Abstract
Experimental studies have shown that prenatal exposure to lead (Pb) produces morphological changes related to extracellular matrix remodelling. To analyse whether the matrix metalloproteinases (MMPs), particularly MMP-2, MMP-9, and the tissue inhibitor of metalloproteinases-2 (TIMP-2), are associated with morphological alterations found in placentas, the expression of these enzymes was evaluated by immunohistochemical and image analyses in placentas of women with histories of environmental exposure to Pb. The median maternal concentration of Pb in blood was 4.68 µg/dL (
Introduction
Lead (Pb) is a well-known toxic metal whose effects on the human body are well documented. In Mexico, it is recommended that concentration of Pb in blood in the general population older than 15 years should be less than 25 µg/dL, whereas in children younger than 15 years and in pregnant women, it should not exceed 10 µg/dL. 1 Nevertheless, several studies have demonstrated that low-level prenatal exposure to Pb, in concentrations of less than 10 µg/dL, can be associated with alterations in cognitive, language, and psychomotor performance, behavioural outcomes, as well as hearing loss or deficiency in auditory capacity.2–4 In the case of pregnant women, it has been demonstrated that the placenta does not represent a barrier for Pb; concentrations of Pb in maternal blood correlate with the levels in cord blood and may predict foetal toxicity.5–7 In the literature, information pertaining to the mechanisms of damage and/or the effects of exposure to Pb in the human placenta is scarce. Experimental studies carried out in murine models have shown that prenatal low exposure to Pb induces histological damage in the placenta that could affect the development of the foetus. 8 Fuentes et al. 8 suggested that alterations found in placentas are a result of the activation of a repair mechanism in which the activity of metalloproteinases may be involved. The extracellular matrix metalloproteinases (MMPs) are Ca2+- and Zn2+-dependent proteolytic enzymes whose expressions are strictly regulated at the transcriptional level by the interaction with tissue inhibitor of metalloproteinases (TIMPs) or by proteolytic activation of latent proenzymes. 9 TIMPs are specific inhibitors that bind MMPs in a 1:1 stoichiometry; under pathological conditions associated with unbalanced MMP activities, changes in the levels of TIMP are considered to be important because they affect the level of MMP activity. 10 Diverse studies have demonstrated that during gestation, MMP-2, MMP-9, and TIMP-2 are constantly expressed in the placenta; these have been implicated in the propagation of labour, both at term or preterm, in gestational trophoblastic diseases, and in pathological processes such as in diabetes mellitus.11–15 Barbosa et al. 16 have demonstrated a positive association between the activity of MMP-9 and the levels of Pb in the plasma of Pb-exposed people.
To analyse whether MMPs, specifically MMP-2, MMP-9, and TIMP-2, are associated with morphological alterations to the placenta, we analysed the expression of these enzymes, by immunohistochemical (IHC) and image analyses, in the placentas of women with histories of environmental exposure to Pb. The present study considered the population living in areas near the American Smelting and Refining Company (ASARCO) metals smelter in Avalos, Chihuahua, Mexico, which is considered to be the main source of exposure to Pb in the state. 17
Materials and methods
Acetone, ethanol, methanol, potassium chloride, potassium phosphate monobasic, sodium chloride, sodium hydroxide, sodium phosphate dibasic, and xylene were purchased from JT Baker (Edo. De México, México). Tris-HCl was obtained from Gibco BRL (Rockville, MD, USA); 3-aminopropyl triethoxysilane, hydrogen peroxide, and paraformaldehyde were purchased from Sigma-Aldrich (St. Louis, MO, USA). Entellan mounting resin was obtained from Merck (Darmstadt, Germany). For IHC analysis, polyclonal goat anti-human metalloproteinases, MMP-2 (C-19) and MMP-9 (M-17), and tissue inhibitor metalloproteinase 2 (TIMP-2; L-17) antibodies were purchased from Santa Cruz Biotechnology, Inc. (CA, USA). The BioGenex ready-to-use kit was obtained from BioGenex Laboratories (San Ramon, CA, USA).
