Abstract
Triptolide (TPL) is a main active compound isolated from Tripterygium wilfordii Hook f. Despite its positive therapeutic effect, the female reproductive toxicity of TPL is still the bottleneck of clinical application. The study was designed to investigate the adverse effects on mice ovary and underlying mechanism of TPL. Adult female NIH mice were treated with two therapeutic doses of TPL (25 and 50 μg/kg/d) for 50 days, respectively. Mice estrous cycle was detected by vaginal cytology method. Half mice from each group were selected randomly to perform superovulation. Quality and quantity of ovulated eggs were evaluated. Other mice from each group were executed for morphological study. Ovarian histological sections were stained by H&E staining for ovarian pathologic detection and follicular counts. Apoptotic granulosa cell (GC) was detected by terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay. Endoplasmic reticulum (ER) stress-related proteins and antiapoptotic X-linked inhibitor of apoptosis protein (XIAP) were detected by immunohistochemical method. Two doses of TPL resulted in estrous cycle disorder and follicles in development reservoir impairment. Quality and quantity of mice ovulated eggs significantly decreased after TPL treatment. Ovarian pathologic examination revealed TPL-induced TUNEL-positive GCs increase and ER stress–related proteins (78-kDa glucose-regulated protein, p-protein kinase-like endoplasmic reticulum kinase, p-eukaryotic initiation factor 2α, and CCAAT/enhancer binding protein homologous protein) expression upregulation. Meanwhile, the expression of antiapoptosis protein XIAP in mice ovary was obviously inhibited by TPL. Our results may demonstrate that therapeutic doses of TPL can injure ovary function, but there is no difference between high-dose and low-dose groups. GCs apoptosis by ER stress pathway and antiapoptotic function impairment may partly mediate TPL-induced ovary toxicity.
Introduction
Tripterygium wilfordii Hook f, extracted from the root xylem of T. wilfordii, was used in traditional Chinese medicine for decades, to treat various rheumatoid arthritis, 1 nephritis, 2 –4 skin disorders, 5 and so on. Triptolide (TPL) is a kind of main potent, biologically active compound, isolated from T. wilfordii Hook f. It is worth noting that T. wilfordii Hook f. tablet has a narrow therapeutic window in clinical. One of their significant adverse effects is ovarian toxicity for females, which manifested as menstrual disorders, amenorrhea, infertility, and so on. 6,7 In previous studies, it is confirmed that the toxic target organ of T. wilfordii Hook f is ovary, 8 indicated by high follicle-stimulating hormone (FSH) acidosis, low estrogen acidosis, and premature ovarian failure. 9 TPL was reported to induce decrease in expression of estrogen receptor α in ovaries. 10 Although the side effects of T. wilfordii Hook f and TPL were reported, the mechanism of their ovary toxicology was understood poorly.
Ovarian follicle development is a process regulated by various factors. 11,12 After puberty, most follicles fail to mature, but undergo the atresia process. As an essential cell for follicular growth, granulosa cell (GC) is becoming the focus of research in recent years. 10,13 GC is a somatic cell of the sex cord that is closely associated with the developing female gamete in the ovary of mammals. Its main functions include producing sex steroids and providing nutrition for oocyte. 14,15 Under various pathologic conditions, GC was considered as an initiator of follicular atresia, which underwent apoptosis in atretic follicles were earlier than oocytes and theca cells. 16
Although accumulated in vitro data confirmed TPL-reduced GCs hormone synthesis and secretion function, 13 the role of GC apoptosis in follicular atresia is unknown. In this study, we mainly focused on the female toxic effect of TPL on ovary and related mechanism.
Materials and methods
Chemicals and reagents
TPL, gonadotropin from pregnant mare serum (PMSG), human chorionic gonadotropin (hCG) were purchased from Sigma-Aldrich Co. (St Louis, Missouri, USA). DeadEndTM Fluorometric terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) system was ordered from Promega Corporation (Madison, Wisconsin, USA). Anti-78-kDa glucose-regulated protein (GRP78) and anti-phospho-eukaryotic initiation factor (eIF) 2α antibodies were ordered from Abcam Ltd (Hong Kong), anti-phospho-protein kinase-like endoplasmic reticulum kinase (PERK), anti-CCAAT/enhancer binding protein homologous protein (CHOP), and anti-X-linked inhibitor of apoptosis protein (XIAP) and all secondary antibodies were obtained from Cell Signaling Technology Inc (Boston, Massachusetts, USA).
