Abstract
In the present study, we aimed to assess the potential protective effect of ascorbic acid (AA) against emamectin benzoate (EMB)-induced hepatotoxicity. For this purpose, biochemical, histopathological and analytical investigations were performed. Male Wistar rats were distributed into three groups, that is, a control group, an EMB group given 10 mg EMB/kg body weight (BW) by gavage and an EMB + AA group given 10 mg EMB/kg BW and vitamin C intraperitoneally (200 mg/kg). The duration of the treatment was 28 days and the duration of the study was 42 days. There was a statistically significant increase of all hepatic biomarkers, that is, aspartate aminotransferase, alanine aminotransferase and gamma-glutamyltransferase activities, and glycemia, in EMB-treated group when compared with the control group. Light microscopic observations revealed variable signs of hepatotoxicity in the EMB group, which were represented by alteration of normal hepatic architecture, inflammatory cell infiltration, hepatocellular steatosis and foci of necrosis at 28 and 42 days post-treatment. However, co-treatment with vitamin C reduced EMB-related liver toxicity and diminished the abnormal biochemical and architectural damage. Emamectin B1a and B1b residues were detectable in all plasma samples of treated rats at 14, 21 and 28 days of treatment. The drug liver tissue concentration was significantly lower in EMB + AA group compared with EMB group at 28 and 42 days. In conclusion, the findings of the present study clearly indicate a significant protective action of vitamin C against EMB hepatotoxicity.
Introduction
Vitamins have an essential role in almost all biochemical reactions and they are ideal antioxidants that are able to increase organ protection from oxidative stress. Vitamin C or ascorbic acid (AA) is involved in a number of metabolic processes in the human body, including those that are important for the optimal functioning of the oxygen energy system. In addition, AA is an important free radical scavenger in extracellular fluids, trapping radicals and protecting biomembranes from peroxide damage. 1 –4
Oxidative stress, which is one of the major mechanisms studied in environmental toxicology, is commonly caused by an imbalance between oxidants and antioxidants within the cells. 5 Because of the health problems induced by many environmental pollutants, including pesticides, much effort has been expended in evaluating the relative ameliorative effect of vitamins.
Emamectin benzoate (EMB) is the active principle of Proclaim®, an avermectin (AVM) insecticide. It is a mixture of at least 90% AVM B1a benzoate (MAB1a) and at most 10% MAB1b salts. These two compounds, B1a and B1b, have very similar biological and toxicological properties. 6 EMB is highly effective against a broad spectrum of lepidopteron pests and is being developed for use on major field crops and vegetables, such as soybean, cotton, cabbage and radish. 7 In Algeria, efficacy of EMB has been reported by Gacemi and Guenaoui on Tuta absoluta Meyrick (Lepidoptera), the most important pest of tomato crops in Algeria since 2008. 8
Recently, the study of El-Sheikh and Galal reported that EMB at 2.5 mg/kg body weight (BW) induced damage in liver and spleen. 9 We have recently demonstrated the toxic effect of EMB and abamectin in rats. 10,11 Our results demonstrated a toxic dose-dependent effect of EMB and abamectin with the accumulation of residues B1a and B1b in plasma and organs. Interestingly, after 14 days of non-treatment period, those residues were eliminated from the plasma, but persisted in kidney and liver tissues. 10,11 It is well known that AVMs are substrates for P-glycoprotein (P-gp)-mediated drug efflux. 12,13 Lespine et al. showed that exposure of cells to P-gp substrates leads to an increase in reactive oxygen species (ROS) due to a higher adenosine triphosphate (ATP) use and the need for higher rates of ATP synthesis. 12 Furthermore, El-Sheikh and Galal demonstrated that toxic effects of EMB are associated with an increased production of ROS. 9 Additionally, Schinkel et al. showed that a strong accumulation of ivermectin occurred in the gall bladder cells in P-gp-deficient mice. 14 Based on the above-mentioned observations, we suggest that toxicity of EMB is probably mediated by oxidative stress, which is due to accumulation of residues in biological matrices; we thus attempted to investigate the possible protective effect of AA supplementation in EMB-treated rats. To the best of our knowledge, in vivo study linking the residue distribution of B1a and B1b isomers of EMB with vitamin C co-treatment in male rats has not been launched as yet. In this article, we report our findings about the subacute hepatotoxic effects of EMB exposure, biochemically and histochemically, and, subsequently, the modulatory effects of vitamin C in diminishing liver alteration and concentrations of EMB residues in rat plasma and liver, using ultra high-performance liquid chromatography coupled with tandem mass spectrometry (UHPLC–MS/MS).
