Abstract
Acetaminophen (APAP) overdose could induce liver damage and lead to acute liver failure. The treatment of APAP overdoses could be improved by new therapeutic strategies. Thymus spp., which has many beneficial effects and has been used in folk medicine, is one such potential strategy. In the present study, the hepatoprotective activity of the main constituents of Thymus spp., carvacrol and thymol, were evaluated in light of APAP-induced hepatotoxicity. We hoped to understand the hepatoprotective mechanism of these agents on the antioxidant system and pro-inflammatory cytokines in vitro. Dose-dependent effects of thymol and carvacrol (25, 50, and 100 µM) were tested on cultured HepG2 cells. N-Acetylcysteine (NAC) was tested as positive control. We showed that APAP inhibited HepG2 cell growth by inducing inflammation and oxidative stress. Incubating APAP-exposed HepG2 cells with carvacrol and thymol for 24 h ameliorated this inflammation and oxidative stress. We also evaluated alanine transaminase and lactate dehydrogenase levels of HepG2 cells. We found that thymol and carvacrol protected against APAP-induced toxicity in HepG2 cells by increasing antioxidant activity and reducing pro-inflammatory cytokines, such as tumor necrosis factor α and interleukin 1β. Taking together high-dose thymol and carvacrol treatment has an effect close to NAC treatment in APAP toxicity, but thymol has better treatment effect than carvacrol.
Introduction
Acetaminophen (APAP; N-acetyl-p-aminophenol; paracetamol) is an antipyretic and analgesic drug commonly used for relieving mild and moderate pain. 1,2 In therapeutic doses, APAP is considered safe, but the main clinical risk of a high dose is liver damage, which ultimately leads to acute liver failure and the need for liver transplantation. 2 In both animals and humans, APAP can induce damage to kidneys and other organs by large single-dose ingestions. 3 In 2008, according to the National Poison Control Center of Turkey, there were 5801 cases of APAP overdose (6.78% of all overdose cases), and it has taken first place in the list of drugs that lead to poisoning. 4 Most APAP is metabolized into sulfate and glucuronide conjugation (>80%), and 2% is excreted unchanged into the urine. A small amount of APAP (10–15%) is converted by hepatic cytochrome P450 isoenzymes (mainly CYP2E1) into the hepatotoxic substance N-acetyl-para-benzoquinone imine (NAPQI). NAPQI is deactivated in the liver by conjugating with the antioxidant molecule glutathione (GSH) and can then be excreted via bile after glucuronidation. In APAP overdoses, however, the glucuronide and sulfate pathways are saturated and the normal GSH levels in liver cells are not adequate to inactivate all NAPQI that is formed. 1,2,5,6 The main mechanism by which NAPQI is thought to cause hepatotoxicity is its transformation into cytotoxic protein adducts by covalently binding to mitochondrial proteins, which leads to liver parenchymal cell death; this also affects the immune system. 2,7 Several complex mechanisms were found to be involved in APAP toxicity; however, the mechanism of APAP toxicity in the liver is not fully understood.
Tumor necrosis factor α (TNF-α) is a pro-inflammatory cytokine released by a number of different cells in the liver. The production of TNF-α is known to be one of the earliest events in the hepatic inflammatory response and is capable of stimulating the production and secretion of several other cytokines, including interleukin (IL)-1 and IL-6. 8,9 TNF-α is also important in modulating hepatocyte proliferation and tissue repair in liver injury. 9 IL-1 is structurally and functionally similar to TNF-α. Both are considered “early response cytokines” that are produced rapidly by macrophages in response to inflammatory stimuli. 10 Studies suggest that cytokines, such as TNF-α and IL-1β, are formed after an APAP overdose. 8,9,11 –14 However, the contribution of inflammation to the mechanism of APAP-induced liver injury is still controversial. Blazka et al. demonstrated that APAP-induced hepatotoxicity in mice results in the increased expression and release of inflammatory cytokines (IL-1α and TNF-α), which play a modulatory role in hepatic necrosis. These cytokines play a key role in many of the immediate pathophysiological events following exposure to APAP. 8 In contrast, Nastevska et al. demonstrated a decrease in the release of TNF-α and TNF-α transcripts in primary cultures of hepatocytes and Kupffer cells upon treatment with subtoxic and toxic concentrations of APAP. 15
N-Acetylcysteine (NAC) is used as an antidote to APAP overdose. Administration of NAC, which is a precursor to GSH, results in increased hepatic GSH concentrations and detoxifies NAPQI. 16 While NAC is still used today as the only approved drug to treat APAP overdose patients, both synthetic and natural compounds are being investigated as new treatment strategies that target different mechanisms of APAP toxicity.
