Abstract
T-2 toxin, a naturally produced Type A trichothecene mycotoxin, has been shown to damage the reproductive and developmental functions in livestocks. However, whether T-2 toxin can disturb the pubertal onset and development following prepubertal exposure remains unclear. To clarify this point, infantile female Sprague–Dawley rats were given a daily intragastric administration of vehicle or T-2 toxin at a dose of 375 μg/kg body weight for 5 consecutive days from postnatal day (PND) 15–19 (PND15–PND19). The days of vaginal opening, first diestrus, and first estrus in regular estrous cycle were advanced following T-2 toxin treatment, indicating prepubertal exposure to T-2 toxin induced the advancement of puberty onset. The relative weights of uterus and ovaries and the incidence of corpora lutea were all increased in T-2 toxin-treated rats; serum hormone levels of luteinizing hormone and estradiol and the messenger RNA expressions of gonadotropin-releasing hormone (GnRH) and GnRH receptor also displayed marked increases following exposure to T-2 toxin, all of which were well consistent with the manifestations of the advanced puberty onset. In conclusion, the present study reveals that prepubertal exposure to a high level of T-2 toxin promotes puberty onset in infantile female rats by advancing the initiation of hypothalamic–pituitary–gonadal axis function in female rats.
Introduction
Puberty, marked by the appearance of secondary sexual characteristics, acceleration of growth, and eventual capacity for fertility, is controlled by the activation of the hypothalamic–pituitary–gonadal (HPG) axis. 1,2 Thus, exogenous or endogenous factors which can dysregulate the HPG axis potentially influence the onset of puberty, resulting in precocious or delayed puberty. Precocious puberty (onset of puberty before age 8 in girls or age 9 in boys) become one of the most popular endocrine-related disorders in children, which may damage both mental behavior and physical development of children. 3 –5 There has been a worldwide scientific discussion on potential pubertal development consequences of children exposed to environmental and food contaminants, and there is a growing body of evidence for the environmental influence (such as environmental endocrine disruptors) on puberty onset. 6 –8
Fusariotoxins (also known as Fusarium mycotoxins), such as trichothecenes T-2 toxin and deoxynivalenol, and zearalenone (ZEA), are commonly found on cereals and their feedstuffs worldwide, posing a health risk to livestocks and humans. 9 –12 Especially, available animal and epidemiologic data have indicated the adverse effects of fusariotoxins on reproduction and development, provoking an increasing health concern. ZEA has been suspected to be a triggering factor for true precocious puberty development in girls, 13,14 and there is evidence that sexual maturation can be advanced by prepubertal exposure to ZEA in female mice. 15,16 T-2 toxin is a Type A trichothecene mycotoxin which has been shown to induce a variety of toxic effects in livestocks and humans. It has been proved that T-2 toxin can pass through the placenta 17,18 and has a strong reproductive and developmental toxicity. 19 –21 It was revealed that exposure to T-2 toxin reduced steroidogenesis and induced alterations in steroidogenic gene expressions in H295R cells, indicating the potential endocrine-disrupting activity of T-2 toxin. 22 Wu et al. recently also reveal that T-2 toxin regulates steroid hormone secretion of rat ovarian granulosa cells through cyclic adenosine monophosphate–protein kinase A pathway. 23 More recently, using GT1-7 cells, an immortalized hypothalamic neuron cell line which can synthesize and secrete gonadotropin-releasing hormone (GnRH), our team reveals that low concentrations of T-2 toxin stimulates GnRH secretion and alters the expressions of associated proteins in GT1-7 cells in vitro, 24 suggesting the potential effect of T-2 toxin on pubertal onset and development.
