Abstract
Many microbial and plant-derived metabolites contribute to the production of inflammatory mediators and the expression of pro-inflammatory molecules. Ophiobolin A (OPA) is a fungal secondary metabolite produced by Bipolaris species. The aim of our study was to examine the acute effects of this compound on inflammatory processes.
Male Wistar rats were treated with 5% ethanol, 0.01 mg/kg OPA, 0.1 mg/kg OPA and 1.0 mg/kg OPA per os. After 24 h of the administrations, inflammatory mediators such as interleukin-6 (IL-6), tumour necrosis factor-alpha (TNF-α) and myeloperoxidase (MPO) enzyme as well as heme oxygenase (HO) activity were measured in both plasma and cardiac tissue, along with serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST). We found that OPA caused a significant elevation in the concentrations of IL-6 and TNF-α, increased MPO activity and decreased HO enzyme activity in the plasma. While OPA induces inflammation in the plasma, it did not change the level of inflammatory mediators in the cardiac tissue and the concentrations of serum ALT and AST. Our findings indicate that rapid release of inflammatory mediators by OPA promotes systemic inflammation. However, this acute OPA treatment does not show toxic effects on the cardiac tissue and the concentrations of liver enzymes.
Introduction
Inflammation is a complex set of interactions among soluble factors (cytokines) and cells that can arise in both tissue and plasma in response to infectious, toxic or traumatic injury. 1,2 The pro-inflammatory cytokines, such as tumour necrosis factor-alpha (TNF-α), interleukin-1-beta (IL-1β) and IL-6, are involved in the initiation and amplification of the inflammatory process. TNF-α and IL-1β act in synergy to enhance IL-6 secretion and may significantly contribute to the overall systemic inflammation. 3 Along with the effects of cytokines, myeloperoxidase (MPO) activity, which synthesized and secreted by neutrophils, is determined as an inflammatory biomarker. The plasma or tissue concentrations of MPO are markers of the neutrophil proliferation/infiltration and severity of inflammation. 4
It has been reported that heme oxygenase (HO) plays a central role in oxidative stress and inflammatory processes. The upregulation of the HO enzyme pathway has significant antioxidant and anti-inflammatory effects in both the circulation and tissue injury. HO enzyme system serves a vital metabolic function as the rate-limiting step in the degradation of heme to generate carbon monoxide (CO), iron and biliverdin, which is converted to bilirubin. HO-1 end products generated from heme degradation may modulate inflammation. Cumulative evidence suggests that CO and bilirubin have anti-proliferative and anti-inflammatory effects via downregulate the production of pro-inflammatory cytokines (e.g. IL-6, IL-1β and TNF-α) and upregulate the anti-inflammatory cytokines (e.g. IL-10). 5,6 The strong interaction between HO enzyme and pro-inflammatory cytokines (which play a pivotal role in pathological conditions) can be major factors to promote systemic inflammation.
Natural compounds, including plant and microbial secondary metabolites, can directly influence the production of inflammatory mediators as well as the expression of transcription factors and key pro- or anti-inflammatory molecules. 7,8
Ophiobolins are sesterterpenoid-type secondary metabolites, a group of pentaprenyl terpenoid compounds whose structures are derivable from geranylfarnesyl diphosphate (C 25). They are characterized by a unique structure of a tricyclic 5-8-5 ring system derived from head to tail linkages of five isoprene units. Ophiobolins are produced by some phytopathogenic fungi, mainly the members of the genus Bipolaris (Cochliobolus) 9 and some species of the genera Drechslera (Pyrenophora), Aspergillus (Emericella, Neosartorya) and others. 10 Ophiobolin A (OPA) was the first member of the group to be isolated and characterized in the mid-1960s. 9 Ophiobolins are known to be effective phytotoxins, and a large number of studies have described the action of OPA in plants. 11,12 Their effect is less known in mammals, though there are some studies also in this field, for example, the biological effects of OPA on human cancer cells 13 –15 and boar spermatozoa 15 were described.
In our present study, we aimed to characterize OPA-mediated acute systemic changes on the inflammatory process. Thus, we tested the effect of different doses (0.01, 0.1 and 1.0 mg/kg) of OPA on inflammatory mediators (IL-6, TNF-α, MPO activity and HO activity) in rat plasma. To clarify the potential cytotoxic effects of OPA, we tested it on both cardiac tissue and liver. The alanine aminotransferase (ALT) and aspartate aminotransferase (AST) markers were measured to analyse the influence of OPA on hepatotoxicity.