Materials and methods
Chemicals
A commercial formulation of EMB (MK244), named Proclaim®, 5% purity of active compound, and produced by Syngenta Company (Switzerland), was used to treat rats. A pure reference standard of EMB (>98.1%, purity, Syngenta Agrochemicals, Greensboro, North Carolina, USA) was used for the analytical procedure based on UHPLC–MS/MS.
Acetonitrile and formic acid (HPLC grade) were purchased from Biosolve Chimie (Valkenswaard, the Netherlands). All biochemical reagents used were obtained from commercial sources (BIOLABO S.A., France). Vitamin C (Pcode: 101132019), the content of which was more than 99%, was purchased from Sigma–Aldrich Chemicals Co. (St. Louis, Missouri, USA). All chemicals used in this study were of analytical grade or higher.
Animals
Healthy adult male Wistar rats weighing 210 ± 10 g were obtained from the CRD Saidal, Medea, Algeria. Animals were housed in cages with controlled temperature (22°C) and 12-h light/12-h dark cycles. The rats were acclimated to the laboratory conditions for 8 days before treatment and had free access to a standard commercial pellet diet (Office National des Aliments de Bétail, Alger, Algeria) and water ad libitum. The study protocol was performed in accordance with the OECD guideline 407 for repeated dose 28-day oral toxicity studies in rodents. 15
Experimental design
Before the acclimatation period, animals were randomly divided into three groups of 7 rats each such that the weight difference within and between groups does not exceed ±20% of the average weight of the total rats as follows: The first group served as a control and received 1 mL of distilled water via oral gavage and 1 mL of saline physiologic solution given daily by intraperitoneal way. The EMB-treated group was given EMB at a dose of 10 mg/kg BW by gavage and 1 mL of saline physiologic solution given daily by intraperitoneal way. The EMB + AA group received EMB by gavage (10 mg/kg BW) and were co-administered with vitamin C intraperitoneally (200 mg/kg/d) 6 h after EMB administration.
All the treatments continued for a period of 28 days. At day 28, 3 rats of each group were allowed to fast overnight and were consecutively anaesthetized with inhaled diethyl ether and then scarified. The remaining rats (4 rats/group) were followed-up for an additional 14-day period to detect the delayed occurrence or persistence of toxic effects and to allow a better evaluation of the vitamin C effects. At the end of experiment (42 days), the remaining rats were then killed by cervical dislocation.
EMB was prepared in distilled water and EMB solutions were administered in the morning (between 8:00 and 9:00 a.m.) to non-fasted rats. Doses of EMB were adjusted according to changes in animal weight. The tested concentration was chosen based on our preliminary experiments, using this experimental model. 11 The selection of vitamin C dose was done according to the study of Fetoui et al. 16
Body and liver weights and tissue sampling
Body weight as well as water and food consumptions were recorded daily throughout the acclimatation, experimental and post-treatment periods. At 28 and 42 days of the experiment, liver tissues were collected from all the rat groups, weighed, cut into pieces and subjected to histopathological and analytical studies. The relative liver weight (liver weight/100 g of BW) of each rat was calculated.
