 h to Masson trichrome staining. The lung tissues were embedded in paraffin and cut into 5 μm sections to transfer on slides. After deparaffinized in xylene and dehydrated in graded ethanol, slides were stained in hematoxylin for 10 min and differentiated in 0.5% hydrochloric acid ethanol after rinsing with distilled water. Following staining in ponceaux scarlet-acid fuchsin for 10 min, slides were rinsed with distilled water and embathed in 1% glacial acetic acid. After differentiation in 1% phosphomolybdic acid aqueous for 2 min, slides were stained in aniline blue for 2 min and embathed in 1% glacial acetic acid. The slides were dehydrated by graded ethanol and cleared by dimethylbenzene, then sealed with neutral gum. The slides were observed and photographed by a light microscope (Olympus, BX53, Japan).
After Masson trichrome staining, which dyed cell nuclear (black), cytoplasm (red), and collagen (blue) in three different colors, semiquantitative analysis were carried out by icalibur biolife pro 3.0 software (Beijing Icalibur Research and Development Center, China). One visual field of alveolar zone was chosen in each rat slide, and five rats in each group, then the collagen positive area percentage was calculated by defining the blue color of collage in control group as the initial color values.
Measurement of hydroxyproline content in lung tissue
The collagen levels were assessed by measurement of hydroxyproline content in lung tissue. According to the detection kit protocol (Nanjing Jiancheng bioengineering institute, China), 60 mg left lung apex was chopped and hydrolyzed at 100°C for 20 min in 1 mL hydrolysate, neutralized to pH 6.8 with sodium hydroxide, diluted to 10 mL with distilled water, and yielded 4 mL samples to add 30 mg activated carbon, then centrifuged at 1200 × g for 10 min at 4°C. The hydroxyproline content was measured at 550 nm wavelength (Jingke, 723N, China), data were expressed as microgram of hydroxyproline per milligram of lung tissue.
Measurement of TGF-β1 content in lung tissue
Each left lobe of rats’ lung tissue was homogenized with 0.9% (w/v) normal saline and centrifuged at 1100 × g for 20 min, then the supernatant was collected and stored at −80°C until further processing. The TGF
Western blot to analyze collagen content in lung tissue
Western blot was performed to measure the protein expression levels of COL1A1 and COL3A1 in lung tissue of five rats for each group, approximately, 100 mg middle lobe of right lung was homogenized in 1.0 mL radioimmunoprecipitation assay containing 10 μL phenylmethanesulfonyl fluoride and centrifuged at 14,000 × g for 5 min at 4°C. Supernatant was collected to quantify total protein concentration using the bicinchoninic acid assay kit (Nanjing Jiancheng Bioengineering Institute, China). Protein samples were mixed with 5× sodium dodecyl sulfate (SDS) buffer and denatured at 100°C for 5 min.
Equal quantities (50 μg) of protein were separated on precasted sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). β-Actin, COL1A1, and COL3A1 were separated by 10%, 10%, and 6% SDS-PAGE, respectively. Then electrophoresis was stopped when the bromophenol blue reached the bottom of the gel. Separated proteins were transferred to the polyvinylidene difluoride membranes (Millipore, Darmstadt, Hessen, Germany) at 250 mA for 150 min in transfer buffer. Membranes were then incubated in Tris-saline-Tween 20 (TBST) buffer containing 5% skimmed milk for 120 min at room temperature. Following rinsing thrice for 10 min in TBST, membranes were incubated with goat anti-rat β-actin (1:200 dilution), COL1A1 (1:500 dilution), and COL3A1 (1:200 dilution) primary antibodies (Santa Cruz Biotechnology Incorporation, Dallas, Texas, USA) overnight at 4°C. Blots were rinsed thrice for 10 min in TBST, then incubated in horse radish peroxidase-conjugated rabbit anti-goat secondary antibody (1:5000 dilution, Santa Cruz Biotechnology Incorporation, Dallas, Texas, USA) for 120 min at room temperature on rocking table. Finally, membranes were rinsed thrice for 10 min in TBST, and incubated with enhanced chemiluminescence reagent (Thermo Scientific, Waltham, Massachusetts, USA) for 2 min and exposed in Molecular Imager ChemiDoc XRS System (Bio-Rad, Hercules, California, USA). Semiquantitative analysis of target immunolabeled bands was performed using Image Pro-Plus 6.0 software (Media Cybernetics, Rockville, Montgomery, USA).
