Abstract
Colorectal cancer (CRC) is a serious health problem throughout the world. 5-Flurouracil, the first-line chemotherapy of colorectal cancer often produces more toxicity to neighboring cells; however, it is still used for CRC treatment. To overcome this, umbelliferone (UMB), a less toxic bioflavonoid has been used to test its anticancer effects on animal model. The objective of the present study is to evaluate the anticancer activity of UMB on 1,2-dimethylhydrazine (DMH)-induced rat colon tumorigenesis to determine the development of aberrant crypt foci (ACF), agyrophylic nucleolar organizer regions (AgNORs), mast cell recruitment, pro-inflammatory cytokines such as tumor necrosis factor (TNF)-α, interleukin (IL)-1β and also study the expressions of inducible nitric oxide synthase, cyclooxygenase (COX)-2, and apoptotic markers. DMH-induced rats showed increased ACF number (incidence), multiplicity and its distribution, counts of AgNORs, mast cells, inflammatory markers and apoptotic proteins. Interestingly, UMB supplementation to DMH-induced rats (group 4) significantly (p < 0.05) suppressed ACF development, AgNORs, mast cells, and inflammatory markers and increased the apoptotic markers as compared to DMH-induced rats (group 2). We concluded that UMB is a potential anticancer agent that can be used for the prevention and treatment of CRC.
Introduction
Colorectal cancer (CRC) is one of the most common malignancies in many parts of the globe. 1,2 The incidence of this cancer has been accelerating in recent years. This is because there are moderate changes in the diet of Indians to “Western type,” which mainly includes high fat, low carbohydrates, and fiber. 3,4
It is widely accepted that 1,2-dimethylhydrazine (DMH) induces CRC by initiating and promoting steps of colon carcinogenesis and had produced colon tumors in rodents. This demonstrates pathological characteristics that are similar to sporadic forms of human CRC. 5,6 DMH is an alkylating agent that targets DNA and stimulates the formation of methyl adducts with DNA bases, point mutations, micronuclei and sister chromatid exchanges yielding microscopically visible neoplasms. 7 DMH is known to induce aberrant crypt foci (ACFs) formation which is a putative preneoplastic focal lesion and was characterized by having thicker lining of epithelial cells than normal crypts. 8 It is picked out as one of the executable tools, a sensitive, reliable, and rapidly appearing biomarker, additionally evidenced by the presence of histopathologic intraepithelial neoplasia. 9 On the other hand, DMH induction showed higher concentration of cholesterol and decreased levels of phospholipids. 10 Similar report was revealed that DMH administration to rodents showed increased cholesterol and cholesterol/phospholipid ratio and decreased the phospholipid metabolism in rats. 11 Hence it was clear that DMH interferes in the fat metabolism in rodents.
In fact, chronic inflammations of the colon have been depicted to enhance the risk of CRC 12 attaining inflammatory signaling pathways, a target for cancer prevention and treatment. Inhibitory protein inhibitor of κB-α (IκB-α) holds nuclear factor kappa light chain enhancer of activated B cells (NF-κB) in an inactive dormant form; predominantly, NF-κB activation involves phosphorylation, ubiquitination, and degradation of IκB granting NF-κB dimers translocate into the nucleus where it binds to the promoter region of several genes, which are pro-inflammatory mediators such as inducible nitric oxide synthase (iNOS), cyclooxygenase 2 (COX-2), tumor necrosis factor α (TNF-α), and interleukin 1β (IL-1β). A prominent body of evidence reported that both COX-2 and iNOS play crucial roles in cancer development by assigning angiogenesis in humans and animal models. 13 COX-2 is an inducible isoform, a rate-limiting enzyme for the production of prostaglandins PGE2 and is associated with inflammation and tumorigenesis. 14 Upregulated iNOS exhibits excess nitric oxide (NO) and results in the production of peroxynitrite by the reaction between NO and superoxide. 15 The inflammatory cytokines IL-1β and TNF-α largely produced by macrophages have all been playing a critical role in the initiation and perpetuation of CRC. 16,17
7-Hydroxycoumarin, also known as umbelliferone (UMB), a major biotransformed product of coumarin (1,2-benzopyrone), is widely distributed in natural products. 18 Our previous study revealed that UMB possesses a promising protective effect against DMH-induced oxidative stress in rat colon carcinogenesis. 19,20 It also reported that UMB has antioxidant, 21 –23 antihyperlipidemic, antidiabetic, and anti-hyperglycemic properties. 24 In addition, UMB is known for its anticoagulant, anti-inflammatory, and analgesic properties. 25 Although it was reported to have many health benefits, the effect of UMB on colon cancer was limited. Hence, in the present study, we intend to analyze the anti-inflammatory and proapoptotic roles of UMB in DMH-induced rat colon carcinogenesis.
