Abstract
Osteoarthritis (OA) is a degenerative joint disease that is characterized by joint pain and a progressive loss of articular cartilage. Kaschin-Beck Disease is a form of endemic OA in China whose etiology is unclear, but epidemiological data indicate a possible link to trichothecenes mycotoxin exposure. In vitro, T-2 toxin, a trichothecenes mycotoxin, has been demonstrated to inhibit aggrecan synthesis and promote aggrecanase and pro-inflammatory cytokine production in cultured chondrocytes. To assess the effects of T-2 toxin on articular cartilage in vivo, Wistar rats were fed a diet containing T-2 toxin (100 ng/kg chow) for six and ten months. Following six months of T-2 toxin exposure, histopathological changes in femorotibial cartilage were characterized by chondrocyte degeneration/necrosis and loss, chondrocyte clones, and loss of proteoglycan staining of articular cartilage, sometimes involving the entire thickness of the cartilage in the tibial plateaus and femoral condyles. By ten months, in addition to these changes, there was evidence of cartilage fibration in some rats. In conclusion, T-2 toxin exposure in rats induced degenerative lesions in articular cartilage similar to spontaneous OA, lending support to an etiologic role of mycotoxins in Kaschin-Beck Disease. T-2 toxin–induced degenerative joint disease may be a useful model of metabolic polyarticular OA.
Introduction
Osteoarthritis (OA) is a degenerative disease of the joints, which has a serious impact on human health. In China, there is endemic form of OA called Kaschin-Beck Disease (KBD) (Cao et al. 2008). Kaschin-Beck Disease is geographically distributed in a narrow zone in China from the northeast to southwest and covers fourteen provinces. Kaschin-Beck Disease usually involves children aged five to fifteen years. To date, more than a million patients have suffered from KBD. Typically, the early stage of KBD is manifested as osteochondrosis, with later stages progressing to OA.
Epidemiological data have supported environmental toxin exposure in KBD, and specifically, T-2 toxin has been implicated (Li et al. 2008; Yang 1995). T-2 toxin is one of the most toxic of the trichothecenes mycotoxins produced by the members of several fungal genera, especially members of the genus Fusarium. Exposure is typically through contaminated food or grains (Mngadi et al. 2008; Ueno 1984). T-2 toxin can cause lesions in hematopoietic, lymphoid, gastrointestinal, and cartilage tissues, especially in physeal cartilage (Nascimento et al. 2001; Rosenstein and Lafarge-Frayssinet 1983). In vitro, T-2 toxin can induce aggrecan degradation in chondrocytes through inhibiting aggrecan synthesis and promoting aggrecanase and pro- inflammatory cytokine production (Li et al. 2008). The chronic effects of T-2 toxin exposure have not been explored. The objectives of the present study were to characterize time-dependent histopathological changes in femorotibial articular cartilage in rats chronically exposed to T-2 toxin to gain further insight into its possible role in KBD and explore it utility as a possible model of metabolic-induced polyarthritis.
Materials and Methods
Induction of OA
The experimental protocol was approved by the Animal Care and Use Committee of Harbin Medical University. Eighty twenty-four-day-old Wistar rats (forty female and forty male, 50–70 g, supplied by Harbin Veterinary Research Institute, China) were allowed to acclimate to the facility for seven days. Animals were randomly divided into two groups according to their body weight: control group and T-2 toxin group. Each group had twenty female and twenty male rats. All animals were housed in stainless steel cages (five per cage) with controlled temperature, humidity, and regular light cycles. Animals in the control group were fed standard rat chow, and animals in the T-2 toxin group were fed standard rat chow containing T-2-toxin. All rats were fed chow and water that were available ad libitum. Twenty rats (ten female and ten male) in each group were maintained on their respective diets for either six or ten months. Animals were clinically monitored throughout the duration of the protocol. Animals retained full mobility and their body weights increased normally during the course of the study.