Animals and treatments
Thirty-six adult female NIH mice (25g ± 5 g, 6–7 weeks) were obtained from Laboratory Animal Center of Southern Medical University (Guangzhou, China). All animal experiments were approved by the University of Guangzhou Traditional Chinese Medicine University Animal Ethics Committee and were conducted according to the University of Guangzhou Traditional Chinese Medicine University Guide for animal experiments. All mice were maintained with 12-h light/12-h dark cycle and free access to food and water. TPL (purity > 99%) was obtained from Sigma-Aldrich and was dissolved in dimethyl sulfoxide (DMSO) before use.
Mice were divided into three groups of 12 animals each randomly. Control group mice were gavaged vehicle (5% DMSO in saline) for 50 days. The low-dose and high-dose TPL groups were intragastrically administered two therapeutic doses of TPL (25 and 50 μg/kg/d) for 50 days, respectively. 17
To determine the effects of the TPL on ovarian function, mice estrous cycle was detected during the last 10 days. Half of the mice from each group were selected randomly and superovulation was performed. Animals were anesthetized with 3.5% (350 mg/kg) chloral hydrate (Sangon Biothch, Shanghai, China) by intraperitoneal injection, before ova harvest process. Superovulated eggs were removed from oviducts, counted, and assessed for maturity and ovulation rate. The mice (excluding mice treated for superovulation) were anesthetized with 3.5% chloral hydrate (350 mg/kg intraperitoneally), and then killed by craniocervical dislocation. After killing, animals were weighed, after that bilateral ovaries were collected and detected.
Estrous cycle detection
Vaginal cytology method was applied in this study. A vaginal swab was collected using cotton-tipped swab, wetted with bacteriostatic sterile saline and inserted into the vagina of mouse. After rolling against the vaginal wall with cotton-tipped swab, we pulled it out quickly. Immediately, the cells were smeared to a dry glass slide by rolling the swab on the slide. The slide was air dried and then fixed in 4% neutral formalin. Vaginal smear slides were stained by papanicolaou staining. The slides were viewed at 200× magnification. The estrous cycle was determined based on the presence or absence of cornified epithelial, leukocytes, and nucleated epithelial cell according to Felicio et al. 18
Superovulation
Six female mice from each group were selected randomly and superovulated. Briefly, mice were intraperitoneally injected with 10 IU PMSG (Sigma-Aldrich) on day 47. 48 hours after PMSG injection, ovulation was induced by injecting the mice with 5 IU hCG (Sigma-Aldrich). The mice were anesthetized 16–18 h after hCG administration. After anesthesia and killing, ovulated oocytes were isolated from the oviductal ampullae into Toyoda 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid medium containing 0.4% bovine serum albumin (BSA) and were counted immediately. 19
Ovarian follicular counts
Collected ovaries were immediately fixed overnight in 4% neutral formalin at 4°C and processed for paraffin embedding for histological analysis. The method was modified as reported by Patrick J et al.. 20 Twelve pieces of serial sections (5 μm) were deparaffinized, rehydrated, and stained by hematoxylin and eosin, dehydrated, coverslipped, and allowed to dry. The structure of follicles was observed by a Nikon light microscope (Nikon 80i, magnification: 200×, Nikon Instruments Inc; Japan). Ovarian follicular count and classification according to the previously described histomorphometric techniques, primordial (single layer of flattened GCs), primary follicle (single layer of cuboidal GCs), secondary follicle (2–4 GC layers), and antral (>4 GC layers with distinct antrum visible) follicles with visible nucleoli were performed on every fifth section in a blinded fashion. Numbers of oocyte-containing follicles at each developmental stage were classified and counted in every 12th section in ovaries.
