Abstract
This study was designed to determine the levels of prolactin, leptin, osteopontin, and follistatin in workers chronically and subacutely exposed to lead compounds. The examined population consisted of three groups. The first group was composed of 56 male workers who were chronically exposed to lead for 13.38 ± 10.38 years. The second group served as a control group and consisted of 24 male administrative workers, while the third group included 32 male workers exposed to lead for 40 ± 3 days. The levels of leptin, osteopontin, and prolactin were significantly lower in the group of workers chronically exposed to lead than in the control group by 42%, 26%, and 41%, respectively. The levels of follistatin did not differ between those groups. The levels of all measured hormones did not change after a short-term exposure to lead compared to baseline. Chronic lead exposure is associated with significantly decreased level of prolactin, leptin, and osteopontin. Lead-induced changes in the levels of these hormones may disturb many functions of the human body, including the immune response, metabolism, reproduction, and bone turnover.
Introduction
Lead is still used in many industries, such as battery manufacturing, ceramics, and dye industry. Exposure to lead results in many adverse health effects. 1 Therefore, both occupational and environmental lead exposures are among the most significant public health problems. 2 It has been established that there is no safe level of exposure to lead. Health effects of acute exposure to lead are related to occupational exposure and are quite uncommon, while chronic toxicity of lead is much more common and manifests clinically at blood lead levels of about 40–60 µg/dl. Severe lead poisoning is characterized by persistent vomiting, encephalopathy, lethargy, delirium, convulsions, and coma. 3
There are many proposed mechanisms underlying clinical manifestations of lead toxicity. For instance, lead has been shown to induce increased release of reactive oxygen species (ROS) that are highly reactive to membrane lipids, proteins, and DNA. In consequence, lead-induced ROS overproduction may result in increased cellular damage. 4 It has been also established that lead modulates the immune response acting as a pro-inflammatory agent. 5 Besides, animal studies have shown that lead is able to promote immune response mediated by the Th2 cells and suppress those mediated by the Th1 cells. 6,7 Additively, there are many reports indicating the impact of lead on the endocrine system function. A negative effect of lead exposure on thyroid function is the most widely studied over the last few decades. Lead poisoning has been also associated with impaired male reproductive function and depressed adrenal and pituitary functions. 2
There is a heterogenic group of biologically active substances, such as prolactin, leptin, osteopontin, and follistatin, which have both hormonal and immunomodulatory activity. Therefore, the possible associations between their levels and lead exposure may bring some new information about the toxicity of lead on the immune and endocrine systems. Therefore, the aim of the present study was to determine the levels of prolactin, leptin, osteopontin, and follistatin in workers chronically and subacutely exposed to lead compounds.
Material and methods
Study population
The experimental setup has been approved by the Bioethics Committee of the Medical University of Silesia in Katowice no. KNW/0022/KB1/108/14.
The examined population consisted of three groups. All of the study subjects provided a written consent to a study. Blood lead level served as a biomarker of lead exposure. Questionnaire data on age, weight, height, medical history, and smoking were obtained.
The first group was composed of 56 male workers who were chronically exposed to lead for 13.38 ± 10.38 years. Their mean age was 39.52 ± 8.55 years. In that group, three persons were diagnosed with hypertension, while one person was diagnosed with diabetes. None of the workers were diagnosed with coronary artery disease.
The second group served as a control group and consisted of 24 male administrative workers aged 42.63 ± 8.65 years. Their blood lead levels were below 10 μg/dl. None of the individuals from this group was occupationally exposed to lead. In that group, two persons were diagnosed with hypertension. None of the workers were diagnosed with diabetes and coronary artery disease.
The third group included 32 male workers exposed to lead for 40 ± 3 days. Their mean age was 41 ± 13 years. To analyze confounding factors, this group was additively divided into pairs of subgroups based on history of occupational exposure to lead before the study, smoking habits, and a median of age and body mass index (BMI). In that group, two persons were diagnosed with hypertension, while one person was diagnosed with coronary artery disease. None of the workers were diagnosed with diabetes.
Laboratory procedures
Blood collection
Blood of chronically exposed workers and those in the control group was drawn once, while blood of subacutely exposed workers was drawn at the beginning of the study for the first time and after a period of short-term exposure to lead for the second time.
Samples of blood obtained from each patient were collected from the cubital vein using vacuum tubes (Vacuette®; Greiner-Bio, Frickenhausen, Germany) that contained K3EDTA to obtain whole blood or plain tubes to obtain serum. Blood samples were frozen and stored at −20°C until tested.
Determination of lead concentration
The assessments of the PbB were performed by graphite furnace atomic absorption spectrometry using an ICE 3400 instrument (Thermo Fisher Scientific, Waltham, Massachusetts, USA). The laboratory met the requirements of proficiency tests (Lead and Multielement Proficiency – CDC in Atlanta). The ClinCal® Whole Blood Calibrator and ClinCal® Serum Calibrator (Recipe, Germany) were used for calibration of the instrument and control materials. ClinCheck Whole Blood Control Levels I, II, and III and ClinCheck Serum Control Levels I and II were used for quality control.
Determination of hormones
The levels of prolactin, leptin, osteopontin, and follistatin were detected in serum using a Bio-Plex 200 System (Bio-Rad Laboratories Inc., Berkeley, California, USA). The Bio-Plex system is based on three core elements. The first core element is a technology that uses fluorescently dyed magnetic microspheres (beads), each with a distinct color code to permit discrimination of individual tests within a multiplex suspension and allows the simultaneous detection of diverse analyte molecules in a single well of a 96-well microplate. Moreover, the magnetic beads allow for magnetic separation during the washing steps. The second core element is a dedicated flow cytometer with two lasers (a 532 nm Nd-Yag laser used to excite phycoerythrin in the assay and a 635 nm solid state laser used to excite the dyes inside the beads to determine their “color” or “region”) and associated optics to measure the different molecules bound to the surface of the beads. The third core element is a higher speed digital signal processor that efficiently manages the fluorescent output.
The principle of these bead-based assays is similar to capture sandwich immunoassays. The samples and standards were incubated with the coupled beads (antibodies directed against the desired cytokines were covalently coupled to internally dyed beads) in the wells of 96-well plates and washed. Next, the biotinylated detection antibodies specific for different cytokine epitopes were added. After incubation and washing, streptavidin (phycoerythrin solution) was added to bind biotinylated detection antibodies on the beads. Next, the suspensions of washed beads were analyzed using the Bio-Plex system. Software presented data as both median fluorescence intensity as well as concentration (pg/ml).
Statistical analysis
The statistical analysis was performed using the Statistica 9.1 PL software program. The statistical analyses included the means and standard deviations of the data. Shapiro–Wilk’s test was used to verify normality, and Levene’s test was used to verify the homogeneity of variances. Statistical comparisons were made using the t test, t test with separate variance estimates, the Mann–Whitney U test, or the χ2 test. Dependent variables were analyzed using Student’s t test and Wilcoxon’s test. The Spearman non-parametric correlation was calculated. A value of p < 0.05 was considered to be significant.
Results
Epidemiologic data and concentrations of lead in the blood are presented in Tables 1 and 2.
Epidemiologic data and lead exposure markers in the group of workers chronically exposed to lead and in the control group.

