Abstract
This study was designed to investigate the alteration of redox status by commonly used antimalarials in Nigeria. Drugs used were artemisinin, artesunate, chloroquine, coartem and quinine at the final concentrations of 0.5–8.0 mg/mL. Blood samples were collected from malarial patients and apparently healthy humans for comparison. Reduced glutathione, catalase, superoxide dismutase (SOD) activities, protein content and lipid peroxidation were determined. All drugs significantly (p < 0.05) increases the protein level relative to control in normal blood, whereas in the infected, a significant (p < 0.05) reduction was observed. In normal blood, the antimalarials dose dependently decreased (p < 0.05) SOD and catalase activities with significant (p < 0.05) increase in the infected. The level of glutathione in normal blood significantly (p < 0.05) increases as compared with control, whereas in the infected, similar observation was made except that the levels were less, relative to control sample. Malondialdehyde level significantly (p < 0.05) increases with increase in drugs concentration even though less than the level in the control with few exceptions. These effects were dose dependent and more pronounced in non-malarial conditions. Commonly used antimalarials might alter the redox status in both healthy and non-healthy subjects thereby inducing oxidative stress.
Introduction
Oxidative stress is a common pathogenic mechanism underlying the development of many diseases and conditions including malaria infection where both the host and the parasite are affected. Adapting to the oxidative stress exerted by the host immune response against malaria infection, Plasmodium falciparum has developed an elaborate reduction–oxidation (redox) system to maintain adequate antioxidant defence mechanisms throughout its complex life cycle. 1 In humans, oxidative changes resulting from malaria infection are central to the host protective response against the malaria parasite as well as other malaria-associated pathophysiology changes. Enhanced oxidative stress reduces erythrocyte deformability, 2 contributing to haemolysis, and the development of anaemia. 3 Although oxidative stress, anaemia and malaria infection are closely linked, their complex relationships and the functional relevance of oxidative stress genes are poorly understood. 4,5
During malaria infections, the oxidative stress results from intraerythrocytic haemoglobin proteolysis by the parasite. Haemoglobin represents the major source of amino acids for plasmodium, and during its degradation in an acidic food vacuole, oxidant haem group is separated from the globin chains and consequently generates a large amount of reactive oxygen species (ROS) and other free radicals. 6,7 Furthermore, during malaria infections, massively recruited and activated monocytes and neutrophils produce increased levels of ROS. 8 These contribute to the pool of an existing stress which if not effectively handled or properly modulated paves way for toxicities around the microenvironment and other vital tissues.
In Africa, the first line of therapy for the treatment of malaria is usually through the administration of antimalarial drugs. At present, the commonly used antimalarial drugs in Nigeria are artemisinin, artesunate, chloroquine, coartem and quinine with different mechanism of actions at the therapeutic doses. However, these antimalarials often leave traces of tissue damages after therapy, as evident in memory impairment after cerebral malaria. 9 Additionally, there have been several scenarios of drug reactions and complains from different users of these drugs. Therefore, understanding the effects of these antimalarial drugs on relevant malaria-induced pathological processes would go a long way in deciphering the biochemical basis for the above-mentioned antimalarial-associated problems. Hence, the present study aims to report the effects of the commonly used antimalarial drugs on the blood redox status of normal and malaria-infected humans.
Materials and methods
Chemicals and reagents
All chemicals/reagents used were of analytical grade except otherwise stated. The drugs (artemisinin, artesunate chloroquine, coartem (artemether and lumefantrine) and quinine) were purchased from a local pharmacy (Kaduna, Nigeria). The drugs were, respectively, grinded into fine powder using a laboratory mortar and pestle. Various concentrations of the drugs (1–16 mg/mL) were prepared using 0.01 M phosphate buffer (pH 7.4). The drug stock solutions were kept at a 4°C until analysis.
Ethical clearance
The study was approved by the Ahmadu Bello University Teaching Hospital Human Research Ethical Committee with an ethical clearance number of ABUTH/HREC/TGR/36. Informed written consents were also obtained from all the subjects.