Methods
Animals and experimental design
All manipulations were performed in accordance with the standards of the European Community guidelines on the care and use of laboratory animals and had been approved by the Institutional Ethics Committee at the University of Szeged.
All the animals were housed in a temperature-controlled facility (23°C) maintained on a 12/12 h of light/dark cycle with food and water provided ad libitum.
Male Wistar rats (weight: 250–350 g; Toxi-Coop Zrt., Dunakeszi, Hungary) were randomly divided into four groups: 5% ethanol vehicle control (n = 18), 0.01 mg/kg OPA (n = 18), 0.1 mg/kg OPA (n = 18) and 1.0 mg/kg OPA (n = 18).
Twenty-four hours after vehicle (once, 1.0 mL of 5% ethanol, p.o.) or OPA (once, 0.01 mg/kg, 0.1 mg/kg or 1.0 mg/kg in 1.0 mL of 5% ethanol, per os [p.o.]) treatment, the animals were killed and blood sample as well as the cardiac left ventricle (LV) was collected for analyses.
Measurement of plasma and cardiac LV IL-6 and TNF-α concentrations
Plasma samples were centrifuged at 2000 × gmax for 20 min at 4°C. The cardiac LV samples were homogenized (Ultra-Turrax T25, IKA-Labortechnik, 2 × 10 s on ice) in 2 mL modified Greenburger buffer (300 mM sodium chloride, 15 mM TRIS, 2 mM magnesium chloride (MgCl2), 2 mM Triton X-100, 0.2 mM phenylmethanesulfonyl fluoride, 100 ng/mL leupeptin, 100 ng/mL aprotinin, 10 mg/L trypsin inhibitor and 100 ng/mL pepstatin-A). Tissue homogenates were sonicated for 10 s and centrifuged at 15000 × g for 15 min. The plasma and cardiac IL-6 and TNF-α levels were determined by means of quantitative enzyme-linked immunosorbent assays (ELISA) according to the manufacturer’s instructions (Quantikine rat IL-6 and TNF-alpha Elisa kit; R&D Systems, Minneapolis, Minnesota, USA). Optical density was measured at 450 nm (Benchmark Microplate Reader; Bio-Rad, Budapest, Hungary). Plasma IL-6 and TNF-α were expressed as pictogram per millilitre plasma and cardiac values were defined as pictogram per milligram protein.
Plasma and cardiac LV MPO activity
Plasma samples were centrifuged at 2000 × gmax for 20 min at 4°C. The cardiac LV tissues were homogenized twice for 30–30 s (Ultra Turrax T25, 13 500 r/min) in ice-cold phosphate buffer (50 mM, pH 6.0), freeze-thawed three times and then centrifuged (15,000 × g for 20 min at 4°C). A 12-μL aliquot of the plasma or cardiac supernatant was mixed with 280 μL of phosphate buffer (50 mM, pH 6) containing 0.167 mg/mL of O-dianisidine dihydrochloride, and the reaction was started with 10 μL of 0.03% hydrogen peroxide and assayed spectrophotometrically at 490 nm (Benchmark Microplate Reader; Bio-Rad) after 90 s of shaking. Plasma MPO activity was expressed as milliunit per millilitre plasma and cardiac values were expressed as milliunit per milligram protein.
Plasma and cardiac LV HO activity
Plasma samples were centrifuged at 2000 × g for 20 min at 4°C. The cardiac LV tissues were homogenized (Ultra Turrax T25; 13 500 r/min; 2 × 30 s) in ice-cold 10 mM N-2-hydroxyethylpiperazine-2-ethanesulfonic acid, 32 mM sucrose, 1 mM dithiotreitol, 0.1 mM ethylene diaminetetraacetic acid disodium salt dihydrate, 10 μg/mL trypsin inhibitor, 10 μg/mL leupeptin and 2 μg/mL aprotinin, at pH 7.4. The cardiac supernatant was collected by centrifugation at 15,000 × g for 20 min at 4°C. The reaction mixture contained the following components in a final volume of 1.5 mL: 2 mM glucose-6-phosphate, 0.14 U/mL glucose-6-phosphate dehydrogenase, 15 μM hemin, 120 μg/mL rat liver cytosol (as a source of biliverdin reductase), 2 mM MgCl2 × 6 H2O, 100 mM KH2PO4 and 150 μL of plasma or cardiac LV supernatant. To start the reaction, 100 μL reduced form of nicotinamide adenine dinucleotide phosphate (150 μM) was added to the samples and they were then incubated in the dark at 37°C for 60 min. The reaction was stopped by placing the samples on ice. The bilirubin formed was calculated from the difference between the optical densities obtained at 464 and 530 nm. Bilirubin solution was used as standard (58.47 μg/mL; 10 μM). One unit of HO activity was defined as the amount of bilirubin (nM) produced per hour per millilitre plasma or the amount of bilirubin (nM) produced hour per milligram protein.