CAD: coronary artery disease; HA: hypertension; DM: diabetes mellitus; PbB: blood lead level; BMI: body mass index.
Epidemiologic data and lead exposure markers in the group of workers subacutely exposed to lead.

CAD: coronary artery disease; HA: hypertension; DM: diabetes mellitus; PbB: blood lead level; BMI: body mass index.
The blood lead level was over 17 times higher in the group of workers chronically exposed to lead than in the control group, while the levels of leptin, osteopontin, and prolactin were significantly lower by 42%, 26%, and 41%, respectively. The levels of follistatin did not differ between those groups (Tables 1 and 3).
The levels of follistatin, prolactin, leptin, and osteopontin in the group of workers chronically exposed to lead and in the control group.

IQR: interquartile range.
The blood lead level increased over 4.5 times after a short-term exposure to lead (Table 2). The levels of all measured hormones did not change after a short-term exposure to lead compared to baseline (Table 4).
The levels of follistatin, prolactin, leptin, and osteopontin in the group of workers subacutely exposed to lead at the baseline and at the end of the study period.

IQR: interquartile range.
In a combined group of workers chronically exposed to lead and males in the control group, the analysis of correlations showed a positive correlation between the level of leptin and BMI (R = 0.60) and a negative correlation between the level of leptin and blood lead level (R = −0.25; Table 5). In the group of workers subacutely exposed to lead, there were no correlations between the cytokine levels and the change of blood lead level due to exposure (data not shown).
Correlations between the levels of determined hormones and age, BMI, and PbB in the combined group of workers chronically exposed to lead and males in the control group. *p: <0.05.

