Abstract
Carbon monoxide (CO) poisoning is a leading cause of toxicity-related mortality and morbidity worldwide. Recent studies focused on CO-induced cardiovascular toxicity. Oxidative stress plays an important role in the pathophysiology of CO toxicity. The aim of this study was to elucidate the relationship between cardiac damage biomarkers and oxidative stress biomarkers in patients with CO-induced cardiotoxicity. This study was carried out on 36 CO-poisoned patients admitted to Zagazig University Hospitals. Forty healthy individuals (age- and sex-matched) were selected as a control group. Clinical examination and electrocardiography (ECG) were performed for CO-poisoned patients. These patients have been investigated for carboxyhaemoglobin percent (COHB%) and cardiac damage biomarkers; cardiac troponin I (cTn-I), heart-type fatty acid-binding protein 3 (H-FABP3). Oxidative stress biomarkers comprising malondialdehyde (MDA), asymmetric dimethylarginine (ADMA), and total antioxidant capacity (TAC) have been also assessed. All biomarkers have been assessed on admission (0 h) and 6 h after treatment of CO-poisoned patients with high-flow oxygen and compared with those of the control groups. ECG findings were abnormal in 31 patients (86.11%), where sinus tachycardia was the commonest finding (58.33%). There was a statistically significant increase of COHB%, MDA, ADMA, and H-FABP3 levels, and a significant decrease of TAC level in CO-poisoned patients compared to controls with no significant changes in cTn-I. Six hours following treatment, all measured parameters were significantly improved except for cTn-I, which was significantly increased when compared with admission status (0 h). Furthermore, H-FABP3 showed a significant positive correlation with COHB%, MDA, ADMA, and a negative correlation with TAC, while cTn-I was significantly correlated with COHB% only. ADMA and MDA seem to be the strongest determinants for the prediction of H-FABP3 changes and hence cardiovascular toxicity. Thus, cardiac damage in patients with CO poisoning could be partially mediated by CO-induced oxidative stress, where H-FABP3 level was directly and strongly associated with MDA and ADMA levels.
Introduction
Carbon monoxide (CO) is a colourless, odourless, non-irritant, toxic gas generated from the incomplete combustion of carboneous compounds. 1 CO poisoning is the most common cause of death from poisoning in the United States. 2 In Egypt, there is little published statistical data on this topic; however, an Egyptian study performed by the Poison Control Center (PCC), Ain Shams University Hospitals in Cairo, showed that CO poisoning represented the sixth most frequent toxic exposure (2.28%) of 25,555 cases admitted to PCC in 2004. 3 CO-induced neurotoxicity has been well described in literature, whereas cardiovascular consequences of CO poisoning is still not well understood and becomes a point of interest in recent studies. 4 –6 CO binds with hemoglobin (HB) and interferes with oxygen transport. 7 Cardiovascular complications are common in CO poisoning owing to high oxygen demand of the myocardium. 8 CO-induced hypoxia does not fully explain cardiac damage in CO-poisoned patients. Previous studies suggested other mechanisms such as inhibition of mitochondrial respiration, oxidative stress, and direct toxic effects. Lipid peroxidation and impaired antioxidant balance are reported to be involved in the toxic effects of CO poisoning. 9,10 Malondialdehyde (MDA) is the main final product of lipid peroxidation. 11 In vascular endothelium, asymmetric dimethyl arginine (ADMA) is regulated in a redox-sensitive fashion. Oxidative stress increases levels of ADMA that was associated with vasodilator dysfunction and increased risks for coronary and arterial diseases. 12
Measurement of the total antioxidant capacity (TAC) is preferred than serum concentrations of different antioxidants measured in laboratories separately because they are time consuming, costly and require complicated techniques. 13 Previous studies demonstrated that CO-induced cardiac damage is not always registered in electrocardiography (ECG) records. Therefore, it becomes necessary to look for other indicators such as biochemical markers. 14 Markers for cardiac tissue damage include myoglobin, creatine kinase-MB, cardiac troponins I and T (cTn-I, -T) and heart-type fatty acid-binding protein 3 (H-FABP3). 15 H-FABP3 is a rapid and sensitive marker of acute ischemia and myocardial infarction. 16,17
Up to our knowledge, few previous studies investigated the relationship between cardiac damage biomarkers and oxidative stress markers in patients with CO-induced cadiotoxicity. 3 So, we aimed here to study the cardiac effects of CO toxicity in CO-poisoned patients through assessment of oxidative stress and cardiac damage biomarkers and the relationship between them.