TUNEL assay
Ovary sections embedded in paraffin were cut at 5 μm thickness. TUNEL assay was performed according to DeadEndTM Fluorometric TUNEL System instruction, apoptosis detection kit as per the manufacturer’s instructions. Simply, the slides were immersed in 4% formaldehyde in PBS for 25 min at 4°C. After rinsing with PBS twice, slides were immersed in 0.2% Triton® X-100 for 5 min to permeabilize. After rinsing with PBS twice, 100 μL equilibration buffer was added for 10 mins. Then 50 μL terminal deoxynucleotidyl transferase reaction mix (50 μL PBS for negative control group) was added to the tissue and the slides were incubated for 60 min at 37°C. After labeling the reaction, 2× soluble sodium citrate (SSC) was used to stop reaction. After rinsing with phosphate-buffered saline (PBS), 4′,6-diamidino-2-phenylindole (DAPI) was added to counterstain nuclei. Fluorescent images were acquired by fluorescence microscopy (original magnification, 100×). TUNEL-positive and DAPI-stained cells quantified using Image J software (Version No.: 1.48u) to minimize subjectivity in manual cell counting. Apoptotic index was calculated as TUNEL-positive cells per thousand GCs. 21
Immunohistochemistry detection
After deparaffinization, hydration, and antigen retrieval in 10 mM sodium citrate, the sections were blocked with block buffer (10% goat serum and 5% BSA in PBS) at room temperature for 1 h and incubated with primary antibodies (anti-GRP78, anti-phospho-PERK, anti-phospho-eIF2α, anti-CHOP, and anti-XIAP) as described at 4°C overnight, followed by peroxidase blocking with 3% hydrogen peroxide for 10 min and incubated with HRP-conjugated secondary antibodies at room temperature for 50 min. Fresh diaminobenzidine (DAB) was prepared and applied for 15 s. After counterstaining with hematoxylin, sections were dehydrated and mounted. 22 Images were captured using Nikon light microscope. Images from randomly selected fields of each sample were examined using light microscope (Nikon 80i, Nikon Instruments Inc) and pictured by a digital camera (Nikon DS-5Mc, Nikon Instruments Inc). All images were obtained by 10× objective lens. The positive follicle area% was captured and then calculated by NIS Element BR software (Nikon Instruments Inc.)
Statistical analysis
Data are presented as mean ± SD. Analysis of variance followed by Student–Newman Keul’s test and t test were used to analyze the significance of differences between groups. A value of p < 0.05 was considered statistically significant.
Results
Mice ovarian pathologic change induced by TPL
As shown in Figure 1(a) (Gross anatomy, white arrow denotes ovary), ovary in the control group was smooth, usually with pink exterior. However, the surface of TPL (two doses)-treated mice ovary looked pale, shrunk, solid, and rough, which was like postmenopausal ovary (Figure 1(a), low-dose (25 μg/kg/d) TPL treatment group (TPL L) and high-dose (50 μg/kg/d) TPL treatment group (TPL H) groups).

Ovarian histological changes of two doses TPL-treated mice compared with mice in the control group. (a) Representative gross anatomy of mouse ovary in each group. White arrow denotes ovary. (b) Representative H&E staining photomicrographs (original magnification, ×40) of mice ovary in each group. Black arrow denotes follicles, CL. TPL: triptolide; H&E: hematoxylin and eosin; CL: corpus luteum; TPL L: low-dose (25 μg/kg/d) TPL treatment group; TPL H: high-dose (50 μg/kg/d) TPL treatment group.
The histological analyses of ovaries showed that there were follicles of different periods distributed in mice ovary of control group, which included primordial follicle, primary follicle, secondary or preantral follicle, and mature follicle (Figure 1(b), control group). Numerous primordial follicles and primary follicles were mainly observed in cortical areas of mice ovary. Contrary to the control group, developing follicles reduced significantly, but atresic follicles and corpus luteums (CLs), especially the latter, were observed everywhere and looked bigger in TPL (both of high- and low-dose groups)-treated mice ovary slides (Figure 1(b), TPL L and TPL H groups, CL). The CL is a temporary endocrine structure in mammal ovary, which is generated during the luteal phase of estrous cycle by ovarian follicles.