BMI: body mass index; PbB: blood lead level; NS: non significant.
Additional comparisons between the subgroups of subacutely exposed workers showed that younger workers have lower follistatin level than older workers (Table 6). Besides, changes of follistatin level due to lead exposure were significantly different when compared the subgroups of smokers and nonsmokers (Table 7). Analysis of confounders also showed that workers with higher BMI have higher level of leptin than those with lower BMI (Table 8). The comparisons between the levels of the remaining measured hormones in subgroups of younger and older workers, smokers and nonsmokers, and workers with higher BMI and lower BMI did not show any significant differences (data not shown). Similarly, the comparisons between the levels of measured hormones in subgroups of workers with a history of occupational exposure to lead and those without such history did not show any significant differences (data not shown).
The comparison between subgroups of younger and older workers based on median of age (36.5 years).
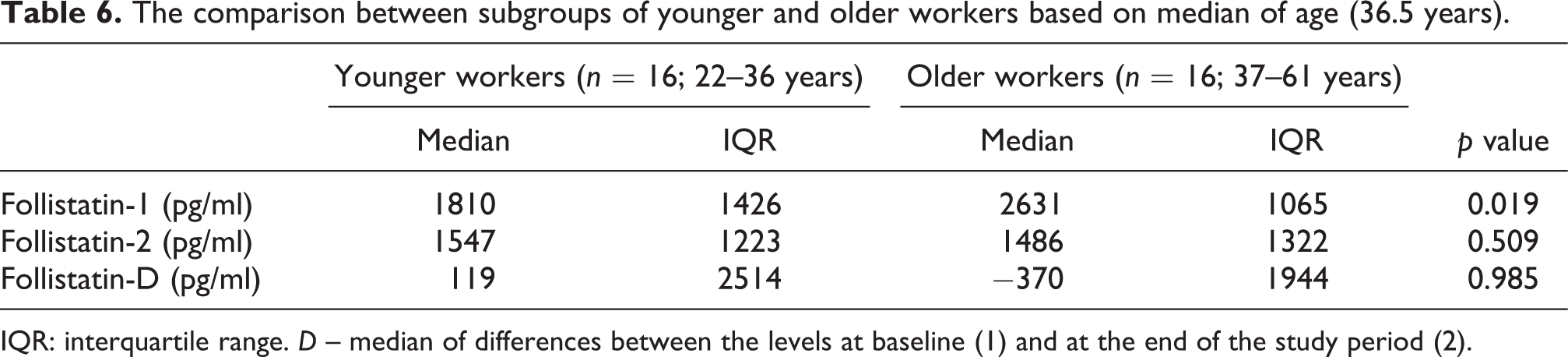
IQR: interquartile range. D – median of differences between the levels at baseline (1) and at the end of the study period (2).
The comparison between subgroups of smokers and nonsmokers.

IQR: interquartile range.
D: median of differences between the levels at baseline (1) and at the end of the study period (2).
The comparison between the subgroups of workers with lower and higher BMI based on median of BMI (25.7 kg/m2).