Subjects and methods
I-Study population
This case control study was conducted on consecutive CO-poisoned patients who had been diagnosed with acute CO intoxication from history, clinical examination, and/or carboxyhaemoglobin percent (COHB%) > 5% at the time of admission to Zagazig University Hospitals from November 2013 to January 2015. The patients with coronary arterial disease or other known heart disease, for example, valvular diseases or rhythm disorders, patients with hepatic or renal failure, and patients subjected to antioxidant drugs or supplements as well as smoker subjects were excluded. According to these exclusion criteria only 36 CO-poisoned patients were enrolled in this study. They were 19 males and 17 females, with their mean age (30.7 ± 9.9 years). All patients received high-flow (12–15 L/min) 100% oxygen therapy with a well-fitting tight face mask for at least 240 min. Forty apparently healthy individuals who were age- and sex-matched and admitted to the hospital for unrelated clinical conditions served as a control group. Informed consents from patients and approval letter were obtained from the Ethical Committee for Research (Institutional Review Board ‘IRB’), Faculty of Medicine, Zagazig University (ZU-IRB # 504, June 2013).
On admission to the emergency department (0th h) and at 6 h after treatment (6th h), blood samples were obtained for analysis of arterial blood gases, COHB%, c-Tn-I, H-FABP3, MDA, ADMA, and TAC. Analysis of arterial blood gases, COHB%, and cTn-I was done in Zagazig University Hospitals laboratories, Zagazig, Egypt. H-FABP3, MDA, ADMA, and TAC analyses were done in Biochemistry Department, Faculty of Medicine, Zagazig University, Zagazig, Egypt.
The evaluation of 12-lead ECG was recorded with a paper speed of 25 mm/s for every patient before and after treatment. Arterial blood gases and COHB% measurements were performed using blood gas analyzer, Bayer 855. COHB% up to 5% were accepted as normal values in non-smoker patients.
Measurement of circulating cTn-I concentrations in serum was done by VIDAS® Troponin I Ultra (TNU, BioMerieux Inc., France), using enzyme-linked fluorescent assay technique by one-step immunoassay sandwich method with a final fluorescent detection. This was done according to methods described by Apple et al. 18 Levels >1.3 ng/mL were accepted as indicators for myocardial damage. 19
H-FABP3 was measured in serum utilizing human cardiac fatty acid-binding protein 3 enzyme-linked immunosorbent assay (ELISA) test kit, (Oxis research, catalogue number: 11230, Oxis International Inc., Foster City, California, USA), according to Wodzig et al. 20 The normal range for H-FABP3 levels was 1.6–19 ng/mL. 21
Serum malondialdehyde (MDA) was determined by measuring thiobarbituric reactive species using the method of Yagi 22 in which the thiobarbituric acid-reactive substances react with thiobarbituric acid to produce a red-coloured complex with peak absorbance at 532 nm.
Serum TAC was determined using commercially available kit (Biodiagnostic, Dokki, Giza, Egypt) by the reaction of antioxidants in the sample with a defined amount of exogenously provided hydrogen peroxide (H2O2). The antioxidants eliminated a certain amount of the provided H2O2. The residual H2O2 was determined colorimetrically by an enzymatic reaction which involved the conversion of 3,5,dichloro-2-hydroxy benzenesulfonate to a colored product. 23
Plasma ADMA was measured by competitive ELISA method according to Schulze et al., 24 using commercial kit purchased from American Laboratory Products Company (ALPCO Diagnostics, Salem, New Hampshire, USA) with a detection limit 0.05 µM/L.
