Abstract
A combination of pentobarbital and phenytoin is used as a veterinary euthanasia drug. Because of its lethal effect, this study described pentobarbital–phenytoin combination veterinary drug human exposures reported to Texas poison centers during 2000–2015. Of 66 exposures, 73% involved female and 27% male patients. The distribution by patient age was 3% 0–5 years, 5% 6–19 years, 91% 20+ years, and 2% unknown. The most common routes were ocular (41%), ingestion (32%), injection (23%), and dermal (18%). The exposure reasons were unintentional (77%) and intentional (23%). The exposure site was the workplace (52%), patient’s own residence (38%), health-care facility (2%), and other/unknown (9%). The management site was managed on site (48%), at/en route to health-care facility (45%), referred to health-care facility (5%), and other (2%). The medical outcomes were no effect (23%), minor effect (30%), moderate effect (8%), major effect (8%), not followed nontoxic (3%), not followed minimal effects (24%), unable to follow potentially toxic (2%), and unrelated (3%). The most common adverse effects were ocular irritation/pain (18%), drowsiness/lethargy (15%), and coma (9%). The most common treatments were dilution/irrigation (70%), intravenous fluids (21%), and oxygen (14%). This study found few pentobarbital–phenytoin combination veterinary drug exposures were reported to Texas poison centers during a 16-year period. Although meant to be administered intravenously, the most common exposure routes were ocular and ingestion. Many of the exposures appeared to be unintentional and occurred at the workplace.
Introduction
Pentobarbital and phenytoin in combination is a veterinary preparation used for animal euthanasia. The preparation is sold under a variety of names such as Euthanasia-III™ (Med-Pharmex, Inc., Pomona, California, USA), Beuthanasia D™ (Merck Animal Health, Madison, New Jersey, USA), and Euthasol™ (Virbac AH, Inc., Fort Worth, Texas, USA). It is administered intravenously or intracardiacally. At lethal doses, pentobarbital produces rapid anesthetic action, unconsciousness, and depression of the respiratory and vasomotor centers; phenytoin causes cardiovascular collapse and/or central nervous system depression. US federal law restricts this drug to use by or on the order of a licensed veterinarian. The products carry warnings to avoid contact of the drug with open wounds or accidental self-inflicted injections and to keep out of reach of children. 1 –3
Due to their use in veterinary practices, adverse human exposures to pentobarbital–phenytoin combination products may occur, either intentionally or unintentionally. A portion of these exposures may lead to severe adverse effects and even death. However, the literature on such exposures is limited primarily to case reports of serious outcomes. A person was found unresponsive in a veterinarian’s office with a bottle of Euthasol nearby. 4 A veterinary clinic employee was found unresponsive with an empty bottle of the drug; exposure was reported as probably oral. 5 The spouse of a veterinarian was found unresponsive after self-administration of Euthasol. 6 In 2013, a veterinarian appeared to have self-injected with either pentobarbital/phenytoin or embutramide/mebezonium iodide/tetracaine in an attempted suicide and subsequently died. 7
US poison centers are telephone consultation services that assist in the management of potentially adverse exposures to various substances. In 2013, 2966 exposures to veterinary drugs without human equivalent were reported to poison centers in the United States. Of the 2769 exposures involving only a single substance, 64 (2.3%) had a moderate or more serious outcome. 7
The intent of this investigation was to describe pentobarbital–phenytoin combination drug exposures reported to a large statewide poison center system.
Methods
This was a retrospective epidemiology study using as a data source the Texas Poison Center Network (TPCN). The TPCN is a network of six poison centers that together service the entire state, a population of over 20 million. All TPCN poison centers use a single electronic database to collect information on all calls in a common manner. The data variables and allowable codes in this database were standardized by the American Association of Poison Control Centers. 8
Cases were all pentobarbital–phenytoin combination exposures reported to the TPCN during 2000–2015. Exposures involving additional substances and those not followed to a final medical outcome were included. The distribution of patients was determined for year or report, urbanization status of caller county, patient age and gender, exposure route, circumstances of (reason for) the exposure, exposure site, presence of additional substances, management site, final medical outcome, and most commonly reported adverse clinical effects and treatments.
For urbanization status, the 254 Texas counties were grouped into those that were rural (n = 172) or urban (n = 82) based on US Office of Management and Budget definitions of metropolitan (urban) and nonmetropolitan (rural). The rate per 1,000,000 population was calculated using the 2000 census as the denominator.