Feed containing T-2 toxin was prepared as follows: T-2 toxin crystals (Lot No.T4887, Sigma, St. Louis, MO, USA) were dissolved in pure ethanol (GR) to prepare a stock solution of 1 mg/mL. The working solution of T-2 toxin (10 µg/mL) was prepared from this stock solution and was applied to the normal rat chow at 100 ng T-2 toxin/g chow, and then vaporized in ethanol.
Morphology and Histochemistry
Rats were euthanized by ether asphyxiation after being maintained on their respective diets for either six or ten months. The femur and tibia were collected without disarticulation at the femoral-tibial joint. The long bones were transected with a rongeur at a distance from the knee joints to avoid damage to and bone fragmentation of the joint area. After removal of the soft tissues, the joint was placed in 10% phosphate-buffered formalin in a position of natural flexion for more than twenty-four hours and then placed in 5% formic acid decalcifier for seven days. The ends of bones were trimmed, and the joint was cut sagittally into approximately equal halves and returned to the decalcifier for a further seven days. The trimmed halves were washed thoroughly in water before processing and embedding in paraffin. Frontal sections (6 µm) of the femorotibial joints were prepared; routinely stained with hematoxylin and eosin (HE), Alcian blue/periodic acid Schiff (PAS), Weigert/van Gieson, and Safranine O/fast green staining (Jocelyn 1974; Noeske 1973; Quintarelli et al. 1964; Rosenbeg 1971); and examined by light microscopy. Microscopic images were collected using the Pathological Image Collection System Software (MOTIC AM 3000; Macaudi) interfaced with a standard light microscope (Olympus BX51, Japan).
Histopathological Evaluation of Articular Cartilage
The degree of femorotibial joint cartilage damage was characterized using two parameters: the extent of damage into the cartilage zones, and the complexity of pathological damage. The articular cartilage was divided into four zones: superficial, middle, deep, and calcification. The score for extent of damage was: 1, one zone only; 2, one or two zones; 3, two or three zones; and 4, entire thickness of the articular cartilage. The pathological damage was divided into four types: (1) chondrocyte degeneration and necrosis; (2) chondrocyte loss; (3) chondrocyte proliferation; and (4) fibration.
The score for complexity of pathological damage was as follows: 1, one type of damage; 2, two types of damage; 3, three types of damage; and 4, all types of damage. The total score was used to evaluate the extent of cartilage damage, which was obtained from the score for extent of damage multiplied by the score for type of pathological damage, with minimum and maximum score of 0 and 16, respectively.
Matrix Proteoglycan Staining Evaluation
Matrix proteoglycan staining was evaluated on Alcian/PAS, Weigert/van Gieson, and Safranine O/fast green stained sections and was used to assess functional activity of articular chondrocytes. Matrix staining (metachromasia) score was: 3, strong staining; 2, slightly reduced staining; 1, significantly reduced staining; and 0, no staining.
Statistical Analysis
Data were analyzed using the Mann-Whitney U test using SPSS, version 13.0. The data were expressed as minimum, median, and maximum, and p < .05 was considered to be significant.
Results
General State of Animals
The median lethal oral dose of T-2 toxin for rats is reported to be 4.0 mg/kg (Bamburg et al. 1969), hence the dose of 100 ng/g used in this experiment was low. Throughout the experimental period, the T-2 toxin–containing diet was well-tolerated. Rats in the two groups had no significant difference in body weights or mobility. However, in the early stage of the experiment, some rats in the T-2 toxin group excreted loose stools. As the experiment progressed, rats in the T-2 toxin group displayed hair loss.
Pathological Changes in Articular Cartilage of Rat Treated with T-2 Toxin
After six months of exposure, H&E–stained sections revealed extensive areas of chondrocyte necrosis in the articular cartilage of femoral condyles and the tibial plateaus from rats fed the T-2 toxin–containing diet. Necrotic chondrocytes were shrunken and had hypereosinophilic cytoplasm and fragmented pyknotic nuclei, consistent with apoptosis. Extensive areas of chondrocyte loss were also seen. Chondrocyte clones were visible in the subjacent cartilage (Figure 1B ). Following ten months of exposure to T-2 toxin, there was significant damage to the articular cartilage surface, with large chondral defects in the weightbearing zones in the femoral condyles (Figure 1C). Extensive fibration was seen in areas of necrosis (Figure 1D).
