Abstract
Oxybuprocaine (OBPC) is a widely used topical anesthetic in eye clinic, and prolonged and repeated usage of OBPC might be cytotoxic to the cornea, especially to the outmost corneal epithelium. In this study, we characterized the cytotoxic effect of OBPC on human corneal epithelial (HCEP) cells and investigated its possible cellular and molecular mechanisms using an in vitro model of non-transfected HCEP cells. Our results showed that OBPC at concentrations ranging from 0.025% to 0.4% had a dose- and time-dependent cytotoxicity to HCEP cells. Moreover, OBPC arrested the cells at S phase and induced apoptosis of these cells by inducing plasma membrane permeability, phosphatidylserine externalization, DNA fragmentation, and apoptotic body formation. Furthermore, OBPC could trigger the activation of caspase-2, -3, and -9, downregulate the expression of Bcl-xL, upregulate the expression of Bax along with the cytoplasmic amount of mitochondria-released apoptosis-inducing factor, and disrupt mitochondrial transmembrane potential. Our results suggest that OBPC has a dose- and time-dependent cytotoxicity to HCEP cells by inducing cell cycle arrest and cell apoptosis via a death receptor-mediated mitochondria-dependent proapoptotic pathway, and this novel finding provides new insights into the acute cytotoxicity and its toxic mechanisms of OBPC on HCEP cells.
Keywords
Introduction
Human corneal epithelium (HCEP), a multilayer of HCEP cells located on the outmost surface of the cornea, plays critical functions in maintaining a proper refractive surface for vision and a functional barrier for protecting ocular tissues. 1,2 Since the epithelium is in direct contact with drugs, anesthetics, pathogens, and biomaterials, disruption of its physical barrier function could result in ocular irritation and may be a risk factor for damages from drug exposure and microbial infections. 3 –5
Oxybuprocaine (OBPC), an ester-type local anesthetic, is often used topically to temporarily reduce or take away sensation in various kinds of ophthalmic diagnostics and surgeries. 6 –9 Unfortunately, prolonged and repeated usage of OBPC often causes side effects on the cornea, such as corneal epithelial fragility, keratopathy, and corneal thickness alteration. 10 –12 Cytotoxicity may cause direct damage of the plasma membrane (PM) and may affect cellular metabolism or indirectly cause cytotoxicity by blocking cell proliferation or inducing cell apoptosis. OBPC was found to exert direct cytotoxicity. 13 However, the cytotoxicity of OBPC on HCEP and its underlying toxic mechanisms have not yet been clarified. One of the obstacles in studying intensively the cytotoxicity of OBPC is the lack of an in vitro model that can be used to investigate the possible cytotoxic mechanisms and the prospective therapeutic interventions. 14 Owing to the recently established non-transfected HCEP cell line, 15 this study intended to investigate the cytotoxic effect of OBPC on HCEP cells and its cellular and molecular mechanisms, providing new insights into the cytotoxicity of topical anesthetics on HCEP in ophthalmology and toxicology.
Materials and methods
Chemicals
The reagents for cell culture were from Invitrogen (Carlsbad, California, USA). Quick Tissue/Culture Cells Genomic DNA Extraction kit (cat. no. CW22985), horseradish peroxidase (HRP)-labeled goat anti-rabbit secondary antibody (cat. no. CW0103), and RIPA lysis buffer (cat. no. CW2333) was from ComWin Biotech (Beijing, China). Rabbit antihuman antibodies against the active form of caspase-2 (cat. no. bs-5802 R), caspase-3 (cat. no. bs-0087 R), and caspase-9 (cat. no. bs-8502 R) were from Biosynthesis biotechnology (Beijing, China). FITC-labeled Annexin V Apoptosis Detection kit I (cat. no. BD556547) and propidium iodide (PI) were from BD Biosciences (San Jose, California, USA). Cytoplasmic/Mitochondrial Protein Extraction kit (cat. no. C500051) was from Sangon Biotech (Shanghai, China). Rabbit antihuman immunoglobulin G (IgG) monoclonal antibodies to β-actin (cat. no. ab20272), Bax (cat. no. ab32503), Bcl-XL (cat. no. ab32370), and apoptosis-inducing factor (AIF; cat. no. ab32516) were from Abcam (Cambridge, UK). OBPC (CAS no. 99-43-4, purity ≥ 99%), 5.5′,6,6′-tetrachloro-1,1′,3,3′-tetraethybenzimida (JC-1, cat. no. D8654), and all other reagents were from Sigma-Aldrich (St Louis, Missouri, USA).