RT-qPCR to analyze mRNA expression in lung tissue
To quantify the mRNA expression levels of critical fibrotic indicators, approximately 95 mg middle lobe of right lung was used for real-time RT-qPCR. Total RNA was extracted from lung tissue using 1 mL Trizol reagent (Invitrogen, UK), and the concentrations of total RNA was measured by NanoDrop 2000C spectrophotometer (Thermo Scientific) at 260 and 280 nm absorbance wavelengths, of which the A 260/A 280 ratio between 1.8 and 2.2 was used in the next step. RNA (1μg) was reverse transcribed into complementary DNA using Fast Quant RT kit (Tiangen Biotech Co., Ltd, China) after genomic DNA clearance. RT-qPCR was performed according to the manufacture’s protocol (Tiangen Biotech Co., Ltd) with IQ5 multicolor RT-PCR detection system (Bio-Rad, USA) under the following conditions: initial denaturation at 95°C for 15 min, following 40 repeats of denaturation at 95°C for 10 s, anneal at 60°C for 20 s, extension at 72°C for 32 s, then increased to 95°C with a heating rate of 0.5°C/s for melting curve. All primers used in this study are listed in Table 1. We quantified the mRNA expression levels of TGF-β1, Smad2, Smad4, MMP-9, TIMP-1, and CTGF with the Pfaffl method. 21
Primer sequences and theoretical amplification length.

TGF-β1: transforming growth factor beta 1; MMP: matrix metalloproteinase; TIMP: tissue inhibitor of metalloproteinase; CTGF: connective tissue growth factor.
Statistical analysis
Data were analyzed using SPSS 20.0 software (IBM, Armonk, New York, USA) and expressed as mean ± standard deviation. For comparisons among multiple groups, one-way analysis of variance was carried out and the significance of differences between two groups was determined by the least significant difference test. A two-tailed p < 0.05 was considered statistical difference.
Results
Particles characterization
As nano NiO can agglomerate into particles larger than 100 nm due to van der Waals force, coexisting ions and static electricity, 22 we confront agglomerability with ultrasonic and vortex oscillation to ensure the particle diameter at nanolevel in suspensions. 23
The morphology of NiO particles was observed by scanning electron microscopy, which showed that most particles had a uniformly scattered situation. The phase identification of the NiO particles was performed by X-ray diffraction, in which a sphere structure appeared. The images obtained by scanning electron microscopy and X-ray diffraction were the same as our previous report. 24 The physicochemical characteristics of NiO particles are shown in Table 2.
Physicochemical properties of NiO particles.

NiO: nickel oxide.
Nano NiO induced pulmonary fibrosis in rats
To verify whether intratracheal instillation of nano NiO could induce pulmonary fibrosis in rats, the histopathological observation, and fibrotic indicators of hydroxyproline and collagen were evaluated in lung tissue.
Light microscope and semiquantitative analysis of fibrotic lesion in lung tissue were performed for the five different experimental groups. Figure 1(a) and (b) shows Masson trichrome staining for fibrotic injury and collagen deposition in lung tissue. We observed normal structure of the lung tissue with 3.98 ± 0.89% positive collagen area in the control group. With the dose increasing, the fibrotic effect became more obvious, namely significantly higher collagen deposited in alveolar septum and cavities than the control group (p < 0.05), and the positive collagen area was up to 14.97 ± 2.63% in 0.24 mg/kg nano NiO group. Compared to the identical dosage of 0.24 mg/kg nano NiO, collagen deposition and widened alveolar septum were alleviated in micro NiO group.