Thymus vulgaris is an aromatic medicinal plant that contains carvacrol and its isomer, thymol. Its essential oil has been used in folk medicine, phytopharmaceutical preparations, food preservatives, and as an aromatic ingredient. 17,18 In folk medicine, Thymus spp. has been used for its anthelminthic, antispasmodic, and antioxidative effects. 18 Several studies have been performed with extracts of Thymus spp., 19,20 and a number of studies have also evaluated the therapeutic effects of carvacrol and thymol. 21 –26 Furthermore, studies have shown that carvacrol and thymol suppress the expression and production of TNF-α and IL-6 in vivo and in vitro, respectively. 27,28 However, there is no study to date on the effects of thymol and carvacrol on APAP-induced hepatotoxicity.
The present study was carried out to investigate the hepatoprotective potential of the active ingredients of Thymus spp.—thymol and carvacrol—against APAP-induced toxicity in HepG2 cell lines via inflammatory cytokine and oxidant–antioxidant mechanisms. The expression of inflammatory markers—namely, TNF-α, IL-1β, and biochemical markers such as alanine transaminase (ALT), lactate dehydrogenase (LDH), GSH, superoxide dismutase (SOD), and malondialdehyde (MDA)—which may be targets of the hepatoprotective reactions, have also been investigated. Also the effect of thymol and carvacrol were compared with NAC. Hence, this study aims to investigate the efficacy of thymol and carvacrol in ameliorating the toxic effects of APAP by inhibiting the oxidative damage and inflammation induced by APAP in HepG2 cell lines.
Materials and methods
Chemicals and reagents
Ethanol (EtOH), thymol, carvacrol, APAP (A5000), and NAC were purchased from Sigma-Aldrich (St Louis, Missouri, USA). Dulbecco’s modified Eagle’s medium (DMEM), cell culture medium, and reagents such as fetal bovine serum, penicillin/streptomycin, and trypsin–ethylenediaminetetraacetic acid were obtained from Gibco (Invitrogen Inc., Grand Island, New York, USA).
Cell culture
HepG2 cells were purchased from American Type Culture Collection (Manassas, Virginia. USA). HepG2 cells were grown in DMEM supplemented with 100 U of penicillin/mL, 100 μg streptomycin/mL, and 10% calf serum in a humidified incubator at 37°C in 95% air atmosphere and 5% carbon dioxide (CO2). Cells were plated for RNA extraction and biochemical investigations in 6-well plates (Falcon, BD Biosciences, Heidelberg, Germany). Cultures were grown for 5–6 days at 37°C in 5% CO2, and the medium was changed the day before treatment.
Real-time monitoring of cell growth and proliferation assay
Cell proliferation was measured with the xCelligence system (Roche Diagnostics, Indianapolis, Indiana, USA), which is a label-free technique for dynamic monitoring of living cells. 29 All substances were dissolved in ethanol, and all cells were exposed to 0.1% final EtOH concentration. Control cells are also exposed to 0.1% EtOH. First, 5000 cells/well were seeded in normal medium; after 24 h, the cells were treated with 25, 50, and 100 µM thymol and carvacrol and 5 mM NAC 1 h before 10 mM APAP administration. Literature investigation showed us that 5 mM NAC concentration was effective against APAP toxicity in vitro. 30 The xCelligence system employs 16-well plates with gold electrodes at the bottom of the wells. The interaction between cells and electrodes generates an impedance response that correlates linearly with a cell index to reflect cell number, adherence, and growth. Automatic measurements were performed every 15 min for the duration of the experiment. Data are presented as a normalized cell index ± standard deviation (SD).
Real-time PCR
Total RNA extraction and cDNA synthesis
Cells were homogenized once during 2 min using the TissueLyser II (Qiagen, Hilden, Germany). Total RNA was purified using RNeasy Mini Kit Qiagen according to the manufacturer’s instructions in Qiaqube (Qiagen). The RNA samples were reverse transcribed into complementary DNA (cDNA) by the high-capacity cDNA reverse transcription (RT) kit (Applied Biosystem, Foster City, California, USA). A total of 10 µL RNA was treated with 2 μL 10× RT buffer, 0.8 μL 25× deoxynucleotide mix, 2 μL 10× RT random primers, 1 μL MultiScribe Reverse Transcriptase, and 4.2 μL diethylpyrocarbonate-treated water. RT was carried out at 25°C for 10 min, followed by 37°C for 120 min, and finally at 85°C for 5 min using a Veriti 96-Well Thermal Cycler (Applied Biosystem). The cDNA concentration and quality were assessed and quantified by the Epoch Spectrophotometer System and Take3 Plate (Biotek, Winooski, Vermont, USA).