Materials and methods
Chemicals
UMB, DMH, dimethyl sulfoxide (DMSO), the polymerase chain reaction (PCR) primers for TNF-α, COX-2, iNOS, IL-1β and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) were purchased from Genetix Biotech Asia Pvt. Ltd. (New Delhi, India). Trizol reagent, random hexamer primers, deoxy nucleotide triphosphate (dNTP), reverse transcriptase (RT) enzyme, and PCR ready master mix were purchased from Bangalore Genei (Bengaluru, Karnataka, India). The antibodies for cytochrome c, Bax, Bcl2, caspase-9, caspase-3, and β-actin were purchased from Santa Cruz Biotechnology (Santa Cruz, California, USA) and Cell Signaling Technology (Danvers, Massachusetts, USA). All other chemicals and solvents used were use of analytical grade.
Animals
Male Wistar rats aged 8–10 weeks, weighing approximately 100–120 g were obtained from Vinayaka Mission College of Pharmacy, Salem, Tamil Nadu, India. The animals were cared for in compliance with the rules and guidelines of the Indian Animal Ethical Committee of Periyar University, Tamil Nadu, India, based on the Indian National Law on Animal Care and Use (reg. no: PU-IAEC/JULY2011/03). The animals were housed in polypropylene cages, with six rats per cage under controlled conditions of 12-h dark/12-h light cycle, 50 ± 10% relative humidity, and 24 ± 2°C room temperature. The rats were held in quarantine for 1 week and had access to tap water ad libitum. The diet composition given to the rats is detailed in Table 1.
Composition of the diet.

Tumor induction
DMH was dissolved in 1 mM ethylenediaminetetraacetic acid (EDTA) prior to use and the pH was adjusted to 6.5 with 1 mM sodium hydroxide to ensure the stability of carcinogen. The rats were given subcutaneous injections of DMH for 15 consecutive weeks at a dose of 20 mg/kg body weight. 26
Preparation of UMB
UMB was freshly prepared daily and dissolved in 10% DMSO at a daily dose of 30 mg/kg body weight (based on the effective dosage fixation studies). 27
Experimental design
Rats were randomly distributed into four groups with six rats in each group. Rats in group 1 received a high fat diet and served as control, group 2 rats received a high fat diet with DMH (20 mg/kg body weight) once a week subcutaneously for the first 15 weeks, which represent the colon cancer-bearing rats. Group 3 rats received UMB via intragastric injection at a daily dose of 30 mg/kg body weight for 30 weeks every day. Group 4 rats received a high fat diet with DMH (20 mg/kg body weight) as in group 2 and received UMB throughout the experimental period. The experimental protocol is shown in Figure. 1.

Schematic representation of experiment design.
At the end of the experimental period of 30 weeks, the rats were anesthetized by ketamine hydrochloride (30 mg/kg body weight, intramuscular), and all the rats were killed by cervical dislocation after an overnight fast. The colons were removed and flushed with phosphate-buffered saline (PBS) and used for further analysis.
Determination of ACF
The fixation, staining, and counting of ACF was adopted by the method described by Ochiai et al. 28 Briefly, the colons were flushed with PBS (0.1 M, pH 7.2). Colons were split open longitudinally and placed on strips of filter paper with their luminal surface open and exposed. Another strip of filter paper was placed on top of the luminal surface. The colon was fixed, set up in a tray-containing 10% buffered formalin overnight. Each segment was placed in a petri dish and stained with 0.2% methylene blue solution for 2 min. The sections were then transferred to another petri dish-containing wash buffer to remove excess stains. The segments were examined using a light microscope at low magnification to score the total number of ACF as well as the number of crypts per focus.