TPL-induced follicles in development reservoir impairment
As we know, folliculogenesis is a continuous process, which occurs throughout the reproductive life. As shown in Figure 2, after treatment with TPL for 50 days, mice ovaries contained reduced follicle populations in developmental stages, especially for primary follicles (control vs. TPL L vs. TPL H: 56 ± 10 vs. 16 ± 3 vs. 17 ± 7, compare with control group, p < 0.01) and secondary follicles (control vs. TPL L vs. TPL H: 47 ± 16 vs. 12 ± 10 vs. 15 ± 7, compare with control group, p < 0.01). Mice primordial follicles and mature follicles populations also decreased after exposure to TPL (primordial follicles: control vs. TPL L vs. TPL H: 72 ± 8 vs. 39 ± 8 vs. 42 ± 3; mature follicles: control vs. TPL L vs. TPL H: 72 ± 8 vs. 39 ± 8 vs. 42 ± 3; compare with control group, p < 0.05) too. Number of atresia follicle from TPL-treated mice increased (control vs. TPL L vs. TPL H: 16 ± 5 vs. 20 ± 5 vs. 22 ± 8), but there was significant difference between high- and low-dose groups. These results implied that TPL inhibited follicle growth, mutation, and promoted follicular atresia.

TPL depleted mice ovarian follicles reservoir. The number of ovarian follicles at various developmental stages after TPL treatment was shown in this columnar statistical figure. Data are reported as mean ± SD. n = 6 mice each group, *p < 0.05; **p < 0.01, compared with the control group, by t test. TPL: triptolide; TPL L: low-dose (25 μg/kg/d) TPL treatment group; TPL H: high-dose (50 μg/kg/d) TPL treatment group.
Estrus change induced by TPL
The murine estrous cycle is approximately 4.5 days. Vaginal cells were examined and the relative number of each cell type is determined by papanicolaou staining. For example, nucleated epithelial cells and part of cornified epithelial cells implied the mouse in proestrus (Figure 3(a), proestrus). Numerous orange cornified epithelial cells indicate that the mouse was in estrus (Figure 3(a), estrus, triangle means nucleated epithelial cell). Leukocytes, few nucleated epithelial cells, and cornified epithelial cells are all present on vaginal smears in metestrus (Figure 3(a), metestrus, rhombuses means cornified epithelial cell). Leukocytes are mainly present in diestrus (Figure 3(a), diestrus, arrow represents leukocytes). As shown in Figure 3(b), compared with estrus cycle in the control group mice, the length of cycle in TPL (both of high and low doses) group mice was longer notably (control vs. TPL L vs. TPL H: 4.4 ± 0.5 vs. 6.3 ± 1.3 vs. 6.4 ± 1.5 days, compare with the control group, p < 0.05). But there was difference in length of cycle between high-dose and low-dose groups. Although TPL extended the estrous cycle of mice, it shortened the estrous phase of mice (data not shown).

TPL-induced mice estrous cycle change. (a) Representative papanicolaou staining of mice vaginal exfoliated cells in different stages of estrous cycle. Triangles mean nucleated epithelial cells, rhombuses mean cornified epithelial cells and arrows denote leukocytes. (b) Length of mice estrous cycle in each group. Data are reported as mean ± SD. n = 6 mice each group, *p < 0.05, compared with the control group, by t test. TPL: triptolide; TPL L: low-dose (25 μg/kg/d) TPL treatment group; TPL H: high-dose (50 μg/kg/d) TPL treatment group.