Determination of serum ALT and AST
Serum samples were centrifuged in 2000 × g for 20 min at 4°C. Serum levels of ALT and AST were determined by ELISA according to the manufacturer’s instructions (SunRed Biotechnology, Shanghai, China) and were expressed as pictogram per millilitre serum.
Determination of protein concentration
Using a commercial protein assay kit (Bio-Rad), aliquots (20 μL) of the diluted samples (15× or 25× with distilled water) were mixed with 980 μL of distilled water with 200 μL Bradford reagent added to each sample. After mixing and following 10 min incubation, the samples were assayed spectrophotometrically at 595 nm. Protein level was expressed as milligram protein per millilitre.
OPA purification
OPA was purified by a multistep high-performance liquid chromatography (HPLC) purification process after an ethyl acetate extraction of the culture supernatant of Bipolaris oryzae SZMC 13003 strain according to the description of Bencsik et al. (http://www.mdpi.com/2072-6651/6/9/2857). Briefly, the concentrated organic phase was first loaded onto an ECO 15/450V3 K glass column (YMC, Germany) filled with Kieselgel 60 (Merck, Hungary) using ethyl acetate/hexane (5:5, V/V, 2 mL/min) as mobile phase, and the OPA contained fractions were pooled and injected onto a reverse-phase Serva ODS (10 × 450 mm, 5 µm) HPLC column with water/acetonitrile (3:7, V/V, 3 mL/min) eluent system to achieve OPA with the purity of up to 96%. During the purification procedure, the identification, quantitation and purity of OPA were tested by analytical HPLC measurements at a wavelength of 230 nm.