Blood sampling and biochemical investigations
To assess the effect of vitamin C on EMB toxicity, blood samples were collected from the orbital sinus vein at 14, 21, 28 and 42 days. After blood collection, plasma was separated by centrifugation at 4000 r/min for 15 min and two separate tubes were collected from each rat and stored at −20°C until analyses. The following hepatic parameters, alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyltransferase (γ-GT) and glucose, were assessed in plasma using a commercially available spectrophotometric enzymatic kit (Biolabo, France) and analyzed by an auto-analyzer (Hitachi 912) instrument (Roche Diagnostics, Mannheim, Germany).
Histopathological examinations
For the light microscopic examination, the liver was excised from all rats and fixed in 10% neutral formalin buffer, processed through graded alcohols and xylene and embedded in paraffin blocks. Liver sections (2–3 μm thick) were cut and stained with haematoxylin and eosin for histopathological studies.
Plasma and liver EMB concentrations
Emamectin (B1a and B1b isomers) concentration was determined in plasma (at 14, 21, 28 and 42 days) and liver tissues (at 28 and 42 days), using a UHPLC–MS/MS method, according to a previously described procedure. 11 In brief, plasma samples were homogenized in acetonitrile. After centrifugation at 10,800 r/min for 8 min, the supernatant was evaporated under nitrogen. The plasma and tissue residues were reconstituted in 100 μL of the mobile phase ((a) (87%) ammonium formate solution and (b) (13%) acetonitrile and formic acid). After centrifugation at 4000 r/min for 10 min, 15 μL of the supernatant was injected into the UHPLC–MS/MS. For tissue sample analyses, liver tissues were cut into small pieces and placed in a glass tube; 1 mL of acetonitrile were sequentially added to liver sample, then vortexed. The mixing and extraction steps were performed using a vortex mixer for 2 min and ultrasonication for 15 min. After centrifugation at 10,800 r/min for 8 min, the supernatant was evaporated under nitrogen. The organ residues were reconstituted in 100 μL of the mobile phase. After centrifugation at 4000 r/min for 10 min, 15 μL of the supernatant was injected into the UHPLC–MS/MS.
Using these conditions, retention times of emamectin B1a, B1b and methylclonazepam (the internal standard) were 2.76 min, 2.59 min and 2.01 min, respectively. The mass transitions used include AVM B1a 886.9 > 158.1, AVM B1b 873.1 > 158.1 and methylclonazepam 330.1 > 284.1.
Statistical analysis
Statistical analysis was performed using Statistica version 10.0 (Stat Soft Inc., Tulsa, Oklahoma, USA). Data were calculated using one-way analysis of variance followed by the Duncan’s post hoc tests. Data were expressed as the mean ± SD. A p-value < 0.05 was considered as the level of significance.
Results
Body and liver weights, food and water consumption
No deaths occurred in any group throughout the experiment. Some signs of general toxicity were apparent in rats treated with EMB. The body weight, as well as the percentage of body weight gain (BWG), average feed and water intake during the experiment are summarized in Table 1.
Effects of EMB and EMB + AA treatments on body weight (BW), body weight gain (BWG) (%), absolute and relative liver weights, average feed and water intake in rats at the acclimatation (Ac; 8 days), treatment (Tr; 28 days) and post-treatment (P-Tr; 42 days) periods.

EMB: emamectin benzoate; AA: ascorbic acid.
a10 mg of EMB/kg/d.
b10 mg of EMB/kg/d + 200 mg of AA/kg 6 h after the administration of EMB.
cResults are given as a mean ± SD for 7, 3 and 4 rats in each group at days 14, 28 and 42, respectively.
dStatistically different from the control group and the EMB + AA group (p < 0.05).
In the macroscopic examination, no significant difference was observed in gross pathology in the livers of EMB- and EMB + AA-treated rats compared to the livers of the control group. The BWG of rats significantly decreased in the EMB group (7%) when compared to the control (20%) and EMB + AA-treated (19%) groups, at day 28.