Histopathological changes
The colons from the experimental animals were microscopically examined for the presence of tumors or other pathological lesions. Tissues with abnormal morphology were fixed at 10% buffered formalin and embedded in paraffin blocks. Histological sections stained with hematoxylin and eosin were used to confirm the presence and type of ACF and tumors by histopathological examination, which was performed by a pathologist unaware of the experimental codes.
AgNOR staining
The AgNOR staining was performed using the one-step staining technique described by Trerè. 29 In brief, hydrated sections were incubated in a freshly prepared solution of one part 2 g/dL solution of gelatin in 1 g/dL formic acid to two parts 50 g/dL aqueous silver nitrate for 40 min at room temperature and counterstained with hematoxylin for 30 s. The AgNORs in 200 nuclei of the basal and lower spinous cells were counted, and the average number of AgNOR per nucleus was estimated for each.
Mast cell staining
Histochemical analysis of mast cells was carried out by the method as described previously. 30 Briefly, the sections of colon were dewaxed in xylene and rehydrated through decreasing concentrations of ethanol. Further the slides were stained with toluidine blue to locate the immature mast cells and counterstain by safranin for mature mast cells. Then they were dehydrated in increasing concentrations through alcohol series and mounted using DPX, and high-power objective field (40×) was chosen for counting the total number of mast cells in 25 different fields/slides.
Reverse transcription PCR
Total cellular RNA was isolated from the snap-frozen colon tumor tissue using guanidinium thiocyanate–phenol–chloroform procedure. 31 Isolated RNA was reverse transcribed into complementary DNA (cDNA) using a reverse transcription system kit (Promega, Madison, Wisconsin, USA) according to the manufacturer’s manual. The cDNA pools from the reverse transcription reaction were subjected to PCR using PCR core system 1 (Promega) and primers specific for rat iNOS, Gene bank accession number NM012611 (sense: 5′-CGA GGA GGC TGC CCT GCA GAC TGG-3′; antisense: 3′-CTG GGA GGA GCT GAT GGA GTA GTA-5′), resulting in an amplified product of 1383 base pair (bp); COX-2, NM011198 (sense: 5′-AAA GCC TCG TCC AGA TGC TAA-3′; antisense: 3′-ATG GTG GCT GTC TTG GTA GG-5′), resulting in an amplified product of 479 bp; IL-1β, NM031512 (sense: 5′-CCA GGA TGA GGA CCC AAG CA-3′; antisense: 3′-TCC CGA CCA TTG CTG TTT CC-5′) resulting in an amplified product of 519 bpbp; TNF-α, NM012675 (sense: 5′-AGA TGT GGA ACT GGC AGA GG-3′; antisense: 3′-CCC ATT TGG GAA CTT CTC CT-5′) resulting in an amplified product of 468 bpbp; and GAPDH, NM017008 (sense: 5′-CCC ATT TGG GAA CTT CTC CT-3′; antisense: 3′-AGG GGC CAT CCA CAG TCT TC-5′) resulting in an amplified product of 595 bp. PCR products were separated on a 1% agarose gel in 1× Tris-borate EDTA and 0.5 µg/mL ethidium bromide in Tris-borate EDTA buffer. Densitometric analysis of band intensity was performed with the gel–doc documentation system and Quantity One software (Bio-Rad, Hercules, California, USA). The results are expressed as a ratio of densitometric score of each gene to GAPDH. Mean ratio and standard error were calculated from the signal obtained from 10 different samples in each group.
Western blotting
Western blots were carried out by the method of Towbin et al. 32 Cells were treated, trypsinized, and the homogenate was estimated for protein by the Lowry method. 33 Colon tissues were washed with cold PBS and homogenized. All samples were loaded on an equal protein basis and analyzed by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis gel. The separated proteins were transferred onto a nitrocellulose membrane and probed with primary antibodies such as caspase 3 (1:5000, v/v), Bax (1:1000, v/v), Bcl-2 (1:1000, v/v), cytochrome c (1:1000, v/v), caspase 9 (1:200), and β-actin. Then the respective secondary antibodies conjugated with horseradish peroxidase were used. Then the bands were developed using a diaminobenzidine (DAB)/hydrogen peroxide H2O2 color development system. Densitometric analysis was carried out using UN-SCAN-IT image digitalizing software.