The medical outcome or severity of an exposure is assigned by the poison center staff and is based on the observed or anticipated adverse clinical effects. Medical outcome is classified according to the following criteria: no effect (no symptoms due to exposure), minor effect (some minimally troublesome symptoms), moderate effect (more pronounced, prolonged symptoms), major effect (symptoms that are life-threatening or cause significant disability or disfigurement), and death. A portion of exposures are not followed to a final medical outcome because of resource constraints or the inability to obtain subsequent information on the patient. In these instances, the poison center staff record the expected outcome of the exposure. These expected outcomes are grouped into the following categories: not followed but judged as nontoxic exposure (symptoms not expected), not followed but minimal symptoms possible (no more than minor symptoms possible), and unable to follow but judged as a potentially toxic exposure. Another medical outcome category is unrelated effect where the exposure was probably not responsible for the symptoms. The analysis of medical outcome was performed for each of these individual medical outcomes as well as grouping the outcomes into those known or expected to not be serious (no effect, minor effect, not followed and judged nontoxic, and not followed and judged minimal effects) and those known or expected to be serious (moderate effect, major effect, death, unable to follow, and potentially toxic).
The Texas Department of State Health Services institutional review board considers this analysis exempt from ethical review.
Results
Sixty-six pentobarbital–phenytoin combination drug exposures were reported to the TPCN during 2000–2015. Fourteen (21.5%) of the exposures were reported from rural counties, or 4.9 per 1,000,000 population, and 51 (78.5%) of the exposures were reported from urban counties, or 2.8 per 1,000,000 population. (For one case, the caller county was unknown.)
Forty-eight (72.7%) of the patients were female and 18 (27.3%) were male. The age distribution was 2 (3.0%) 5 years or less, 3 (4.5%) 6–19 years, 60 (90.9%) 20 years or more, and 1 (1.5%) unknown age.
A given exposure might occur by more than one route. The exposure route was ocular alone (n = 22, 33.3%), ingestion alone (n = 16, 24.2%), injection alone (n = 12, 18.2%), dermal alone (n = 6, 9.1%), ocular + dermal (n = 3, 4.5%), ingestion + injection (n = 2, 3.0%), injection + rectal (n = 1, 1.5%), ingestion + dermal (n = 1, 1.5%), ocular + ingestion + dermal (n = 1, 1.5%), ocular + ingestion + dermal + inhalation (n = 1, 1.5%), and unspecified other (n = 1, 1.5%). The most common routes that occurred alone or in combination were ocular (40.9%), ingestion (31.8%), injection (22.7%), and dermal (18.2%). The exposure occurred at the workplace (n = 34, 51.5%), patient’s own residence (n = 25, 37.9%), health-care facility (n = 1, 1.5%), unspecified other site (n = 5, 7.6%), and unknown site (n = 1, 1.5%). The distribution by circumstances of (reason for) the exposure was 51 (77.3%) unintentional (23 (34.8%) unintentional general (unintentional exposures that are not otherwise specifically defined), 21 (31.8%) unintentional occupational, and 7 (10.6%) unintentional misuse) and 15 (22.7%) intentional (11 (16.7%) intentional-suspected attempted suicide, 2 (3.0%) intentional misuse, 1 (1.5%) intentional abuse, and 1 (1.5%) intentional unknown).
No other substances were reported in 56 (84.8%) of the exposures.
The highest proportion of patients was managed on site with only three of the patients referred to a health-care facility by the poison center (Table 1). Table 2 presents the distribution of cases by medical outcome. Fifty-three (82.8%) of the total cases and 49 (90.7%) of the cases without other substances did not result in serious outcomes. No deaths were reported.
Management site of pentobarbital–phenytoin combination exposures reported to the Texas Poison Center Network during 2000–2015.

HCF: health-care facility.
Medical outcome of pentobarbital–phenytoin combination exposures reported to the Texas Poison Center Network during 2000–2015.