The administration of EMB resulted in a significant increase in the absolute liver weight of rats in the EMB group at 28 days and a significant decrease after 14 days without treatment, whereas no change was observed for the EMB + AA group when compared with the control group. Furthermore, EMB + AA-treated rats exhibited no significant alteration in their absolute and relative liver weights in comparison with that of control rats after 28 and 42 days.
Average feed and water intake during the treatment significantly decreased at 28 and 42 days for the EMB group, whereas no significant change was observed for the EMB + AA group, when compared with the control group.
Biochemical results
Table 2 shows results of liver biomarkers (transaminases (AST and ALT), γ-GT and glucose levels) that were measured in plasma samples. All liver biomarkers were significantly increased in the EMB-treated rats as compared to those of control rats at days 14, 28 and 42, strongly suggesting the hepatotoxic effects of EMB in rats. Co-administration of vitamin C to the EMB-treated rats resulted in a partial recovery of the liver biomarkers, although AST and ALT activities, as well as glucose levels, were still significantly higher than those of the control rats at days 28 and 42. It is noteworthy that the γ-GT activity was found to be significantly increased in EMB group compared to that of the control group, whereas it was not altered in the EMB + AA-treated rats at any period of the study.
Effects of EMB and EMB + AA treatments on plasma biochemical parameters in rats.

EMB: emamectin benzoate; AST: aspartate aminotransferase; ALT: alanine aminotransferase; γ-GT: gamma-glutamyltransferase; AA: ascorbic acid.
a10 mg of EMB/kg/d.
b10 mg of EMB/kg/d + 200 mg of AA/kg 6 h after the administration of EMB.
cResults are given as a mean ± SD for 7, 3 and 4 rats in each group at days 14, 28 and 42, respectively.
dStatistically different from the control group (p < 0.05).
Histological results
The liver of control groups revealed an expected normal histological architecture with normal hepatocytes arranged in cords that are separated from each other by sinusoids (Figure 1).
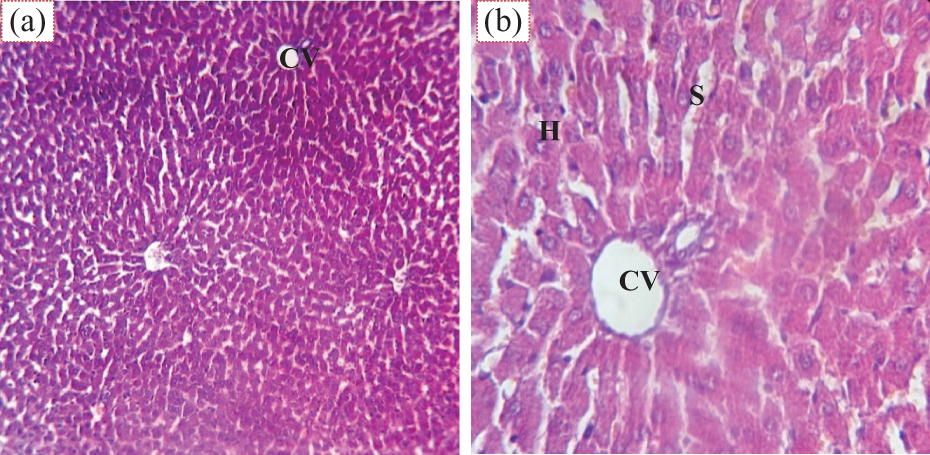
Photographs of a liver section of control rats showing normal architecture of the hepatocytes (H), hepatic cords radiating from the central vein (CV) and separated by the hepatic sinusoids (S) (H&E: (a) ×100 and (b) ×400). H&E: haematoxylin and eosin.