Immunohistochemical analysis
Paraffin-embedded tissue sections were rehydrated first in xylene and then in graded ethanol solutions. The slides were then blocked with 5% bovine serum albumin (BSA) in Tris-buffered saline (TBS) for 2 h. The sections were then immunostained with primary antibodies such as Bcl-2, Bax, caspase 3, caspase 9 (Santa Cruz Biotech) were diluted in 1:300 to 5% BSA in TBS and incubated overnight at 4°C. After washing the slides thrice with TBS, the sections were then incubated with respective secondary antibodies and incubated for 2 h at room temperature. Sections were then washed with TBS and incubated for 5–10 min in a solution of 0.02% DAB-containing 0.01% H2O2. Counterstaining was performed using hematoxylin, and the slides were visualized under a light microscope.
DNA fragmentation assay
The pattern of DNA cleavage was analyzed by agarose gel electrophoresis as described previously. Briefly, different groups of animal tissue samples were lysed with 0.5 mL lysis buffer (5 mM Tris-hydrochloric acid (pH 8.0), 0.25% Nonidet P40, and 1 mM EDTA), followed by the addition of RNase A (Sigma, St Louis, Missouri, USA) at a final concentration of 200 μg/mL. After incubation for 1 h at 37°C, cells were treated with 300 μg/mL proteinase K for an additional hour at 37°C. A volume of 20 μL samples in each lane was subjected to electrophoresis on a 1.5% agarose at 50 V for 3 h. DNA was stained with ethidium bromide.
Transmission electron microscopy
The colon tissue samples were fixed in Karnovsky’s fixative immediately after euthanization of rats for 6–8 h at 4°C. These were postfixed in 1% osmium tetroxide in 0.1 M phosphate buffer for 2 h at 4°C, dehydrated in ascending grades of acetone, infiltrated and embedded in Araldite CY212, and polymerized at 60°C for 72 h. Thin (60–70 nm) sections were cut with an ultramicrotome. The sections were mounted on copper grids and stained with uranyl acetate and lead citrate and observed under a Philips CM 10 transmission electron microscope (Philips Scientifics, Eindhoven, the Netherlands).
Statistical analysis
Values are given as mean ± SD. Data were analyzed by one-way analysis of variance and any significant difference among treatment groups was evaluated by Duncan’s multiple range test. The results were considered statistically significant at p < 0.05. All statistical analysis was created using SPSS 16.0 software package (SPSS, IBM product, Chicago, Illinois, USA).