Study population
This cross-sectional study was carried out in the city of Chihuahua, in northern Mexico, between 2003 and 2006. The study protocol was approved by the ethics committee of the Mexican Social Security Institute (IMSS) in the city of Chihuahua, Mexico. Women who were working in the manufacture industry associated with Pb use and/or who had their residence in the area of Avalos (at least 5 years ago) were included in the study.
Women were recruited when they were admitted for their obstetric and delivery care, signed a letter of consent to participate in the study, and a structured questionnaire was administered to obtain information about obstetric antecedents, maternal occupation, zone of residence, and maternal health. Patients with any complications during gestation or who had antecedents of chronic illnesses were excluded from the study. A total of 334 mother–newborn pairs were obtained at the beginning of the study, because it is a new subject and there are no specific records to calculate an adequate sample size, a subsample of n = 29 placentas (≈10%) was selected and analysed.
Blood samples
Samples of 5 mL of venous maternal blood were drawn with the consent of the mother, and a similar volume of cord blood was collected immediately after delivery. Samples were transported to the laboratory and stored at 4°C. Levels of Pb in cord and maternal blood were used to quantitatively reflect the status of prenatal exposure to Pb. Levels of Pb in all of the blood samples were determined by atomic absorption spectrophotometry using pyrolytically coated furnace tubes and L’vov platforms according to the methodology recommended in Mexican Official Policy. 1 The detailed methodological issues have been provided elsewhere. 18
Placenta samples
All placentas included were treated in the same manner. Subsequent to birth, the cord was clamped, the maternal coagulum was removed, and the placenta was weighted without trimmed membranes and cord. Placental samples from the central area were dissected, the sites of selection were determined macroscopically free of lesion, and were immediately fixed in a 3.8% formaldehyde solution. Paraffin sections were stained with haematoxylin and eosin to analyse histological placental alterations.
IHC analysis
Paraffin sections (4 µm thick) from each placenta were mounted on 3-aminopropyl triethoxysilane-treated glass slides, deparaffinised with xylene, rehydrated in a graded series of ethanol, and equilibrated in a phosphate-buffered saline (PBS) bath for 5 min. IHC was performed with a BioGenex ready-to-use kit following the manufacturer’s instructions. Endogenous peroxidase quenching treatment was done by incubating the samples for 10 min at room temperature in absolute methanol containing 3% hydrogen peroxide. After blocking (PBS pH 7.4 containing 1% nonfat milk), slides were incubated separately for 12 h at 4°C with the polyclonal goat anti-human MMP-2 or MMP-9 or with TIMP-2 antiserum (1:100 dilution in PBS pH 7.4 containing 1% nonfat milk). Slides were washed and exposed to the secondary affinity-purified biotinylated rabbit anti-goat IgG antibody for 1 h at room temperature. The signal was revealed using avidin peroxidase and a freshly prepared diaminobenzidine substrate. Stained slides were dehydrated for permanent coverslipping with Entellan resin. All the reagents were used as recommended by the manufacturer. Negative controls were mounted using a nonrelated antiserum and/or by omitting the primary antibody.
Image analysis of MMP-2, MMP-9, and TIMP-2 expression
Optical density measurements of MMP-2, MMP-9, and TIMP-2 immune detection were performed on slides using IMAGE pro-plus 4.1 software (Media Cibernetics, Silver Spring, MD, USA) and a BX41 Olympus microscope equipped with a Pixera-CCD camera. The area on the slide without any microscopic lesions was selected for the measurements of optical density. Six representative microphotographs of placental villi from each sample and IHC stained were obtained using the ×10 objective lens. Microphotographs were transformed to greyscale and five measurements of optical density from the signal present on the syncytiotrophoblast were obtained for each photograph using a bar of 0.05 mm (n = 30 for each sample, n = 870 for each protein; n = 420 for group with levels of Pb less than that of the median, and n = 450 for group with levels of Pb more than or equal to the median). Measurements were made after calibrating the microscope with an individual pixel resolution of 175 grey levels. All determinations were made the same day to diminish calibration or lighting errors. Image analysis was conducted by a single researcher to whom the exposure status of the women was unknown.