Histological examination of the liver sections of the EMB-treated groups at 28 days revealed considerable dilated hepatic sinusoids, steatosis, and, consequently, a loss of normal hepatocytes architecture. At 42 days, the hepatic sinusoids appeared congested, but less dilated, and some hepatocytes showed microvesicular steatosis and necrotic changes including pyknotic nuclei (Figure 2).
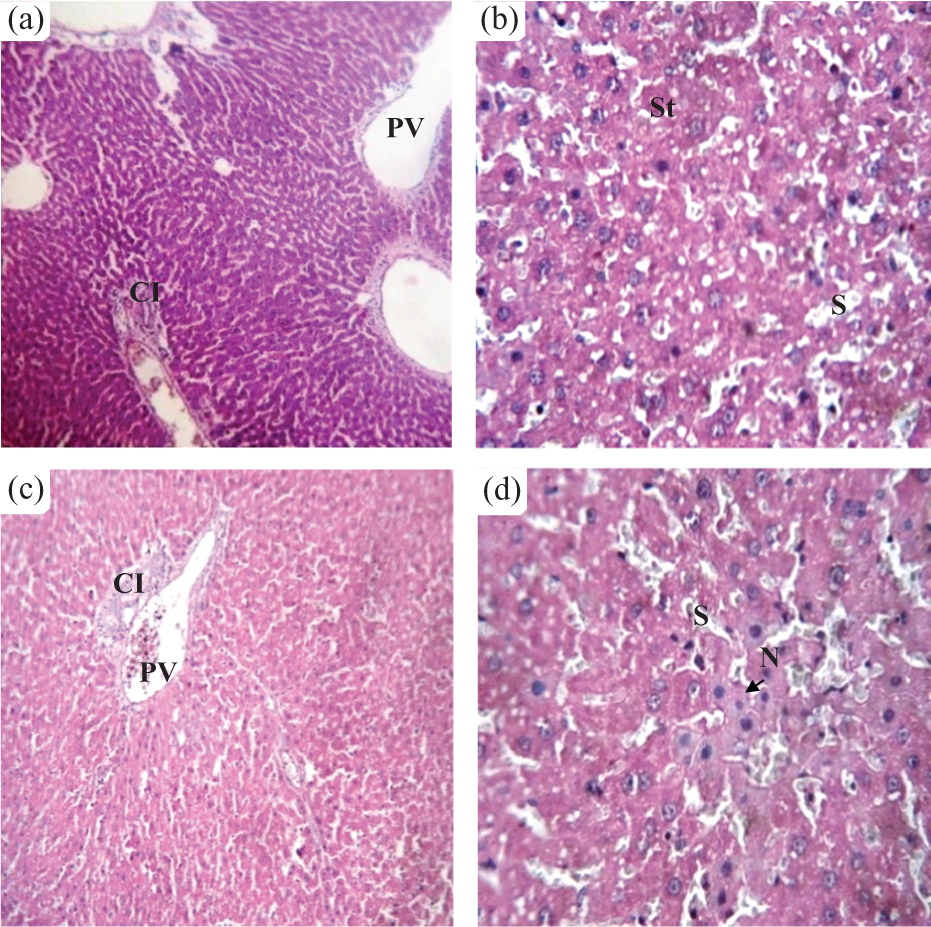
Effects of emamectin benzoate on the structure of rat liver tissues. Photographs of a liver section of EMB-treated group at 28 days (a and b), showing loss of the normal architecture of the hepatocytes and micro-vesicular steatosis (St), dilated portal vein (PV), dilated hepatic sinusoids (S) and cell infiltration (CI). Photographs of a liver section of EMB-treated rats at 42 days (c and d) showing dilated and congested portal vein (PV), dilated hepatic sinusoids (S), cell infiltration (CI) and some hepatocytes showed necrosis (N). H&E: (a and c) ×200 and (b and d) ×400. H&E: haematoxylin and eosin; EMB: emamectin benzoate.