The collected data were computerized and statistically analyzed using Statistical Package for the Social Sciences (SPSS) programme version 15. Quantitative data were summarized as mean ± standard deviation (X ± SD). Qualitative data were summarized as percentage. Test of significance for qualitative data was χ2 (chi-square) test. Test of significance for quantitative data was done using student t test for comparison between groups’ means and paired t test for comparison of repetitive measurements. Pearson correlation test was used to evaluate the association between two variables. Logistic regression analysis was performed to identify the independent predictors of variables. The significance level was considered at p value < 0.05.
Results
There was a non-significant difference between CO-poisoned patients and controls regarding the personal data (Table 1). There was a statistically significant difference in the heart rate, systolic, diastolic blood pressure, temperature, and respiratory rate between the study group and the control group (Table 2). The most frequent cardiovascular manifestations in CO-poisoned patients on admission were palpitation in 32 cases (88.88%) and dyspnea in 27 cases (75%). Chest pain was present only in 25 cases (69.44%). ECG was abnormal in 31 patients (86.11%; Figure 1). Abnormal ECG findings were sinus tachycardia in 21 cases (58.33%), ST segment changes in 9 cases (25%), prolonged Q–T interval in 12 cases (33.33%) and prolonged Q–T interval & ST segment changes with inverted T-wave in 2 cases (5.56%). Regarding control group, no ECG changes were found except for sinus tachycardia in 5 subjects (12.5%).
Personal data among the study and control groups.
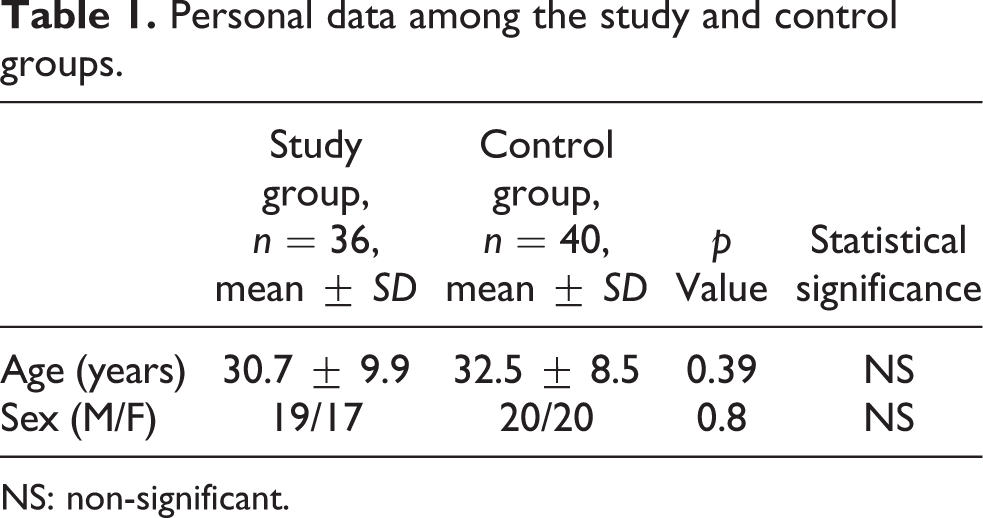
NS: non-significant.
Vital signs among the study and control groups.