Relative quantification of gene expression
Relative TNF-α and IL-1β messenger RNA (mRNA) expression analyses were performed with the StepOne Plus real-time polymerase chain reaction (PCR) system (Applied Biosystem) using cDNA synthesized from HepG2 RNAs. Real-time PCR was performed using primers generated for human TNF-α forward: 5′-AGG TTC TCT TCC TCT CAC ATAC-3′; reverse: 5′-ATC ATG CTT TCA GTG CTC ATG-3′; human IL-1β forward: 5′-TGG CAA TGA GGA TGA CTT GTTC-3′; reverse: 5′-CTG TAG TGG TGG TCG GAG ATT-3′; and for human β-actin forward: 5′-GCA AGC AGG AGT ATG ACG AGT-3′; reverse: 5′-CAA GAA AGG GTG TAA CGC AAC TAA-3′ (Primer Design Ltd, Southampton, UK). Results were expressed as relative fold and compared with the control groups. Expression data of β-actin in each cell group were used as endogenous controls. For each cell group, triplicate determinations were performed for both targets in a 96-well optical plate using 9 µL of cDNA (100 ng), 1 µL of Primer Perfect Probe mix, and 10 µL of QuantiTect Probe PCR master mix (Qiagen) in each 20 µL reaction. The plates were heated for 2 min at 50°C, 10 min at 95°C, and a subsequent 40 cycles of 15 s at 94°C and 60 s at 60°C. All data were expressed as fold change in expression compared with the cell groups using the
Biochemical investigations
ALT and LDH measurements
The collected culture medium or culture medium with cell was used to determine ALT levels and LDH leakage with an auto analyzer (ChemWell 2910-ELISA&Biochemistry analyzer, Awareness Technology, Inc., Palm City, FL, USA). All data are presented as mean ± SD results based on per liter.
SOD, GSH, and MDA assay procedure
SOD activity, 32 GSH levels, 33 and MDA levels 34 from each cell supernatant and standards were measured at room temperature with an enzyme-linked immunosorbent assay reader in duplicate according to the modified methods. The average absorbances of each sample and standard were calculated, a standard curve was plotted, and the equation for the absorbance of the standards was obtained. Linear SOD, GSH, and MDA concentrations were calculated according to this equation, and all data are presented as the mean ± SD per milligram protein.
Protein determination
The protein concentrations were determined by Lowry’s method using commercial protein standards (Sigma Aldrich, Total protein kit-TP0300-1KT).
Statistical analysis
All data were subjected to one-way analysis of variance using the IBM SPSS Statistics 20.0 software program (SPSS Inc., Chicago, Illinois, USA). Differences among the groups were determined using the Duncan’s multiple comparison test and were considered significant when the p value was less than 0.05 in a 95% confidence interval. All results were given as mean ± SD.
Results
Cell growth and proliferation
The addition of 10 mM APAP caused a significant decrease in the viability of HepG2 cells after 24, 48, and 72 h (21%, 44%, and 58% of the cells were dead, respectively). NAC treatment as positive control to APAP-treated cells caused significant elevations when compared with APAP-treated cells in all time intervals. The addition of thymol at concentrations 25, 50, and 100 µM for 24, 48, and 72 h caused significant elevations in cell viability. While 100 µM thymol gave the best results for 24 h treatment, 50 µM and 100 µM thymol did not show significant differences after 48 h and 72 h (Figure 1). Cell viability of APAP groups also increased significantly after treatment of 25, 50, and 100 µM carvacrol when compared to APAP-treated cells for each time period (Figure 2). Interestingly, treating cells with 25, 50, and 100 µM carvacrol and thymol without paracetamol administration did not cause cytotoxic effects. It has been observed that 100 µM of thymol increased the viability of cells for 24, 48, and 72 h (Figures 1 and 2). All doses of thymol showed significantly better increase at 24 h, but this elevation decreased at 72 h in contrast to NAC which showed better cell proliferation at the period of time (72 h). Besides, all doses of carvacrol did not show significantly higher cell proliferation for 24, 48, and 72 h.

The viability of the cells treated with APAP and/or thymol. Columns with the same letters are not significantly different; column with different letters indicate significant differences between the groups according to Duncan’s test (α = 0.05). APAP: acetaminophen.