At 28 and 42 days, liver sections of rats co-treated with EMB + AA revealed a clear restoration of normal appearance and structure of hepatocytes, with minimal vacuoles. However, mild mononuclear cell infiltration and vascular congestion were also observed (Figure 3).

Effects of EMB plus vitamin C on the structure of rat liver tissues. Photographs of liver sections of EMB + AA-treated rat at 28 days (a and b) showing restoration of normal appearance and structure of hepatocytes (H), hyperplasia of Kupffer cells (K) and mild vascular congestion (VC). Photographs of liver sections of EMB + AA-treated rat at 42 days (c and d) showing normal structure of hepatocytes (H), central vein (CV) and mild mononuclear cell infiltration (CI). H&E: (a and c) ×200 and (b and d) ×400. H&E: haematoxylin and eosin; EMB: emamectin benzoate.
Plasma EMB concentrations
Emamectin B1a and B1b were detectable in plasma samples of EMB- and EMB + AA-treated rats between days 14 and 28 (Table 3). However, B1a and B1b concentrations were significantly lower in the EMB + AA-treated rats compared to those in the EMB-treated rats. The calculated percentages of residues removed (PRR) in the plasma of EMB + AA rats are also presented in Table 3. At 42 days, there were no more EMB residues detectable in the plasma of all treated rats
Plasma emamectin benzoate (B1a and B1b, ng/mg) concentrations and the PRR at 14, 21, 28 and 42 days after administration of 10 mg/kg/d (EMB group) and co-administered with vitamin C (EMB + AA group) daily for 28 days to male rats.
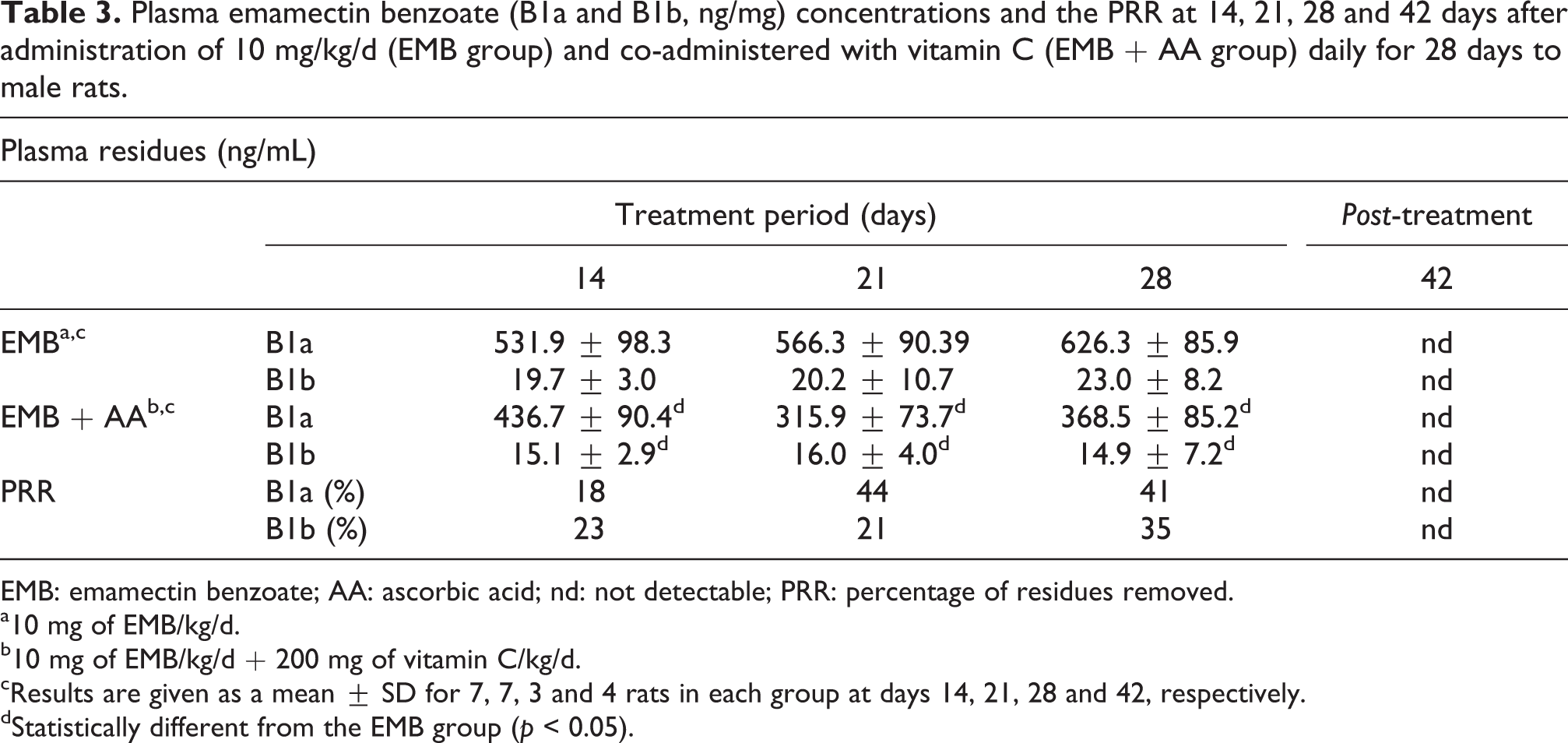