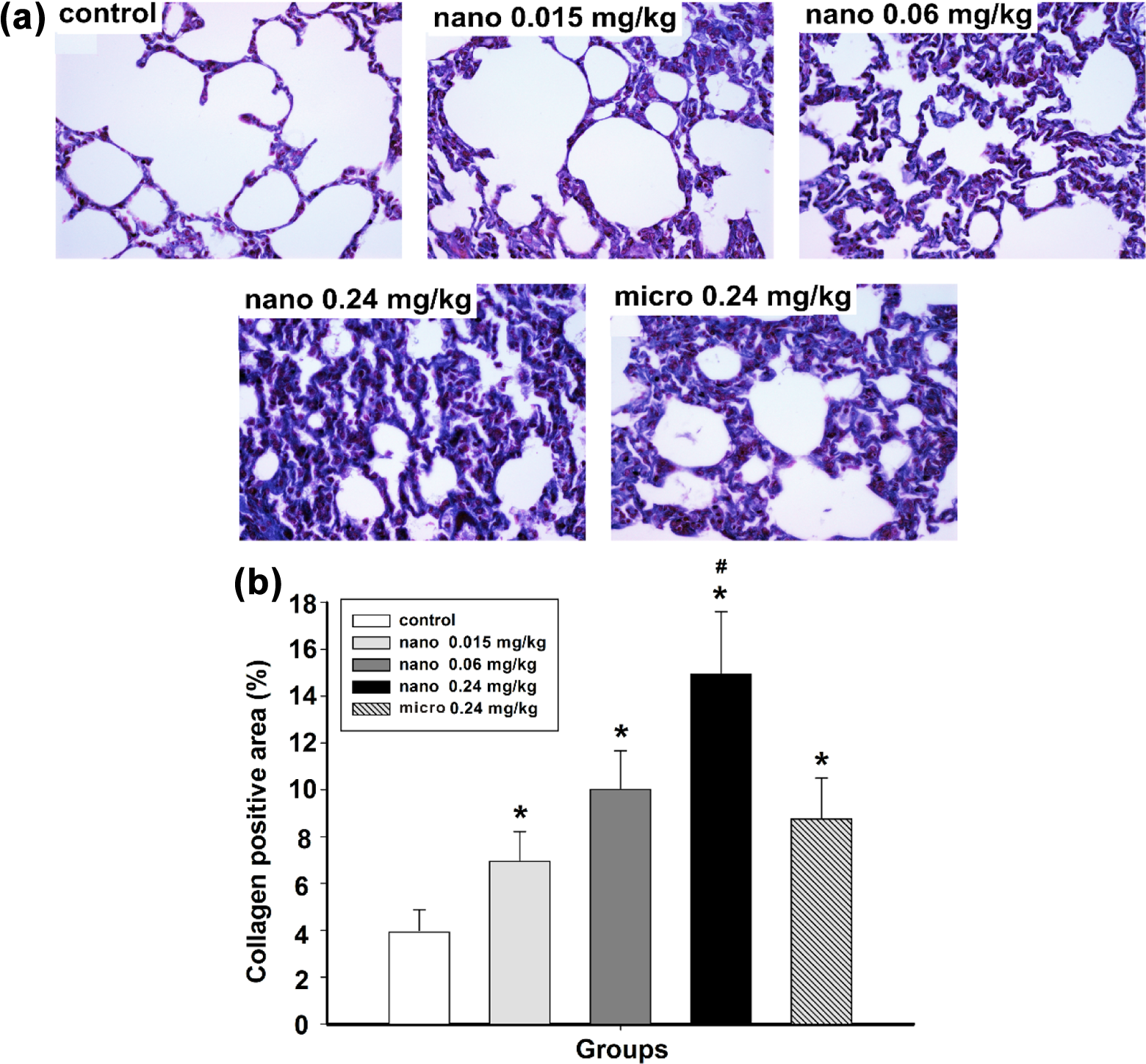
The collagen deposition in rat lung tissue after intratracheal instillation of NiO particles: Masson trichrome staining (a) and semiquantitative analysis (b), magnification ×400. Data are expressed as mean ± standard deviation, n = 5. *Significant difference from the control group, p < 0.05. #Significant difference from the 0.24 mg/kg micro NiO group, p < 0.05. NiO: nickel oxide.
Hydroxyproline, a specific marker of collagen, was measured to assess the development of pulmonary fibrosis. As shown in Figure 2, the hydroxyproline content was 0.33 ± 0.02 μg/mg in control group, which was significantly lower than 0.06, 0.24 mg/kg nano NiO groups and 0.24 mg/kg micro NiO group (p < 0.05). In addition, the hydroxyproline content in 0.24 mg/kg micro NiO group was up to 0.40 ± 0.05 μg/mg, which was still significantly lower than 0.24 mg/kg nano NiO groups (p < 0.05).

Changes of hydroxyproline content in rat lung tissue after NiO particle exposure. Data are expressed as mean ± standard deviation, n = 8. *Significant difference from the control group, p < 0.05. #Significant difference from the 0.24 mg/kg micro NiO group, p < 0.05. NiO: nickel oxide.