BMI: body mass index; IQR: interquartile range.
D – median of differences between the levels at baseline (1) and at the end of the study period (2).
Discussion
Prolactin is mainly produced by lactotroph cells of the anterior pituitary gland. However, there are extra-pituitary sites of prolactin release, such as adipocytes and immune, decidual, mammary, and epithelial cells. The secretion of prolactin is regulated by hypothalamic hormones, including thyrotropin-releasing factor and dopamine. The receptor of prolactin is a member of the cytokine receptor superfamily which includes also receptors for interleukin (IL)-2, IL-6, granulocyte-macrophage colony-stimulating factor (GM-CSF), and leptin. Prolactin modulates water and electrolyte balance, growth, metabolism, brain function, reproduction, and the immune response. Prolactin influences the function of macrophages, granulocytes, natural killer cells, dendritic cells, and T and B lymphocytes. As a result, prolactin’s action may result in increased release of ROS and increased secretion of the pro-inflammatory cytokines, chemokines, and antibodies. Therefore, many authors postulate that hyperprolactinemia may be related to the development of autoimmune diseases, such as systemic lupus erythromatosus, rheumatoid arthritis, multiple sclerosis, autoimmune thyroiditis, myasthenia gravis, and diabetes mellitus, however, data on this topic are not conclusive. 8,9
There are in vivo and in vitro studies suggesting that the neurotoxicity of lead may be attributed to its influence on the dopaminergic system. It has been proposed that lead may decrease synaptosomal dopamine release via altering the synthesis of dihydroxyphenylacetic and homovanillic acids in the mesolimbic and nigrostriatal regions. Besides, lead may modify the number or sensitivity of the dopaminergic receptors in the tuberoinfundibular region. Because the tuberoinfundibular dopaminergic system controls prolactin secretion, proposed lead-induced inhibition of the dopamine release should theoretically result in hyperprolactinemia. However, the results of studies concerning the associations between blood lead levels and urine levels of homovanilic acid, an end-product of dopamine metabolism, are inconsistent. Therefore, a biphasic dose-dependent effect of lead on the dopaminergic neurotransmission has been postulated as well. 10 An existence of such mechanism may explain also discordant results of studies on prolactinemia in lead exposure. In the present study, prolactin level was significantly lower in workers chronically exposed to lead than in the control group, while a short-term exposure to lead did not significantly affect its level. Consistently, an inverse relation between prolactin and lead levels was found in two groups of 219 11 and 240 12 men environmentally exposed to lead. Analogical results were obtained in a group of 101 pregnant women, 13 while no significant association between prolactin and lead levels was found in 400 children environmentally exposed to lead. 10 However, Govoni et al. 14 showed higher level of prolactin in a group of male workers occupationally exposed to lead with blood lead levels higher than 40 µd/dl than in those with blood lead level below this value. Similarly, Lucchini et al. 15 found higher prolactin level in a group of 66 lead-exposed workers with mean blood lead level 27.50 ± 28 µg/dl than in an unexposed control group with mean blood lead level of 8.11 ± 4.47 µg/dl.
Similar to prolactin, leptin regulates many functions of the human body. Generally, leptin is synthesized and secreted by white adipose tissue. The hypothalamus is believed to be the main site of leptin action. Leptin plays important roles in the regulation of food intake, energy balance, and metabolism. 16 It has been observed that serum leptin level increases in proportion to body fat percentage. Consequently, obese patients display elevated leptin concentrations relative to lean controls. 17 Results of the present study are in concordance with these findings because leptin level was significantly higher in the subgroup of workers with higher BMI than in those with lower BMI. In addition, leptin level correlated positively with BMI (R = 0.6) in a combined group of workers chronically exposed to lead and those in the control group.
It has been postulated that obesity and lead exposure may be co-occurring risk factors for decreased bone mass. Lead toxic action has been inversely correlated with femoral bone density and associated with osteoporosis, while leptin has been proposed to negatively influence bone mass accrual indirectly through serotonin suppression. In a study conducted on mice, leptin levels were significantly elevated by both high-fat diet and lead exposure, but without any interactions between them. 18 By contrast, in the present study, leptin level was significantly lower in workers chronically exposed to lead than in the control group. There was also a negative correlation between leptin and lead levels (R = −0.25) in a combined group of workers chronically exposed to lead and those in the control group. Subacute exposure to lead did not significantly affect leptin level. Because leptin is not only an antiobesity hormone but also influences hematopoiesis, thermogenesis, reproduction, angiogenesis, and more importantly the immune response, 19 lead-induced alterations of leptin levels may disturb function of many systems of the human body. Leptin modulates both innate and adaptive immunity being a pro-inflammatory molecule necessary for full functionality of the immune cells. 16,20 Leptin has been shown to promote Th1-mediated immune response. 19 Therefore, a decrease in leptin level may explain lead-induced skewing toward Th2-mediated immune response postulated in experimental studies by Heo et al. 6,7
Analogous to leptin, osteopontin has been shown to enhance T cell differentiation along the Th1 pathway and suppress formation of Th2 cells. 21 As a result, observed in the present study, an inverse association between chronic lead exposure and osteopontin level may contribute to the postulated lead-induced promotion of the Th2-mediated immune response. Osteopontin is a multi-functional member of the small integrin-binding N-linked glycoprotein family of proteins involved in many physiological and pathological processes, such as bone remodeling, calcification, inflammation, carcinogenesis, fibrosis, and cell survival. On the one hand, osteopontin acts as an anti-oxidant via inhibiting both iNOS and the NADPH oxidase complex which results in decreased release of reactive oxygen and nitrogen species. On the other hand, osteopontin exerts pro-inflammatory properties and promotes formation of the atherosclerotic lesions. Besides, osteopontin expression in malignant tumors correlates with an unfavorable prognosis because this molecule promotes invasive and metastatic progression of many carcinomas. 22,23 In light of this, decreased osteopontin level due to lead toxicity may be in some cases regarded as a paradoxically beneficial effect of lead action.
Follistatin is the next hormone that affects immune system function. The suppression of activin was the first known ability of follistatin. It is a glycosylated single-chain protein functionally associated with the members of the transforming growth factor-β superfamily and controls proliferation, differentiation, and apoptosis of many cell types. 24 Follistatin together with activin has immunomodulatory properties and acts as an acute-phase protein. 25 Results of the present study did not confirm that there are links between exposure to lead and serum follistatin levels. However, it is not possible to exclude the influence of lead on the follistatin’s paracrine or autocrine action. Additively, the analysis of confounders showed that smoking habits may differentiate the effect of subacute exposure to lead on the follistatin level. Besides, we showed higher follistatin level in the population of older workers than in younger. This difference is likely related to the aging processes. However, age does not seem to interfere with investigated lead impact on follistatin level as smoking does.
To sum up, chronic lead exposure is associated with significantly decreased level of prolactin, leptin, and osteopontin, while a short-term exposure to lead does not induce changes in the levels of these hormones. Follistatin level seems to be not influenced by both chronic and subacute lead exposure. Lead-induced changes in the levels of the examined hormones may disturb many functions of the human body, including immune response, metabolism, reproduction, and bone turnover.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