In vitro culture and treatment
Passage 80 HCEP cells, from a non-transfected HCEP cell line (utHCEPC01) established previously in our laboratory, 15 were maintained and cultured in 10% heat-inactivated fetal bovine serum (FBS) containing DMEM/F12 medium at 37°C in 25 cm2 culture flasks or plates (all from Corning, New York, USA). For treatment, the culture medium of HCEP cells at logarithmic phase was replaced entirely with the medium containing OBPC at various concentrations ranging from 0.0125% to 0.4%. HCEP cells cultured in the same medium without any OBPC at the same time point were used as blank controls in all experiments.
Light microscopic observations
HCEP cells were inoculated into a 24-well culture plate and cultured in 10% FBS-DMEM/F12 medium at 37°C in a humidified 5% carbon dioxide atmosphere. Logarithmic HCEP cells were treated with OBPC at concentrations ranging from 0.0125% to 0.4% as described earlier. The cell morphology and cytopathic effect (CPE) were monitored continuously with an inverted light microscope (model TS100; Nikon, Tokyo, Japan) every 2–4 h.
Cell viability assay
Cell viability was measured by thiazolyl blue tetrazolium bromide (MTT) according to the method described previously. 16 Briefly, HCEP cells were seeded into a 96-well culture plate at a density of 1 × 104 cells/well and were treated with OBPC at concentrations ranging from 0.0125% to 0.4% as described earlier. At each time point, the culture medium was replaced by 200 μL serum-free DMEM/F12 medium containing 1.1 mM MTT and then incubated for 4 h at 37°C in dark. After 150 µL of dimethyl sulfoxide was added, the 490 nm absorbance of each well was measured using a microplate reader (model Multiskan GO; Thermo Scientific, Waltham, Massachusetts, USA).
PM permeability assay
PM permeability was measured by acridine orange (AO)/ethidium bromide (EB) double staining according to the method described previously. 16 HCEP cells were cultured in 24-well culture plate and treated with OBPC as described earlier. At each time point, the cells were harvested by 0.25% trypsin digestion (1–2 min) and centrifugation (200 × g, 10 min). The cell pellet was resuspended with 100 μL serum-free DMEM/F12 medium and 4 μL of AO/EB solution (100 μg/mL AO: 100 μg/mL EB = 1:1) was added, mixed, and stained for 1 min at room temperature. About one drop of stained cell suspension from each group was placed onto a glass slide and covered with a coverslip. The AO/EB-stained HCEP cells were observed under a fluorescent microscope (model Ti-U; Nikon, Tokyo, Japan). The cells with red or orange nuclei were designated as apoptotic cells while those with green nuclei designated as non-apoptotic cells according to the previous report, 17 and the apoptotic rate of HCEP cells was calculated on condition that at least 400 cells were counted in each group. The apoptotic rate of HCEP cells was calculated according to the formula: “Apoptotic ratio (%) = Apoptotic cells/(Apoptotic cells + non-apoptotic cells) × 100.”
DNA fragmentation assay
DNA fragmentation was examined by agarose gel electrophoresis as described previously. 18 Briefly, HCEP cells in 25 cm2 flasks were treated with OBPC and harvested every 4 h as described earlier. After washed once with ice-cold phosphate-buffered saline (PBS) by centrifugation (200 g, 10 min), the cells were resuspended in 200 μL PBS and their DNA was isolated with a Quick Tissue/Culture Cells Genomic DNA Extraction kit following the manufacturer’s instructions. The DNA preparation from each group was electrophoresed on a 1% agarose gel (200 mA, 260 min) using the D10000 standard molecular-weight marker. After stained with 0.005% EB for 10 min, the gel was observed with an imaging system (model EC3 300; UVP, LLC Upland, California, USA).
Transmission electron microscopy
The ultrastructure of HCEP cells was characterized by transmission electron microscopy (TEM) following the method reported previously. 16 In brief, HCEP cells in 25 cm2 flasks were treated with 0.1% OBPC and harvested every 4 h as described earlier. The cells were fixed with 4% glutaraldehyde in 0.1 M sucrose with 0.2 M sodium cacodylate buffer (pH 7.4) overnight at 4°C. After washing with sodium cacodylate buffer and post-fixing with 1% osmium tetroxide for 1.5 h, the fixed cells were dehydrated and embedded in epoxy resin. Ultrathin sections were stained with 2% uranyl acetate–lead citrate and observed by a TEM (model H700; Hitachi, Tokyo, Japan).