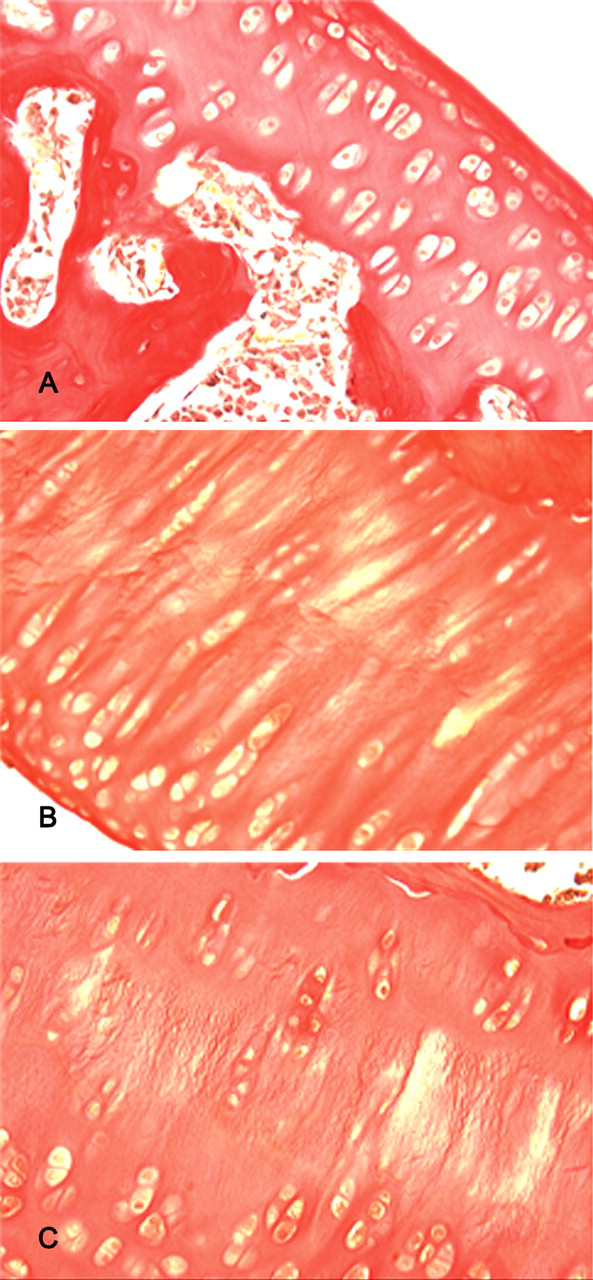
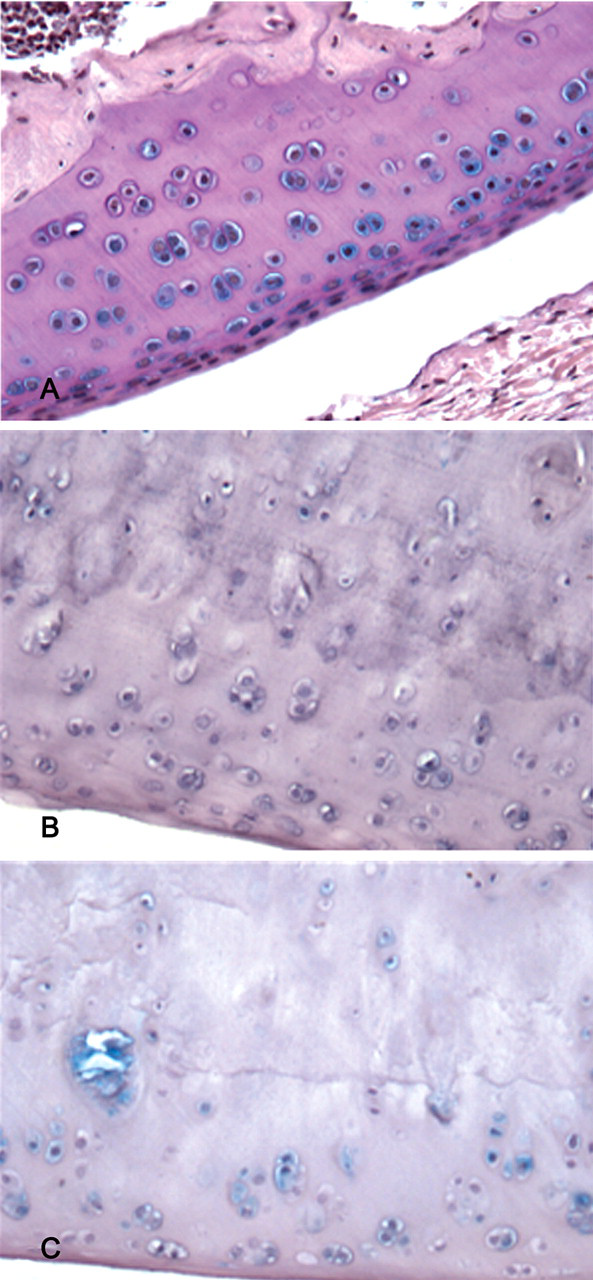
Articular cartilage from Wistar rat femorotibial joint. HE, ×200. (A) Cartilage from a joint from a rat fed standard rat chow. (B) Six months after feeding with T-2 toxin, there were extensive areas of chondrocyte degeneration, necrosis, and loss in the tibial plateau. (C) Ten months after feeding with T-2 toxin, there were large chondral defects of the weightbearing zones of the femoral condyles, with surrounding chondrocyte degeneration/necrosis, loss, and cloning. (D) Ten months after