Results
UMB suppress cell proliferation
The regional distribution of AgNORs, mast cell counts, histology ACF, and ACF counts of control and experimental rats are shown in Figure 2. Silver-stained AgNORs of control and positive control showed normal crypts. Table 2 shows the mean number of AgNORs/nucleus in group 2 was the heaviest when compared to UMB-treated DMH-induced rats (group 4). The effect of UMB greatly reduces cell proliferation and was more pronounced and evident in group 4.
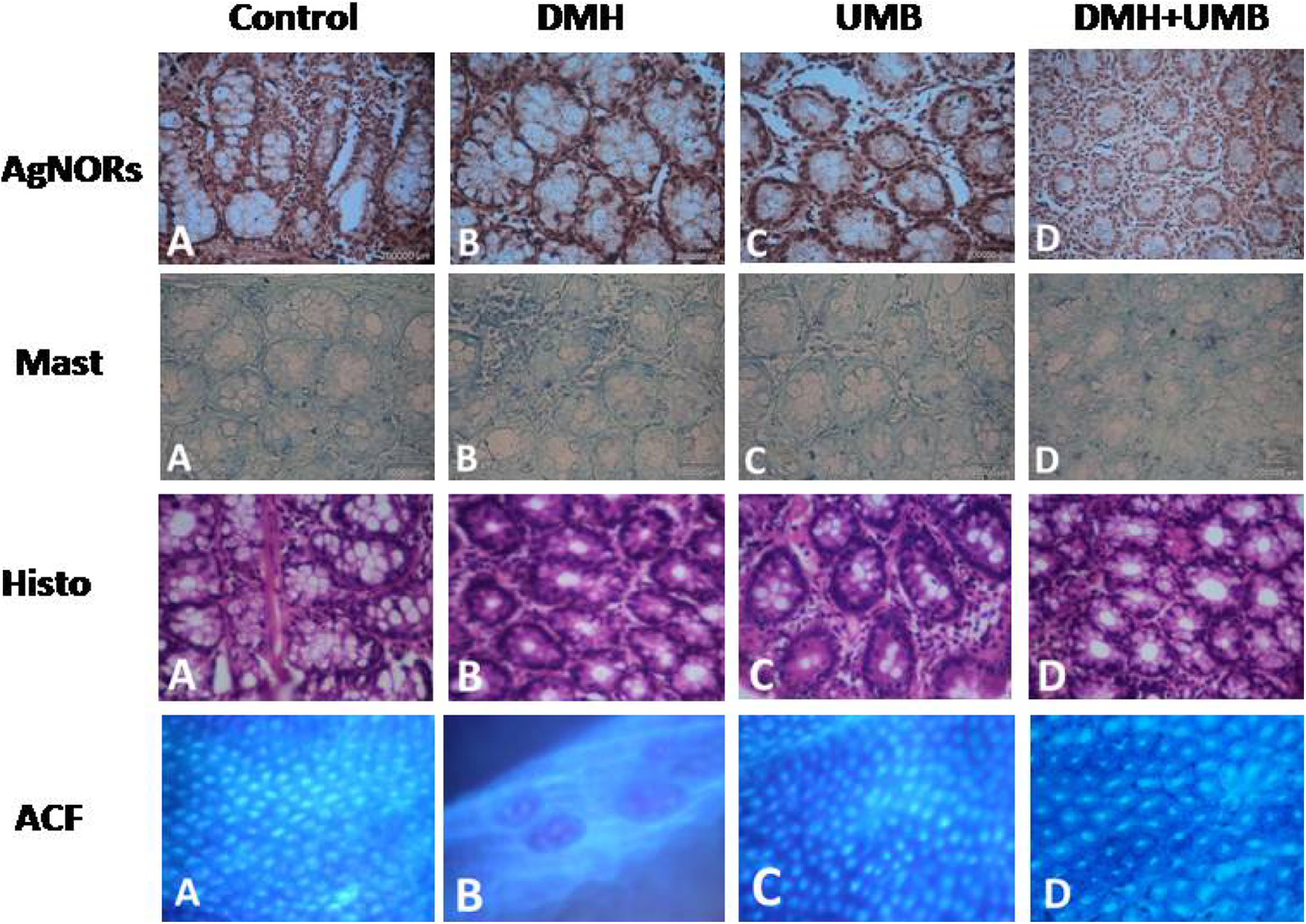
Silver staining of AgNORs (×40). (a and c) Normal colonocytes lack silver positive black dots. (b) Adenocarcinoma of the colon with irregular glands exhibiting cluster of intense silver positive black dots. (d) Silver staining was decreased significantly upon UMB supplementation. Toluidine staining of mast cells counts. (a and c) Colonic crypts are looking normal lacking more mast cells. (b) Adenocarcinoma sequence of colonic crypts exhibit significantly increased concentration of mast cells. (d) Colonic crypts showing significantly decreased concentration of mast cells. HE staining of ACF. (a) Control rats showing normal crypt appearnce, (b) DMH-induced colon rats showing more than 2 aberrant crypt foci having nearly 10 crypts per foci, (c) UMB-treated rats showing one or two crypts looking like control, and (d) UMB-treated DMH-induced rats showing normal crypts like that of control. Methylene blue staining of colonic tissue. (a) Control shows normal crypt, (b) DMH-induced rat showed two aberrant crypt foci having more than seven crypts, (c) positive control showing normal crypts, and (d) UMB-treated DMH-induced rats showing no aberrant crypt and looks like normal. AgNOR: agyrophylic nucleolar organizer region; UMB: umbelliferone; HE: hematoxylin and eosin; DMH: 1,2-dimethylhydrazine; ACF: aberrant crypt foci.
Effect of DMH and UMB on the number of AgNORs in crypt cell nuclei.a

UMB: umbelliferone; DMH: 1,2-dimethylhydrazine; AgNOR: agyrophylic nucleolar organizer region; ANOVA: analysis of variance; DMRT: Duncan’s multiple range test.
aValues are expressed as mean ± SD. Values not sharing common superscript letters (b to e) are significant with each other at p < 0.05 (ANOVA followed by DMRT).
