Abstract
Objectives:
Clinical application of gentamicin may cause nephrotoxicity and ototoxicity. Our study is the first study to investigate the protective effects of edaravone against the gentamicin-induced ototoxicity. We investigated the protective effect of intraperitoneal (i.p.) edaravone application against gentamicin-induced ototoxicity in guinea pigs.
Methods:
Fourteen guinea pigs were divided into two equal groups consisting of a control group and a study group. One-hundred sixty milligrams per kilogram subcutaneous gentamicin and 0.3 mL i.p. saline were applied simultaneously once daily to seven guinea pigs in the control group (group 1). One-hundred sixty milligrams per kilogram gentamicin was applied subcutaneously and 3 mg/kg edaravone was applied intraperitoneally once daily for 7 days simultaneously to seven guinea pigs in the study group (group 2). Following the drug application, auditory brainstem response measurements were performed for the left ear on the 3rd and 7th days.
Results:
Hearing threshold values of the group 1 and group 2 measured in the 3rd day of the study were detected as 57.14 ± 4.88 and 82.86 ± 7.56, respectively. This difference was statistically significant (
Conclusion:
A statistically significant difference between the average threshold values of edaravone-administered group 2 and that of group 1 without edaravone was found. These differences show that systemic edaravone administration could diminish ototoxic effects of gentamicin and the severity of the hearing loss.
Introduction
Aminoglycosides are highly effective antimicrobial agents. The most frequently used aminoglycosides are gentamicin, streptomycin, tobramycin, and amikacin. These are used for severe infections caused by aerobic gram-negative bacteria and
Reactive oxygen species (ROS) are involved in the etiologies of many pathological conditions, such as ototoxicity, cardiovascular disease, acoustic trauma, aging, and infection. 2,3 ROS scavengers have been shown to protect the inner ear from the detrimental effects of aminoglycosides, cisplatin, bacterial toxin, and acoustic trauma. 4 –7 Edaravone (MCI-186, 3-methyl-1-phenyl-pyrazo-line-5-one), a free radical scavenger, has already been used in clinical treatment of acute myocardial infarction, rheumatoid arthritis, and ischemic cerebral infarction. 8 –10
Our study is the first study to investigate the protective effects of edaravone against the gentamicin-induced ototoxicity. The aim of this study was to determine whether edaravone could be used to prevent cochlear damage caused by gentamicin.
Material and method
Chemicals and animals
Gentamicin and edaravone were dilute in sterile phosphate-buffered saline (PBS) solution.
A total of 14 guinea pigs were used.
Experimental design
The study was approved by the local ethic committee. Male albino adult guinea pigs (
ABR measurements were made to evaluate the initial level of hearing and were recorded. Left pinna (negative), right pinna (positive), and vertex (ground) subdermal needle electrodes were placed. Auditory brainstem potentials were used to warn of click stimuli (stimulus rate: 31 interstimulus intervals: 12 min). Evoked potentials were amplified, filtered between 50 and 2500 Hz, and their averages of 2000 sweeps (Amplaid Mk 10, Multisensory System Machine; Milan, Italy) were recorded. All audiometric data obtained were collected by an audiometrist who had no information about the groups.
Experimental procedure
Animals (14 guinea pigs) were divided into two groups as follows:
Control group (
ABR was evaluated by determining the hearing threshold changes before the first injection and after 3 and 7 days injection of gentamicin or a mixture of gentamicin with edaravone. Before each measurement, tympanic membranes and middle ears were inspected through an operating microscope (Zeiss, Germany). After the completion of the study, all animals were killed.
Statistical Analysis
Data from this study have been expressed as mean ± standard deviation minimum and maximum values: SPSS (version 13) statistics package program was used for calculations. Mann–Whitney
Results
Table 1 shows the ABR thresholds for click stimuli before and after drug administration for each guinea pig.
ABRs thresholds before and after treatment.

C: control animal; S: study animal; dB: decibel; dB SPL: dB of sound pressure level; ABR: auditory brainstem response.
The mean ABR thresholds on the 3rd and 7th days in the group 1 were significantly higher than pretreatment hearing thresholds (
ABR threshold data within the group 1 (with gentamicin + saline).