feeding with T-2 toxin, there were extensive areas of fibration and chondrocyte degeneration.
The total histology score of the T-2 toxin group was markedly increased in T-2 toxin–exposed rats compared to controls (Z6m = –3.844, p = .000; Z10m = –3.830, p = 0.000) (Figures 5 and 6 ).
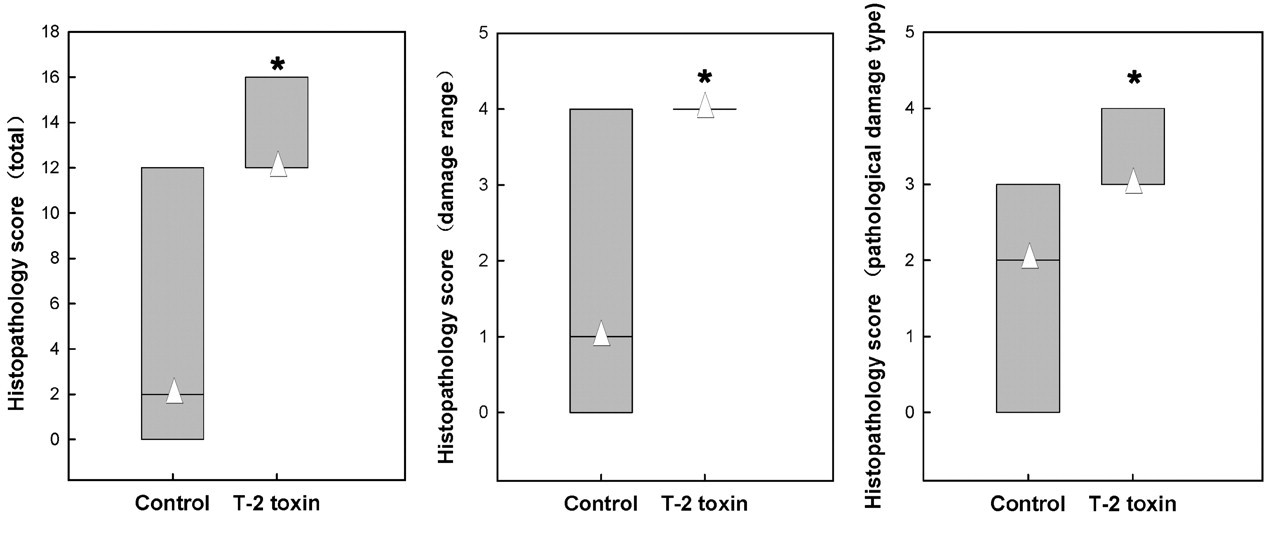
Articular cartilage damage score from a Wistar rat femorotibial joint at six months after T-2 toxin feeding. Compared to the control, the damage score, type score, and total score in the T-2 toxin group significantly increased.* Δ denotes the median.