Flow cytometry analysis
Flow cytometry (FCM) analysis was used to analyze the cell cycle progression, phosphatidylserine (PS) externalization, and mitochondrial transmembrane potential (MTP, ΔΨm) as reported previously. 16 Briefly, HCEP cells in 6-well culture plates were treated with 0.1% OBPC and harvested every 4 h as described earlier. The cell pellet was resuspended and washed twice with 1 mL PBS by centrifugation (200 × g, 10 min). For cell cycle assay, 70% alcohol was added and mixed for fixation overnight at 4°C. After the cells were washed twice with PBS as described earlier, 500 μL PI was added and incubated for 30 min in dark. For PS externalization assay, 5 μL FITC-labeled Annexin V and 5 μL PI were added into 500 μL of the cell suspension and incubated for 30 min in dark using FITC Annexin V Apoptosis Detection Kit I following the manufacturer’s instructions. For MTP assay, 5 μL JC-1 was added into 500 μL of the cell suspension and incubated for 20 min in dark. The stained HCEP cells were detected by a flow cytometer (model FACScan; BD Biosciences, San Jose, California, USA).
Caspase activation assay by ELISA
Caspase activation was detected by enzyme-linked immunosorbent assay (ELISA) as described previously. 16 In brief, HCEP cells in 25 cm2 flasks were treated with 0.1% OBPC and harvested every 2 h as described earlier. Whole-cell protein extracts were prepared with a RIPA lysis buffer following the manufacturer’s instructions and coated into the wells of a 96-well microtiter plate. Following three washes with PBS containing 0.05% Tween-20 (PBST), the wells were blocked with 5% nonfat milk, incubated with 100 μL rabbit antihuman caspase-2, -3, and -9 (active form) antibodies (1:500) and conjugated 100 μL HRP-labeled goat anti-rabbit secondary antibody (1:3000), respectively, at 37°C for 2 h. After being washed three times with PBST, a colorimetric reaction was induced by the addition of 100 μL chromogenic substrate (1% tetramethylbenzidine, 100 mM acetate buffer, pH 5.6, and 1 mM urea hydrogen peroxide) for 25 min at room temperature in dark. Color development was stopped by adding 50 μL 0.5 M sulfuric acid, and the 490 nm absorbance of each well was measured using a microplate reader (model Multiskan GO; Thermo Scientific).
Western blotting
Expression of apoptotic proteins was analyzed by Western blotting according to the method described previously with minor modifications. 19 Briefly, HCEP cells in 25 cm2 flasks were treated 8 h with 0.1% OBPC and harvested as described earlier. Whole-cell protein extracts and cytoplasmic protein extracts were prepared using a RIPA lysis buffer and cytoplasmic/mitochondrial protein extraction kit, respectively, following the manufacturer’s instructions. The protein extract (about 30 μg) from the same number of cells in each group was electrophoresed by SDS-PAGE and transferred to polyvinylidene fluoride membranes. The membrane was blocked for 1 h at room temperature using 5% nonfat milk in 0.05% Tween-20 containing PBS (PBST) buffer (10 mM Tris, 150 mM PBS, pH 8.0). Following three washes with PBST buffer, the membrane was incubated with rabbit antihuman IgG monoclonal antibody to β-actin (1:1000), Bax (1:2000), Bcl-xL (1:1000), and AIF (1:1000), respectively, overnight at 4°C. Following three washes with PBST buffer, the membrane was incubated with 100 μL of HRP-conjugated goat anti-rabbit IgG monoclonal antibody (1:3000) for 1 h at room temperature. Following three washes with PBST buffer, the membrane was incubated with DAB solution (3 mg DAB, 10 μL of 30% H2O2, 4.5 mL of 10 mM Tris buffer) for 25 min at room temperature in dark and observed with an EC3 imaging system (UVP, LLC Upland, California, USA).
Statistical analyses
Each experiment was repeated three times independently (n = 3). Data were presented as mean ± standard error of the mean and analyzed for statistical significance with one-way analysis of variance. Differences to blank controls were considered statistically significant when p < 0.05.
Results
OBPC induces morphological abnormality
To evaluate the cytotoxicity of OBPC on HCEP cells, cell morphology and CPE were monitored by light microscopy. Our results showed that HCEP cells treated with 0.025–0.4% of OBPC exhibited typical CPE, retarded growth, cell atrophy, and death in a dose- and time-dependent manner, while no obvious difference was observed between those treated with 0.0125% OBPC and controls (Figure 1).