Adverse effects of TPL on mouse ovulation function
After superovulation, eggs in oviduct were separated and observed by light microscopy. As shown in Figure 4, lots of bright and fertile eggs were observed in the control group (Figure 4(a), control group). Nevertheless, the size of superovulated eggs from TPL-treated mice was significantly smaller than that in the control group mice, especially for high-dose TPL group (Figure 4(a)). In terms of quantity, the number of eggs sharply reduced after treatment of TPL (control vs. TPL L vs. TPL H: 52.3 ± 6.6 vs. 18.8 ± 6.7 vs.18.2 ± 2.2 per oviduct). The results showed that both the low and high doses of TPL injured the mouse ovulation function seriously. But there was difference in the number of superovulated eggs between high-dose and low-dose groups.

TPL seriously injured the mice ovulation function. (a) Representative superovulated eggs (black arrows denote) photographs of mice ovary in the control group and TPL-treated group. (b) Superovulated egg number in each group mice. Data are reported as mean ± SD, n = 6 mice each group, **P<0.01, compared with the control group, by t test. TPL: triptolide; TPL L: low-dose (25 μg/kg/d) TPL treatment group; TPL H: high-dose (50 μg/kg/d) TPL treatment group
Apoptotic GCs induced by TPL
Apoptosis in ovarian follicles was assessed in histological sections by TUNEL assay. As shown in Figure 5, there were no apoptotic GCs within the ovarian follicles of the control group. Positive TUNEL reaction mainly occurred in CL in ovaries of the control group. Compared with the control group, confocal fluorescence microscopy revealed more TUNEL-positive reaction mainly in primary follicles and secondary follicles after TPL (both of high- and low-dose groups) treatment, mostly in GCs area. Compared with 19.3 ± 6.3‰ in the control group, the apoptotic index of GC reached 74.0 ± 12.0‰ and 82.8 ± 7.1‰ in GCs from low-dose and high-dose TPL-treated mice, respectively. But there was significant difference between high- and low-dose TPL groups.

TPL-induced mice ovary apoptotic granulosa cells increase. (a) Representative TUNEL-positive and (green) GC and total cell (DAPI, blue) stained ovary sections showing multiple follicles (original magnification, ×200, white cycle denotes follicles, CL.). (b) Apoptotic index of GCs from the control, TPL L and TPL H groups. Data are reported as mean ± SD. n = 6 mice each group, **p < 0.01, compared with the control group, by t test. TPL: triptolide; DAPI: 4′,6-diamidino-2-phenylindole; CL: corpus luteum; TPL L: low-dose (25 μg/kg/d) TPL treatment group; TPL H: high-dose (50 μg/kg/d) TPL treatment group; GCs: granulose cells.
ER stress involved in TPL-induced GCs apoptosis
To determine the possible mechanism of GCs apoptosis induced by TPL, we analyzed GC expression of GRP78, phospho-PERK (p-PERK), and phospho-eIF2α (p-eIF2α), and CHOP proteins. PERK is an eIF2α kinase and is implicated in endoplasmic reticulum (ER) stress-induced cell death, partly through upregulation of proapoptotic CHOP. As shown in Figure 6, all of GRP78, p-PERK, p-eIF2α, and CHOP proteins’ expressions were elevated in TPL (both of high- and low-dose groups)-treated mice GCs, especially for secondary follicles and mature follicles (Figure 6(a), the circle represents positive expression follicle of GRP78, p-PERK, p-eIF2α, and CHOP). Some proteins’ (GRP78, phospho-eIF2α, and CHOP) expressions were upregulated in mice CL too. There was, however, no difference in above proteins’ expression between high-dose and low-dose groups. The results demonstrated that the branch of ER stress, GRP78-PERK-eIF2α-CHOP signal pathway was activated in this model and may mediate TPL-induced GC apoptosis.