Factors such as xenobiotic pollutants that damage the reproductive and developmental systems may also regulate puberty onset, just like ZEA. To the best of our knowledge, no study has described the effect of T-2 toxin on puberty onset. Given the adverse effects of T-2 toxin on the reproductive and developmental targets and its endocrine disruptive activity mentioned above, we hypothesize that T-2 toxin may disturb the onset and development of puberty. The sex development process of rodents and humans are similar, both experiencing similar central activation and endocrine changes, and the established time of puberty in rats is short. Therefore, the present study aims to explore the effect of prepubertal exposure to T-2 toxin on the timing of puberty onset in immature female rats and the alterations in HPG axis which determines the puberty onset are also detected.
Materials and methods
Animals
Healthy Sprague–Dawley rats were purchased from Laboratory Animal Center of Academy of Military Medical Sciences (Beijing, China), male rats weighed 300–350 g, and nonpregnant female rats weighed 250–300 g. The animals were maintained in a room with controlled illumination (12-h light/12-h dark cycle), temperature (20–25°C), and humidity (40%–70%) and were given free access to regular rat diet and water. All the procedures were carried out according to the guidelines provided by the Animal Care and Facilities Committee, Institute of Disease Control and Prevention, Academy of Military Medical Sciences (Beijing, China).
Chemicals and reagents
T-2 toxin was kindly provided by the Institute of Pharmacology and Toxicology, Academy of Military Medical Sciences (Beijing, China). The toxin was biosynthesized and purified by the Institute of Pharmacology and Toxicology, and the purity was more than 99.5%. T-2 toxin solution was prepared in corn oil at a concentration of 150 μg/mL. Rat luteinizing hormone (LH), follicle-stimulating hormone (FSH), and estradiol enzyme-linked immunosorbent assay (ELISA) kits were obtained from RapidBio Lab (RapidBio, Hercules, California, USA). Trizol reagent was obtained from Invitrogen Life Technologies Corporation (Carlsbad, California, USA). Complementary DNA (cDNA) synthesis kit was obtained from Fermentas International Inc. (Burlington, Ontario, Canada). All the other chemicals were of analytical grade and were commercially available.
Experimental procedures
Mating and experimental design
The females were mated (2 females per male), and the pregnant rats were housed individually. After birth, 10 female offspring were selected and raised by their consanguineous mother. If the number of female offspring was less than 10, it would be complemented by male ones. The female offspring were randomly assigned to 4 groups (groups 1, 2, 3, and 4) of 10 animals each, of which 2 groups (group 1 and group 2) were designed for experiment I, and the other 2 groups (group 3 and group 4) were for experiment II. From postnatal day (PND) 15 to 19 (PND15–PND19), infantile female rats of group 1 in experiment I and of group 3 in erxperiment II received a daily intragastric administration of T-2 toxin at a dose of 375 μg/kg body weight (bw), and the other group in each experiment received corn oil only (vehicle control). The dosing volume of T-2 toxin solution or corn oil was 2.5 mL/kg. Daily dosing was performed regularly in the morning from 8:30 to 9:00. The animals were weaned on PND21, then were examined daily for vaginal opening (VO) and vaginal smears afterward.
The dose of 375 μg/kg bw used in the present study was chosen mainly on the basis of the provisional maximum tolerable daily intake (PMTDI) of 60 ng/kg bw/day proposed by the WHO/FAO Joint Expert Committee on Food Additives (JECFA) for T-2 toxin, 25 and the tolerable daily intake (TDI) of 100 ng/kg bw/day established by the European Food Safety Authority (EFSA) for the sum of T-2 and HT-2 toxins. 26 The dose chosen was briefly justified as follows. A PMTDI or TDI for a food additive or contaminant is traditionally established based on the most critical no observed adverse effect level (NOAEL), which is derived from toxicological studies with laboratory animals, taking the uncertainties into consideration. 27 The uncertainty factor of 100 or higher is conventionally adopted, considering the extrapolation differences (such as extrapolation from animals to humans and extrapolation from high to low intake levels) and interindividual variability. And the completeness and precision of the toxicological data from which a NOAEL is derived should also be considered when allocating an uncertainty factor. 27 Therefore, in the present study, we utilized a reverse deduction method, a NOAEL or “tolerable daily exposure dose” for T-2 toxin in rats was deduced from the existing PMTDI and TDI for humans by applying a suitable uncertainty factor. Considering the extrapolation differences in species and doses, interindividual variability, and weak precision of the existing long-term toxicological data of T-2 toxin, we adopted a higher uncertainty factor of 1000 to estimate an exposure dose in the present study. Accordingly, a NOAEL or tolerable daily exposure dose for T-2 toxin in rats was estimated in the range of 60–100 μg/kg bw/day. We thus chose a slightly higher dose of 375 μg/kg bw/day, which exerted no significant adverse effects on the general behavior and growth of immature female rats in our preliminary test.