EMB: emamectin benzoate; AA: ascorbic acid; nd: not detectable; PRR: percentage of residues removed.
a10 mg of EMB/kg/d.
b10 mg of EMB/kg/d + 200 mg of vitamin C/kg/d.
cResults are given as a mean ± SD for 7, 7, 3 and 4 rats in each group at days 14, 21, 28 and 42, respectively.
dStatistically different from the EMB group (p < 0.05).
Liver EMB concentrations
EMB residues B1a and B1b were detectable in all liver tissue samples of EMB- and EMB + AA-treated rats at day 28, with the B1a residue concentration systematically higher than that of the B1b residue (Table 4). However, liver samples from EMB + AA-treated rats exhibited significantly lower B1a and B1b residue concentrations compared to those of the EMB-treated rats at day 28, suggesting an increased elimination or a reduced accumulation of EMB residues related to vitamin C co-treatment. The PRR in the liver of EMB + AA rats were 83% and 73% for B1a and B1b, respectively.
Liver emamectin benzoate residue (B1a and B1b, ng/mg) concentrations and the percentage of residues removed (PRR) at 28 and 42 days after administration of 10 mg/kg/d (EMB group) and co-administered with vitamin C (EMB + AA group) daily for28 days to male rats.

EMB: emamectin benzoate; AA: ascorbic acid; nd: not detectable.
a10 mg of EMB/kg/d.
b10 mg of EMB/kg/d + 200 mg of vitamin C/kg/d.
cResults are given as a mean ± SD for 3 and 4 rats in each group at days 28 and 42, respectively.
dStatistically different from the EMB group (p < 0.05).
After 14 days of non-treatment (day 42), the liver residue concentrations were drastically reduced in all EMB-treated rats and there were no more detectable residues in the EMB + AA liver samples.