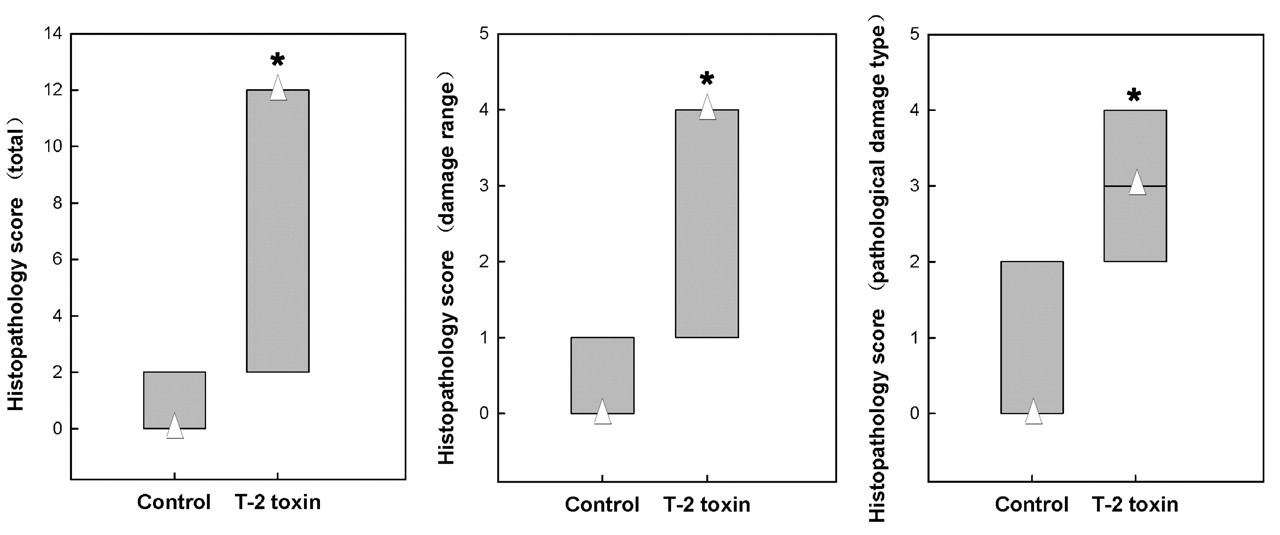
Articular cartilage damage score from Wistar rat femorotibial joint, ten months after T-2 toxin feeding. Compared to the control, the damage score, type score, and total score in the T-2 toxin group significantly increased.* Δ denotes the median.
