Abstract
Tri-ortho–cresyl phosphate (TOCP) has been widely used as plasticizers, plastic softeners, and flame retardants in industry and reported to have delayed neurotoxicity and reproductive toxicology in animals. However, it remains to be elusive whether TOCP induces liver injury. In this study, male mice were orally administered different concentrations of TOCP (100, 200, or 400 mg/kg/day) for 28 days. Histological examination showed that TOCP led to serious hepatocellular injury. In addition, administration of TOCP induced a marked elevation in the serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels in mice. The content of malondialdehyde (MDA) was increased significantly in the liver after the mice were treated with TOCP; while there was a dramatic decrease in the content of glutathione (GSH) and the activities of antioxidative enzymes superoxide dismutase (SOD) and glutathione peroxidase (GSH-PX). TOCP inhibited viability of mouse liver cancer Hepa 1-6 cells in a dose-dependent manner. Meanwhile, TOCP significantly increased MDA content and inhibited GSH content and the activities of SOD and GSH-PX in the cells, respectively. Oxidative stress dramatically inhibited viability of Hepa 1-6 cells; while inhibition of oxidative stress by N-acetyl-
Introduction
Reactive oxygen species (ROS) are chemically reactive molecules containing oxygen, which include both free radicals, such as nitric oxide (NO•), superoxide (O2•−), hydroxyl radical (OH•) and peroxides. 1 ROS can be formed as a natural byproduct of oxygen metabolism, and can be scavenged by reduced glutathione (GSH) and antioxidant enzymes such as SOD and glutathione peroxidase (GSH-PX). When there is an imbalance of ROS production and the cellular antioxidant defense system, oxidative stress occurs. 2
ROS are shown to be involved in modulating transition from G1 to S phase, which needs a temporary increase in mammalian cells during the transition; high concentration of ROS, however, leads to cell cycle arrest. 3 Oxidative stress can also induce autophagy and apoptosis in mammalian cells. 4 –6 Oxidative stress has been implicated as a critical pathophysiological mechanism of hepatocytic toxicity from environmental chemicals and organophosphorus compounds (OPs). 7,8 It shows that oxidative stress can be induced by tri-ortho–cresyl phosphate (TOCP) in cerebrum, spinal cord and sciatic nerve of hens. 9
TOCP has been widely used as plasticizers, plastic softeners, jet oil additives, and flame retardants in industry. 10,11 TOCP mainly induces a delayed neurodegenerative condition known as OP-induced delayed neuropathy, while does not exert their acute toxic effects by suppressing acetylcholinesterase. TOCP affects both the central and the peripheral nerves in sensitive species such as humans and hens. 12 –15
It has been reported that TOCP has been shown to induce reproductive toxicology, besides neurotoxicity in animals. TOCP has been shown to decrease sperm density in the cauda epididymal, inhibit testicular enzyme activities and disrupt the seminiferous epithelium in rats and mice. 16 –19 TOCP can also decreases the fertility index in Swiss (CD-1) mice, and lead to decrease in the number of live-born pups per litter. 20 We recently found that TOCP can inhibit viability of rat spermatogonial stem cells and induce autophagy of the cells, without affecting cell cycle and apoptosis. 21 Recent studies showed that TOCP can also induce autophagy in human neuroblastoma SH-SY5Y cells. 22,23 Moreover, autophagy is involved in TOCP-induced delayed neuropathy in hens. 24
The liver is a vital organ in vertebrates, which has essential physiological functions, including clearance and inactivation of drugs, detoxification, synthesis of plasma proteins, and regulation of carbohydrate and lipid metabolism. 25 It needs a lot of enzymes to maintain these physiological functions, which is essential for survival. Cytochrome P450, one of the most important enzymes in liver, can convert TOCP into saligenin cyclic-o-tolyl phosphate (SCOTP), which is more toxic to the body. 26 –28 However, whether TOCP induces liver injury and the precise mechanisms remain to be elusive.
The aim of the present study is to investigate whether TOCP induces liver injury in male mice and whether oxidative stress is involved in TOCP-induced hepatocellular injury.