Statistical analysis
All data are presented as means ± standard error of mean. Statistical difference between two groups was analysed with the two-tailed Student’s t-test. p Values less than 0.05 were considered significant.
Results
Plasma IL-6 and TNF-α concentrations
The plasma level of IL-6 was increased after treatment with OPA. The administration of 1.0 mg/kg of OPA led to a significant increase compared to the vehicle control group (55.8 ± 5.97 vs. 77.9 ± 5.51 pg/mL plasma, *p < 0.05). Data are presented in Figure 1.
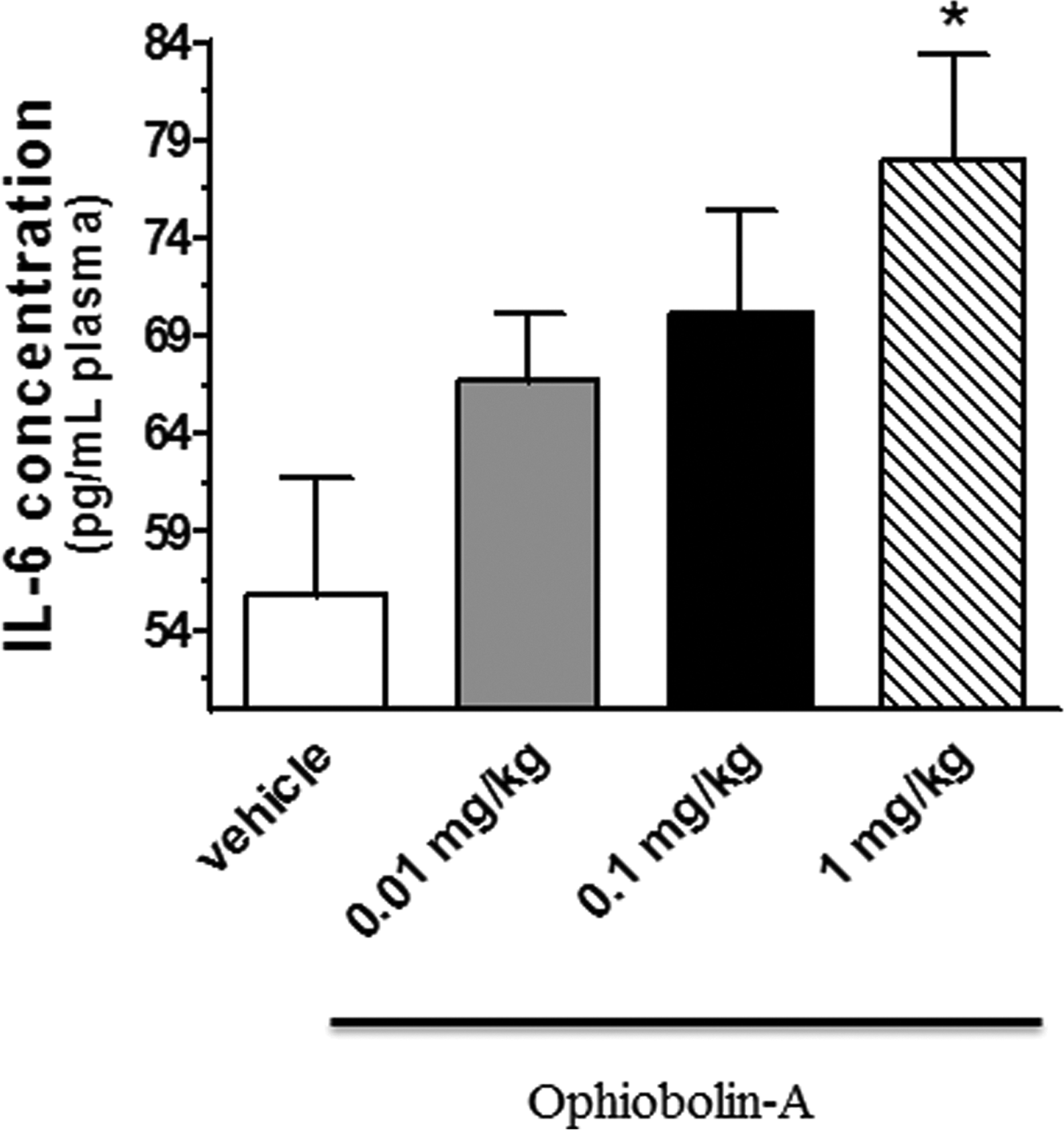
The concentrations of plasma IL-6 (expressed as pg/mL plasma) after 24 h of treatment with 0.01, 0.1 and 1 mg/kg OPA. Data are means ± SEM, n = 5. Statistical significance: *p < 0.05 relative to the vehicle control group. OPA: ophiobolin A; IL-6: interleukin-6; SEM: standard error of mean.
Similarly to the IL-6 values, the TNF-α level reached the highest concentration at 1 mg/kg OPA and showed a significant difference compared to the vehicle control (5.59 ± 0.55 vs. 7.41 ± 0.57 pg/mL plasma, *p < 0.05) and to 0.1 mg/kg OPA-treated (5.82 ± 0.26 pg/mL plasma, p+ < 0.05) groups. Data are shown in Figure 2.
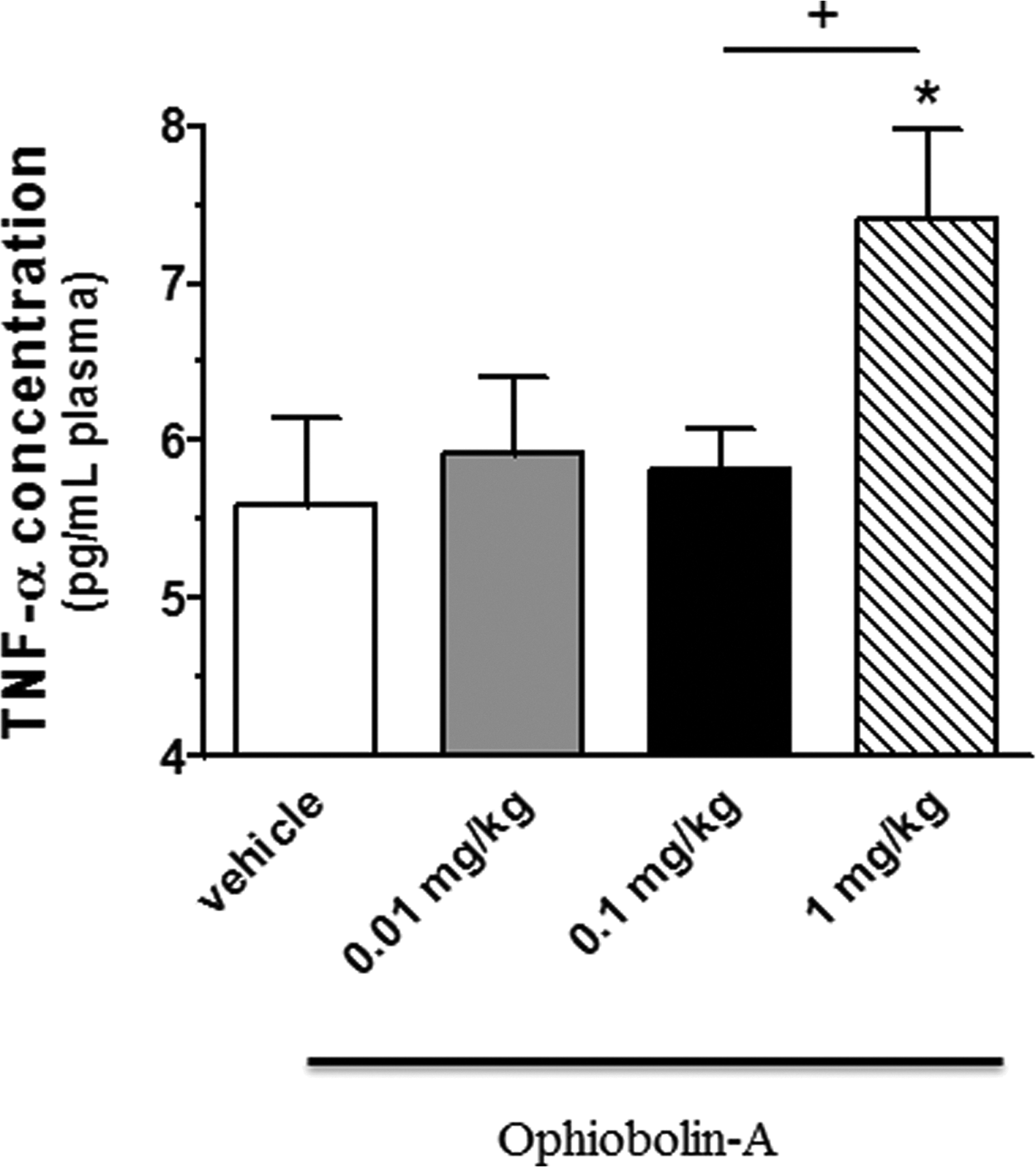
The plasma levels of TNF-α (expressed as pg/mL plasma) in vehicle control and OPA-treated groups. Data are means ± SEM, n = 6–9. Statistical significance: *p < 0.05 relative to the vehicle control group; +p < 0.05 a significant difference between animals treated with 0.1 and 1 mg/kg OPA. TNF-α: tumour necrosis factor-alpha; OPA: ophiobolin A; SEM: standard error of mean.