Sample collection and treatments
Blood samples were collected from the consented P. falciparum-infected patients and apparently healthy human subjects between the ages of 20 and 35 years by venipuncture and stored in ethylenediaminetetraacetic acid containers. The treatment was for 4 h incubation period at room temperature and the drugs were used at the final concentrations of 0.5, 1.0, 2.0, 4.0 and 8.0 mg/mL.
Malaria screening test
The malaria screening test was carried out using SD-BIOLINE test kit (Standard Diagnostics, Inc., Republic of Korea). Only confirmed positive malaria blood samples harbouring P. falciparum were considered for the study (except for the apparently healthy group).
Determination of total protein
Biuret method 10 was adopted in the determination of the total protein level of blood. Briefly, 100 µL of the blood sample was diluted with 100 µL of the phosphate buffer (pH 7.4) followed by the addition of 100 μL of each drug (0.5–8.0 mg/mL; the final concentration of drugs was reduced by half from the initial stock solutions) to the experimental tubes accordingly except control in which phosphate buffer was added to normalize the experimental condition. The content of each tube was gently mixed and allowed to incubate for 4 h. Thereafter, 0.1 mL of the above treated sample was added to 0.9 mL of distilled water and 4 mL Biuret reagent was then added. The absorbance of the reaction mixture was read at 540 nm after 30 min incubation period.
Determination of SOD activity
This was carried out according to the method of Misra and Fridovich. 11 Here, 100 µL of the blood sample was diluted with 100 µL of the phosphate buffer (pH 7.4) followed by the addition of 100 μL of each drug (0.5–8.0 mg/mL; the final concentration of drugs was reduced by half from the initial stock solutions) to the diluted blood except for the control tube where phosphate buffer was added. These were allowed to stand for 4 h. Thereafter, 0.9 mL distilled water was added to 0.1 mL of the treated sample. Then, 2.5 mL of 0.05 M carbonate buffer (pH 10.2) was added to 0.2 mL of the mixture followed by 0.3 mL of 0.3 mM adrenaline. The absorbance of the reaction mixture was read at 480 nm after every 30 s for five times.
Determination of catalase activity
Catalase activity was determined according to the method of Brannan et al. 12 Here, 100 µL of the blood sample was diluted with 100 µL of the phosphate buffer (pH 7.4) followed by the addition of 100 μL of each drug (0.5–8.0 mg/mL; the final concentration of drugs was reduced by half from the initial stock solutions) to the diluted blood except the control tube where 100 μL of phosphate buffer (pH 7.4) was added. The mixture was allowed to stand for 4 h. Thereafter, 0.9 mL of distilled water was added to 0.1 mL of the treated sample. Then, 2.5 mL of phosphate buffer (pH 7.4) was added to 0.5 mL of the mixture followed by the addition of 2 mL of 0.2 M hydrogen peroxide (H2O2). It was mixed thoroughly and 1 mL of dichromate/acetic acid solution was added at the interval of 1 min for 3 min. It was incubated in water bath for 10 min at 80°C. The absorbance was read at 570 nm.
Determination of reduced glutathione concentration
The reduced glutathione was determined according to Ellman’s method. 13 Here, 100 µL of the blood sample was diluted with 100 µL of the phosphate buffer (pH 7.4) followed by the addition of 100 μL of each drug (0.5–8.0 mg/mL; the final concentration of drugs was reduced by half from the initial stock solutions) to 100 µL of the blood except to the control tube where 100 µL of the phosphate buffer saline (pH 7.4) was used. These were allowed to stand for 4 h. A total of 1.5 mL of 10% trichloroacetic acid was added to all tubes containing 150 µL of the mixture and centrifuged at 1500g for 5 min. Then, 0.5 mL Ellman’s reagent was added to 1 mL of the supernatant, followed by the addition of 3 mL of 0.2 M phosphate buffer (pH 8.0). The absorbance was read at 412 nm.