UMB reduces mast cell density
Mature mast cell density and distribution is normal in control and positive control rats. DMH-induced (group 2) rats revealed a significant increase in the count of mast cells in the colonic tissue. UMB-supplemented colon cancer-bearing rats (group 4) showed significant reduction in mast cell count.
UMB improves histological alterations
Histological view of ACF in control and positive control were normal. DMH-induced colon rats (group 2) showed more than 2 aberrant crypt foci having nearly 10 crypts per foci. The frequency of ACF/colon in the UMB-supplemented colon cancer rats (group 4) were significantly reduced than that of DMH alone-treated rats (group 2). Pathological studies showed a great degree of variation in the different groups.
UMB suppresses ACF formation
Methlyene blue-stained colonic tissue of control and positive control rats showed normal crypts. DMH-induced colonic crypts were stereoscopically distinct from normal crypts as evidenced by their darker staining, larger size, ovate shape, thicker epithelial lining, and larger pericryptal zone (Figure 2). DMH-induced rats (group 2) had a significantly higher number of total aberrant crypts compared with the control rats. Administration of UMB to colon cancer-bearing rats (group 4) significantly reduced the total number of aberrant crypts. There was a total reduction of 75% in ACF incidence in rats administrated with UMB (group 4), while there was no ACF incidence in control and positive control rats as clearly presented in Table 3.
Incidence of ACF at the end of 30-week study.
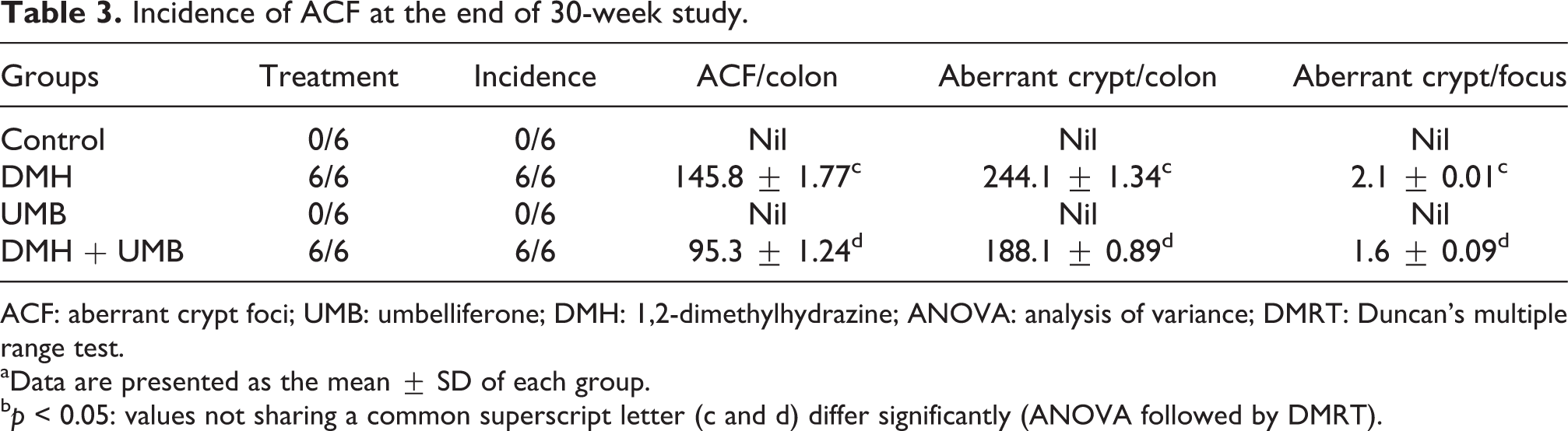
ACF: aberrant crypt foci; UMB: umbelliferone; DMH: 1,2-dimethylhydrazine; ANOVA: analysis of variance; DMRT: Duncan’s multiple range test.
aData are presented as the mean ± SD of each group.
b p < 0.05: values not sharing a common superscript letter (c and d) differ significantly (ANOVA followed by DMRT).