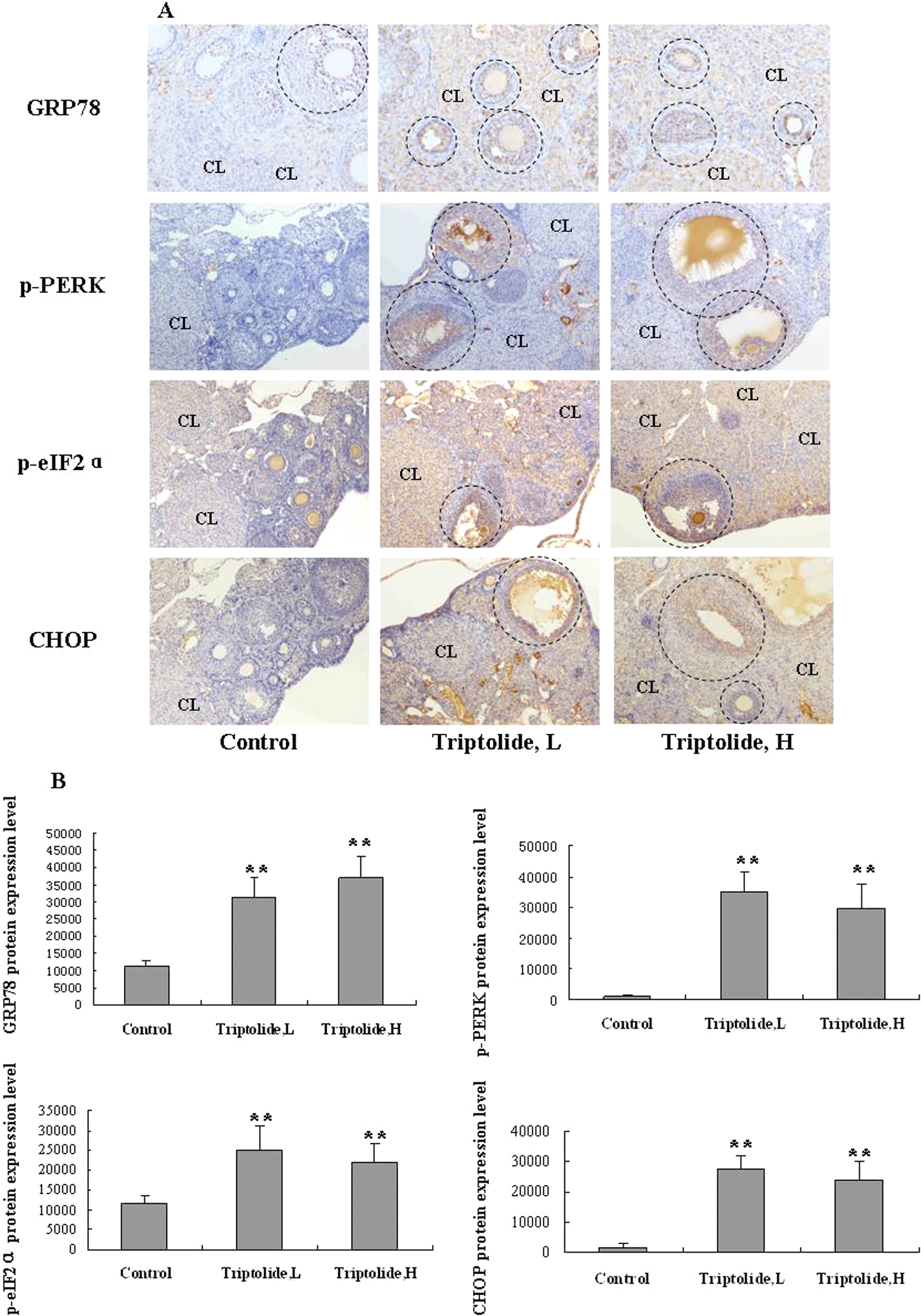
TPL-activated ER stress signal pathway in GCs. (a) Representative photomicrographs of ER stress–related proteins of ovaries in each group (original magnification, ×100, cycle denotes follicles, CL). The branch of ER stress signal pathway contains GRP78, p-PERK, p-eIF2α, CHOP proteins, and so on. Phosphorylation of PERK (p-PERK) and eIF2α (p-eIF2α) serves as a marker for their activation status. (b) Quantitative analysis of ER stress–related proteins (GRP78, phospho-PERK, phospho-eIF2α, and CHOP proteins) expression of follicle areas from the control, TPL L and TPL H groups. Data are reported as mean ± SD. n = 6 mice each group, **p < 0.01, compared with the control group, by t test. TPL: triptolide; ER: endoplasmic reticulum; GCs: granulose cells; TPL L: low-dose (25 μg/kg/d) TPL treatment group; TPL H: high-dose (50 μg/kg/d) TPL treatment group; CL: corpus luteum; GRP: 78-kDa glucose-regulated protein; PERK: protein kinase-like endoplasmic reticulum kinase; eIF: eukaryotic initiation factor; CHOP: CCAAT/enhancer binding protein homologous protein.