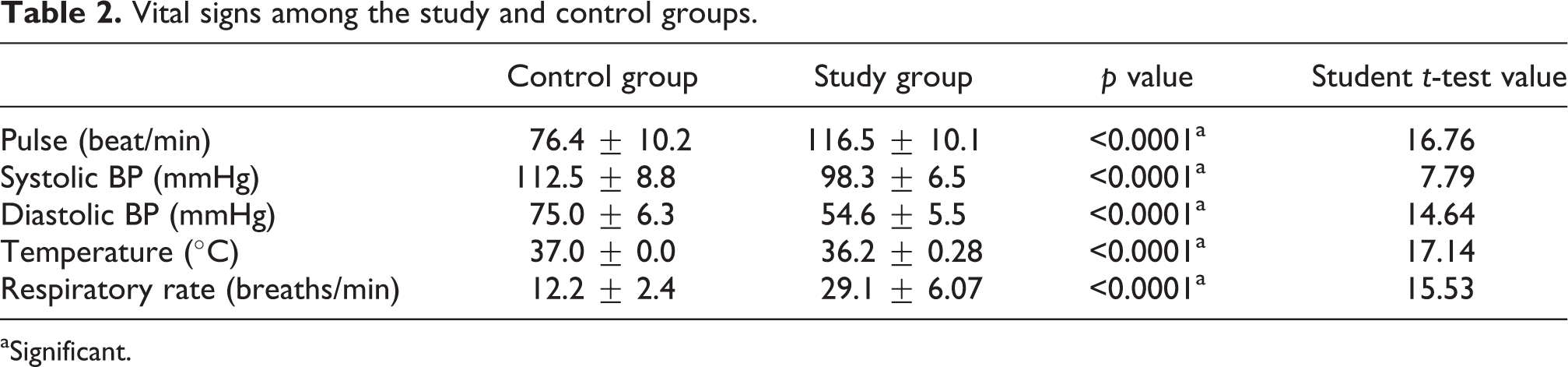
aSignificant.

Some examples of ECG changes in CO-poisoned patients on admission. ECG: electrocardiography; CO: carbon monoxide.
Results of arterial blood gases of the CO-poisoned patients were statistically significantly different compared to those of the control group (Table 3).
Arterial blood gases parameters among study and control groups on admission (0 h).

aSignificant; PCO2: partial pressure of carbon dioxide; PO2: partial pressure of oxygen; SaO2: the percentage of oxygen saturation of arterial blood; HCO3: bicarbonate level.
Laboratory findings of COHB%, cTn-I, H-FABP3, MDA, ADMA, and TAC in CO-poisoned patients on admission compared to controls are presented in Table 4. All parameters were significantly higher than those of the control group except for cTn-I, which was not significantly affected and TAC showed a significant decrease.
Laboratory parameters of study and control groups on admission (0 h).

aSignificant; NS: non-significant; COHB%: carboxyhaemoglobin percent; cTn-I: cardiac troponin I; H-FABP3: heart-type fatty acid-binding protein 3; MDA: malondialdehyde; ADMA: asymmetric dimethylarginine; TAC: total antioxidant capacity.
Laboratory findings of CO-poisoned patients 6 h after treatment are presented in Table 5. COHB% and H-FABP3 levels were significantly decreased, while cTn-I was significantly increased after treatment. Regarding oxidative stress biomarkers, both MDA and ADMA were decreased by treatment while serum level of TAC was significantly increased.
Laboratory parameters of study group at 0 h and 6 h of treatment.

aSignificant; COHB%: carboxyhaemoglobin percent; cTn-I: cardiac troponin I; MDA: malondialdehyde; ADMA: asymmetric dimethylarginine; TAC: total antioxidant capacity.
A positive correlation was observed between cTn-I and COHB%. There was no correlation among cTn-I and MDA, ADMA, and TAC (Table 6, Figure 2). We also found that H-FABP3 was positively correlated with COHB%, MDA, and ADMA and negatively correlated with TAC (Table 7, Figure 3). When we introduce all significant variables (COHB%, MDA, ADMA) to multiple regression analysis (the model), the variables that persisted significant (the strongest significant) in the model were MDA and ADMA. Therefore, MDA and ADMA seem to be significant determinant factors for the prediction of H-FABP3 levels (Table 8).
Correlation of troponin I and other parameters at 0 h.

aSignificant; NS: non–significant; COHB%: carboxyhaemoglobin percent; MDA: malondialdehyde; ADMA: asymmetric dimethylarginine; TAC: total antioxidant capacity.