Plasma MPO and HO enzyme activity
The increasing doses of OPA caused a dose-dependent elevation in plasma MPO activity in each group compared to the vehicle control animals (vehicle control: 45.3 ± 2.04 mU/mL plasma, *p < 0.05; 0.01 mg/kg OPA: 53.3 ± 2.86 mU/mL plasma, *p < 0.05; 0.1 mg/kg OPA: 56.9 ± 2.22 mU/mL plasma, **p < 0.01 and 1.0 mg/kg OPA: 59.2 ± 2.5 mU/mL plasma, **p < 0.01). Data are shown in Figure 3.

Changes in plasma MPO activity (expressed as mU/mL plasma) after 24 h of OPA treatment. Data are means ± SEM, n = 7–9. Statistical significance: *p < 0.05 and **p < 0.01 relative to the vehicle control group. MOP: myeloperoxidase; OPA: ophiobolin A; SEM: standard error of mean.
After 24 h of treatment with 1.0 mg/kg OPA, we observed a significant decrease in HO enzyme activity compared to the vehicle control group (74.1 ± 9.03 vs. 48.3 ± 1.77 nM bilirubin/h/mL plasma, *p < 0.05). In addition, we also observed significant differences between 1.0 mg/kg and lower dose OPA-treated groups (0.1 mg/kg OPA: 55.8 ± 1.42 nM bilirubin/h/mL plasma, +p < 0.05 and 0.01 mg/kg OPA: 63.4 nM bilirubin/h/mL plasma, aap < 0.01). Data are shown in Figure 4.

Effects of OPA treatment on plasma level of HO activity (expressed as nM bilirubin/h/mL plasma) after 24 h. Data are means ± SEM, n = 6–7. Statistical significance: *p < 0.05 relative to the vehicle control group; +p < 0.05 a significant difference between animals treated with 0.1 and 1.0 mg/kg OPA; aap < 0.01 a significant difference between animals treated with 0.01 and 1.0 mg/kg OPA. OPA: ophiobolin A; HO: heme oxygenase; SEM: standard error of mean.