Experiment I
In experiment I, the female rats of group 1 and group 2 (10 animals per group) were used to observe the days of VO, first diestrus (D1), and first estrus (E1) in regular estrous cycles. Vaginal smears were checked daily at the same time (8:00–8:30 a.m.) from the day of VO until consecutive regular 4- or 5-day estrous cycles were established in all rats. On the day of consecutive regular estrous cycles were observed in the last rat, all rats were subject to euthanasia by cervical dislocation after deep anesthesia, followed by autopsy. The thymus, liver, heart, kidney, spleen, uterus, ovary, bone, and other tissues/organs were harvested. The samples were fixed in 10% buffered formalin for 24 h at room temperature, embedded in paraffin, sectioned at 4 μm, stained with hematoxylin and eosin, and examined by light microscopy.
Experiment II
In experiment II, group 3 and group 4 (10 animals per group) were used. Once VO was detected in rats of vehicle control group (group 4), these control rats were subject to euthanasia at the day, and as the matches, the same number of T-2 toxin-treated rats (group 3) were randomly selected and killed as well.
The blood was collected from the femoral artery before euthanasia, then was centrifuged at 3500g for 15 min to obtain the serum. The resulting serum was frozen at −80°C until hormone determination. Uterus, ovaries, and vagina were dissected out of the surrounding fats and weighed, and their relative weights were calculated. The incidence of corpora lutea was calculated with stereomicroscope (Olympus, Japan). Then ovaries, uterus, and vagina were fixed in 10% buffered formalin for the histopathological examination. The brain was immediately removed and the hypothalamus (limited anteriorly by the optic chiasma, laterally by the hypothalamic fissures, posteriorly by the mammillary bodies and in depth by the subthalamic sulcus) and pituitary glands were dissected. Tissue samples were snap-frozen in liquid nitrogen, then were stored at −80°C for the determinations of messenger RNA (mRNA) expressions of GnRH and GnRH receptor (GnRHR).
Examination and biochemical determinations
VO examination
VO was determined by daily inspection at the same time (8:00–8:30 a.m.) from PND20 using surgical loupes. The day of VO was recorded as the day on which the vaginal orifice transitioned from tightly closed to patent. 28
Hormone assays
Levels of LH, FSH, and estradiol in the serum obtained from the rats of groups 3 and 4 in experiment II were measured by ELISA methods (rat LH, FSH, and estradiol ELISA kits, RapidBio Lab). The results for LH, FSH, and estradiol were expressed as ng/mL, ng/mL, and pg/mL, respectively; and the sensitivity limits for these hormones were 0.3 ng/mL, 1 ng/mL and 5 pg/mL, respectively.