Statistical analysis
Univariate analysis was done for each demographic and clinical characteristic studied. Levels of Pb are shown as means ± SD. Based on the levels of Pb in the cord blood, placentas were classified into two groups below (n = 14) or above (n = 15) the median (4.68 µg/dL), the former one considered as the control group.
Comparisons between the groups were performed using a Student’s t test and/or Wilcoxon range test according to the data distribution. In all the cases, statistical difference was considered significant when p < 0.05. Data analyses were carried out with the STATA 9.0 program for Windows (Stata Statistical Software, Release 9.0, Stata Corporation, PA, USA). For image analysis correlation, interobserver and intraobserver coefficients were calculated using n = 120 and 180 measurements of optic density, respectively, obtaining values of 0.8729 and 0.8878, respectively. 19
Results
As shown in Table 1, there were no differences in gestational or demographic parameters between the total population and the subsample analysed in the present study. Neither placental nor newborn weights showed a significant difference from normal full-term newborns from the general population. Similarly, no significant difference was found in the weight of the placenta or the newborn in relation to the concentration of Pb in maternal or umbilical cord blood.
Sociodemographic and gestational parameters of the population included in the study

a Data are shown as means ± SD.
b Comparisons between groups were performed using a Student t test.
Levels of Pb in maternal blood were 5.85 ± 6.48 µg/dL (range from 0 to 29.26 µg/dL) and 4.38 ± 5.4 µg/dL (range 0–24.59 µg/dL) in umbilical cord blood. These levels were relatively low because only 17% of mothers had levels of Pb >10 µg/dL. 1 However, there was a correlation between the concentrations of Pb in cord blood and in maternal blood (r = 0.95, p < 0.01 for total population and r = 0.80, p < 0.01 for subsample).
To analyse the differences in both the placental histological findings and the expression of MMPs, the median of maternal blood concentration of Pb was determined (4.68 µg/dL) and based on this two groups were defined: the first one with the levels of Pb below the median (control) and the second one with the levels of Pb above the median. Variables were then compared between these two groups.
Gross examination of the placentas revealed no serious anatomical changes; however, at the microscopic level, the most frequent anomalies were vascular congestion (Figure 1A), microcalcifications (Figure 1B), and fibrosis, which, in some cases, affected a great percentage of the villous stroma (Figure 1C and D); in some samples, fibrosis limited the presence of villous vessels and epithelial plates available for maternal–foetal exchange (Figure 1E), while in other samples, areas with fibrosis showed abundant fibroblasts and collagen fibres with no epithelial plates (Figure 1F).

Morphologic alterations found in placentas of women environmentally exposed to Pb. Microphotographs showing (a) stem villi with vascular congestion; (b) appearance of microcalcifications present in the villi; (c) and (d) deposits of fibrinoid material present in placental villi; original magnification: ×10; (e) normal appearance of the stroma of placental villi; (f) abundant collagen fibres plus fibroblasts in areas of fibrinoid deposits; original magnification: ×40. fe: foetal erythrocytes; mb: maternal blood; V: villi; MC: microcalcifications; VC: vascular congestion; Fi: fibrinoid material; fb: fibroblasts; St: stroma.
When we analysed the percentage of morphological findings detected in placentas according to the median value of maternal concentration of Pb (4.68 µg/dL), results showed that the placentas of women with levels of Pb <4.68 µg/dL had a greater number of microcalcifications than those found in the placentas of women with the levels above the median (Table 2). Other morphological changes showed no significant relationship with maternal levels of Pb.