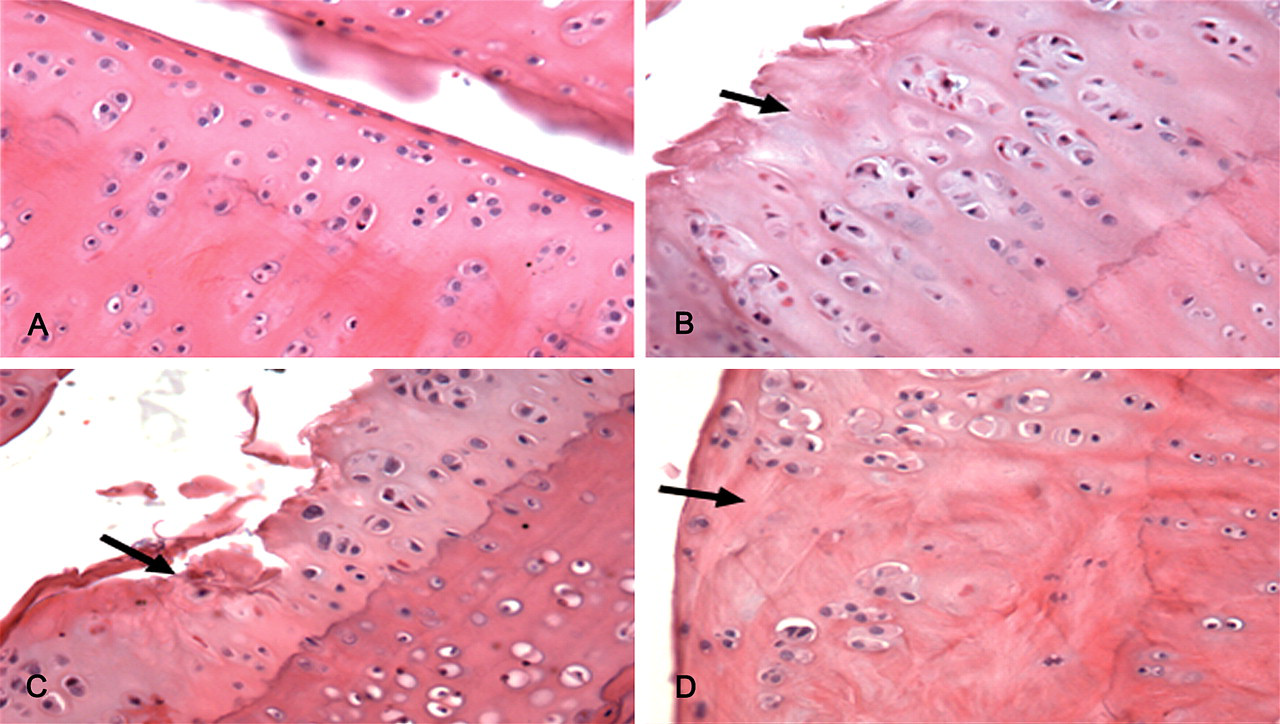
Comparison of cartilage lesions at two different time points indicated changes were progressive. Examination of Alcian/PAS– and Safranine O/fast green–stained sections showed progressive loss of proteoglycan staining following T-2 toxin exposure (Figures 2 and 4). The cartilage matrix staining (Alcian/PAS, Weigert/van Gieson and Safranine O/fast green) score at the two time points is shown in Tables 1 and 2 . Weigert/van Gieson staining revealed progressive collagen fiber disruption, with proteoglycan and water loss, the so-called phenomenon of collagen fasciculi (Figure 3B ), which is an early pathological lesion of OA. As the experiment progressed, the lesions became more severe (Figure 3C).
Articular cartilage from Wistar rat femorotibial joint. Alcian/PAS, ×200. (A) Cartilage from a joint of a rat fed standard rat chow. There was abundant proteoglycan in the cytoplasm, and blue staining was strong. (B) Six months after feeding with T-2 toxin, there was extensive chondrocyte degeneration, with little proteoglycan in the cytoplasm and extracellular matrix; the blue staining was weak. (C) Ten months after feeding with T-2 toxin, there was extensive chondrocyte loss and loss of proteoglycan staining.

Articular cartilage from Wistar rat femorotibial joint. Safranine O/fast green, ×200. (A) Cartilage from a joint of a rat fed standard rat chow. Note the clear red matrix staining. (B) Six months after feeding with T-2 toxin, there was extensive chondrocyte degeneration and loss; the red staining was weaker. (C) Ten months after feeding with T-2 toxin, there were extensive areas of chondrocyte and proteoglycan staining loss.