Determination of malondialdehyde concentration
This was carried out according to the method of Ohkawa and Ohishi. 14 Here, 100 µL of the blood sample was diluted with 100 µL of the phosphate buffer (pH 7.4) followed by the addition of 100 μL of each drug (0.5–8.0 mg/mL; the final concentration of drugs was reduced by half from the initial stock solutions) to all tubes except the control which contained 100 μL of phosphate buffer (pH 7.4). They were incubated for 4 h. Thiobarbituric reagent (2 mL) was added to 150 μL of the mixture and incubated in water bath for 1 h at 90°C and allowed to cool. It was later centrifuged at 3000g for 5 min and the supernatant was collected. The absorbance of the pink supernatant (tert-butyl alcohol-malondialdehyde (MDA) complex) was measured at 535 nm. MDA formed was then calculated using the molar extinction coefficient of 1.56 × 105 cm−1 M−1
Statistical analysis
To address the biological variability and stability of the samples, each and every experiment was repeated at least three times and the results were expressed as mean ± standard deviation. Differences between the groups were analysed by Student’s t-test for comparison with the aid of Statistical Package for Social Sciences (SPSS) software, standard version 20.0 (SPSS Inc., Chicago, Illinois, USA). Values of p < 0.05 were considered significant for differences in mean.
Results
Determination of total proteins revealed (Figure 1(a)) that all the drugs caused a dose dependent and significant (p < 0.05) increase in the total proteins level as compared with control in all the concentration used except for the artemisinine (at 0.5 and 1 mg/mL), artesunate (at 0.5 mg/mL) and chloroquine (at 0.5 and 1 mg/mL), respectively. Contrariwise, there was a significant (p < 0.05) reduction on the proteins level when compared with untreated samples but within the treatments group, a dose-dependent increase was observed (Figure 1(b)). Comparatively, the total proteins level seems to be higher in the infected blood samples than the apparently healthy blood samples.

Effects of commonly used antimalarial drugs on total protein ex vivo. (a) Blood of apparently healthy humans; (b) malaria-infected blood. *p < 0.05: statistically significant as compared with the control using Student’s t-test.
A significant (p < 0.05) decrease in the activity of superoxide dismutase (SOD) was observed in the apparently healthy treated samples (Figure 2(a)) relative to control, except in few cases where by artemisinine and chloroquine (at 0.5–2 mg/mL), artesunate and coartem (at 0.5–1 mg/mL) and quinine (at 0.5 mg/mL) differ not significantly (p > 0.05) as compared with the control. Interestingly, the activity (Figure 2(b)) was significantly (p < 0.05) increase with the decrease in drugs concentration when compared with untreated samples. Comparatively, the activity was on the trend of A ≥ B.

Effects of commonly used antimalarial drugs on SOD activity ex vivo. (a) Blood of apparently healthy humans; (b) malaria-infected blood. *p < 0.05: statistically significant as compared with the control using Student’s t-test. SOD: superoxide dismutase.
All the drugs dose dependently decreased (p < 0.05) the catalase activity (Figure 3(a)). However, in Figure 3(b), a significant (p < 0.05) increase in the activity when compared with untreated samples was observed. Comparatively, the activity was on the trend of A ≥ B.

Effects of commonly used antimalarial drugs on catalase activity ex vivo. (a) Blood of apparently healthy humans; (b) malaria-infected blood. *p < 0.05: statistically significant as compared with the control using Student’s t-test.
As depicted in Figure 4(a), the level of reduced glutathione dose dependently (p < 0.05) increase as compared with control, whereas similar observation was made in Figure 4(b) except the higher glutathione level in the untreated sample than the treated ones.

Effects of commonly used antimalarial drugs on reduced glutathione level ex vivo. (a) Blood of apparently healthy humans; (b) malaria-infected blood. *p < 0.05: statistically significant as compared with the control using Student’s t-test.
All drugs significantly (p < 0.05) and dose dependently caused decreases in MDA level (Figure 5(a)) as compared with control. Conversely, in Figure 5(b), the level increases with the increase in drugs concentration approaching the control level. Comparatively, the induction trend was in the order of B ≥ A.