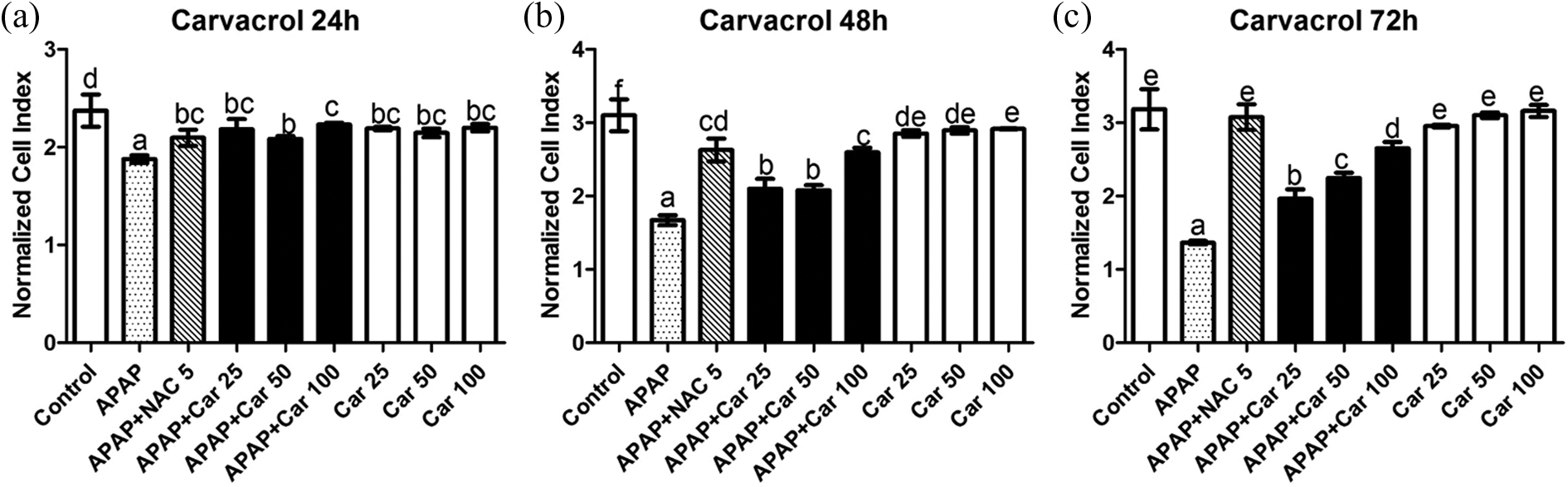
The viability of the cells treated with APAP and/or carvacrol. Columns with the same letters are not significantly different; column with different letters indicate significant differences between the groups according to Duncan’s test (α = 0.05). APAP: acetaminophen.
Oxidative stress
The activity of SOD and the levels of GSH and MDA were evaluated. As shown in Tables 1 and 2, the studies performed in cell culture medium showed that MDA levels, an indicator of lipid peroxidation, were increased in the APAP group compared with the control group cells. NAC administration to APAP-treated cells as positive control increased GSH levels and SOD activity and decreased MDA levels. Addition of thymol and carvacrol to APAP-exposed cells significantly decreased the MDA levels. Levels of SOD activity and GSH, indicators of the antioxidant system, were significantly decreased in the APAP-exposed cells when compared with the control cells. Thymol and carvacrol administration to APAP-exposed cells ameliorated this decrease in SOD activity and GSH levels but not in the same extent than that of NAC treatment. Thymol of 100 µM ameliorates oxidative stress close to NAC effect.
Effect of thymol on biochemical parameters in the control and toxicity induced by APAP in HepG2 cells.