Further, in order to assess collagen deposition, we analyzed the protein expression levels of COL1A1 and COL3A1 in lung tissue. As shown in Figure 3(a) and (b), with the dose increasing, the COL1A1 and COL3A1 contents in 0.06 and 0.24 mg/kg nano NiO groups were significantly higher than control group and micro NiO group (p < 0.05). In addition, the COL1A1 and COL3A1 contents presented no significant difference between control group and micro NiO group (p > 0.05).

Relative protein expression levels of COL1A1 and COL3A1 in rat lung tissue after NiO particle exposure: western blot (a) and semiquantitative analysis (b). Data are expressed as mean ± standard deviation, n = 5. *Significant difference from the control group, p < 0.05. #Significant difference from the 0.24 mg/kg micro NiO group, p < 0.05. NiO: nickel oxide.
TGF-β1 content and mRNA expression in lung tissue
To explore whether TGF-β1 was activated by nano NiO in lung tissue, we measured the TGF-β1 content by ELISA kit and the mRNA expression level of TGF-β1 by RT-qPCR. Compared to control group (Figure 4(a)), the TGF-β1 content was significantly increased in 0.06 and 0.24 mg/kg nano NiO groups and micro NiO group, respectively (p < 0.05). As shown in Figure 4(b), with the dosage increasing, the mRNA expression levels of TGF-β1 were upregulated, and there were significant differences in 0.06 and 0.24 mg/kg nano NiO groups when compared to control group (p < 0.05). The mRNA expression level of TGF-β1 in 0.24 mg/kg nano NiO group was higher than micro NiO group.

TGF-β1 content (a) and relative mRNA expression levels of TGF-β1 (b) in rat lung tissue after NiO particles exposure. Data are expressed as mean ± standard deviation, n = 8. *Significant difference from the control group, p < 0.05. #Significant difference from the 0.24 mg/kg micro NiO group, p < 0.05. TGF-β1: transforming growth factor beta 1; NiO: nickel oxide; mRNA: messenger RNA.
mRNA expression of fibrosis factors in lung tissue
To detect whether pulmonary fibrosis factors were abnormal after nano NiO exposure, we detected the mRNA expression levels of Smad2, Smad4, MMP-9, TIMP-1, and CTGF in lung tissue by RT-qPCR. As shown in Figure 5, with the dose increasing, the mRNA expression levels of Smad2, Smad4, MMP-9, TIMP-1, and CTGF were upregulated, and there were significant differences in 0.06 and 0.24 mg/kg nano NiO groups when compared to control group (p < 0.05). The mRNA expression levels of Smad2, Smad4, and CTGF in 0.24 mg/kg nano NiO group were higher than micro NiO group.

Relative mRNA expression levels of Smad2, Smad4, MMP-9, TIMP-1, and CTGF in rat lung tissue after NiO particles exposure. Data are expressed as mean ± standard deviation, n = 8. *Significant difference from the control group, p < 0.05. #Significant difference from the 0.24 mg/kg micro NiO group, p < 0.05. MMP: matrix metalloproteinase; TIMP: tissue inhibitor of metalloproteinase; CTGF: connective tissue growth factor; mRNA: messenger RNA.
Discussion
The present study chose intratracheally instilled nano NiO to construct rat pulmonary fibrosis model for the following reasons. (1) Nano NiO has a wide range of applications such as catalysts, cosmetics, and sensors, and the high consumption leads to environment pollution and health hazard. 25,26 (2) As an insoluble compound, nano NiO can exert toxicity via cellular uptake and subsequent mobilization of nickel ions. (3) Lung is an important target organ of inhaled nano NiO, 27 and International Agency for Research on Cancer has classified NiO particles as Group I carcinogenic substance, for its association with the development of lung cancer. (4) To date, there have been few reports on the studies on pulmonary fibrosis induced by nano NiO, not to mention its potential mechanisms.
Compared to 1–2 μm NiO, 20 nm NiO exerted stronger cytotoxicity to inhibit viability and colony-forming ability in A549 cells. 28 Another study showed that nano NiO induced more collagen deposition in alveolar duct and pleura after a single intratracheal instillation of 2.7 μm or 27 nm NiO in rats. 10 Since nano NiO own smaller particle size, higher specific surface area, and reactivity than micro NiO, it could release more nickel ions in the liquid solution for intracellular uptake and mobilization, thus exerting stronger bioavailability. 5 As shown in our study, nano NiO indeed induced higher hydroxyproline content, collagen deposition, and gene expression of profibrotic factors in rat lung tissue, when compared to micro NiO at the same dosage.