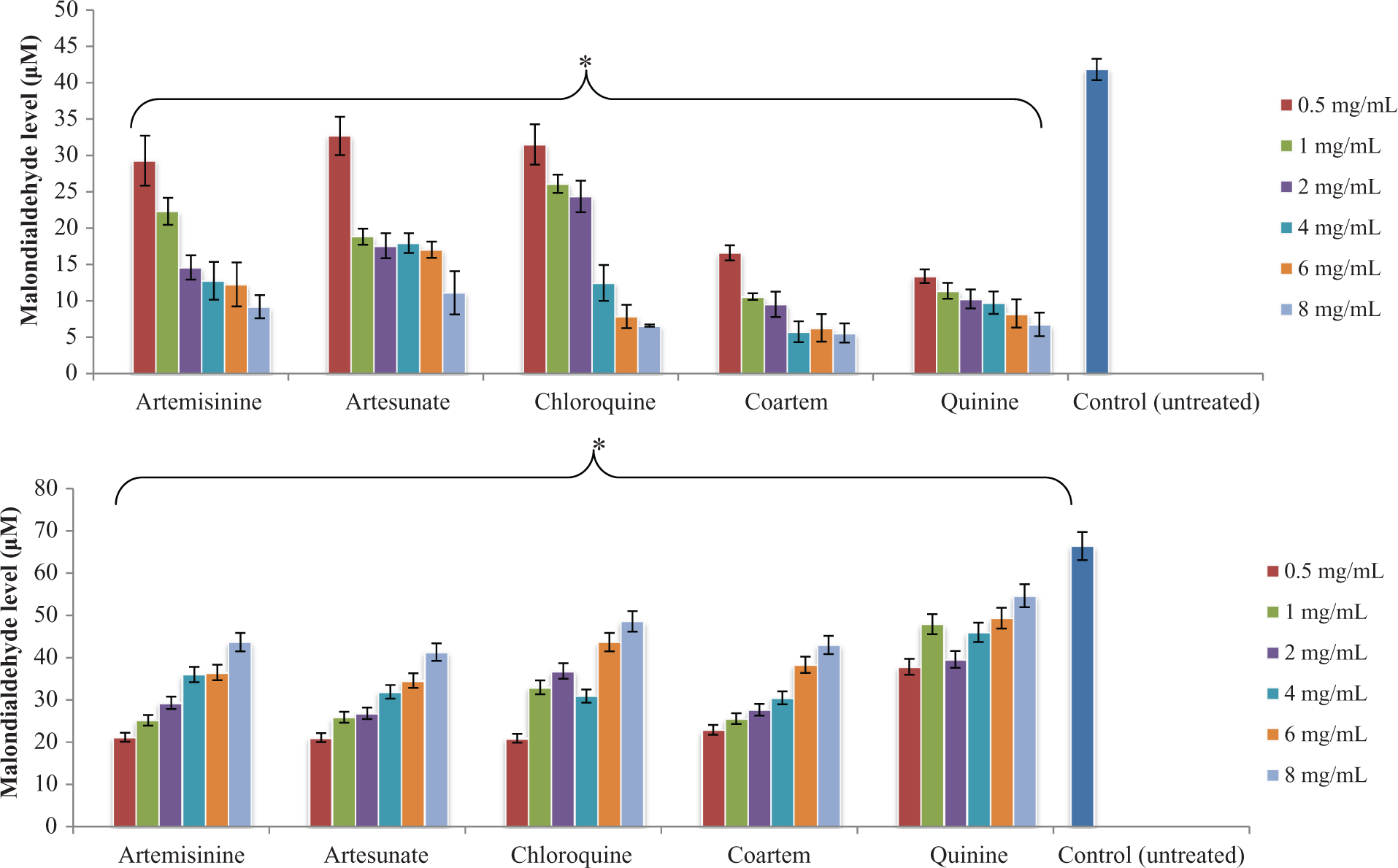
Effects of commonly used antimalarial drugs on MDA level ex vivo. (a) Blood of apparently healthy humans; (b) malaria-infected blood. *p < 0.05: statistically significant as compared with the control using Student’s t-test. MDA: malondialdehyde.