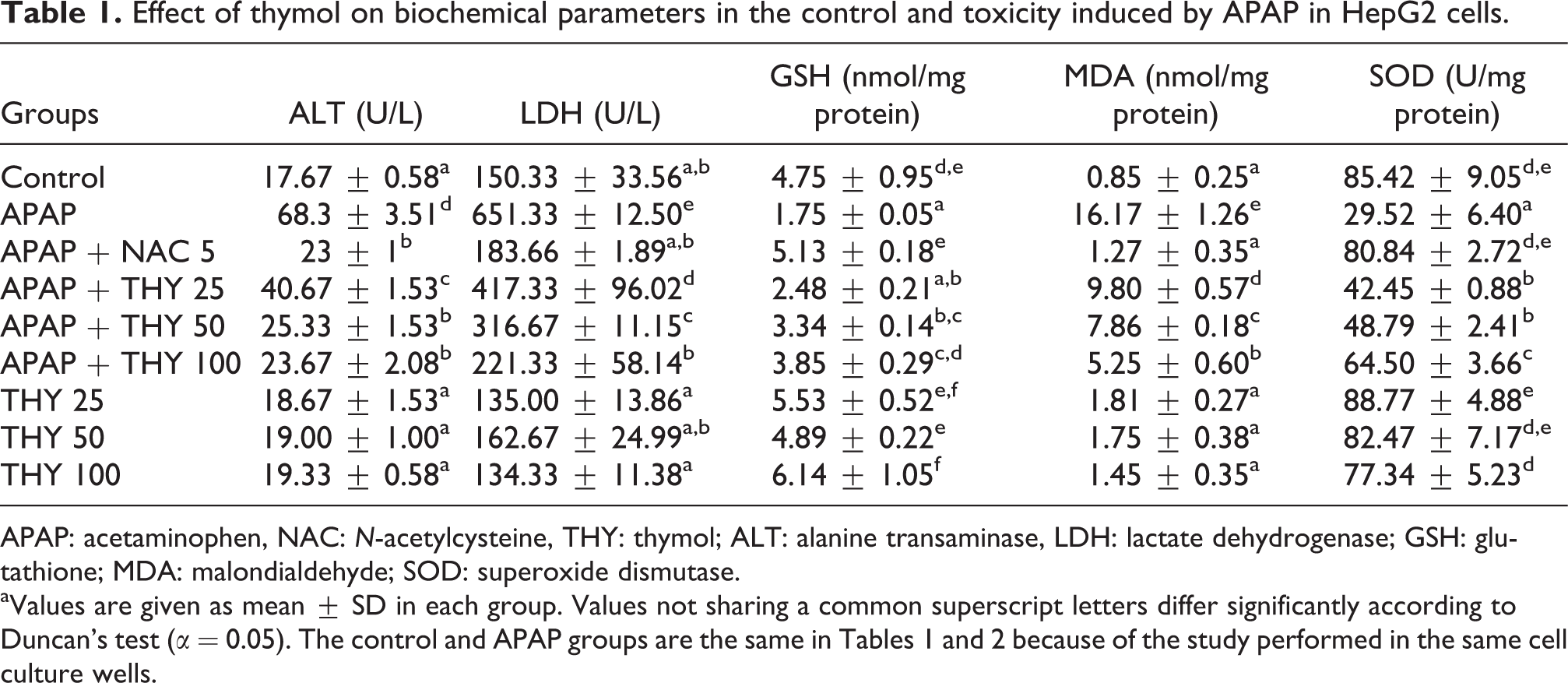
APAP: acetaminophen, NAC: N-acetylcysteine, THY: thymol; ALT: alanine transaminase, LDH: lactate dehydrogenase; GSH: glutathione; MDA: malondialdehyde; SOD: superoxide dismutase. aValues are given as mean ± SD in each group. Values not sharing a common superscript letters differ significantly according to Duncan’s test (α = 0.05). The control and APAP groups are the same in Tables 1 and 2 because of the study performed in the same cell culture wells.
Effect of carvacrol on biochemical parameters in the control and toxicity induced by APAP in HepG2 cells.

APAP: acetaminophen; NAC: N-acetylcysteine; CRV: carvacrol; THY: thymol; ALT: alanine transaminase, LDH: lactate dehydrogenase; GSH: glutathione; MDA: malondialdehyde; SOD: superoxide dismutase. aValues are given as mean ± SD in each group. Values not sharing a common superscript letters differ significantly according to Duncan’s test (α = 0.05).
ALT and LDH levels
ALT and LDH levels are presented in Tables 1 and 2. ALT and LDH levels in APAP-treated cells were increased compared with the control groups, and the increases were statistically significant (p < 0.05). Thymol, carvacrol, and NAC administration significantly decreased the levels of ALT and LDH when compared to APAP-treated cells. The effect of 100 µM thymol and carvacrol reduced LDH activity in the same magnitude close to NAC effect. In addition, 100 µM thymol has an impact on ALT levels in the same magnitude than that of NAC treatment.
Studies of gene expression
The gene expression levels of TNF-α and IL-1β for treated and untreated HepG2 cells were analyzed. TNF-α and IL-1β mRNA expression levels were significantly higher in the cells exposed to APAP than those in control groups. Treatment of HepG2 cells exposed to APAP with 25, 50, and 100 µM concentrations of thymol and carvacrol significantly decreased mRNA expression of TNF-α and IL-1β when compared to APAP-treated cells. NAC also caused significant decrease in pro-inflammatory cytokines when compared to APAP-treated cells. The best protective effect on cytokine expression levels was shown by the addition of 100 µM of thymol. Thymol of 100 µM concentration and carvacrol treatment decreased mRNA expression of TNF-α better than NAC treatment. The effect of 100 µM thymol and carvacrol on mRNA expression of IL-1β is the same as for the NAC treatment. While treatment with 25 µM carvacrol caused a significant decrease in TNF-α mRNA expression levels, 100 µM caused the most significant decrease in IL-1β levels. Treatment of cells with thymol and carvacrol alone did not cause any significant elevations of cytokine mRNA expression levels (Figures 3 and 4).