OBPC-induced abnormal morphological changes of HCEP cells. The concentration and exposure time of OBPC were indicated at the top left of each light microscopic photograph. The dose- and time-dependent growth retardation, morphological shrinkage, and CPE (#) of OBPC-induced HCEP cells were shown. OBPC: oxybuprocaine; HCEP: human corneal epithelial; CPE: cytopathic effect.
OBPC induces cell viability decline
The MTT assay was used to confirm the cytotoxicity of OBPC on HCEP cells, and results revealed that the cell viability of OBPC-treated HCEP cells decreased with time and concentration, while that of HCEP cells treated with 0.0125% OBPC showed no significant difference compared to controls (Figure 2). The cell viability of HCEP cells decreased to 4.91 ± 0.79% (p < 0.01), 39.02 ± 6.37% (p < 0.01), and 50.57 ± 1.19% (p < 0.01) after treated 2 h by OBPC at the concentration of 0.4, 0.2, and 0.1, respectively.

OBPC-induced viability decline of HCEP cells. Data of MTT assay was expressed as the mean ± standard error of the mean (n = 3), and the relative cell viability of each group treated with the indicated concentration and exposure time of OBPC was expressed as percent (%) compared to its corresponding control. *p < 0.05 versus blank control. OBPC: oxybuprocaine; HCEP: human corneal epithelial; CE: cytopathic effect; MTT: thiazolyl blue tetrazolium bromide.
OBPC induces cell cycle arrest
To postulate the possible proliferation inhibiting mechanisms of OBPC on HCEP cells, cell cycle progression was assayed by FCM using PI staining. Our results showed that the number of HCEP cells in G1 phase decreased from 71.20 ± 1.29% of control to a constant number of about 63.40 ± 1.10% (p < 0.01), that in S phase increased gradually with time (p < 0.01), and that in G2/M phase decreased gradually with time (p < 0.05 or 0.01), after treated with 0.1% OBPC for 4, 8, and 12 h, respectively (Table 1).
Effect of OBPC on cell cycle parameters of HCEP cells.a
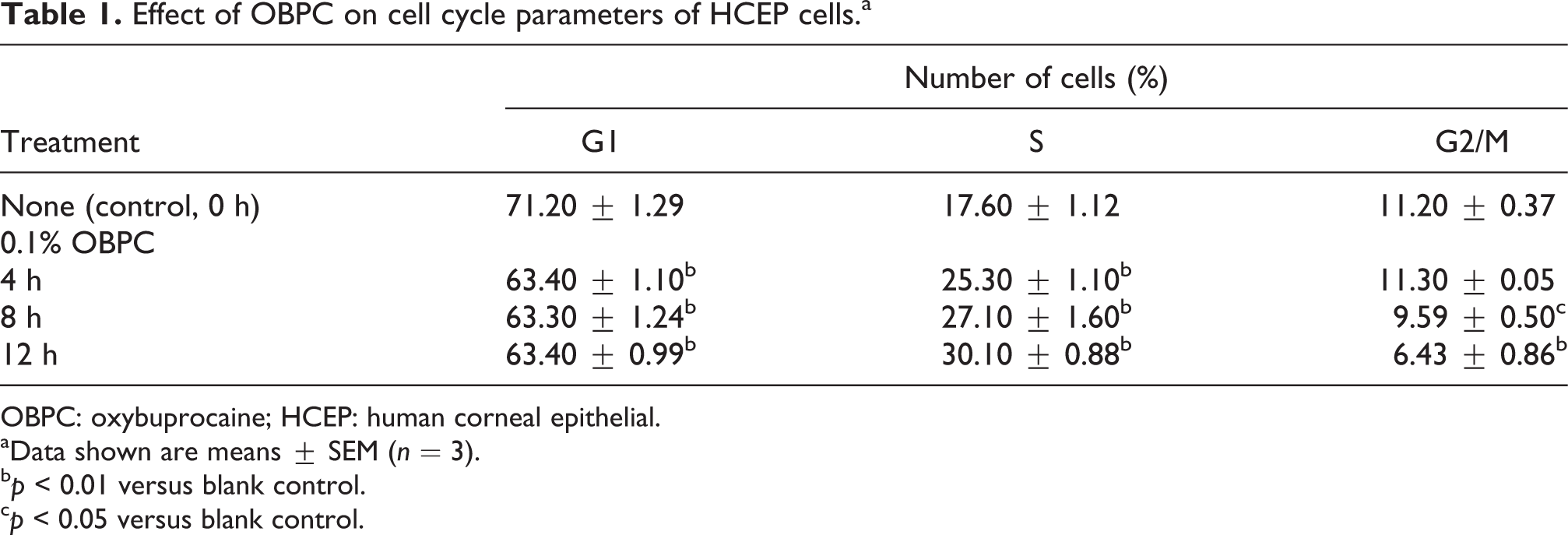
OBPC: oxybuprocaine; HCEP: human corneal epithelial.
aData shown are means ± SEM (n = 3).
b p < 0.01 versus blank control.
c p < 0.05 versus blank control.