Correlation between cTn-I and COHB% in the study group on admission. cTn-I: cardiac troponin I; COHB%: carboxyhaemoglobin percent.
Correlation of H-FABP3 and other parameters at 0 h.

aSignificant; COHB%: carboxyhaemoglobin percent; H-FABP3: heart-type fatty acid-binding protein 3.
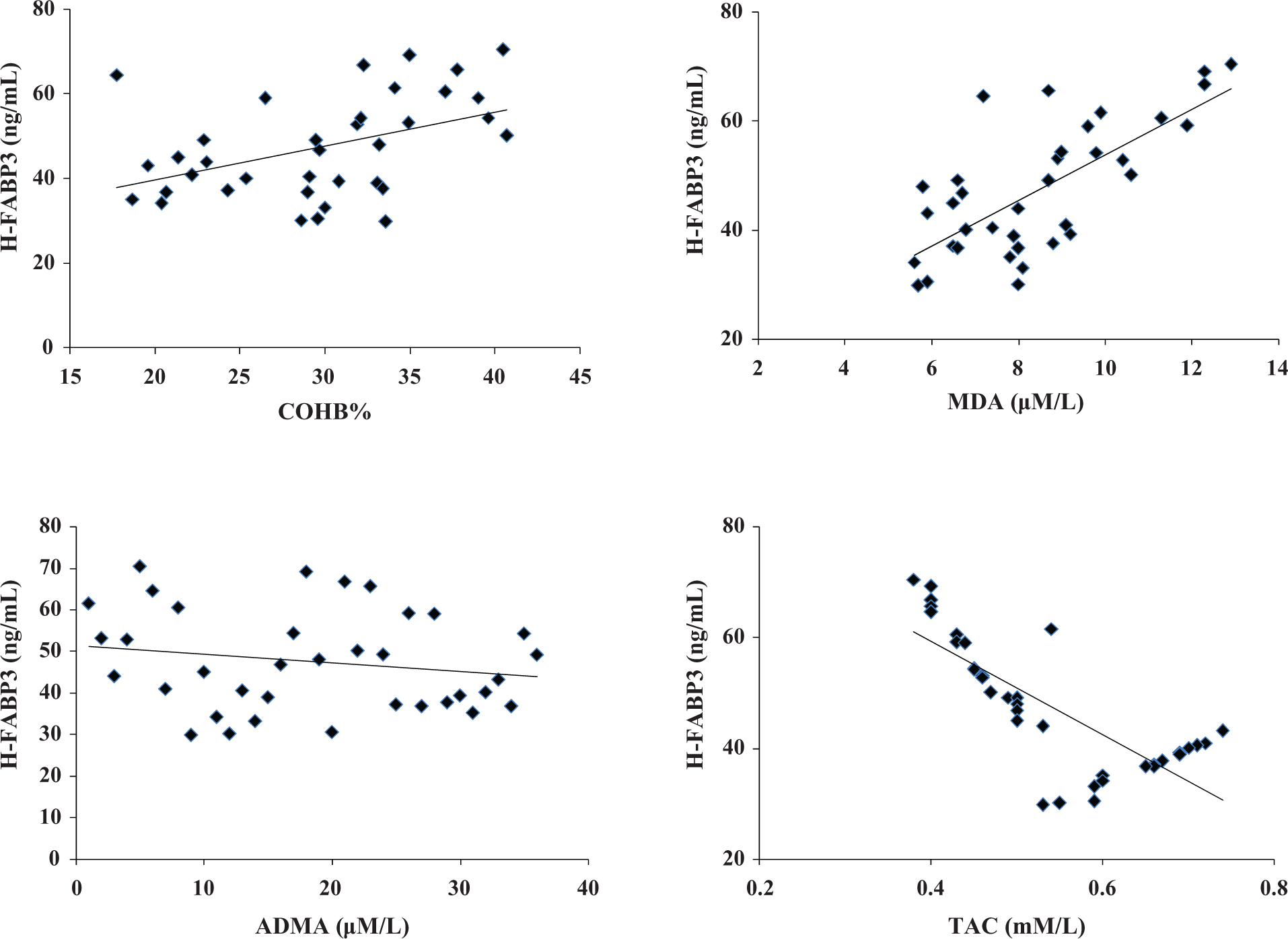
Correlation between H-FABP3 and other parameters in the study group on admission. H-FABP3: heart-type fatty acid-binding protein 3.