Histological findings in human placentas according to levels of Pb in maternal blood

a Difference between the groups according to the median value of maternal concentration of Pb (4.68 µg/dL); p < 0.01.
With regard to the expression of MMP-2, MMP-9, and TIMP-2, immunolocalisation of these proteins showed a similar pattern of expression. The signal was located mainly in the syncytiotrophoblast layer of secondary and terminal villi (Figure 2A). Signals for these proteins were absent in the villous stroma. Changes in protein localisation associated with maternal levels of Pb were not detected; however, differences in signal intensity for both MMPs and TIMP-2 were observed between different samples (Figure 2C and D). In an attempt to analyse these differences in the expression, the signal for each protein on the syncytiotrophoblast layer was measured in arbitrary units of optical density using the Image Pro Plus software according to the methodology previously reported by our research group. 20
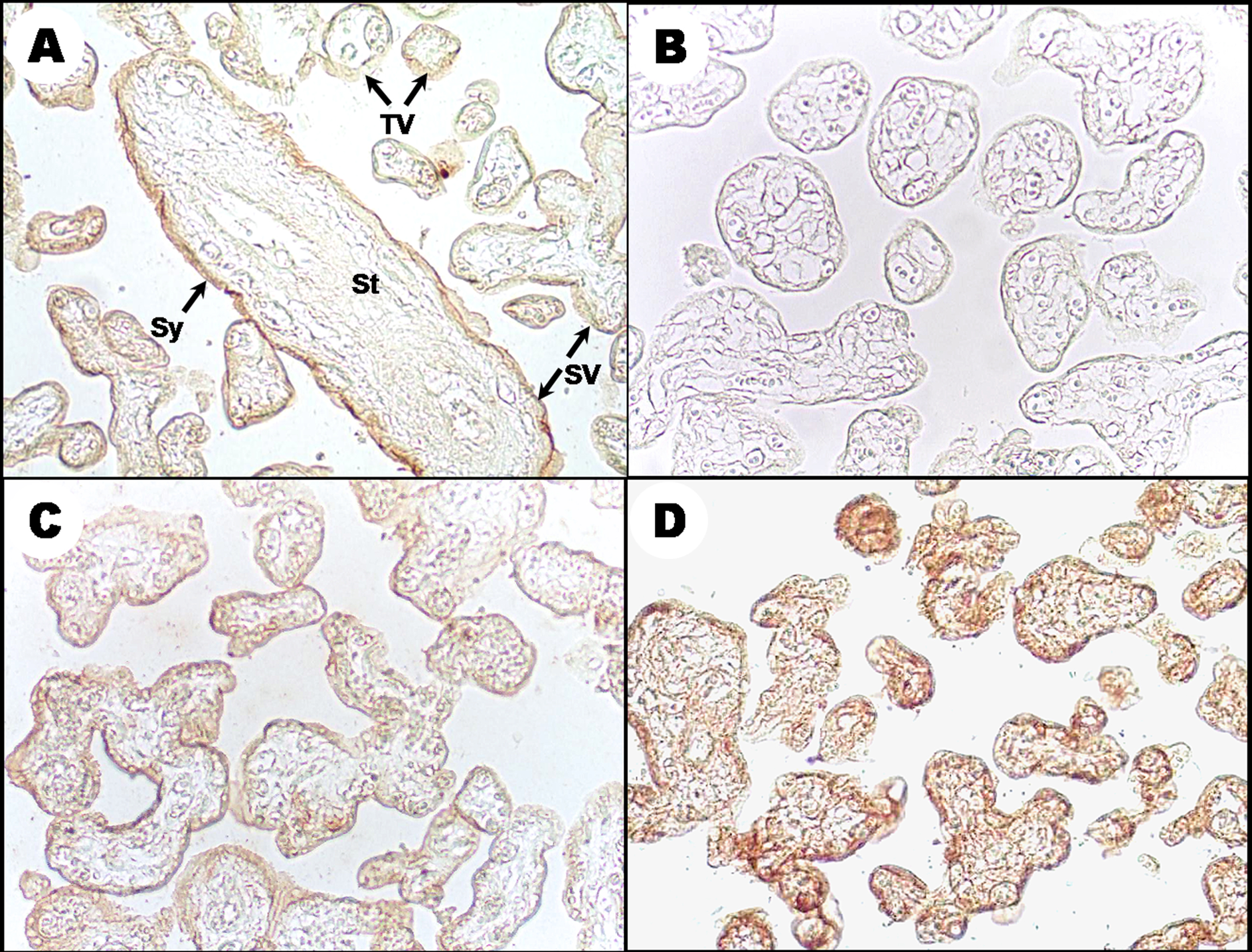
Immunolocalisation of MMPs in placentas of women environmentally exposed to Pb. The photomicrograph is a representative of the location of MMPs; in this case, the images correspond to the signal obtained for MMP-2. (a) Arrows indicate the positive signal on the syncytiotrophoblast layer of the villi. (b) Negative control. (c) and (d) MMP-2 expression in placenta from women with levels of Pb below or above the median, respectively. Original magnification: ×10. St: stroma; Sy: syncytiotrophoblast layer; SV: secondary villi; TV: tertiary villi.
When we analysed MMP-2 expression in placentas of women with the concentration of Pb ≥4.68 µg/dL, we found that MMP-2 expression was significantly greater than that found in women with the levels of Pb below the median (Figure 3; p = 0.01). Similarly, a significant increase in MMP-9 expression was detected in placentas of women with the levels of Pb above the median (Figure 3; p = 0.03).

Expression of MMP-2 and MMP-9 in placentas of women environmentally exposed to Pb. Each bar indicates the
Similar to MMP-2, TIMP-2 was expressed in placental tissue, which guarantees that MMP-2 can be activated when required. TIMP-2 showed a lower expression in placentas with higher levels of Pb (median = 1.05 (5th/95th = 0.30/2.71) vs. median = 1.14 (5th/95th = 0.48/2.44); p = 0.01). Furthermore, the MMP-2/TIMP-2 ratio was significantly higher in placentas of women with the levels of Pb above the median (median = 4.10 (5th/95th = 1.25/19.05) vs. median = 3.58 (5th/95th = 1.05/10.2); p < 0.01; Figure 4), balance that favours proteolytic activity.