RNA extraction and RT-PCR
Total RNA in hypothalamus and pituitary glands from the rats of groups 3 and 4 in experiment II was extracted using Trizol reagent (Invitrogen) according to the manufacturer’s instructions. The synthesis of cDNA and the analyses of the results obtained were performed as previously described. 29 Before analytical procedures, the purity and integrity of the RNA were examined spectroscopically (USA Technologies Inc, Malvern, Pennsylvania, USA) and by gel electrophoresis (BioRad, Hercules, California, USA). RNA samples (1 μg) were reverse transcribed by adding Oligo d(T) (Fermentas, Canada) and nuclease-free water in a volume of 12 μL for 5 min at 65°C. Reaction buffer, RNAase inhibitor, deoxynucleotide, and Moloney Murine Leukemia Virus reverse transcriptase were subsequently added and incubated for 60 min at 42°C, then denatured for 5 min at 70°C, and stored at −20°C. Polymerase chain reaction (PCR) primers and annealing temperature are shown in Table 1. All primers were synthesized by InvitrogenTM (Beijing, China). PCR cycling conditions were as follows: denaturation at 95°C for 10 min, 40 cycles of denaturation at 95°C for 30 s, annealing at 56°C for 30 s, and extension at 72°C for 30 s. Twenty microliters of each PCR product was electrophoresed on a 1.5% agarose gel (with ethidium bromide) for 20 min at 120 V and then photographed (BioRad). Densitometry was performed using Quantity One 6.0 software. The data were recorded as the ratio of the sample to the internal standard β-actin.
PCR primers and annealing temperature.

GnRH: gonadotropin-releasing hormone; GnRHR: gonadotropin-releasing hormone receptor.
Statistical analysis
All data were expressed as mean ± SD, and the differences between control and toxin-treated group were analyzed by t-test using SPSS 16.0 statistical software. Significance was set at p < 0.05 or p < 0.01.
Results
General observation of animals
No abnormal signs in general appearance and mortality were observed in all the infantile female rats during the experiments. All the rats displayed normal activities and growth, and histological (macroscopic and microscopic) examinations of main tissues/organs, such as thymus, liver, heart, kidney, spleen, and bone, showed no any abnormalities or lesions in any rat. No significant difference in body weight was identified between control and T-2 toxin-treated groups when compared at the same age (PND15, PND19, and PND35; Table 2).
Effect of prepubertal exposure to T-2 toxin on the body weight of female rats.a

PND: postnatal day.
aThe body weights of all groups were measured on PND15, PND19, and PND35, and data were expressed as means ± SD (n = 10).
Effects of T-2 toxin on the days of VO, D1, and E1 of the female rats
VO and the emergence of the D1 have been used as efficient indicators for puberty onset in female rats in many studies. 30 –32 As shown in Figure 1, as compared to the vehicle control group (group 2 in experiment I), the days of VO, D1, and E1 of the female rats were all advanced in T-2 toxin-treated group (group 1 in experiment I) (p < 0.05, p < 0.05, p < 0.05, respectively), suggesting that prepubertal exposure to T-2 toxin promotes the onset of puberty.
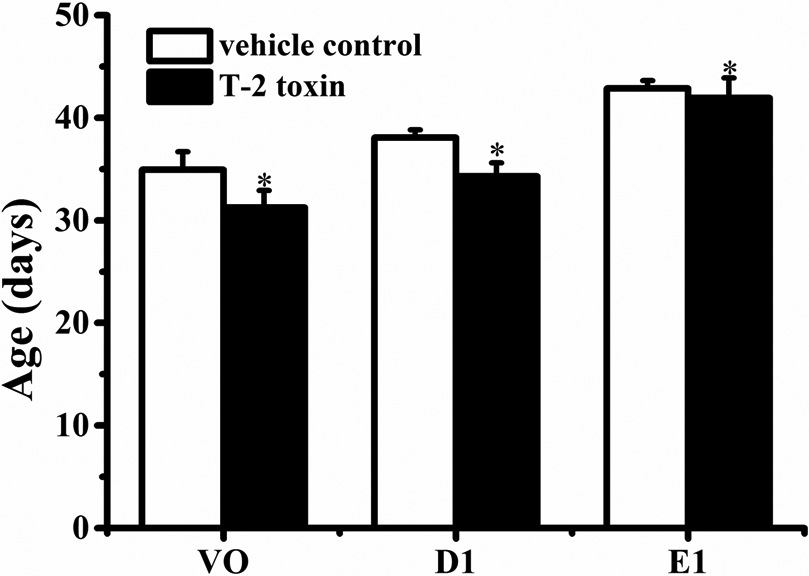
Effects of prepubertal exposure to T-2 toxin on the ages of VO, D1, and E1 of female rats from groups 1 and 2 in experiment I. Data are means ± SD (n = 10). *p < 0.05: significantly different from vehicle control values. VO: vaginal opening; D1: first diestrus; E1: first estrus; SD: standard deviation.