Discussion
Apart from their medicinal properties, health-promoting or disease prevention ability, antimalarial drugs can be very toxic to the body having a negative effect on the biological system leading to malfunctioning of some vital tissues and organs. 9 One of the mechanisms that pave way for the toxicity is oxidative stress. This is because most of these drugs by their pharmacodynamics nature may induce tissue damage directly and/or indirectly by generating an electrophilic reactive intermediates or respiratory burst characterized by ROS generations. 15 In this study, we report the alteration of redox status by commonly used antimalarial drugs in the north-western region of Nigeria.
Our findings indicate that the activity of SOD was low in normal blood samples and high in the infected samples. Moreover, the activity of catalase was reduced in healthy subjects and elevated in the malarial infected, whereas reduced glutathione level was elevated in healthy subjects and depleted in the infected patients. MDA level was dose dependently observed to be low in normal and high in infected blood due to antimalarials. In both the cases, the MDA levels were less than that of control. Alteration of redox status has been implicated during malarial infection and application of antimalarials that specifically alleviate plasmodium-induced oxidative stress and its associated complications largely yielded a desired outcome. 9,16 However, above the therapeutic dose, which may consistently induce ROS generation not only in the red blood cells but other lineages like macrophages and neutrophils, the ultimate results could be fatally characterized by molecular and cellular damages around the microenvironment and other distant vital tissues. 17 –19 Overall, these biochemical events could further amplify the intensity of an oxidative burst leading to fatal consequences on macromolecules in both the host and parasite. Phagocytes respond to particulate or soluble stimuli by the oxidative burst associated with increased production of ROS. The NADPH oxidase located in the plasma membrane and in the membranes of specific granules produces superoxide anions from which other free radicals arise. The ROS are released into the surrounding medium or into a membrane-enclosed subcellular organelle. 15 Dismutation of O2·−, either spontaneously or catalysed by SOD, results in the formation H2O2, which acts as a substrate for the myeloperoxidase system and this H2O2 converts to hypochlorous acid, 20 the latter may contribute to the observed alteration of redox status seen in the current study. In addition, several antimalarial drugs have been shown to influence the biochemical environment within and around the plasmodium-infected erythrocytes with variable outcomes. 21 Furthermore, antimalarials have also been implicated to cause haemolysis which invariably leads to release of haemoglobin and ultimately iron due to haem catabolism. The iron released (substrate for Fenton and Haber Weiss reactions) generates dangerous radicals like OH· that has far reaching effects on cellular materials. 16,22 This might explain the high level of MDA observed in the infected blood samples (untreated) as against the treated samples because OH· generated due to inability of the catalase to reduce H2O2 released by SOD-based reaction or via other oxidative means is key to lipid peroxidation. The observed decrease in the MDA level of the treated samples is a clear indication of possible antioxidant properties of antimalarials. 23,24 Overall, these may likely be the reason behind the reduction of SOD and catalase activities as well as MDA level in the present study because oxidative stress is a consequence of malarial pathogenicity. 6,8
Conclusion
Commonly used antimalarial drugs altered the redox status by inducing oxidative stress which may have a cellular and molecular consequence depending on the regulatory mechanism in place. In addition, the drugs could be biotransformed to reactive intermediates having far-reaching effects on the cellular materials. However, the effects are dose dependent and more pronounced in non-malarial conditions compared to otherwise. Hence, adequate care should be taken during prescription with total avoidance for self-medications and/or drugs abuse as a result of their adverse effects within the red blood cells and its immediate microenvironment.
Footnotes
Acknowledgement
We appreciate the consistent laboratory assistance by Messrs. Reuben Baba, John Shaffa and Bashir Musa at the Department of Biochemistry, Ahmadu Bello University, Zaria.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was fully funded by the authors.