OBPC induces PM dysfunction
To verify whether OBPC induces a dysfunctional change of the plasma membrane, the PM permeability and PS externalization of OBPC-treated HCEP cells were examined by AO/EB staining and FCM using Annexin V-FITC/PI staining, respectively. Results of AO/EB staining showed that the membrane permeability of HCEP cells exposed to 0.025–0.4% OBPC was elevated with concentration and time, while that of HCEP cells exposed to 0.0125% OBPC showed no significant difference compared to controls. The apoptotic ratio of OBPC-treated HCEP cells was 100% (p < 0.01) at 0.4% for 1 h and 0.2% for 6 h (Figure 3(a)). Our results of FCM with AnnexinV-FITC/PI staining showed that the number of PS externalized apoptotic HCEP cells treated with OBPC increased from an average of 3.54 ± 1.31% of controls to 45.44 ± 3.13% (p < 0.01), 78.43 ± 5.84% (p < 0.01), and 99.54 ± 4.73% (p < 0.01) at 4, 8, and 12 h, respectively (Figure 3(b)).

OBPC induces PM abnormality of HCEP cells. (a) PM permeability assay of HCEP cells treated with the indicated concentrations and exposure time of OBPC using AO/EB double staining. Data was expressed as the mean ± SEM (n = 3), and the apoptotic ratio of each group was expressed as percent (%) compared to its corresponding control. *p < 0.05 versus control. (b) FCM detection of PS externalization of HCEP cells treated with 0.1% OBPC for the indicated exposure time using FITC-Annexin V/PI staining. Data was expressed as the mean ± SEM (n = 3), and the number of PS-externalized cells in each group was expressed as percent (%) compared to the total cell number. *p < 0.05 versus blank control. PS: phosphatidylserine; OBPC: oxybuprocaine; HCEP: human corneal epithelial; AO/EB: acridine orange/ethidium bromide; PM: plasma membrane; FCM: flow cytometry; PI: propidium iodide; SEM: standard error of the mean.
OBPC induces DNA fragmentation and ultrastructural abnormality
To validate whether OBPC has a proapoptotic effect on HCEP cells, DNA fragmentation and ultrastructural changes of the OBPC-treated cells were also examined by agarose gel electrophoresis and TEM, respectively. Our results showed that the DNA isolated from HCEP cells treated at 4–24 h with 0.1–0.2% OBPC was fragmented into a highly fragmented state, and typical DNA ladders appeared, while no DNA ladder was found in those of controls at 24 h (Figure 4(a)). Results of TEM observations revealed that HCEP cells treated 4 h with 0.1% OBPC exhibited ultrastructural abnormities associated with early stage of apoptosis, such as microvillus lost, structural disorganization, cytoplasmic vacuolation, and mitochondrion swelling, while those treated 8 h with 0.1% OBPC exhibited ultrastructure disorders associated with late stage of apoptosis, such as chromatin condensation, nuclear envelope breakdown, large-scale mitochondrion swelling, and disaggregation into a lot of apoptotic bodies (Figure 4(b)).

OBPC induces DNA fragmentation and abnormal ultrastructural changes of HCEP cells. (a) DNA electrophoresis. Genomic DNA isolated from HCEP cells treated 4–24 h with 0.1–0.2% OBPC was electrophoresed in 1% agarose gel, and the emerged DNA ladders were shown. (b) TEM photographs. The concentration and exposure time of OBPC were indicated the top of each TEM photograph. The microvillus lost, structural disorganization, cytoplasmic vacuolation, mitochondrion swelling, chromatin condensation, and apoptotic body formation were shown. a: apoptotic body; m: mitochondrion; mv: microvillus; N: nucleus; v: vacuole; TEM: transmission electron microscopy; OBPC: oxybuprocaine; HCEP: human corneal epithelial.
OBPC induces caspase activation
To determine whether caspases were involved in OBPC-induced apoptosis and reveal the possible proapoptotic signaling pathway, we measured the caspase activation in OBPC-treated HCEP cells by ELISA using caspase-2, -3, and -9 antibodies against their active forms. Our results showed that caspase-9 was first activated at 2 h, and caspase-2 and caspase-3 activated subsequently at 6 h in 0.1% OBPC-treated HCEP cells (Figure 5). The difference of activations of caspase-2, -3, and -9 between OBPC-treated cells and their corresponding controls was significant at 2–12 h (p < 0.01 or 0.05), while no significant difference was found at 14 h.