Effect of T-2 toxin on the vaginal estrus of the female rats
In the current study, irregular estrous cyclicity at an early age was observed in all female rats. However, the irregularities were transient and succeeded by periods of normal 4- and 5-day cycles (including estrus, metestrus, diestrus, and proestrus) in all groups. Also the performance of vaginal smears and the cycle length in each group were similar (data not shown).
Effect of T-2 toxin on the development of reproductive organs of female rats
The development of reproductive organs is often used as a reliable indicator of the appearance of secondary sexual characteristics in female rats. In experiment II, the days of VO for all 10 vehicle control rats (group 4) were observed between PND33 and PND36, which is consistent with a previous study, which showed that the day of VO of female rats was around PND34. 33 Accordingly, as depicted in Materials and Methods section, the control rats were killed on their respective VO days, and the same number of T-2 toxin-treated rats (group 3) were randomly killed as the matches. As shown in Table 3, the relative weights of uterus, ovaries, and vagina in T-2 toxin-treated group were slightly increased as compared to the control ones, and the incidence of corpora lutea was remarkably increased as much as by 50% (p < 0.05).
Effects of T-2 toxin on the development of reproductive organs of female rats.a

VO: vaginal opening; PND: postnatal day.
aIn experiment II, the days of VO for vehicle control rats (group 4) were observed between PND33 and PND36. The control rats were killed on their VO days, and as the matches, same number of T-2 toxin-treated rats (group 3) were randomly selected and killed. Uterus, ovaries and vagina were dissected and weighed, and the relative organ weight was calculated and expressed as organ weight (mg)/body weight (g). Data were presented as the means ± SD of 10 animals.
bp < 0.05: significantly different from vehicle control group.
Histopathological examination of reproductive organs of female rats
In experiment II, on the day of VO observed in control rats, the reproductive organs of uterus, ovaries, and vagina in control rats and their matched toxin-treated ones were subject to histopathological examination following the measurement of organ weights.
Uterus
As shown in Figure 2(a) and (b), the thickness of myometrium and the number of endometrial glands was significantly increased in T-2 toxin-treated rats as compared to vehicle control ones. Meanwhile, glandular cavity was expanded obviously in T-2 toxin-treated group.

Representative histological pictures stained by H & E for reproductive organs in vehicle control group (group 4) and T-2 toxin treatment group (group 3) in experiment II. The days of VO for vehicle control rats were observed between PND33 and PND36. The control rats were killed on their VO days, and as the matches, same number of T-2 toxin-treated rats were randomly selected and killed. (a) and (b) were for uterus in vehicle control and T-2 toxin treatment group, respectively, the arrows indicate endometrial glands (magnification ×100); (c) and (d) were for ovaries in vehicle control and T-2 toxin treatment group, respectively, the arrows indicate the corpus luteum (magnification ×40); and (e) and (f) were for vagina in vehicle control and T-2 toxin treatment group, respectively. H & E: hematoxylin & eosin; VO: vaginal opening; PND: postnatal day.
Ovaries
In vehicle control rats, the major ovarian follicles were secondary follicles, and there was little corpus lutea. In contrast, the incidence of corpus lutea and mature follicles were significantly increased in T-2 toxin-treated rats (Figure 2(c) and (d)).