Materials and methods
Materials
TOCP (purity > 99.0%) was purchased from BDH Chemicals Co. Ltd (Poole, England). Commercial oxidation–antioxidation assay kits of malondialdehyde (MDA), GSH, SOD and GSH-PX were bought from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Mouse liver cancer Hepa 1-6 cells were purchased from the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China). Cell culture reagents were obtained from Gibco BRL (Grand Island, New York, USA). Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) kits were obtained from Nanjing Jiancheng Bioengineering Institute. N-acetyl-
Animal studies
Adult male Kunming mice (8 weeks old and 20–25 g in size) were purchased from the Shanghai Laboratory Animal Center, Chinese Academy of Sciences. The mice were maintained under standard conditions with access to water and food ad libitum and acclimatized for 1 week prior to the start of the experiment. All animal procedures were performed in compliance with current China legislation and approved by the Animal Ethics Committee of Nanchang University.
Animal administration
Mice were orally administered at different concentrations of TOCP (0, 100, 200, and 400 mg TOCP/kg/day) dissolved in dimethyl sulfoxide (DMSO). 18 On completion of evaluations at 28 days postdosing, the mice were killed by cervical dislocation after anesthesia with sodium pentobarbital. Blood samples were collected and liver tissues were quickly dissected and frozen in liquid nitrogen before storing at −80°C.
Histology
Male mice liver tissues were dissected, fixed in formalin solution, embedded in paraffin, sectioned (4 μm), and stained with hematoxylin and eosin as described. 29
Measurement of serum enzymes
The blood samples were centrifuged at 12,000g at 4°C for 30 min to separate serum. The activities of serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were determined according to the method of Reitman–Frankel using commercially available assay kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China).
Measurement of oxidative stress in liver tissue
Liver samples were homogenized, and then centrifuged at 600g for 10 min at 4°C, and the supernatants were analyzed for the contents of MDA and GSH and the activities of SOD and GSH-PX using kits according to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Protein concentration was assayed using the Bradford protein assay. The concentrations of MDA and GSH were expressed as nmol/mg protein and mg/g protein, respectively. The activities of SOD and GSH-PX were both expressed as U/mg protein.
Cell culture
Mouse liver cancer Hepa 1-6 cells were grown and maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum, 100 IU/mL penicillin and 100 μg/mL streptomycin. Incubations were carried out at 37°C in a humidified atmosphere of 5% CO2 in 95% air atmosphere. The cells were maintained in the logarithmic phase of growth and subcultured at 3–4 days intervals.
MTT reduction assay
The cells (1 × 104 cells/well) were seeded in a 96-well culture plate and were incubated with fresh medium containing 0, 100, 200 and 400 Mm hydrogen peroxide (H2O2) for 48 h or 0–1.0 mM TOCP in absence or presence of 5 mM NAC for 48 h. TOCP were dissolved in DMSO, the final concentration of DMSO in the culture medium was 0.1% (v/v). Forty-eight hours later, cell viability was assessed by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Cell medium containing 0.5 mg/mL MTT was replaced to each well and incubated at 37°C in 5% CO2 in 95% air atmosphere for 4 h. The formed insoluble formazan was dissolved in DMSO, and the absorbance was measured in a spectrophotometer at 570 nm with a background reading of 660 nm.
Measurement of oxidative stress in Hepa 1-6 cells
Mouse liver cancer Hepa 1-6 cells (1 × 106 cells) were seeded in a 60 mm culture plate and were incubated with fresh medium containing 0–1.0 mM TOCP for 48 h. The cells were homogenized and centrifuged at 600g for 10 min at 4°C, and then the supernatants were analyzed for the contents of MDA and GSH and the activities of SOD and GSH-PX using kits according to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Protein concentration was assayed using the Bradford protein assay.
Statistical analysis
Values are expressed as means ± SE. Data were evaluated by one-way analysis of variance (ANOVA) with a Newman–Keuls multiple range test. For each test, p values less than 0.05 (p < 0.05) were considered statistically significant.
Results
TOCP induces liver tissue injury
In order to investigate the effect of TOCP on liver tissue, male mice were administrated with indicated concentrations of TOCP (0, 100, 200, and 400 mg TOCP/kg/day) for 28 days. As shown in Figure 1, the vehicle-treated mice showed normal liver tissue arrangement; while the liver tissue showed mild deranged histoarchitecture and edema in mice receiving 100 and 200 mg TOCP/kg/day group. In the 400 mg TOCP/kg/day group, marked changes in liver tissues were observed, and the liver sections showed disorganized liver architecture, severe edema, and vacuolar degeneration. The disruption and atrophy of the liver tissues suggested that TOCP exposure caused liver injury in male mice.