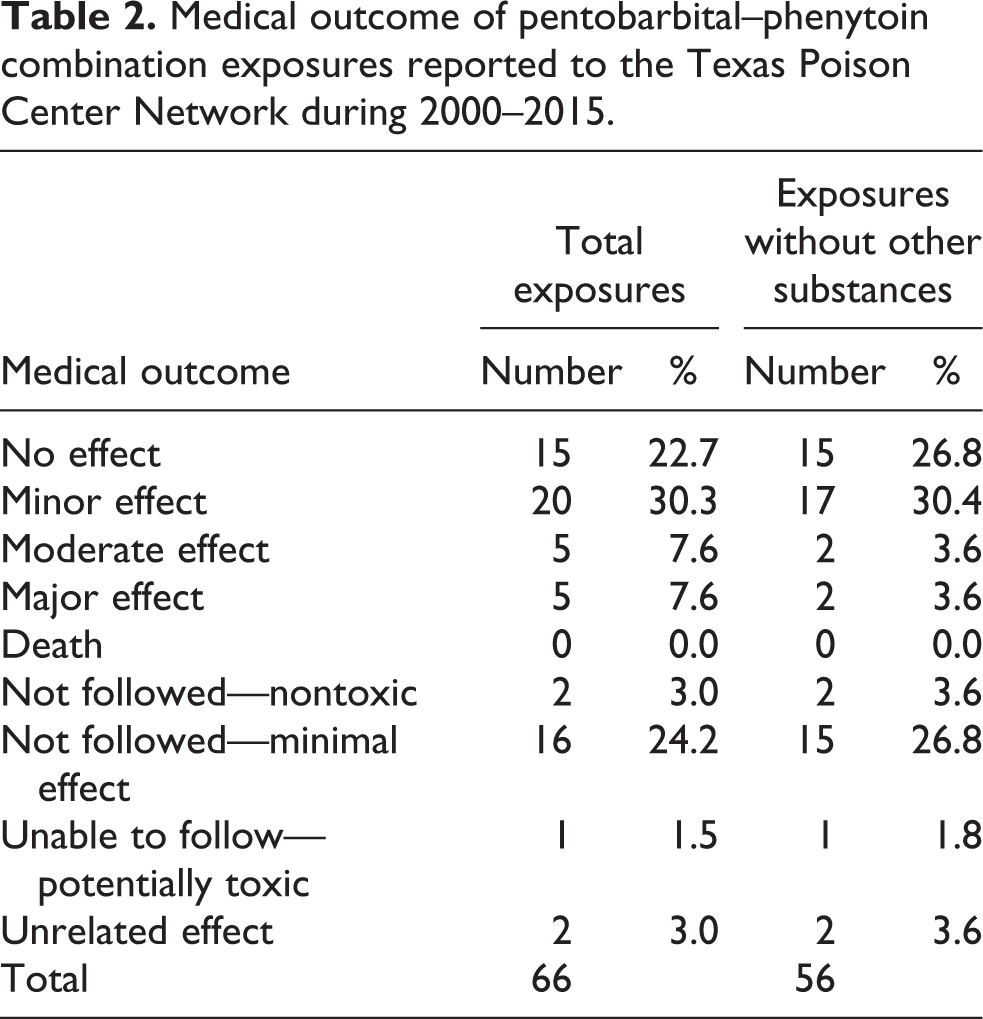
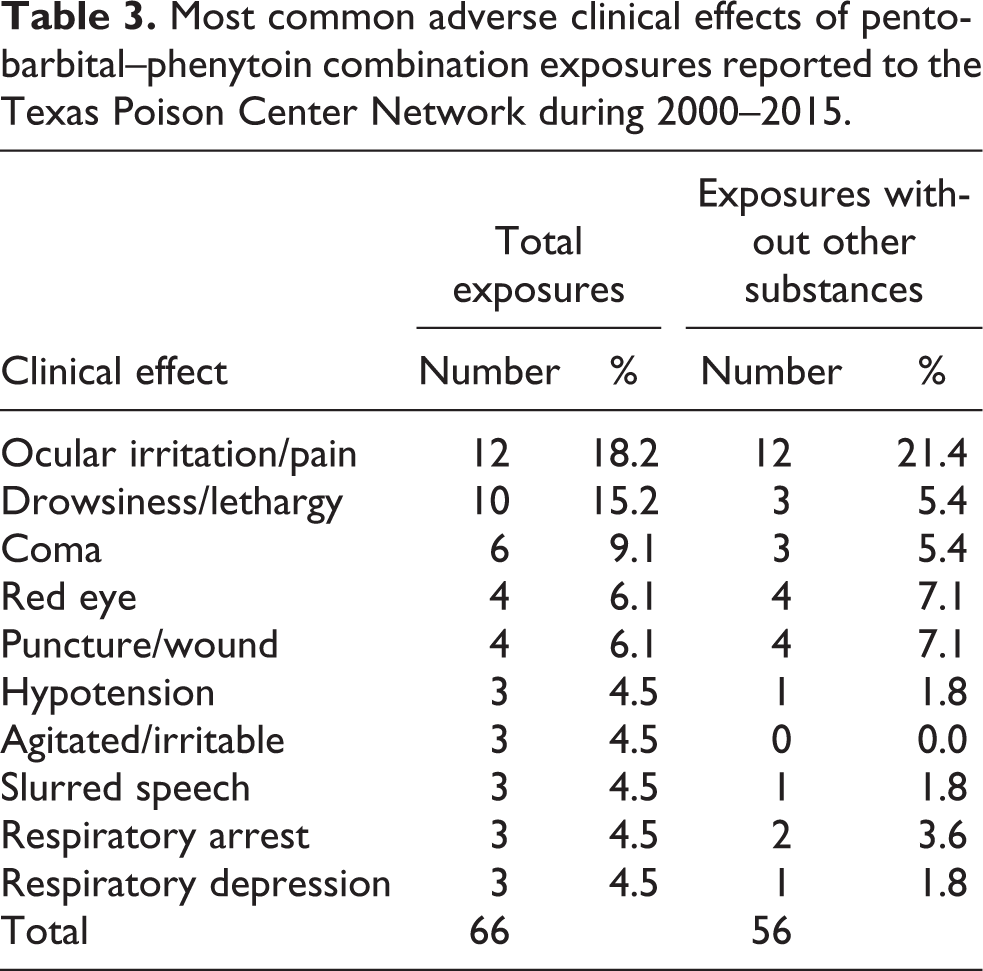
No specific adverse clinical effect was reported in more than a fraction of the cases (Table 3). The most commonly reported clinical effects were ocular irritation/pain, drowsiness/lethargy, and coma. The most frequently reported treatments were decontamination by dilution/irrigation/wash and administration of intravenous fluids and oxygen (Table 4).