TNF-α and IL-1β levels of the cells treated with APAP and/or thymol. Columns with the same letters are not significantly different; column with different letters indicate significant differences between the groups according to the Duncan test (α = 0.05). TNF: tumor necrosis factor; IL: interleukin; APAP: acetaminophen.

TNF-α and IL-1β levels of the cells treated with APAP and/or carvacrol. Columns with the same letters are not significantly different; column with different letters indicate significant differences between the groups according to Duncan’s test (α = 0.05). TNF: tumor necrosis factor; IL: interleukin; APAP: acetaminophen.
Discussion
In the present study, we evaluated the hepatoprotective effects of thymol and carvacrol on inflammatory cytokines and oxidative stress mechanisms after APAP-induced toxicity in HepG2 cell lines. We also investigated the effects of different doses of thymol and carvacrol; all were seen to exert significant decreases on inflammatory cytokine levels and ameliorated the oxidative stress. Cell viability, cytokine levels, sensitive markers (ALT and LDH) of liver injury and oxidative stress and lipid peroxidation parameters in cells were also investigated. Using xCelligence to evaluate the viability of cells and real-time PCR to evaluate cell TNF-α and IL-1β mRNA expression levels, we could confirm that APAP exposure mediates cytotoxicity in HepG2 cells and found that both thymol and carvacrol attenuated APAP-mediated cell death due to their antioxidative properties. NAC being used as positive control to compare the effect of thymol and carvacrol administration.
Medicinal plants have been used by humans for thousands of years and have various effects on living organisms. There is growing interest in these natural products and their potential benefits to human health. In vivo and in vitro studies have reported that plants or plant-derived agents can significantly contribute to the treatment of APAP-induced hepatic injury. 35 –38 Thymus spp. has various beneficial effects including antiseptic, antimicrobial, antioxidative, and anti-inflammatory effects. 18 Grespan et al. demonstrated that T. vulgaris essential oil (including carvacrol and thymol) is hepatoprotective against APAP-induced hepatic damage in mice. 36 But this study was limited by the lack of information on the nature of T. vulgaris constituents and the molecular mechanism underlying the observed activity. Carvacrol is the major constituent of Thymus spp., possessing anti-inflammatory, antioxidant, and hepatoprotective potential. 17 Hepatoprotective effect of carvacrol has been shown in studies of d-galactosamine- and thioacetamide-induced hepatotoxicity in rats 22,39 while that of effect of thymol has also been reported in studies of carbon tetrachloride-induced hepatotoxicity in mice. 25 Our study is the first one investigating the effects of thymol and carvacrol in an APAP-induced in vitro hepatotoxicity model.
HepG2 cell lines have been selected because of their unlimited life span in culture and their drug-metabolizing enzymes are more stable than primary cultures of hepatocytes. In addition, such cells lines are preferred in metabolism and toxicity studies related to hepatotoxic drugs. 30,40 The effect of thymol and carvacrol on cell viability were assessed in different in vitro models. Ferraz et al. evaluated the cytotoxic activity of thymol on HepG2, K562, B16-F10, and peripheral blood mononuclear cell (PBMC) cell lines. Thymol was found only cytotoxic in B16-F10 and its half maximal inhibitory concentration (IC50) values are higher than 25 µM in other cell lines including HepG2. 41 Yin et al. and Melusova et al. evaluated the antiproliferative effect of carvacrol on HepG2 cell line and they found the IC50 value 400 and 425 µM, respectively. 42,43 Cabello et al. evaluated the wide range concentration of thymol and carvacrol in Caco-2 cell line. According to this study, thymol did not have cytotoxic effects up to 250 µM and 460 µM half maximal effective concentration value defined for carvacrol. 44 Finally, Deb et al. showed no cytotoxic effect on healthy PBMCs up to 100 µM. 45 The different results seen in these studies may probably be due to different metabolic activities of the cells and the methods that were used to measure the cytotoxic activity. Some phenolic compounds are oxidized and can produce highly reactive phenoxyl radicals, which lead to the generation of reactive oxygen species (ROS). At higher concentrations, cells cannot cope with these radicals and these antioxidant compounds are prooxidant and affect the cell viability at high concentrations. 46 According to this studies we defined our highest dose 100 μM for both carvacrol and thymol which has no cytotoxic effect on healthy cells and also other cell lines. We demonstrated that HepG2 cells are susceptible to APAP exposure, which leads to decreased cell viability compared with control cell cultures. This effect is similar to data previously published in a study about APAP toxicity in HepG2 cells. 47 With continued exposure to APAP, time-dependent cytotoxic effects on HepG2 cells were seen. Ozkan and Erdogan reported that carvacrol and thymol reduced hydrogen peroxide (H2O2)-induced cytotoxicity in HepG2 cells. 48 Similarly, we found that all doses of thymol and carvacrol significantly increased cell viability when added to APAP-exposed cell cultures at all time intervals. Despite their effect on cell proliferation, NAC treatment still increased cell proliferation better than all doses of thymol and carvacrol administration in APAP-treated cells at 72 h.