OBPC induces caspase activation in HCEP cells. Caspase activation of the cells treated with 0.1% OBPC for the indicated exposure time was detected by ELISA using the active form of caspase-2, -3, and -9 antibody. Data was expressed as the mean ± standard error of the mean (n = 3), and the relative caspase activity of each group was expressed as percent (%) compared to its corresponding control. *p < 0.05 versus blank control. HCEP: human corneal epithelial; OBPC: oxybuprocaine; ELISA: enzyme-linked immunosorbent assay.
OBPC induces apoptosis involving mitochondrion
To postulate whether a mitochondrial signaling pathway was involved in OBPC-induced apoptosis, the expression of Bcl-2 family proteins and cytoplasmic amount of mitochondria-released apoptosis regulating proteins along with MTP (ΔΨm) in OBPC-treated HCEP cells were further examined by Western blotting and FCM, respectively. Results of Western blotting demonstrated that the expression of Bcl-xL was downregulated while that of Bax upregulated, and the cytoplasmic amount of AIF was upregulated significantly after 8 h of 0.1% OBPC treatment, respectively (Figure 6(a) and (b)). Results of FCM using JC-1 staining showed that the number of MTP-disrupted cells (JC-1 positive cells, in green fluorescence) increased from an average of 1.63 ± 0.17% of controls to 16.88 ± 1.21% (p < 0.01) at 4 h, 32.33 ± 0.77% (p < 0.01) at 8 h, and 70.31 ± 1.00% (p < 0.01) at 12 h, respectively, after treated with 0.1% OBPC (Figure 6(c)).
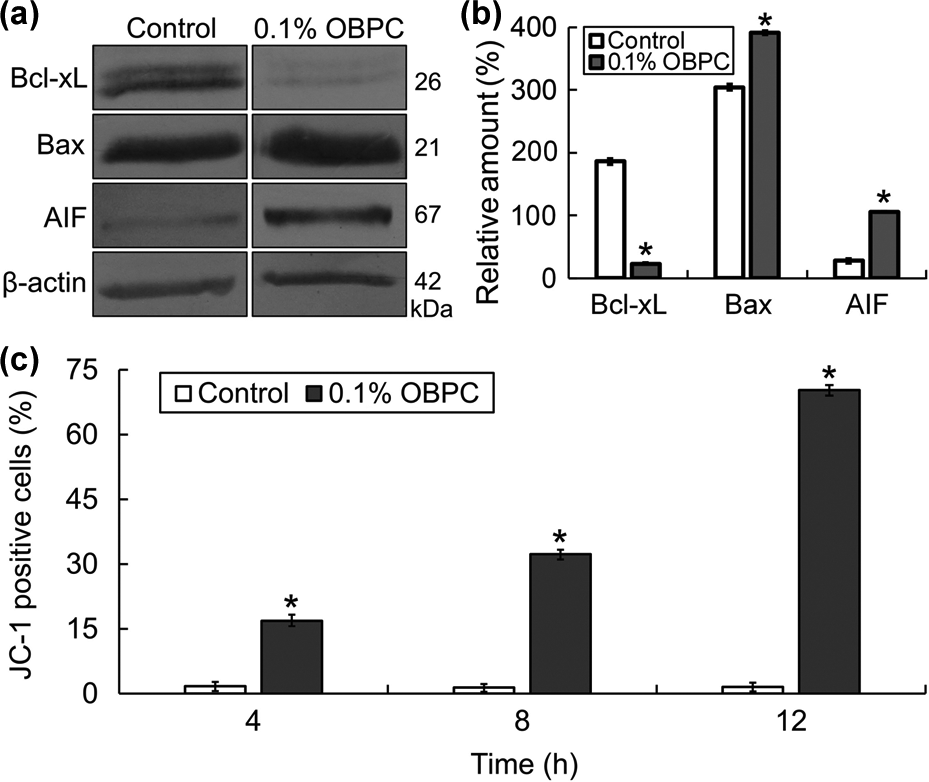
OBPC induces expressional changes of mitochondrion-related apoptotic proteins and disruption of MTP in HCEP cells. (a) Western blotting of mitochondrion-related apoptotic proteins from the cells treated with 0.1% OBPC for 8 h using β-actin as an internal control. AIF, apoptosis inducing factor. (b) Densitometry analyses. The relative level of protein amount was expressed as percentage (mean ± SEM) of protein band density compared to an internal control of β-actin (n = 3). *p < 0.05 versus blank control. (c) FCM detection of MTP of cells treated with 0.1% OBPC for the indicated exposure time using JC-1 staining. Mitochondria with depolarized MTP make JC-1 (monomer) positively stained in green fluorescence. Data is expressed as the mean ± SEM (n = 3), and the number of JC-1 positive cells of each group was expressed as percent (%) compared to its corresponding control. *p < 0.05 versus blank control. MTP: mitochondrial transmembrane potential; OBPC: oxybuprocaine; HCEP: human corneal epithelial; FCM: flow cytometry; SEM: standard error of the mean.