TOCP induces liver tissue injury. Livers were obtained from male mice treated with 0 mg (a and b), 100 mg (c and d), 200 mg (e and f) or 400 mg (g and h) TOCP/kg/day for 28 d. The livers were stained with hematoxylin and eosin and then were visualized under a IX51 Olympus microscope. Arrow indicated edema and vacuolar degeneration in liver tissue. Magnification: (a, c, e, and g) ×200; (b, d, f, and h) ×400. TOCP: tri-ortho–cresyl phosphate.
To further determine the hepatotoxicity of TOCP in mice, serum ALT and AST levels were observed. Compared with the control, TOCP induced a marked elevation in the serum ALT and AST levels in mice (Figure 2). These results showed that administration of TOCP indeed induced liver injury of male mice.

Effect of TOCP on serum ALT and AST levels. Male mice were treated with 0–400 mg TOCP/kg/day for 28 days; then serum ALT (a) and AST (b) levels were observed. The experiment was done in triplicate and repeated three times. Data were analyzed by one-way ANOVA. *p < 0.05. TOCP: tri-ortho–cresyl phosphate; ALT: alanine aminotransferase; AST: aspartate amino transferase; ANOVA: analysis of variance.
TOCP induces oxidative stress in liver tissue
To investigate whether TOCP exposure induces oxidative stress in the liver of the mice, MDA was determined in the liver tissues after the mice were treated with indicated concentration of TOCP (0–400 mg/kg/day) for 28 days. We found that MDA significantly increased in the liver tissue of the mice receiving 200 and 400 mg TOCP/kg/day group, which indicated that TOCP induced hepatic lipid peroxidation Meanwhile, TOCP decreased GSH content in a dose-dependent manner. The activities of antioxidant enzymes SOD and GSH-PX were also dramatically decreased in the TOCP-treated group (Figure 3). These results indicated that TOCP could induce oxidative stress in liver tissue.
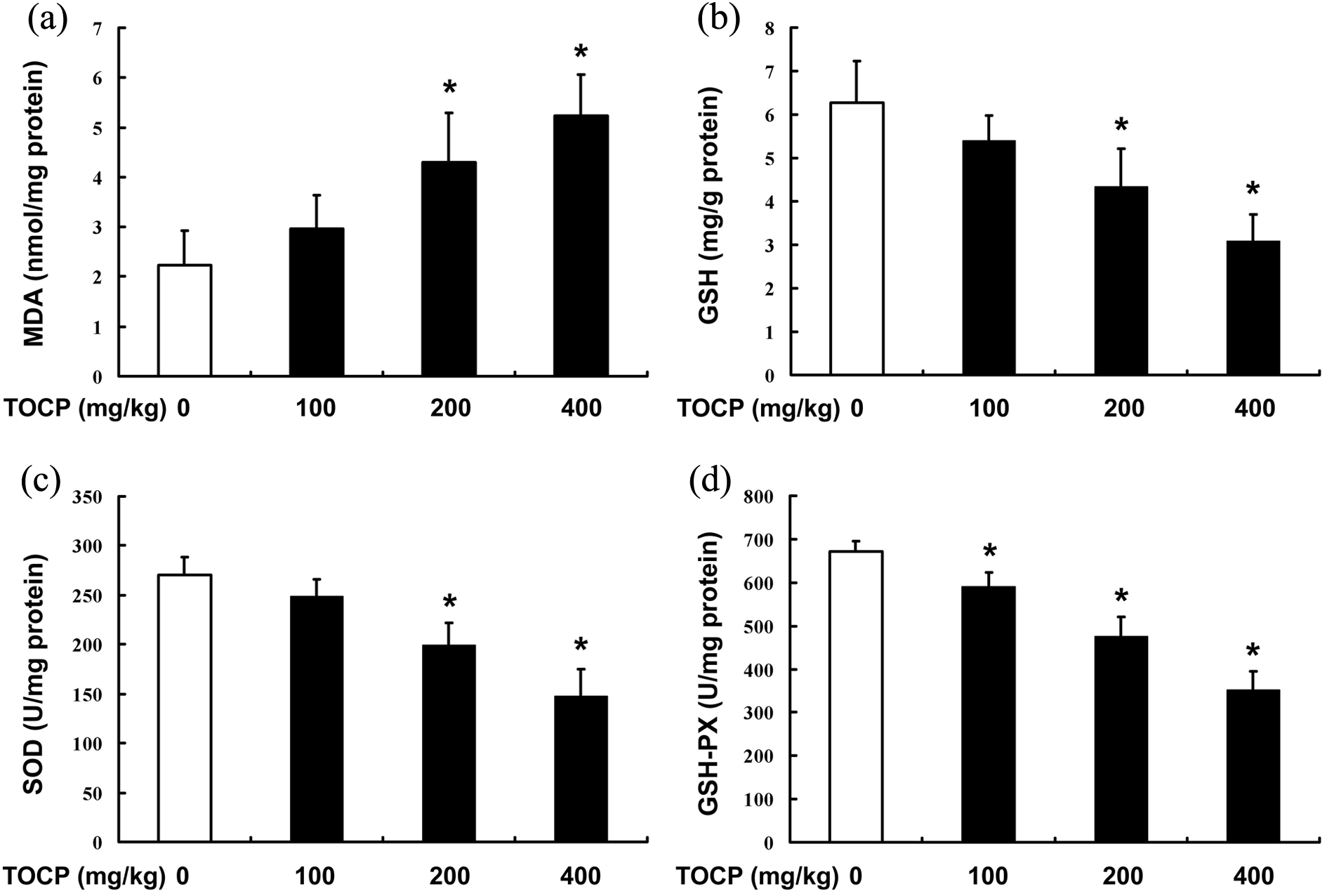