Pulmonary fibrosis is an irreversible and lethal pathological process resulted in lung architecture distortion, gas exchange disruption, and respiratory failure. 29 Previous studies on the pathogenesis of pulmonary fibrosis have focused on the final phases of fibrosis, especially myofibroblast, which secrete excessive extracellular matrix to deposit in lung interstitium. Among these components, hydroxyproline is a unique gelatin hydrolysate to maintain cell structure and function, also the main component in collagen. 30 Moreover, collagen is the most abundant in extracellular matrix, and collagen types I and III are the main collagen of lung interstitial. An in vitro study reported 300 mmol/L nickel(II) chloride suppressed activity of prolidase, a critical enzyme for collagen degradation, and increased 3H-proline incorporation in human dermal fibroblast for 72 h. 31 An in vivo study discovered that the hydroxyproline was increased in rat lung tissue after a single intratracheal instillation of 3.5 mg/kg cerium oxide nanoparticles. 32 Morimoto et al. 12 reported that inhaled nano NiO for 4 weeks (6 h/d) could upregulate gene expression of collagen type I in rat lung tissue. Our findings showed obvious fibrogenetic effect (Figure 1), and increased hydroxyproline content, COL1A1, and COL3A1 protein expression in rat lung tissue after nano NiO intoxication (Figures 2 and 3). The results verified that nano NiO could induce pulmonary fibrosis in rats.
TGF-β1, a pleiotropic mediator derived from macrophage and epithelial cells, is considered to be critical in pulmonary fibrosis. 33 TGF-β1 was increased in bronchoalveolar lavage fluid and lung tissue after multiwall carbon nanotubes exposure in rats. 14,34 In addition, TGF-β1 has been proved to be associated with the synthesis of hydroxyproline and collagen. In a subacute study, hydroxyproline was increased in human gingival fibroblasts treated with TGF-β1 or CTGF. 35 The gene and protein expression of collagen types I and III was increased in A549 cells treated with TGF-β1 for 72 h. 36 Xie et al. 37 reported that paraquat induced collagen oversynthesis in A549 cells was suppressed by SB431542, a specific inhibitor of TGF-β1. We found the increasing content and gene expression of TGF-β1 in rat lung tissue, implying that TGF-β1 activation was related to pulmonary fibrosis induced by nano NiO.
Excessive TGF-β1 binds to its receptors and leads to cascade reaction with Smad2 and Smad4 to induce pulmonary fibrosis. 38 In bleomycin induced rat pulmonary fibrosis, the protein expression of TGF-β1, phosphorylated Smad2 and Smad4 were upregulated in lung tissue. 39,40 Following the activation of TGF-β1, excessive extracellular matrix would deposit in lung interstitium, thus alveolar septa became widened and inversely narrowed alveolar cavities. 41 TGF-β1 induces the expression of TIMP-1. 42 MMP-9 is a key factor for degrading collagen and can be inhibited by endogenous TIMP-1, thus regulate the synthesis and degradation of extracellular matrix in pulmonary fibrosis. 43,44 Ma et al. 45 reported that MMP-9 and TIMP-1 were increased in rat lung tissue and bronchoalveolar lavage fluid after a single intratracheal instillation of cerium oxide nanoparticles. In addition, CTGF, a key profibrotic factor to enhance collagen synthesis, also promote fibroblasts migration and proliferation. 46 In an acute study treated rats with CTGF, increased hydroxyproline content, and upregulated gene expression of procollagen, fibronectin, TGF-β1, and TIMP-1 were observed in lung tissue. 47 Our study showed the upregulated gene expression of Smad2, Smad4, MMP-9, TIMP-1, and CTGF in rat lung tissue, which might be related to TGF-β1 activation induced by nano NiO. However, the precise and integrated role of Smad2, Smad4, MMP-9, TIMP-1, and CTGF in nano NiO induced pulmonary fibrosis should be investigated further in vivo and in vitro.
Conclusion
In summary, this study has demonstrated that intratracheally instilled nano NiO for 6 weeks could induce pulmonary fibrosis in rats. The potential mechanism of pulmonary fibrosis induced by nano NiO could be related to TGF-β1 activation. However, the precise and integrated mechanisms of pulmonary fibrosis need to be further verified by in vitro studies.
Footnotes
Author Contribution
XH Chang and A Zhu contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundamental Research Funds for the Central Universities (lzujbky-2015-186, lzujbky-2016-204, and lzujbky-2016-205) and the National Natural Science Foundation of China (81402703).