Cardiac LV IL-6 and TNF-α concentrations
As shown in Figures 5 and 6, OPA treatment did not cause significant changes in IL-6 and TNF-α concentrations in cardiac LV.

The concentrations of IL-6 (expressed as pg/mg protein) in the cardiac LV after 24 h of treatment with 0.01, 0.1 and 1.0 mg/kg OPA. Data are means ± SEM, n = 6–8. LV: left ventricle; OPA: ophiobolin A; IL-6: interleukin-6; SEM: standard error of mean.

The levels of TNF-α (expressed as pg/mg protein) in the cardiac LV of vehicle control and OPA-treated animals. Data are means ± SEM, n = 6–8. OPA: ophiobolin A; TNF-α: tumour necrosis factor-alpha; LV: left ventricle; SEM: standard error of mean.
Cardiac LV MPO and HO activity
The results show that administration of OPA did not produce changes in the activity of MPO and HO enzymes in cardiac LV. Data are shown in Figures 7 and 8.

MPO activity (expressed as mU/mg protein) in the cardiac LV of vehicle control and OPA-treated animals. Data are means ± SEM, n = 7–8. OPA: ophiobolin A; LV: left ventricle; MPO: myeloperoxidase; SEM: standard error of mean.

HO activity (expressed as nM bilirubin/h/mg protein) in the cardiac LV of vehicle control and OPA-treated animals. Data are means ± SEM, n = 7. OPA: ophiobolin A; LV: left ventricle; HO: heme oxygenase; SEM: standard error of mean.
Serum ALT and AST concentrations
No differences in serum ALT and AST concentrations in response to increasing doses of OPA were observed between vehicle control and OPA-treated animals. Data are shown in Figures 9 and 10.

The concentrations of serum ALT (expressed as mU/mL serum) in vehicle control and OPA-treated groups. Data are means ± SEM, n = 11–12. ALT: alanine aminotransferase; OPA: ophiobolin A; SEM: standard error of mean.
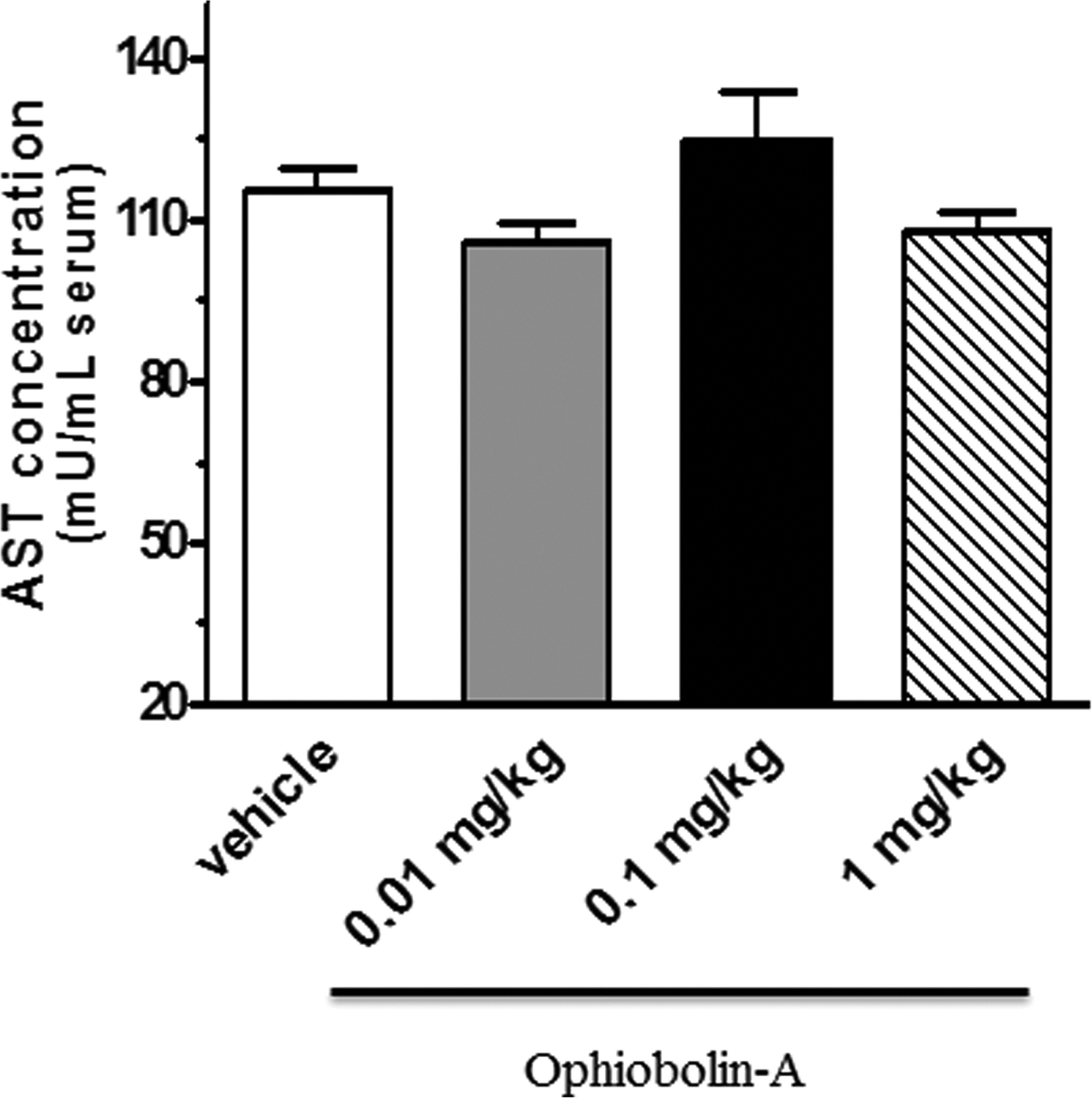
The levels of serum AST (expressed as mU/mL serum) in vehicle control and OPA-treated groups. Data are means ± SEM, n = 5–12. AST: aspartate aminotransferase; OPA: ophiobolin A; SEM: standard error of mean.
Discussion