Matrix staining (metachromasia) score (6m).

Abbreviation: f, collagen fasciculi by VG stain.
Matrix staining (metachromasia) score (10m).

Abbreviation: f, collagen fasciculi by VG stain.
Articular cartilage from Wistar rat femorotibial joint. Weigert/van Gieson, ×200. (A) Cartilage from a joint of a rat fed standard rat chow. The matrix staining was even and strong. (B) Six months after feeding with T-2 toxin, some collagen was disrupted and some was thickened. (C) Ten months after feeding with T-2 toxin, there were extensive areas of proteoglycan and water loss, and collagen fasciculi.
Discussion
T-2 toxin induced significant articular cartilage degeneration when fed to rats for six and ten months. Cartilage lesions were characterized by chondrocyte degeneration, necrosis, and loss; chondrocyte cloning; fibration; loss of stainable proteoglycan matrix; and exposure of underlying collagen fibers. Thus, we successfully demonstrated that chronic exposure to T-2 toxin induces degenerative articular cartilage lesions similar to spontaneous OA.
T-2 toxin is one of the most toxic naturally occurring trichothecene mycotoxins. It can produce toxic syndromes in humans or animals after ingestion of only tiny amounts, as low as micrograms or nanograms per gram of food (Mngadi et al. 2008). Furthermore, it can destroy joint cartilage of chickens during the development and proliferation stage and can lead to OA (Nascimento et al. 2001; Yang 1995). In China, KBD is an endemic form of OA that is characterized by joint thickening, deformation, short limbs, and dwarfism. Kaschin-Beck Disease has a major detrimental effect on child growth and development. In China, many epidemiological investigations and experimental studies on KBD have been conducted, and T-2 toxin has been implicated in the etiopathogenesis. We have demonstrated that T-2 toxin can damage articular cartilage and induce polyarticular OA-like lesions that are similar to those in human primary OA (Wang et al. 2009; Yang 1994), supporting a potential role of T-2 toxin in KBD.
The mechanisms of cartilage damage induced by T-2 toxin are complex and can involve DNA damage and apoptosis that results from oxidative stress and lipid peroxidation (Huo et al. 1998). In a previous experiment, we found that T-2 toxin induced chondrocyte DNA breakage, necrosis, and apoptosis. The morphological changes in chondrocytes in the present study showed that T-2 toxin induced chondrocyte degeneration, necrosis, and apoptosis. In addition, T-2 toxin could destroy the oxidation–antioxidation balance and decrease the level of total anti-oxidation capability (T-AOC).
The reproducibility, simplicity, and polyarthritic-nature of T-2 toxin–induced degenerative joint disease make it a potentially useful animal model of OA. This model bears similarities to the monosodium iodoacetate (MIA) model of OA (Combe et al. 2004; Guingamp et al. 1997; Guzman et al. 2003). The MIA model is a metabolic model, and the histopathology of the joint degeneration is similar to that seen in the human disease (Janusz et al. 2001). However, a limitation of the MIA model is that it is monoarticular. The T-2-toxin–induced model of OA described in the present study reproduced polyarticular cartilage lesions with loss of proteoglycan matrix. The results of the current study suggest that six months of feeding is sufficient to reproduce the T-2 toxin model of OA in rats. Further refinement of the model may allow for more rapid induction of lesions.
Footnotes
Acknowledgments
We thank members of the key lab of etiology and epidemiology, education bureau of Heilongjiang Province, and the Ministry of Health, China for their scientific input.
The study was supported by: Chinese National Natural Scientific Foundation, contract N30671800; Heilongjiang Provincial Youth Scientific and Technological Foundation, contract NQC06C052; Bureau of Health Foundation, contract N2006–284; and Provincial Government Post-MD Foundation, contract N LBH-Z05199.