TOCP induces oxidative stress in liver tissue. Male mice were treated with 0–400 mg TOCP/kg/day for 28 d; then the contents of MDA (a) and GSH (b) and activities of SOD (c) and GSH-PX (d) in the liver samples were determined. The experiment was done in triplicate and repeated three times. Data were analyzed by one-way ANOVA. *p < 0.05: compared with control. TOCP: tri-ortho–cresyl phosphate; MDA: malondialdehyde; GSH-PX: glutathione peroxidase; SOD: superoxide dismutase; ANOVA: analysis of variance.
TOCP inhibits viability of mouse liver cancer Hepa 1-6 cells
To observe whether TOCP induces hepatocellular injury, cell viability was determined after mouse liver cancer Hepa 1-6 cells were treated with 0–1.0 mM TOCP for 48 h. As shown in Figure 4, TOCP inhibited cell viability in a dose-dependent manner, which indicated that TOCP could induce hepatocellular injury.

TOCP inhibits viability of Hepa 1-6 cells. Hepa 1-6 cells were treated with 0–1.0 mM TOCP for 48 h; then cell viability was observed by MTT assay. The experiment was done in triplicate and repeated three times. Data were analyzed by one-way ANOVA. *p < 0.05. TOCP: tri-ortho–cresyl phosphate; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; ANOVA: analysis of variance.
Involvement of oxidative stress in TOCP-induced hepatocellular injury
To investigate the potential mechanism of TOCP inhibiting viability of mouse liver cancer Hepa 1-6 cells, the contents of MDA and GSH and the activities of SOD and GSH-PX were determined after the cells were treated with the indicated concentration of TOCP (0–1.0 mM) for 48 h. As shown in Figure 5(a), TOCP dramatically increased the MDA level in the cells in a dose-dependent manner; while GSH significantly decreased in the cells treated with TOCP (Figure 5(b)); the activities of antioxidant enzymes SOD and GSH-PX were also inhibited dramatically in the TOCP-treated cells (Figure 5(c) and (d)), which suggested that TOCP could induce oxidative stress of Hepa 1-6 cells.