Recently, much interest has been generated for a wide range of compounds from microbial sources with reports demonstrating their role in the modulation of inflammatory responses. Among these, several microbial metabolites show potent anti-inflammatory action; 16 some derivatives contribute to chronic inflammation and inflammatory diseases.
The aim of our study was to clarify the effects of OPA, a phytotoxic sesterterpenoid of fungal origin, on the modulation of pro-inflammatory cytokines such as IL-6 and TNF-α. Furthermore, its effect on the inflammatory biomarker MPO activity as well as on the activation of HO was also investigated.
In the current investigation, OPA was shown to promote the inflammatory processes. We demonstrated that administrations of 1.0 mg/kg of OPA led to significant increases in the plasma levels of IL-6. Similarly to the IL-6, the TNF-α level reached the highest concentration at 1 mg/kg OPA. All these inflammatory cytokines play a critical role in controlling many inflammatory responses. 17 In a previous study, Nagashima et al. observed elevation of serum IL-6 level in 24-h rubratoxin B-treated mouse model. 18 Dugyala et al. found that TNF-α plays a role in the mechanism of fumonisin B toxicity. 19 Gene expression of inflammatory cytokine leads to another stage of the inflammatory cascade and facilitates the recruitment of effector cells such as monocytes and neutrophils to the site of disturbance. Neutrophils create a cytotoxic environment by releasing noxious chemicals. MPO is a hemoprotein that is stored in azurophilic granules of polymorphonuclear neutrophils and macrophages. MPO catalyses the conversion of chloride and hydrogen peroxide to hypochlorite and is secreted during inflammatory condition. 20 The elevated MPO concentrations of both plasma and tissue have been used as a marker of polymorphonuclear leukocytosis in sepsis and inflammation. 21 –23 Our results show that a single treatment with increasing doses of OPA caused a dose-dependent increase in the plasma. The administration of 1 mg/kg OPA induced 75% increase in the MPO activity. MPO is linked to both inflammation and oxidative stress by catalysing the formation of oxidizing agents.