Vagina
When compared with vehicle control rats, microscopic examination of the vagina showed that the thickness of vaginal wall, the layer of mucosal epithelial and keratinization of the epithelium were increased, and the lamina propria was loosened and congestive in T-2 toxin-treated rats (Figure 2(e) and (f)).
These abnormal histological findings of the reproductive organs, along with the observations of increased relative weights of uterus, ovaries, and vagina and increased incidence of corpora lutea in T-2 toxin-treated group, suggested that prepubertal exposure to T-2 toxin advanced the development of reproductive organs.
Effects of T-2 toxin on the levels of serum hormones
Gonadotropins are responsible for the development of reproductive function and secondary sexual characteristics. Therefore, serum LH, FSH, and estradiol levels are also effective targets to determine the onset of maturity. In the present study, in experiment II, on the day of VO observed in control rats, the serum levels of LH and estradiol were increased by 23% and 11% in T-2 toxin-treated group (group 3; p < 0.01, p < 0.05) in comparison with vehicle control values (group 4), respectively. Serum FSH concentration also showed an increase, despite no significant difference (Table 4). The elevated serum gonadotropins indicated that T-2 toxin advanced the onset of maturation, which is well consistent with the advanced development of reproductive organs.
Effects of T-2 toxin on serum levels of LH, FSH, and estradiol in female rats.a
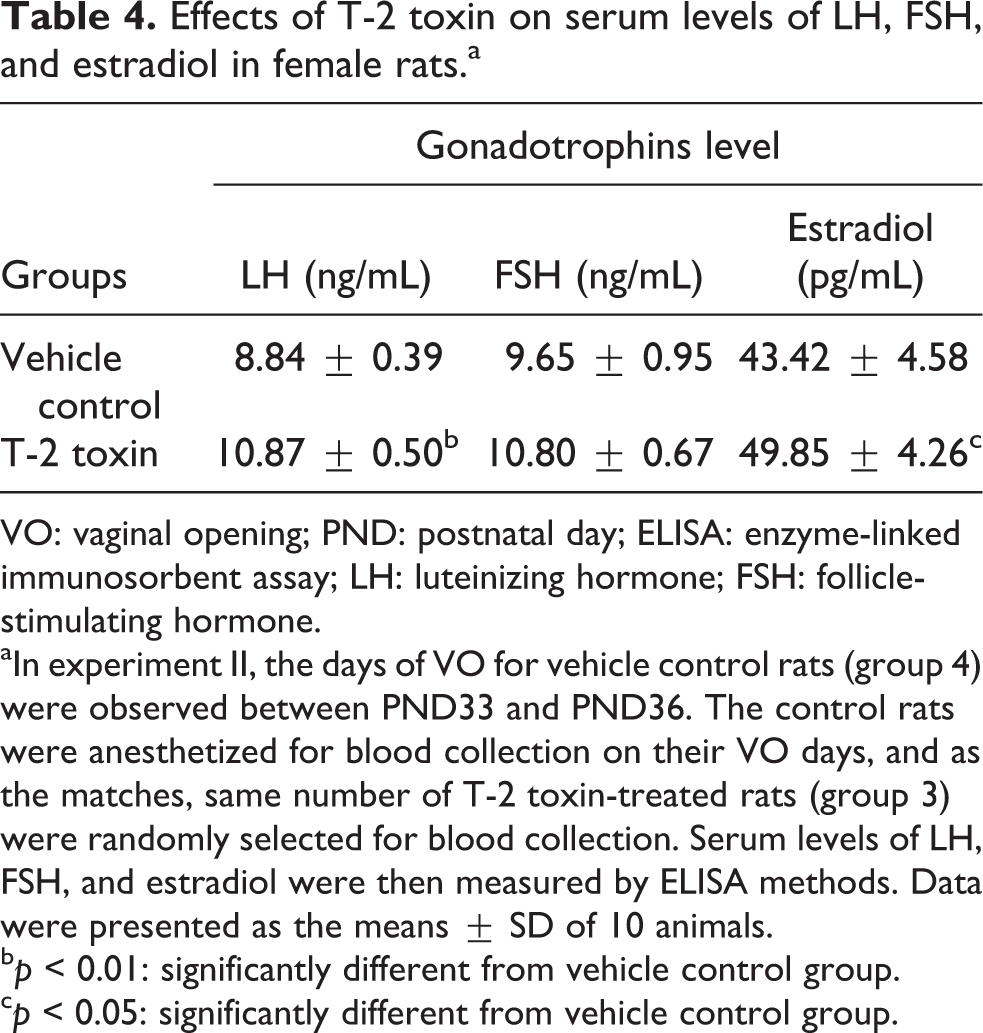
VO: vaginal opening; PND: postnatal day; ELISA: enzyme-linked immunosorbent assay; LH: luteinizing hormone; FSH: follicle-stimulating hormone.