Discussion
Damage to HCEP cells by topical anesthetics might result in corneal edema and even blindness, 12,20 suggesting the significance of cytotoxic studies of topical anesthetics on HCEP cells. 13 Although some clinical evidences suggest a potential toxicity of OBPC to HCEP, no data are available on the cytotoxic effect of OBPC on HCEP cells and its underlying mechanisms. In this study, we characterized the cytotoxic effect of OBPC and its possible cellular and molecular mechanisms using an in vitro model of non-transfected HCEP cells. 15 Here, we took advantage of a series of in vitro assays, which are suitable for evaluation of basic biological aspects relating to cytotoxicity allowing to obtain rapid and reliable results without using laboratory animals. 21
First, we characterized the cytotoxicity of OBPC on HCEP cells based on cell morphology and viability. We found that OBPC above and at a concentration of 0.025% (1/16 of its clinical therapeutic concentration) has a dose- and time-dependent cytotoxicity to HCEP cells by inducing cell atrophy, growth retardation, CPE, and cell viability decline, which is supported by our previous report on lidocaine. 16 Occurrence of cytotoxicity may exhibit a discrepancy between the results of in vitro and in vivo because of microenvironmental differences of HCEP cells. The in vitro cytotoxicity of OBPC observed in these studies is subject to confirmations by in vivo models, it at least provides evidences that OBPC might have cytotoxicity to HCEP cells.
As demonstrated, cell viability decline and growth retardation are useful for the comparison of cytotoxicity that is often related with cell cycle arrest and cell apoptosis triggered by chemotherapeutic agents. 22,23 Our results of OBPC-induced growth retardation, morphological abnormality, and cell viability decline of HCEP cells imply that the cytotoxicity of OBPC might be achieved by inducing cell cycle arrest and cell apoptosis in these cells. To investigate the cytotoxic mechanisms of OBPC, the cell cycle progression was then detected by FCM using PI staining and results showed that OBPC could induce S phase arrest of HCEP cells. Our results of the cell viability decline and S phase arrest coincide with previous reports on OBPC-induced corneal epithelial fragility in vivo 10 and ofloxacin induced cytotoxicity to HCEP cells in vitro. 24
Since cellular atrophy, cell viability decline, and cell cycle arrest are often associated with cell apoptosis induced by chemotherapeutic agents, 16,24 we hypothesized that the cytotoxicity of OBPC to HCEP cells might be also realized by inducing cell apoptosis. To verify the proapoptotic effect of OBPC, we analyzed the abnormal changes of plasma membrane, DNA, and ultrastructure of OBPC-treated HCEP cells because these are the hallmark features of apoptosis. 25 –27 Our results showed that OBPC at concentrations above 0.025% could induce dose- and time-dependent elevation of PM permeability, PS externalization, DNA fragmentation, and apoptotic body formation of HCEP cells. As well known, the formation of apoptotic bodies with PS externalization in PM is a vital feature for macrophages to clear away the apoptotic cells by phagocytosis, which is also a key difference between apoptosis and necrosis. 25,27 Therefore, all these results suggest that OBPC has a proapoptotic effect on HCEP cells, and the cytotoxic effect of OBPC on HCEP cells is most probably achieved by inducing apoptosis in the cells.