Discussion
Vitamin C has shown remarkable protective effect against hepatotoxicity induced by certain drugs and chemical agents. 4 In the present study, we used biochemical, histological and analytical methods to investigate the potential hepatotoxic mechanisms induced by EMB in rats and the ability of vitamin C to protect the liver from EMB-related damages. Our result shows that vitamin C reduces the hepatotoxicity induced by EMB probably by reducing the oxidative damage and the lipid peroxidation and/or by preserving the antioxidant defence system. 17
During the experiment, the BWGs were reduced in the EMB-treated rats. Moreover, after the 14 days post-treatment period, the body weight of the EMB-treated rats remains lower than that of the EMB + AA and control rats. This result is consistent with our previous study showing that subchronic exposure to EMB significantly reduced BWG of rats. 11
Based on the results of the present study, lower mean food and water consumptions were noted in the EMB group. This may have been due to the toxic effect of EMB causing decreased intake and absorption of nutrients by the gastrointestinal tract and altered efficiency of food conversion. 18,19
Interestingly, the EMB + AA-treated rats showed no significant difference in body and liver weights, as well as in food and water consumptions, when compared to the control group. These results are in agreement with the findings of other studies which demonstrated that co-treatment with vitamin C improves body weight and food intake in rats, 16,20 rabbits 21 and humans. 22
The significant elevation of the glucose levels, as well as ALT, AST and γ-GT activities, in the plasma of EMB-treated rats is in accordance with our previous results, 10,11 and with the results of Hsu et al. who demonstrated that AVM exposure (abamectin and EMB) has a significant potential to induce damages in rat liver by increasing plasma transaminases and glycaemia. 23 The effects of EMB and its combination with vitamin C on hepatic biomarkers in rats indicate that vitamin C provides only partial hepatoprotection towards EMB toxicity.
In disagreement with our findings, Zhen et al. reported that Fe2+ coupled with vitamin C can enhance glucose metabolism and decrease blood glucose levels in alloxan-induced diabetic mice. 24 The study of Shalan et al. showed that ethanol co-treatment of male rabbits with vitamin C and vitamin E normalized AST and ALT activities. 21 Wang et al. also evaluated the protective effect of AA and thiamine supplementation at different concentrations against lead-induced toxicity in mice. 25 They reported that the administration of vitamin C and thiamine inhibited lead acetate-induced apoptosis in mice liver. However, in our study, we have used only vitamin C as an antioxidant and probably addition of other supplement could have better normalized the biochemical parameters in EMB-treated rats.
These findings were correlated with the histopathological changes observed in the liver of rats. Rats treated with EMB presented extensive damage of parenchyma, with loss of the normal architecture of the hepatocytes and microvesicular steatosis, dilated hepatic sinusoids and presence of hemorrhagic areas and necrosis. However, animals co-treated with vitamin C presented a reduction of necrosis and a restoration of normal appearance and structure of hepatocytes. Therefore, we deduce that vitamin C protects liver against EMB-induced liver damage. By comparison, many studies have demonstrated that vitamin C is able to reverse the histopathological and biochemical changes in liver tissue after xenobiotic intoxication using experimental models. 1,4,16,26
In addition to the biochemical and histopathological investigations, the residues of EMB in both rat plasma and liver samples were monitored by measuring the two isomers of EMB, B1a and B1b, by UHPLC–MS/MS. After EMB exposure, B1a and B1b residues in plasma and liver samples were clearly detectable in EMB- and EMB + AA-treated rats. However, the B1a and B1b concentrations were significantly higher in the EMB-treated group when compared to those in the EMB + AA group, in both biological matrices. The information on the vitamin C effect in metabolism of xenobiotics remains limited. The results of the present study suggest that vitamin C probably binds to EMB isomers directly and prevents their absorption in liver tissue; although the underlying mechanisms remain to be studied, Chui and Lu reported that the main site of AVM biotransformations is the liver, with less biotransformation occurring in fat. 27 Some articles have reported that AVMs induce major oxidant stress in mammals. 28 Therefore, detoxification and removal of free radicals depend on enzymatic and non-enzymatic antioxidant defence molecules in tissues, such as vitamin C.
Conclusion
In conclusion, vitamin C supplementation in EMB-exposed rats ameliorates food and water intake, significantly reduces plasma and liver EMB residue concentrations and partially reduces the hepatotoxicity of EMB.
Footnotes
Acknowledgements
This study is partially achieved in the laboratory of C.R.D Saidal Médea, Algeria, and the Laboratory of Toxicology, CHRU Lille, France. The corresponding author thanks the laboratory for support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