aIn experiment II, the days of VO for vehicle control rats (group 4) were observed between PND33 and PND36. The control rats were anesthetized for blood collection on their VO days, and as the matches, same number of T-2 toxin-treated rats (group 3) were randomly selected for blood collection. Serum levels of LH, FSH, and estradiol were then measured by ELISA methods. Data were presented as the means ± SD of 10 animals.
bp < 0.01: significantly different from vehicle control group.
cp < 0.05: significantly different from vehicle control group.
Effects of T-2 toxin on mRNA expressions of hypothalamic GnRH and pituitary GnRHR
The mRNA levels of hypothalamic GnRH and pituitary GnRHR were assayed by reverse transcription (RT) PCR. In experiment II, on the day of VO observed in control rats, hypothalamic GnRH mRNA expression was obviously elevated (p < 0.05) by 51% in T-2 toxin-treated rats (group 3 in experiment II) as compared to the vehicle control ones (group 4 in experiment II) (Figure 3). Similarly, pituitary GnRHR mRNA expression was significantly increased by 45% (p < 0.01) in T-2 toxin-treated group. These results are well consistent with the manifestations of advanced puberty onset, including increased serum gonadotrophins, advanced development of reproductive organs, and advanced VO.

The mRNA expressions of hypothalamic GnRH and pituitary GnRHR by RT-PCR analysis. In experiment II, the days of VO for vehicle control rats (group 4) were observed between PND33 and PND36. The control rats were killed on their VO days, and as the matches, same number of T-2 toxin-treated rats (group 3) were randomly selected and killed. The brain was immediately removed, and the hypothalamus and pituitary glands were dissected and used for the determinations of mRNA expressions of GnRH and GnRHR, respectively. The upper picture shows the gel electrophoresis of the RT-PCR products for the hypothalamic GnRH and pituitary GnRHR amplified from the total RNA isolated from the vehicle control rats and T-2 toxin-treated ones. The results of GnRH and GnRHR were normalized to β-actin level. Data are expressed as the means ± SD of 10 animals. *p < 0.05; **p < 0.01: significantly different from vehicle control values. mRNA: messenger RNA; GnRH: gonadotropin-releasing hormone; GnRHR: gonadotropin-releasing hormone receptor; VO: vaginal opening; PND: postnatal day; RT-PCR: reverse transcriptase polymerase chain reaction.
Discussion
The present findings clearly show that prepubertal exposure to T-2 toxin causes advanced onset of puberty in infantile female rats without showing any visible toxicity, as indicated by the consistent alterations in the HPG axis that dominates the pubertal onset and development, from upstream hypothalamic GnRH synthesis increase (Figure 3), to midstream serum gonadotrophins elevations (Table 4), until downstream reproductive organs development advancement (Table 3 and Figure 2).