ALT is commonly used as a standard biomarker to diagnose drug-induced liver injury. ALT released from the hepatocytes due to the damage. Released enzyme enter into the circulation and serum ALT levels increased due to the hepatic injury. 49 In the present study, significantly increased ALT levels were observed in APAP-treated cells due to the damaged of the cells when compared with the control cells. In all doses of carvacrol and thymol, the levels of ALT were significantly decreased when compared to APAP-treated cells. Previous studies performed in vivo and in vitro on chemically induced hepatotoxicity obtained similar ALT results. 22,36,38,50 In this study, ALT levels were used to indicate hepatotoxicity. NAC treatment decreased the ALT levels in the APAP-treated cells and showed protective effect. The decline in ALT levels with thymol and carvacrol treatment suggests that thymol and carvacrol attenuated cell injury, which is the primary evidence of the hepatoprotective effect of these compounds. The concentrations of 50 and 100 µM thymol showed similar decrease with NAC treatment. LDH is an indicator of cytotoxicity; it is released due to lysis or leakage from injured cells. APAP toxicity also leads to the leakage of LDH into the cell supernatant, which indicates the destabilization of the cellular membrane. In line with our results, Manov et al. 30 reported that leakage of LDH increased in cultured Hep3B cells after 24 h exposure to APAP and that treatment with NAC led to reduced LDH levels. Thymol and carvacrol administration to APAP-exposed cells was also found to decrease leakage of LDH with attenuated cell injury. Only 100 µM thymol and carvacrol showed similar recovery with NAC treatment on LDH and ALT levels.
Another possible indicator of liver injury is oxidative stress. In previous studies, overdoses of APAP have been reported to cause oxidative stress and lipid peroxidation. 12 –14 Depletion of GSH levels by toxic metabolite NAPQI and GSH is the cofactor of GSH peroxidase enzyme, which detoxified peroxides. Due to the GSH depletion, intracellular peroxide levels are increased and highly reactive hydroxyl radicals oxidized lipids in the cell membranes. 51 Peroxidation of lipids leads to changes in membrane structure, tissue damage, and failure of antioxidant defense mechanisms to prevent the formation of free radicals. 52 Free radicals attack lipids containing carbon–carbon double bound with oxygen insertion and cause lipid peroxyl radicals and hydroperoxides formation. Lipid peroxidation produces a wide variety of oxidation products. 53 MDA, which is an important end product of lipid peroxidation, is often used as a biomarker to measure the level of lipid peroxidation. 52 In APAP-induced toxicity, depletion of GSH may lead to increased intracellular peroxide levels and cause the formation of the highly reactive hydroxyl radical, which may in turn oxidate lipids. 51 In the present study, MDA levels were dramatically increased in cells treated with APAP when compared with control cell cultures. This situation is closely related to APAP-induced lipid peroxidation that leads to cell damage because of oxidative stress. The conventional treatment NAC led to decreased MDA level in APAP-exposed cells. This study also found treatment with thymol and carvacrol to significantly inhibit these MDA levels but neither thymol nor carvacrol decreased MDA levels in the same extent with NAC treatment. This effect may be due to the antioxidant activities of thymol and carvacrol, which have also been demonstrated in previous studies. 22,25 Therefore, thymol and carvacrol are thought to have many preventative effects against lipid peroxidation and oxidative stress.
GSH is a tripeptide nonenzymatic antioxidant that scavenges ROS such as H2O2 and superoxide radicals. 54 It plays a key role in the detoxification of the cytotoxic, highly electrophilic APAP metabolite, NAPQI. When APAP overdose depletes GSH in the liver, NAPQI reacts with cellular lipids, proteins, and other cell structures, leading to liver damage. 2 In APAP overdose, clinical and experimental studies have shown depleted GSH levels. 12,30,50 In the present study, GSH level in the APAP-treated cells substantially decreased when compared with the control group. Similarly, Nafees et al. 22 demonstrated that carvacrol leads to increased GSH levels in thioacetamide-induced hepatotoxicity in Wistar rats. NAC is a precursor of GSH and caused significant increase of GSH levels in APAP-treated cells. Cells treated with all doses of thymol and carvacrol exhibited significant increases in GSH when compared with the APAP group but not as well as NAC treatment. Apart from the treatment with 100 µM thymol, better results with NAC treatment were only observed in cell proliferation despite the depletion of GSH observed with thymol and carvacrol treatment. This seem to be originated from replacement of GSH by NAC. 16 However, thymol and carvacrol reduce better pro-inflammatory cytokines TNF-α and IL-1β than NAC.