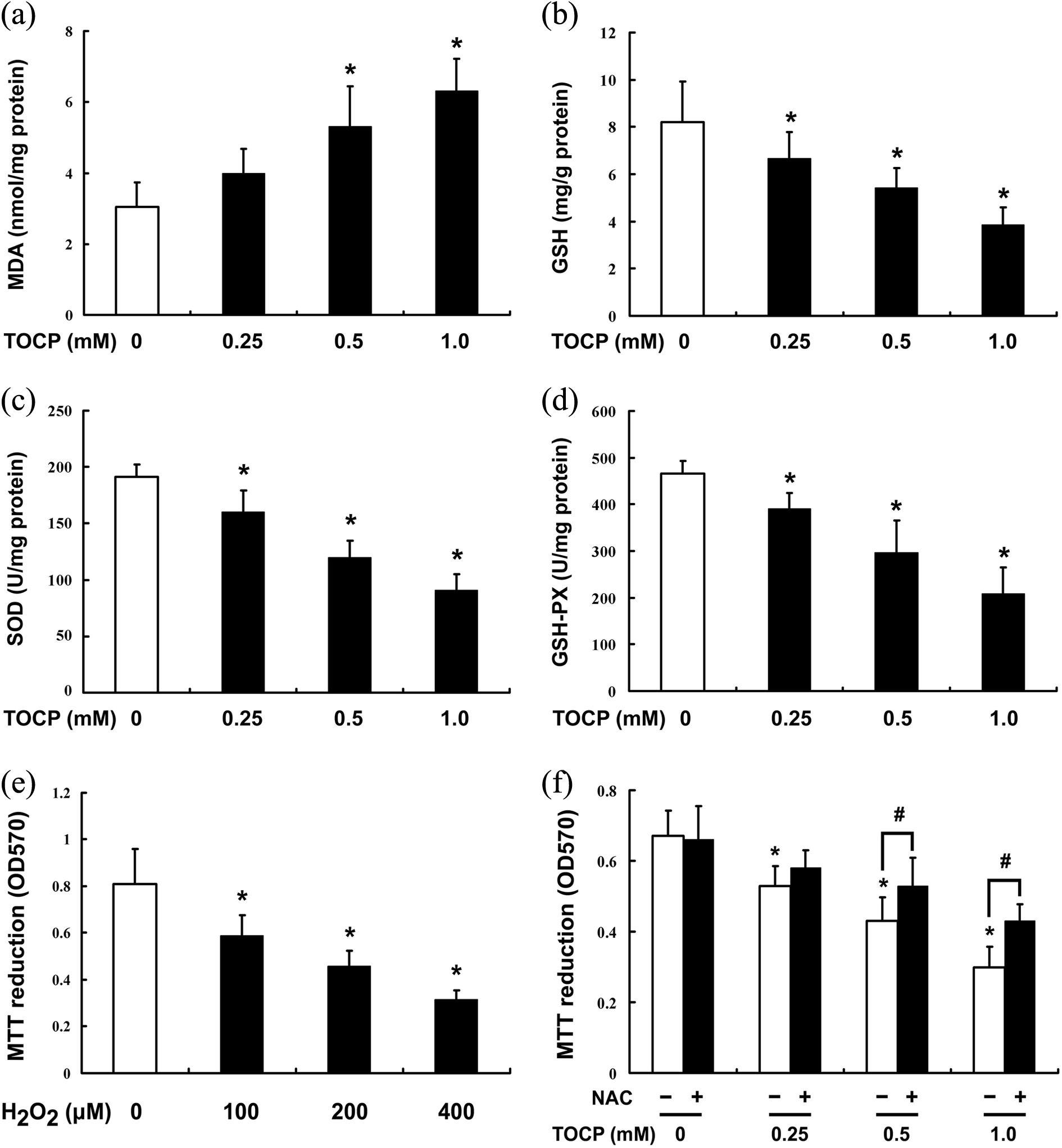
Oxidative stress is involved in TOCP-induced hepatocellular injury. Hepa 1-6 cells were treated with 0–1.0 mM TOCP for 48 h; then the contents of MDA (a) and GSH (b) and the activities of SOD (c) and GSH-PX (d) were determined. The cells were treated with 0-400 μM H2O2 for 48 h (e) or 0–1.0 mM TOCP for 48 h in the absence or presence of 5 mM NAC (f); then cell viability was observed by MTT assay. These experiments were done in triplicate and repeated three times. Data were analyzed by one-way ANOVA. *p < 0.05: versus 0 mM TOCP in the absence of NAC. #
p < 0.05: compared with the same TOCP concentration without NAC treatment. TOCP: tri-ortho–cresyl phosphate; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; MDA: malondialdehyde; GSH-PX: glutathione peroxidase; SOD: superoxide dismutase; ANOVA: analysis of variance; NAC: N-acetyl-
To further identify whether oxidative stress was involved in TOCP-induced hepatocellular injury, cell viability was determined after Hepa 1-6 cells with treated with 0, 100, 200, and 400 μM H2O2 for 48 h or treated with 0–1.0 mM TOCP for 48 h in the presence or absence of 5 mM NAC. As shown in Figure 5(e), H2O2 inhibited cell viability in a dose-dependent manner, inhibition of oxidative stress by NAC, however, could rescue the cell viability inhibited by TOCP to a certain degree (Figure 5(f)), which indicated that oxidative stress was involved in TOCP-induced hepatocellular injury.