Expression of TIMP-2 and MMP-2/TIMP-2 ratio in placentas of women environmentally exposed to Pb. Each bar indicates the median of arbitrary units of optical density obtained for placentas grouped according to the median maternal blood levels of Pb. TIMP-2 showed a lower expression in placentas with higher levels of Pb (median = 1.05 (5th/95th = 0.30/2.71) vs. median = 1.14 (5th/95th = 0.48/2.44); p = 0.01). MMP-2/TIMP-2 ratio was significantly higher in placentas of women with the levels of Pb above the median (median = 4.10 (5th/95th = 1.25/19.05) vs. median = 3.58 (5th/95th = 1.05/10.2); p < 0.01). n = 870 for each protein; n = 420 for group with levels of Pb less than that of the median, and n = 450 for group with levels of Pb more than or equal to the median. Comparisons between groups were performed using Wilcoxon range test; *p < 0.01.
Discussion
To date, there have been no reports on the effect of Pb exposure on the expression of placental MMP enzymes. To our knowledge, this is the first study that shows that environmental exposure to Pb is associated with an increase in the expression of MMP-2 and MMP-9 and a decrease in the expression of TIMP-2 in human placentas at term. The functional implications of the level of MMP expression associated with Pb exposure in placenta of women may be important to consider as a mechanism for generating placental abnormalities and in the induction of preterm delivery or abortion. Although specific studies on exposure to Pb have been not reported to date, high plasma levels of MMP-9 have been previously reported with respect to pre-eclampsia and gestational hypertension. 21 Moreover, Barbosa et al. 16 demonstrated a positive association between activity of pro-MMP-9 and blood levels of Pb, suggesting a possible mechanism through which low exposure to Pb may increase susceptibility to cardiovascular and neoplastic diseases. Additionally, in subjects environmentally exposed to mercury, Jacob-Ferreira et al. 22 found a positive association between blood mercury concentration and MMP-9/TIMP-1 ratio levels that were related to (CA)(n) polymorphism of the MMP-9 gene.