TPL-induced GC antiapoptosis function decline
The inhibitor of apoptosis (IAP) protein family acts as anti-apoptotic factors. XIAP is a key member of IAP and is the most potent IAP protein. To determine the role of XIAP in TPL-induced follicular fate change, we detected the XIAP protein expression in mice follicles. In the control group mice, strong positive XIAP expression was observed in mice ovary tissue, including follicles and CLs (Figure 7(a), control group). Compared to the control group, low-dose and high-dose TPL significantly reduced XIAP expression of mice ovary tissue (Figure 7(a), TPL L and TPL H groups). But there is no difference in positive expression rate between low-dose and high-dose groups.

TPL impaired antiapoptotic function of ovary. (a) Representative photomicrographs of XIAP protein of ovaries in each group. (b) Quantitative analysis of XIAP protein expression of follicle areas from the control, TPL L and TPL H groups. Data are reported as mean ± SD. n = 6 mice each group, **p < 0.01, compared with the control group by t test. TPL: triptolide; TPL L: low-dose (25 μg/kg/d) TPL treatment group; TPL H: high-dose (50 μg/kg/d) TPL treatment group; XIAP: X-linked inhibitor of apoptosis protein.
Discussion
During intake of T. wilfordii Hook F for treatment, male oligomenorrhea, amenorrhea, even sterility was found frequently. TPL was characterized as the main active substance from the T. wilfordii Hook F. Its adverse effects on rat ovary had been reported by Jing Liu et al. previously. 8 They confirmed that various doses of TPL (100, 200, and 400 μg/kg) could induce various reproductive parameter of female rats change, including altered estradiol (E2), progesterone (P), FSH and luteinizing hormone, reduced ovary/uterus weight, atretic follicles increasing and disturbance in the estrous cycle, and so on. But the potential mechanisms were unclear. In our study, therapeutic doses of TPL (25, 50 μg/kg/d) were adopted in vivo, according to the method reported by Gao Q et al. 17 The shrinkage of ovaries occurred after TPL administration. The phenomenon may be caused by ischemia induced by TPL. We mentioned above that TPL-treated ovarian presented grain appearance, which liked postmenopausal ovary (Figure 1(a), TPL group). The impaired ovarian appearance maybe caused by an analogous mechanism of an acute ovarian injury due to ischemia. The ovarian size change was similar to doxorubicin-induced ovarian insult. 19 By counting ovarian follicles, we estimated the ovarian follicles reservoir after TPL treatment. We found that TPL decreased developed follicle numbers significantly, especially for primary follicles and secondary follicles. The phenomenon made it sense that T. wilfordii Hook f could induce sex hormones change in clinical. Estrus cycle change induced by TPL also partially reflected indirectly its adverse effect on sex hormone. Jing Liu reported that subnormal levels of estradiol and progesterone estradiol and progesterone might disturb the estrous cycle in TPL-treated rats 8 Our study confirmed that TPL could downregulate the expression of estrogen receptor and progesterone receptor (Data not shown). Meanwhile, the quality and quantity of eggs released from ovaries obviously diminished following exposure to TPL. In brief, therapeutic doses of TPL dramatically damaged mice ovary structure and function.