As well-known, the physical changes associated with puberty are caused by prior activation of HPG axis. The initiation of HPG axis requires an increase in pulsatile release of GnRH from the hypothalamus. Thereafter, GnRH binds with high affinity to its specific receptor GnRHR on the surface of gonadotrope pituitary cells, leading to the synthesis and release of gonadotrophins such as LH and FSH from the anterior pituitary gland. 34 LH and FSH, which in turn stimulates GnRH secretion, 35 elicit the secretions of the gonadal hormones estrogen and testosterone which are responsible for the development of reproductive function and secondary sexual characteristics. Studies have indicated that the onset of puberty in humans and rodents is highlighted by the augmentation of pulsatile GnRH secretion. 36,37 In our present study, increased mRNA expression of hypothalamic GnRH was observed following prepubertal exposure to T-2 toxin, which is consistent with the increased serum gonadotrophins, advanced development of reproductive organs, and the manifestations of advanced puberty onset (VO and estrous cycle). Previous studies found that T-2 toxin could increase the levels of 5-hydroxy-3-indoleacetic acid, serotonin, and norepinephrine in the brain, 38,39 thus playing an important role in the regulation of the HPG axis. 40 More recently, our team reveals that low concentrations of T-2 toxin stimulates GnRH secretion in hypothalamic GT1-7 cells. 24 Taken together, the advanced onset of puberty in female rats induced by prepubertal exposure to T-2 toxin might be ascribed to its regulatory activity on hypothalamic function.
The synthesis and secretion of GnRH is regulated by a complex and systematic network in hypothalamus, and Kiss-1 gene product of kisspeptins and their G protein-coupled receptor 54 (GPR54) are recognized as essential gatekeepers in control of GnRH secretion and reproductive function. 41 –43 In recent years, increasing studies have revealed the key role of kisspeptins in several environmental endocrine disruptors-induced adverse effects on puberty onset and reproductive function. 44 –46 As described above, our team found that T-2 toxin had potential stimulating effects on GnRH secretion in hypothalamic GT1-7 cells. A further study revealed that T-2 toxin-induced GnRH secretion in hypothalamic GT1-7 cells was further increased in the presence of exogenous kisspeptin-10, which can activate GT1-7 cells to secrete GnRH. 24 In other words, kisspeptin-10 pretreatment boosted the reactivity of GT1-7 cells to T-2 toxin in light of GnRH secretion. In addition, the change trends of protein expressions of GPR54 following T-2 toxin treatment were consistent with the alterations of GnRH secretion in the absence or presence of exogenous kisspeptin-10. These in vitro findings provide us a clue that T-2 toxin may promote puberty onset in immature female rats by the advanced activation of kisspeptins/GPR54 signaling pathway.
In conclusion, our present results reveal that prepubertal exposure to a high level (about 3000 times of the TDI for humans) of T-2 toxin advances the puberty onset in infantile female rats. However, there are several limitations or deficiencies in the present study. Firstly, since this is a single-dose experiment, the lack of dose–response limits the usefulness of the data to some extent. Well-designed experiments with more reasonable exposure doses should be performed to obtain the NOAEL or Bench Mark Dose for the perturbation activity of T-2 toxin on puberty onset in infantile female rats. Secondly, the key point remains to be illuminated is whether the promoting effect of T-2 toxin on puberty onset is mediated by the activation of hypothalamic kisspeptins/GPR54 signaling pathway. Nevertheless, since there are few epidemiological and experimental data on the adverse effects of T-2 toxin on pubertal onset and development, this single-dose study provides new information for the reproductive and developmental toxicity of T-2 toxin. And in recent years, special emphasis has been placed on the evaluation of dietary intake of T-2 toxin in high-risk population, especially children. It has been reported that exposure to T-2 plus HT-2 toxins in children from France and Catalonia (Spain) is relatively high and occasionally exceeds the TDI of 100 ng/kg bw/day. 47,48 Considering the vulnerability of developing children to food contaminants and the relative high level of chronic dietary intake of T-2 toxin in children, we think the findings of our present study may provide valuable information for the health risk assessment of this trichothecene mycotoxin in children, though extreme higher exposure level than TDI was tested.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (no. 81172699).