Yoon et al. 23 reported that elevated levels of MMP-8 in amniotic fluid can be considered as an important predictor of preterm birth (<32 weeks). Similarly, increased levels of MMP-2 and MMP-9 were reported in placentas from rats with streptozotocin-induced diabetes mellitus type 2. 24 Additionally, in that model, nitric oxide overproduction by the activity of nitric oxide synthase was found to be responsible for modulating the activities of MMP-2 and MMP-9. 25
Although further studies are needed in this regard, the increase in MMP-2 and MMP-9 expression in placentas found in this study may involve the production of reactive oxygen species and nitric oxide, factors that have been shown to be heavily implicated in the regulation of these enzymes in diabetic pathology.24,25
The activity of MMPs is controlled by TIMPs, and although TIMP-1 regulates almost all MMPs, it has higher affinity for MMP-9, whereas TIMP-2 binds to both the zymogen and the active form of MMP-2. 13 The decrease in TIMP-2 expression, detected in placentas of women with higher levels of Pb, generates an imbalance in the MMP-2/TIMP-2 ratio, favouring proteolytic enzyme activity and contributing to the production of placental abnormalities.
A decrease in the expression of TIMP-2 coupled with the increase in activity of MMP-2 activity has been reported in gestational trophoblastic diseases in which there has been a greater invasive capacity of the trophoblast. 26 Similarly, a decrease in the expression of TIMP-2 in response to damage caused by Pb could favour the activity of MMP-2 for the repair and remodelling of the placental tissue. Furthermore, a decrease in the levels of TIMP has been reported during labour, during which the increase in the activity of MMP enzyme facilitates the breakdown of extracellular matrix in foetal membranes and decidua during parturition. 27 Changes in the balance of MMP/TIMP may alter the stability of the foetal membranes and Pb to preterm birth 28 ; thus, the effect that we reported in this study on the changes produced by exposure to Pb in the expression of MMP-2, MMP-9, and TIMP-2 may also have implications at this level.
One limitation of our study was that the measurement of the expression by determining the optical densities of a signal did not permit us to differentiate between active or zymogen enzymes; however, this methodology allowed us to discriminate between the enzyme present in other cell types and in the syncytiotrophoblast, which is not possible when placental tissue homogenates are used to determine the proteolytic activity of MMPs.
In conclusion, our results suggest that this increased expression of MMPs may be induced to aid in repairing damaged placental tissue caused by the exposure to Pb and that TIMP-2, which is involved in MMP-2 regulation, decreases its expression to permit tissue repair.
However, additional studies in experimental models are being carried out to analyse the mechanisms by which Pb induces these changes in the placental tissue and the role of MMPs in the induction of preterm delivery or abortion due to the exposure to Pb. This could help to elucidate the mechanism for which the Pb is a risk for spontaneous abortion 29 and is also an additional evidence for supporting the need for lowering the level of action of blood Pb, 30 especially in women of reproductive age.
Footnotes
Acknowledgements
The authors thank Dr Ricardo Gómez Flores for critically reading the manuscript.
Funding
This work was partially supported by the National Council of Science and Technology (CONACyT) grants SIVILLA 9502-121 and FOMIX-Gobierno del Estado de Chihuahua-CONACyT (CHIH-2008-C01-92074). E.G.-P. was the recipient of a MSc scholarship from CONACyT (Reg. 184904).