We mentioned that TPL significantly decreased the number of primary follicles and secondary follicles. We assumed that above ovarian insults induced by TPL might due to the GC injuries. As mentioned above, GC is a cell surrounding the oocyte, which associated with the developing oocyte of mammals. The main function of GCs contains production of sex steroid and providing nutrition for oocyte. Dynamic GC proliferated with folliculoogenesis. Because apoptosis in GCs represents the earliest event described during the onset of atresia, some studies of follicle loss have focused on this type cell death. 19,23 It was reported that TPL could mediate some type cell apoptosis in some pathological process and inhibit transcription of many genes. 24,25 Nevertheless, pro-apoptotic effects on ovary tissue were not understood. To determine the role of GC apoptosis, TUNEL assay was applied in this study. The results showed that increased apoptotic GCs were observed in developing follicles of mice treated by TPL. Apoptosis of GCs, rather than oocyte may explain that in clinical, partial females, who exposed to T. wilfordii and then suffered from ovarian function failure, could recover after withdrawal ultimately.
Although accumulating data confirmed that various apoptotic genes were activated in GCs during follicular development, 26 but we still believed that TPL itself accelerated GCs apoptosis, independent of sex hormonal change. ER stress was confirmed to be involved in GC apoptosis during follicular atresia in goat ovaries. 27 Yang Y et al. also reported that a novel ER stress–regulated gene, luman recruiting factor might be involved in inducing apoptosis of GCs through the ER stress pathway, which might have a key role in mouse follicular selection. 28 In this study, we mainly detected ER stress–related proteins, GRP78, PERK, and CHOP. The GRP78 is a stress-inducible protein ubiquitously expressed in animal cells, which is typical hallmarker protein of ER stress. PERK/eIF2α/ATF4 UPR branch was a main signal pathway of ER stress. PERK (protein kinase-like ER kinase) is an eIF2α kinase. ER stress increases the activity of PERK, which then phosphorylates eIF2α to reduce translation. Phosphorylation of PERK at Thr980 serves as a marker for its activation status. During ER stress, CHOP expression elevated and CHOP functions to mediate programmed cell death. Too much PERK signaling also promotes cell death, in part through its induction of the CHOP proapoptotic transcription factor. 29 But the role of PERK/eIF2α/CHOP branch in TPL-induced GC apoptosis was understood. In this study, we found that TPL could induce GRP78, p-PERK, p-eIF2α, and CHOP protein expression upregulation in GC. These results clarified that the TPL induced GC apoptosis partly via ER stress pathway.
Balance of pro- and antiapoptotic machineries decided the cell’s fate ultimately. Numerous functional genes behave as antiapoptotic sequences, such as the IAP. X-linked inhibitor of apoptosis gene is most potent antiapoptotic gene. XIAP stops apoptotic cell death, which is induced either by viral infection or by overproduction of caspases. XIAP binds to and inhibits caspase 3, 7 and 9. Hiramatsu N et al reported that PERK downregulates XIAP synthesis through eIF2α and promotes XIAP degradation through ATF4. 30 Loss of XIAP leads to increased cell death, whereas XIAP overexpression significantly enhances resistance to ER stress–induced cell death. In our study, we also detected XIAP in TPL-treated mice ovaries. The XIAP expression of mice ovaries decreased after TPL treatment significantly. Although the relationship of XIAP and ER stress proteins was still unclear, the expressions between them presented a negative correlation in this model.
In summary, the data presented here demonstrated that the ovarian function of NIH mice treated by two therapeutic doses of TPL, was injuried obviously, which were mainly characterized as shrunken ovary, follicular atresia, estrous cycle disorder, ovulation function failure, and so on. The changes were partly contributed to TPL-induced GC apoptosis. PERK/eIF2α/CHOP UPR branch of ER stress confirms the involvement in promoting GC apoptosis in this pathologic model. In addition, TPL-induced GC antiapoptotic function decline involved in this pathological process. Of note, there was no significant difference between high and low doses of TPL. The phenomenon suggested that double therapeutic doses may not increase the side-effect of TPL. This research is helpful to understand the female reproductive side effect of T. wilfordii. Although the adverse effects of TPL have been clarified, the mechanism of ovary function recovery after withdrawal needs further research.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangdong Province National Natural Science Research Grant [Grant number S2012010010969]; and the Shenzhen Science and Technology Project Plan [JCYJ20120830095257926].