As well elucidated, apoptosis is regulated by intricate pathways that instruct a cell to suicide, and the triggering of cell apoptosis requires the cooperation of signal molecules, death receptors, caspases, and the key proteins converged on the membranes of mitochondria. 28 –30 In general, apoptosis is often triggered by two pathways, one is the extrinsic pathway that is activated by the ligation of death receptors 31 and the other is the intrinsic pathway emerges from mitochondria, endoplasmic reticulum, and DNA damages. 32 Both pathways result in the activation of initiator caspases (e.g. caspase-2, -8, -10, and -9) and executioner caspases (e.g. caspase-3, -6, and -7). 29,33 To postulate the possible proapoptotic pathways involved in OBPC-induced cell apoptosis, we characterized caspase activation by ELISA using caspase-2, -3, and -9 antibodies against their active forms. Our results indicated that caspase-9, -2, and -3 were activated successively in HCEP cells after OBPC treatment. Since caspase-2 is an important regulator of tumor necrosis factor receptor 1 (TNFR1)-mediated extrinsic proapoptotic pathway, while caspase-9 is one of the key regulators of the mitochondria-dependent intrinsic proapoptotic pathway, 29 our results suggest that the proapoptotic effect of OBPC on HCEP cells might be triggered by the death receptor TNFR1-mediated mitochondrion-dependent pathway. The involvement of both extrinsic TNFR1-mediated and intrinsic mitochondria-dependent proapoptotic pathways as shown here has also been reported for other anesthetics and chemicals. 24,34,35
Since the expression of mitochondria-dependent proapoptotic proteins is thought to promote apoptosis triggered by chemotherapeutic agents, 29,34,35 we further examined the expression level of Bcl-2 family proteins (Bcl-xL and Bax as a representative of antiapoptotic and proapoptotic Bcl-2 family proteins, respectively) and cytoplasmic release of mitochondria-sequestered apoptosis regulating proteins (AIF as a representative) in HCEP cells in order to verify the involvement of mitochondria in OBPC-induced apoptosis. Our results indicated that OBPC could downregulate the expression of antiapoptotic Bcl-xL, upregulate the expression of proapoptotic Bax, and upregulate the cytoplasmic amount of mitochondria-released AIF in HCEP cells. As well known, antiapoptotic Bcl-xL functions as a gatekeeper of mitochondria to prevent the release of mitochondria-sequestered apoptosis regulating proteins, 36 proapoptotic Bax induces the release of AIF and cytochrome c by interacting with mitochondrial membrane to promote the release of proapoptotic regulators, 37 and AIF is a mitochondrial apoptosis-inducing flavoprotein with a unique capacity to induce caspase-independent peripheral chromatin condensation and large-scale DNA fragmentation by activating poly(ADP ribose) polymerase-1. 38,39 Our results on the upregulation of Bax expression and downregulation of Bcl-xL expression along with the upregulation of cytoplasmic AIF released from mitochondria suggest that HCEP cell apoptosis induced by OBPC is most probably triggered by a mitochondrion-dependent proapoptotic pathway.
Since the disruption of MTP (ΔΨm) is an indispensable prerequisite for triggering mitochondrial release of mitochondria-sequestered apoptosis regulating proteins, such as AIF, cytochrome c, and Smac/Diablo. 28,32,38 To confirm the involvement of a mitochondria-dependent proapoptotic pathway in OBPC-induced apoptosis, we finally investigated the disruption of MTP in OBPC-exposed HCEP cells by FCM with JC-1 staining. Our results of the disruption of MTP induced by OBPC confirmed that a mitochondria-dependent proapoptotic pathway is involved in OBPC-induced apoptosis of HCEP cells. Therefore, our results of MTP disruption, combined with caspase-9 activation and mitochondrial release of AIF, indicate that the proapoptotic effect of OBPC on HCEP cells is triggered and regulated via a mitochondria-dependent proapoptotic pathway. The mitochondrion-dependent proapoptotic pathway has also been reported in the apoptosis triggered by the other topical ophthalmologic drugs. 16,24,40
Conclusions
This study demonstrated that OBPC has significant cytotoxicity to HCEP cells by inducing cell cycle arrest and cell apoptosis, which is triggered by a death receptor-mediated mitochondria-dependent pathway. To our knowledge, this is the first attempt of studying the cell cycle arrest and proapoptotic effect of OBPC on HCEP cells at cellular and molecular levels, and our findings provide new insights into the cytotoxicity of OBPC on HCEP cells and its underlying cytotoxic mechanisms as well. Even though these findings are considered particularly relevant in deciding the optimal topical anesthetic to be applied in the clinic, they do not allow to predict clinical inferences directly without further verifications in vivo. Future investigations will be essential to predict human risks of corneal health upon exposure to OBPC in eye clinic. However, these findings remind clinicians that this topical anesthetic is most probably cytotoxic to HCEP and should be used with great caution in clinical situations.
Footnotes
Authors’ Contributions
The authors Wen-Yi Fan and De-Ping Wang contribute equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National High Technology Research and Development Program (“863” Program) of China (no. 2006AA02A132) and received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