Antioxidants play an important role in protecting against APAP-induced injury by preventing oxidative damage.
55,56
SOD is an antioxidant enzyme that protects cells against oxidative damage induced by superoxide radicals.
54
Our previous studies have demonstrated decreases in SOD activity in APAP toxicity.
12
–14
SOD reduces the toxic effect caused by superoxide anions by scavenging this anion to form H2O2. In the present study, SOD enzyme activity in cells was significantly lower in the APAP-treated cells. This reduction in SOD activity in APAP toxicity may be due to the overproduction of superoxide radical anions and the result of futile cycling of CYP caused by NAPQI, which reduces NADPH with the molecular superoxide anion radical,
In many inflammatory diseases, production of ROS plays a central role acting as both signaling molecules and mediators of inflammation. 57 TNF-α is a pro-inflammatory cytokine with an important role in inflammation. One of the main pathways in TNF-α signaling is its proapoptotic pathway—complex II and ROS, both functioning as downstream mediators. ROS generation contributes to TNF-α–induced cell death. ROS involved in both cell survival and cell death, with induction of mitochondrial membrane permeabilization and sustained c-Jun N-terminal kinase activation. 58 TNF-α is assumed to act as a mediator in toxic liver injury. TNF-α transcript concentrations and serum TNF-α concentrations in overdoses of APAP have shown conflicting results under different experimental and clinical conditions. 15 IL-1β is another pro-inflammatory cytokine where increased levels of IL-1β are also associated with APAP overdoses. 59 Inhibition of TNF-α and IL-1β can partially improve APAP-induced liver toxicity. 8 In our study, we examined cells for TNF-α and IL-1β mRNA expression using real-time PCR, and we investigated the role of thymol and carvacrol on TNF-α and IL-1β in the pathogenesis of APAP-induced toxicity in HepG2 cells and compare the effect with NAC. Expression levels of both cytokines were significantly higher in APAP-treated cells than the control group. The increase in TNF-α and IL-1β mRNA expression in APAP-treated cells shows that APAP induces inflammation. The data from the present study are consistent with previous studies. James et al. showed that IL-1β was significantly increased in APAP-treated mice. 59 Similarly, Blazka et al. reported that neutralizing antibodies of TNF-α or IL-1 were partially hepatoprotective in the APAP-treated mice. 8 Thymol and carvacrol were also shown to reduce TNF-α and IL-1β expression in previous studies. 24,27,28 According to our data, all doses of thymol and carvacrol have a beneficial effect on TNF-α and IL-1β expression when compared with the APAP group. Thymol (100 µM) and carvacrol (100 μM) treatment have significantly decreased TNF-α mRNA expressions compared to NAC treatment and the effect on IL-1β mRNA expressions does not significantly differ from NAC treatment. The present study suggests that thymol and carvacrol can downregulate the expression of pro-inflammatory cytokines and the effect on TNF-α expressions is better than that observed in NAC treatment.
In conclusion, these results suggest that a high dose of APAP alters biomarkers related to oxidative stress, decreases antioxidant activity, and elevates pro-inflammatory cytokines. Thymol and carvacrol protect HepG2 cells against APAP-induced toxicity by increasing antioxidant activity and reducing TNF-α and IL-1β. In addition, thymol and carvacrol exert protective effects against experimentally induced APAP toxicity in vitro. The protective effects of thymol and carvacrol are possibly due to decreased oxidative stress, improved antioxidant defense, and inhibition of pro-inflammatory cytokines. We suggest that pro-inflammatory cytokines in APAP-induced cytokines are upregulated and thymol and carvacrol protected cell damage by inhibiting these cytokines. This TNF-α inhibition has better outcome than that observed with NAC treatment in high doses of thymol and carvacrol. The mechanisms of thymol and carvacrol in this model may be mediated by suppression of the inflammatory response of cells, which would reduce liver injury. Taking all together, high doses of thymol and carvacrol has similar effect in reducing APAP toxicity than NAC treatment. Thymol has better treatment effect than carvacrol. However, the protective effects of thymol and carvacrol against APAP toxicity still needs further investigation to fully elucidate their molecular mechanism.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific Research Council of Atatürk University (number 2014/22).