UMB reduces the expressions of inflammatory mediators
An inflammatory response is characterized by the expression of a number of pro-inflammatory mediator genes. Therefore, anti-inflammatory activity of UMB was monitored by evaluating the gene levels of inflammatory-related enzymes (iNOS and COX-2) and some pro-inflammatory cytokines (TNF-α and IL-1β), using RT-PCR (Figure 3). As shown in our results, DMH induction led to markedly elevated levels of these pro-inflammatory mediator genes in the inflammation-induced colon cancer-bearing rats. Remarkably, treatment with UMB (30 mg/kg body weight) suppressed the gene expression of the above pro-inflammatory genes and markers significantly (p < 0.05) in experimental rats.

Dynamic expression of the COX-2, iNOS, TNF-α, and IL-1β mRNA of control and experimental rats. Data are presented as the mean ± SD of each group. p < 0.05: the values not sharing a common superscript letter (a, b, c, d, and e) are significantly different from the DMH-treated groups (analysis of variance followed by DMRT). COX-2: cyclooxygenase 2; iNOS: inducible nitric oxide synthase; TNF-α: tumor necrosis factor α; IL-1β: interleukin 1β; mRNA: messenger RNA; DMH: 1,2-dimethylhydrazine; DMRT: Duncan’s multiple range test.
UMB induces apoptosis
Immunohistochemical analysis of Bax, Bcl-2, caspase 3, and caspase 9 in the control and experimental groups of animals are represented in Figure 4. Induction with DMH (group 2) decreased the expressions of Bax, caspase 3, and caspase 9 and increased the expression of Bcl-2 was observed. Administration of UMB extensively increased the expressions of Bax, caspase 3, and caspase 9 and decreased the expression of Bcl-2 which is observed as dense brown invasion throughout the cells. Control and UMB-treated rats, however, exhibited few positive expressions. The quantification of Bax, Bcl-2, caspase 3, and caspase 9 is represented in Figure 4.

Effect of UMB on Bax, Bcl2, caspase 9, and caspase 3 in control and experimental rats. Normal expression of Bax, Bcl2, caspase 9, and caspase 3 were observed in controls (a) and positive controls (c). DMH-induced rats (b) showed very high expression of Bcl2 and with complete loss of Bax, caspase 9, and caspase 3 were observed. UMB-treated DMH-induced rats restore the normal levels of Bcl2 as well as increased the concentrations of Bax, caspase 9, and caspase 3 (magnification, ×40). UMB: umbelliferone; DMH: 1,2-dimethylhydrazine.
Figure 5 illustrates the immunoblot analysis of p65-NF-κB, Bax, Bcl-2, caspase 9, caspase 3, and actin in the control and experimental rats. We observed an increase in the expressions of p65-NF-κB, Bcl-2 and decreased expressions of Bax, caspase 9, and caspase 3 were observed in the DMH-induced rats. Conversely, administration of UMB to treat DMH-induced rats showed a notable difference in the expression pattern of the above proteins (group 4).

Immunoblot analysis of NF-κB, Bax, Bcl-2, caspase 9, and caspase 3 in the colon tissue of control and experimental rats. Densitometric analysis of NF-κB, Bax, Bcl-2, caspase 9, and caspase 3 normalized with GAPDH and are expressed in fold changes compared with control group. Lane 1: control, lane 2: DMH; lane 3: UMB; lane 4: DMH + UMB. NF-κB: nuclear factor κB; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; UMB: umbelliferone; DMH: 1,2-dimethylhydrazine.
DNA fragmentation ladder is not present in control and DMH-induced rats (Figure 6, lanes 1, 2, 3). Interestingly, we observed ladder of DNA fragmentation in UMB-treated DMH-induced CRC rats. (Figure 6, lane 4). Hence, we confirmed that UMB can induce apoptosis since DNA fragmentation is considered as a hallmark of apoptosis.

Electrophoretic pattern of DNA isolated from the colon tissue of control and experimental rats determined by agarose gel electrophoresis. M: marker; lane 1: control; lane 2: DMH; lane 3: UMB; lane 4: UMB + DMH. UMB: umbelliferone; DMH: 1,2-dimethylhydrazine.