Logistic regression analysis for factors predicting H-FABP3.

H-FABP3: heart-type fatty acid-binding protein 3; MDA: malondialdehyde; ADMA: asymmetric dimethylarginine; CI: confidence interval.
aSignificant, where logistic regression analysis determined that the strongest factors that added significant to the model were MDA and ADMA.
Discussion
The pathophysiology of CO poisoning is complex and incompletely understood. The affinity of CO to HB is 210 times greater than that of oxygen. It decreases both the oxygen-carrying and oxygen-delivery capacity of HB. 6 Moreover, CO intoxication causes cardiovascular problems as a result of diffuse tissue hypoxia and direct action of CO on the heart. 8
Elevated COHB% measurements are used to confirm a clinical diagnosis of CO poisoning and to assess the severity of the condition. 25 In the current study, blood COHB% was increased in the study group on admission compared to the control group, and these results were in agreement with Hampson and Hauff. 14 The level of COHB is poorly correlated with cardiac damage. Moreover, CO-induced cardiac damage is not always recorded in ECG findings. To determine CO-induced cardiac damage and its severity, a proper diagnostic approach is required. Therefore, it becomes necessary to look for other indicators such as biochemical markers. 26
Electrocardiographic evaluation in this work showed abnormal ECG findings including sinus tachycardia, ST segment changes, prolonged Q–T interval, prolonged Q–T interval, and ST segment changes with inverted T-wave. In the control group, no ECG changes were found except for sinus tachycardia in 5 subjects (12.5%). These findings agree with Satran et al., 2 who reported that CO-poisoned patients had diagnostic ischemic changes recorded in the ECG which were in the form of ST-segment or T-wave changes ‘flat or inverted T-wave’, ST-segment elevation and non-specific ST-changes, either elevation or depression of the segment, as well as no ischemic changes in a study to detect the cardiovascular manifestations of CO poisoning. The ECG changes in cases of acute CO poisoning can be attributed to the direct toxic effect of CO on the heart or due to CO-induced depression of respiratory and central nervous systems causing cardiac affection. 27 On the other hand, many authors suggested that CO-induced myocardial dysfunction might take place without ECG changes which could be attributed to myocardial stunning. 28,29
Cardiac troponins are markers of cardiac muscle damage, not only limited to acute myocardial infarction (AMI). Other conditions that directly or indirectly lead to cardiac muscle damage can also increase troponin levels. 30 They appear in serum within 4–8 h after symptom onset, remain elevated for as long as 7–10 days post-AMI. 31 In the present study, cTn-I was not significantly elevated in the study group at 0th h compared to the control group. These results are in accordance with Henry et al. 32 Moreover, cTnI level became significantly higher after 6 h of treatment. These results could be explained by that cardiac cellular energy metabolism is blocked for prolonged periods even after returning to normal COHB levels. 6
Serum concentration of H-FABP3 is very low in the circulation (<5 mg/L) under non-pathological conditions. 33,34 However, H-FABP3 secretion into the interstitial space is induced by increased permeability of the myocardial cell membrane associating myocardial damage. Several studies have shown that H-FABP3 monitoring can reliably diagnose AMI within 1 h with no false-negative results. 35 –37 A number of experimental and clinical studies have investigated H-FABP3 levels in CO poisoning and showed that H-FABP3 levels were higher following CO exposure. 38 –40 The molecule of H-FABP3 is smaller than that of other cardiac damage markers, permitting its leak from damaged cardiomyocyte earlier. 4,41 Erenler et al. 39 measured serum H-FABP3 levels in CO-poisoned patients and control subjects. Its level was higher in CO-poisoned patients compared to controls. They suggested that H-FABP3 level may be used in the early diagnosis of cardiotoxicity in patients with CO poisoning. In our study, we measured H-FABP3 levels at admission and after 6 h of oxygen therapy. We clearly observed that serum level of H-FABP3 was significantly higher in the study group at admission compared to the control group, where serum level of cTn-I level was not significantly elevated in the study group at 0th h compared to the control group. These results suggest that H-FABP3 is an acute reactant and an earlier marker than cTn-I in patients with CO-induced cardiotoxicity. Our results were supported by earlier results of Valle and his colleagues 42 who found that H-FABP3 is a useful and sensitive tool in the early assessment of acute coronary syndrome.