Discussion
In the present study, we provided the first direct evidence that TOCP led to liver injury and induced oxidative stress; and inhibition of oxidative stress can protect TOCP-induced hepatocellular injury.
TOCP has been widely used in the industry, which can induce neurotoxicity, 10,11 reproductive toxicology, 17 –21 and immunotoxicity 30,31 in animals. TOCP can be activated into SCOTP by cytochrome P450 in the liver, which indicates that the liver might be injured by exposure of TOCP. However, it is still unknown whether TOCP can lead to liver injury.
In order to evaluate whether TOCP induces liver injury, male mice were administered with indicated concentrations of TOCP. Liver tissues showed disorganized liver architecture, severe edema, and vacuolar degeneration after the mice were treated with TOCP. To further explore the effect of TOCP on liver injury, the serum levels of aspartate amino transferase (AST) and alanine amino transferase (ALT), the well-known biomarkers for the hepatic injury or disorders, were determined. 32 Compared with the control, TOCP induced a significant elevation in the serum ALT and AST levels in mice. These results showed that TOCP exposure caused liver injury in male mice.
TOCP is shown to induce oxidative stress in cerebrum, spinal cord and sciatic nerve of hens. 9 Oxidative stress can be induced by environmental chemicals and OPs, which lead to hepatocytic toxicity. 7,8 However, it remains to be elusive whether involvement of oxidative stress in TOCP-induced liver injury in male mice.
To investigate whether administration of TOCP induces oxidative stress in the liver of the mice, MDA, a lipid peroxidation indicator, was determined in the liver tissues after the mice were treated with TOCP. We found that TOCP significantly increased MDA content in the liver tissues. Meanwhile, there was a decrease in GSH content and the activities of antioxidant enzymes SOD and GSH-PX in the TOCP-treated group. These results indicated that TOCP could induce oxidative stress in liver tissue.
To further confirm that whether TOCP induced hepatocellular injury, cell viability was determined in mouse liver cancer Hepa 1-6 cells after the cells were treated with 0–1.0 mM TOCP for 48 h. TOCP was shown to inhibit viability of Hepa 1-6 cells in a dose-dependent manner, which suggested that TOCP might induce hepatocellular injury. To further observe whether oxidative stress is involved in TOCP-induced hepatocellular injury, the contents of MDA and GSH and the activities of SOD and GSH-PX were also determined in Hepa 1-6 cells after the cells were treated with 0–1.0 mM TOCP for 48 h. TOCP significantly increased MDA content and decreased GSH level, respectively; while the activities of SOD and GSH-PX were both inhibited by TOCP treatment. Treatment the cells with H2O2 indeed inhibited cell viability; inhibition of oxidative stress by NAC, however, can rescue the cell viability inhibited by TOCP to a certain extent. These results suggested that oxidative stress was involved in TOCP inhibiting viability of Hepa 1-6 cells.
In summary, oxidative stress might be involved in TOCP-induced hepatocellular injury in male mice. This study sets in motion our future investigation of the mechanisms underlying TOCP-induced hepatocellular injury.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant from National Natural Science Foundation of China (No. 81200433, No. 81360098), Jiangxi Provincial Natural Science Foundation (20114BAB215020) and Jiangxi Provincial science and technology support plan project (No. 20112BBG70029).