Most common adverse clinical effects of pentobarbital–phenytoin combination exposures reported to the Texas Poison Center Network during 2000–2015.
Most common treatments of pentobarbital–phenytoin combination exposures reported to the Texas Poison Center Network during 2000–2015.

IV: intravenous.
Table 5 provides details on the 11 cases with serious outcomes. All except one of the patients were adults aging 23–50 years, and the majority were female. Most of the exposures occurred by ingestion or injection. The majority of exposures were suspected attempted suicides and were managed at a health-care facility.
Patients with serious medical outcomes after pentobarbital–phenytoin combination exposures reported to the Texas Poison Center Network during 2000–2014.a

IV: intravenous.
aSerious medical outcome: moderate effect, major effect, unable to follow—judged as a potentially toxic exposure.
Discussion
This investigation described pentobarbital–phenytoin combination drug exposures reported to a large poison center system during a recent 16-year period. There is little published information on such exposures, primarily consisting of case reports of serious exposures. 4 –7
Only 66 exposures were reported among a population of over 20 million. This might suggest that such exposures are not common. Alternately, it may be that persons are unlikely to report pentobarbital–phenytoin combination drug exposures to poison centers. Unfortunately, data on sales of pentobarbital–phenytoin combination drugs in Texas are not available for comparison.
The preponderance of patients were adults with only two patients 5 years or younger. This might be expected considering that federal law limits pentobarbital–phenytoin combination drugs to use by or on the order of a licensed veterinarian and that there are warnings that the products be kept out of reach of children. 1 –3 Thus, young children might be unlikely to have access to these drugs.
Pentobarbital–phenytoin combination drugs are meant to be injected. Considering that the two most common routes of exposure were ocular and ingestion, this might suggest that in many of the cases the exposure was accidental. In fact, over 77% of the exposures were reported to be unintentional. While over half of the exposures occurred at the workplace, the next most frequent site of the exposure was the patient’s own residence.
The majority of exposures were known or expected to not result in a serious outcome. As a consequence, it might be expected that most patients would not require treatment at a health-care facility. Almost half of the patients were managed outside of a health-care facility with only three patients referred to a health-care facility by the poison center.
Specific adverse clinical effects were reported in a fraction of the total cases; the most common of these clinical effects were ocular or neurological in nature. The presence of specific clinical effects might depend on the particular route of the exposure, and route might need to be taken into account when managing a patient exposed to the pentobarbital–phenytoin combination drug.
Ten (15%) of the 66 cases involved other substances in addition to the pentobarbital–phenytoin combination drug. Moreover, of the 11 cases with serious outcomes, 6 involved other substances. This suggests that the presence of additional substances needs to be taken into account when managing exposures to the pentobarbital–phenytoin combination drug, as these might result in more serious outcomes.
This study is subject to various limitations. Reporting of potentially adverse exposures to pentobarbital–phenytoin combination drugs to the TPCN is voluntary. Those exposures that are reported might not be representative of all such exposures that occur in Texas. Exposure was primarily based on report of the caller and not verified by the clinical analysis. Dose was not reported in the majority of cases and thus could not be analysed. Details of the circumstances of the exposures, for example, whether the patient was a veterinarian, were not provided in the narrative for many of the cases.
In conclusion, few pentobarbital–phenytoin combination veterinary drug exposures were reported to Texas poison centers during a 16-year period. Most patients were adults and female. Although meant to be administered intravenously, the most common exposure routes were ocular and ingestion. Many of the exposures appeared to be unintentional and occurred at the workplace. Almost half were managed outside of a health-care facility. Although almost 20% of the exposures were known or suspected to have serious outcomes, no deaths were reported. This study adds to the limited information on potentially adverse exposures to these drugs and might provide useful for preventing such exposures in the future.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