It has been reported that HO plays a central role in regulating the levels of intracellular heme by catalysing the oxidative degradation of heme and by generating CO, biliverdin and bilirubin. 24 Three isoforms of HO have been described: an inducible isoform, HO-1 and two constitutively expressed isoforms, HO-2 and HO-3. In animal and human studies, HO-1 may affect many pathways and cytokines. During physiological processes, HO activity plays a role in the inhibition of apoptosis and oxidative processes, with significant reductions in inflammatory events including production of inflammatory cytokines. HO-1 and its products such as CO, biliverdin/ bilirubin and free iron may modulate inflammation. 24 CO can downregulate the production of pro-inflammatory cytokines (e.g. IL-1β, IL-6 and TNF-α). These effects were attributed to alterations of mitogen-activated protein kinase (MAPK) activities, including p38 MAPK and c-Jun kinase. 25,26 Furthermore, HO-1/CO activation downregulates the inflammatory response by blocking the release of NO from inducible nitrogen monoxide synthase and expression of the granulocyte macrophage colony-stimulating factor from macrophages and smooth muscle cells. 27 Jung et al. reported that CO can modulate the activation of the NLRP3 inflammasome, which regulates the production of pro-inflammatory cytokines. 28 Devey et al. demonstrated that HO-1 inhibits the production of TNF-α and IL-6 in the liver. 29 In another model, Kapturczak et al. showed that HO-1 deficiency leads to increased production of pro-inflammatory cytokines. 30 Our data indicate that treatment with the fungal phytotoxin, OPA, increased the concentrations of IL-6, TNF-α and MPO inflammatory mediators in blood plasma. The plasma HO activity showed a significant reduction 24 h after administrations of OPA. In agreement with previous observations, our results show that systemic inflammation suppresses the level of HO.
Elevated circulating concentrations of pro-inflammatory cytokines and adhesion molecules suggest that inflammatory processes are occurring systemically and play a role in the development of inflammatory disorders, including allergic reactions, arthritis, cancer, atherosclerosis and ischemic heart disease. 31,32 Although, many studies have detected a positive correlation between inflammation and cardiovascular diseases in different models, we did not observe inflammation-related changes in cardiac tissue after 24 h of acute OPA administrations. Animal studies on TNF-α and development of atherosclerosis have produced mixed results. Inhibition of TNF-α in apoE-deficient mice resulted in significant decrease of atheromatous lesions 33 in wild-type animals, it produced no improvements. 34 In another inflammation-related disease model, Bury et al. revealed that this fungal metabolite, OPA, displays in vivo antitumor activity in mammalian. 35
Since we obtained that OPA treatment led to systemic inflammatory changes measured in plasma, our aim was to clarify if there is any potential toxic effect of OPA on both liver and cardiac tissue. In addition to inflammatory changes, we found a significant HO activity reduction in plasma. The decrease in circulating HO enzyme activity promotes oxidative processes which can trigger hepatic damage and modulate pathways that control normal biological functions. 36,37
To analyse the potential hepatotoxic effects of OPA, ALT and AST markers were also measured. It has been demonstrated that a great number of toxins, microbial metabolites and natural products could cause liver damage. Transaminases are located in liver cells and leak out into the circulation when liver cells are injured. Ozer et al. reported that serum/plasma ALT and AST biomarkers in humans and rats have potential to be utilized as bridging markers to monitor liver injury in early clinical trials. The ALT is a more specific indicator of liver inflammation than AST, which is also found in the heart and skeletal muscle. 38 Hepatic dysfunction accompanied by elevated ALT and AST levels show a complex relationship with cardiac diseases. 39 In our study, we did not observed changes in the serum ALT and AST concentrations after 24 h of treatment with 0.01, 0.1 and 1 mg/kg OPA.
In summary, the current report highlights the acute role of OPA in systemic and cardiac-specific inflammatory responses. While the inflammation-inducing effects mediated by OPA in the plasma seem related to the activation of inflammatory markers (e.g. IL-6, TNF-α and MPO) and the suppression of HO enzyme, in the cardiac tissue, OPA treatment did not cause changes in the inflammatory responses. Furthermore, OPA did not show cytotoxic effects in the serum ALT and AST levels.
Footnotes
Authors’ contribution
All authors participated in the design and interpretation of the studies, in the analysis of the data and in the drafting and final approval of the article. Renáta Szabó and Anikó Pósa contributed equally to this article as first authors.
Acknowledgements
This work was supported by PIAC_13-1-2013-0201, ÚNKP-16-4 (Anikó Pósa), ÚNKP-16-3 (Renáta Szabó), TÁMOP 4.2.2.A-11/1/KNOV-2012-0035, AGR_PIAC_13-1-2013-0008 and DE-KUTEGY. This research was realized in the frames of TÁMOP 4.2.4. A/2-11-1-2012-0001 “National Excellence Program: Elaborating and Operating an Inland Student and Researcher Personal Support System.” The project was subsidized by the European Union and cofinanced by the European Social Fund. This work was connected to the project GINOP-2.3.3-15-2016-00006.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