In CO-poisoned patients, an altered balance between pro-oxidants and antioxidants has been reported. Oxidative stress is among factors involved in the pathogenesis of acute CO poisoning and particularly appear to play a role in CO-induced cardiotoxicity. 10,43 Supporting this notion, in the current study, MDA and ADMA were significantly higher in patients with CO poisoning compared to the control group. Following 6 h of treatment these indices were significantly decreased. However, TAC was significantly decreased when compared with controls and increased after 6 h of treatment. In confirmation, Kavakli et al. 10 showed that oxidative stress indices were reduced after therapy during CO poisoning. On the contrary, Garrabou et al. 44 found that oxidative stress indices tended to increase after oxygen therapy in patients with CO poisoning.
ADMA is synthesized by protein arginine methyltransferases (PRMTs) from arginine and degraded by dimethylarginine dimethylaminohydrolase (DDAH). 45 The activity of both PRMTs and DDAH is regulated in a redox-sensitive fashion. Oxidative stress increases the activity of PRMTs and inhibits the DDAH, leading to increased levels of ADMA. 12 Increased levels of ADMA have recently been implicated in endothelial vasodilator dysfunction and coronary and arterial diseases, 46,47 which could be explained by ADMA inhibition of nitric oxide synthase leading to decreased nitric oxide. 48 Nitric oxide is a potent vasodilator and its altered biosynthesis has been implicated in the pathogenesis of cardiovascular diseases. Moreover, ADMA can uncouple nitric oxide synthase leading to the generation of superoxide and peroxynitrite free radicals that can further inactivate mitochondrial enzymes and damage the vascular endothelium. The end result was membrane lipid peroxidation. 49,50 In our study, although MDA and ADMA were dependent predictors of serum H-FABP3, they were not associated with cTn-I. These results are partially in line with a recent experimental study of Ibrahim 38 who found that increased H-FABP3 level was associated with ADMA increase in rats exposed to CO poisoning. A probable explanation for this observation could be free radical scavenger activity of H-FABP3. FABP is very likely to be an effective endogenous antioxidant, because it has high affinity and capacity to bind long-chain fatty acid oxidative products. It also has a very favourable protein structure and redox conditions. The concentration of total methionine residues in FABP is high. Methionine and cysteine amino acids are regarded as cellular scavengers of oxidative stress induced by xenobiotics. 51
In conclusion, this study mainly deals with the relationship between cardiac damage markers and oxidative stress markers. H-FABP3 seems to be an early indicator for CO-induced myocardial damage. CO-poisoning-induced oxidative stress causes increased ADMA levels. A significant association between ADMA levels and H-FABP3 levels was observed, thereby indicating that increased ADMA levels may play a role in myocardial damage in patients with CO poisoning. However, this study has been conducted on a small sample size, so it is felt that further larger clinical trials should be conducted in the future to clarify the role of H-FABP3 and ADMA in CO-induced cardiotoxicity. Moreover, a detailed evaluation of cardiac function with trans-thoracic ECG would be of value.
Footnotes
Acknowledgement
Great thanks and gratefulness to Prof. Dr. Ahmad Mohmmad Ahmad Omar, Professor of Clinical Toxicology and Director of Poisoning Treatment Unit, Zagazig University Hospitals for providing facilitation of the clinical part of the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