SD: standard deviation; ABR: auditory brainstem response.
aWilcoxon test.
The mean ABR thresholds on the 3rd and 7th days in the group 2 were significantly higher than those recorded before the treatment thresholds (
ABR threshold data within the group 2 (with gentamicin + edaravone).
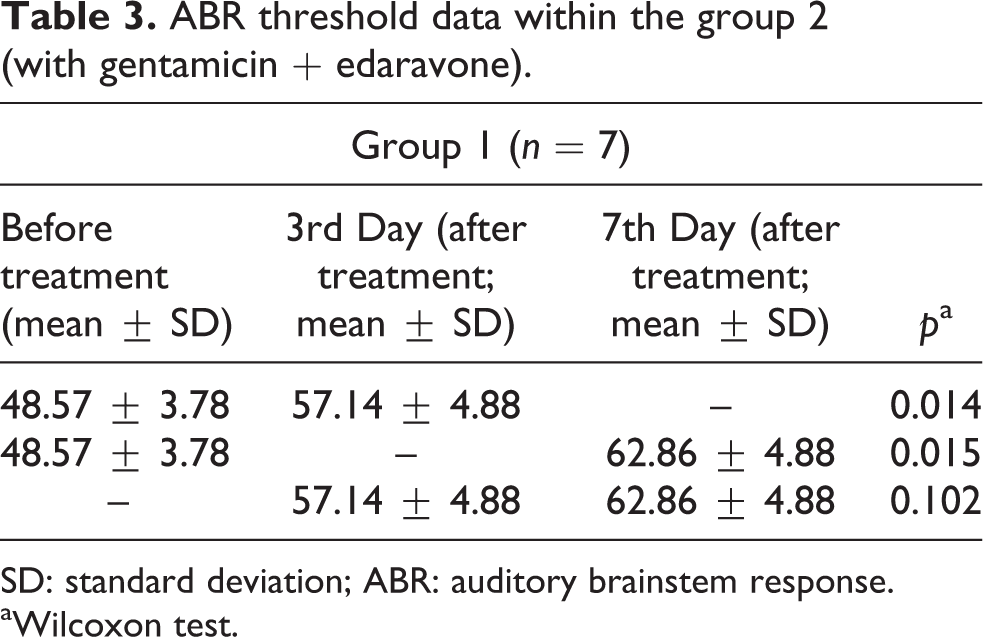
SD: standard deviation; ABR: auditory brainstem response.
aWilcoxon test.
When pretreatment ABR thresholds were compared between the two groups, there was no significant difference (
Comparison of ABR threshold data between group 1 (with gentamicin and saline, control group) and group 2 (with gentamicin and edaravone).

SD: standard deviation; ABR: auditory brainstem response.
aMann–Whitney
Discussion
Aminoglycoside antibiotics enjoy widespread clinical use, but a clinical limitation to the use of these drugs is ototoxicity. Aminoglycoside concentrations in the inner ear have been considered to correlate closely with ototoxicity. It has been suggested that aminoglycosides are ototoxic because they generate free radicals. 11 The generation of ROS is believed to be the initiating step of aminoglycoside ototoxicity, followed by a cascade of events that ultimately results in programmed cell death. 12 –15 Evidence is accumulating that generation of ROS is an important factor in inner ear damage. 16 –18 Aminoglycoside ototoxicity is considered to be mediated by the formation of an aminoglycoside–iron complex. Free radicals can attack cell membranes, proteins, and DNA, causing irreparable damage and ultimately cell death. 15,19 ROS, including superoxide radicals and hydroxyl radicals, can deplete cochlear tissues of antioxidant protective molecules. This may allow lipid peroxidation that can increase calcium influx and apoptosis in cells of the cochlea. 20 Studies on the localization of aminoglycosides in the inner ear have been performed, showing that gentamicin is selectively taken up by hair cells especially basal coil hair cells. Hayashida et al. demonstrated that type I hair cells are more sensitive to aminoglycoside than type II hair cells. 21 –23
Edaravone is a neuroprotective agent with potent free radical scavenging and antioxidant actions that do not cause any serious side effects.
24
Asplund et al. and Takumida and Anniko reported that treatment with edaravone (3 mg/kg) has a protective effect against
Because of toxic effect on the cochlea, our study used gentamicin. Gastrointestinal absorption is not good, thus general use local or parenteral application is used. These uses may cause toxicity to the inner ear. Parenteral administration was preferred in our study. Dorman et al. used a dose range from 10 to 160 mg/kg to investigate aminoglucoside ototoxicity in rats. 28 In our study, a unique dose of 160 mg/kg gentamicin was sufficient to induce ototoxicity all guinea pigs tested. At this dose, no statistically significant difference was observed in the ototoxicity severity between early period and the late period in group 1.
In addition, due to its low molecular weight (174.2) and its fat- and water-soluble nature, cell membrane permeability of edaravone is very good. 29 Despite its gradual decrease in its pharmacological effect, edaravone activity remains stable for a period of 24 h after its dissolution in saline. 30 As shown in our study, systemic administration of edaravone was found to be protective against gentamicin ototoxicity.
Effects of edaravone has been demonstrated in various studies.
6,18,25,30
It has been shown that edaravone is an effective protective agent of cochlear function against
As shown in Table 4, both the groups were similar in relation to auditory functions in the beginning. Mean threshold of hearing values was significantly higher in the group 1 than the group 2 in days 3 and 7. This result supports the preventive effects of edaravone against gentamicin ototoxicity. In addition, we have noticed that mean hearing threshold values of the group 1 measured in 3rd and 7th days were higher than the thresholds measured before the treatment. This confirms the cochlear damage related to gentamicin with gentamicin administration in animals. Compared to the threshold values observed before treatment, high level of mean hearing threshold observed in the group 2 suggests that edaravone does not completely prevent cochlear damage related to gentamicin despite the significant reduction of its severity. Preventive effect of edavarone was reported only in one study where the use of tobramycin and edaravone protect rats against hearing loss with ABR. 25
Edavorone is known as antioxidant and non-ototoxic agent. In order to avoid unnecessarily killing of animal, we did not test this compound alone on animals for its potential effects. This could constitute a limitation for our study.
Conclusion
The results of our study support that antioxidant effects of edaravone decrease the severity of gentamicin toxic effects in inner ear and the severity of hearing loss. Simultaneous administration of edaravone with aminoglycosides and other ototoxic agents commonly used in clinical practice could reduce ototoxic side effects. Experimental animal studies with increased number of cases and placebo control human studies are needed to confirm the benefit of mutual administration of edaravone with ototoxic agents.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