UMB modulates the ultrastructural changes in colon
Figure 7 shows ultrastructural changes in the colonic tissues of control and experimental groups of rats. Control (group 1) and UMB control (group 3) showed a normal array of epithelial cells without any distribution. DMH-induced rats (group 2) showed cells with multiple irregular-shaped nuclei close to each other with irregular cytoplasm and dislodged nuclear membrane. This indicates the cell division that supports the tumorigenic events. Morphological changes of apoptosis were seen in the colonic cells of DMH + UMB pretreated rats (group 4). Colonic cells with shrunken nucleus and condensed chromatin undergoing apoptosis with collapsed nuclear membrane were seen in antiapoptotic effect of UMB (group 4).

Ultrastructural examination of colonic tissues of control and experimental rats were determined by TEM analysis. (a) Control rats showing normal nucleus and cytoplasm (×6000). (b) DMH-induced rats showing multiple irregular-shaped nuclei close to each other with irregular cytoplasm (×6000). (c) UMB control rats showing normal nucleus with intact endoplasmic reticulum (×6000). (d) Morphological changes of apoptosis were seen in DMH-induced UMB treatment in an experimental model (×8200). TEM: transmission electron microscopy; UMB: umbelliferone; DMH: 1,2-dimethylhydrazine.
Discussion
In recent times, CRC is the malignant disease with high mortality rate. Treating CRC using potential natural compounds is one of the crucial ways to eliminate CRC. The recent studies reported that a bioflavonoid has the capacity to inhibit the pathogenesis of CRC via modulating multiple signaling pathways. 34,35 UMB is a crucial natural product that is slowly evolving as an anticancer agent. Importantly, when it was administered to rats, it was excreted in the urine partly unchanged but mainly as a glucoronide and/or sulfate conjugate. It was also metabolized to 2,4-dihydroxyphenylpropionic acid and, following intraperitoneal dosage, to a very small amount of 3,7-dihydroxycoumarin. 36 So, we intend to use UMB to treat DMH-induced CRC in rats.
The present study furnishes evidence that UMB could act as a stiff, strong inhibitory compound against DMH-induced CRC in rats. DMH is believed to induce enormous production of reactive oxygen species and nitrogen species that results in gene mutation and thereby the formation of ACF and epithelial carcinomas in the colon. The present study has also been corelated with previous findings 37 and confirmed that DMH-treated rats showed an increased in the number and size of crypts. Interestingly, UMB supplementation significantly inhibited colonic ACF formation in rats treated with DMH and also brought down the number of ACF counts in the distal and middle colon. The histological analysis of ACF in control and experimental rats also corelates with the methylene blue staining of ACF.
It has been suggested that the number of AgNOR dots in a nucleus may reflect the status of cell activation and therefore is a useful index to assess cell proliferation in the cancer. 38 AgNOR quantification by image analysis is a useful tool for the evaluation of the rate of cell proliferation. 39,40 In our study, increased numbers of AgNORs dots were seen in DMH-induced rat colon carcinogenesis, while on treatment with UMB, the number of AgNORs dots were reduced. This clearly indicates that UMB inhibits the cell proliferation in DMH-induced CRC (Figure 2). UMB was shown to inhibit cell proliferation in DMH-induced rats.
An interaction between tumor cells and mast cells in their microenvironment is important for their growth and survival. The connection between these occurs in three forms as described: (i) its effect on tumor angiogenesis, (ii) for mediating the tissue remodeling, and (iii) mast cell-dependent immune regulation. 41 An increase in the number of infiltrating inflammatory mast cells in DMH-induced rats creates a tumor microenvironment that manifests a persistent inflammatory state, thereby encouraging tumor progression. Upon treatment with UMB (group 4), it was discovered that it significantly decreased the number of mast cell density when compared to DMH-induced rats. This might be due to the anti-inflammatory property of UMB.
Epidemiological indication brings together the CRC to chronic intestinal inflammation; 42 also perennial inflammatory reactions within the colon might create and progress the CRC pathogenesis. Concisely, the present study disclosed that anti-inflammatory effect of UMB greatly suppresses the inflammatory markers of DMH-induced animal models. Our determinations present that an elevation in the intestinal inflammatory markers (TNF-α, IL-1β, iNOS, and COX-2) through p65-NF-κB-mediating response occurs at continued DMH inductions in experimental animals (Figures 3 and 5).
Upon metabolism of DMH, the production of excess ROS ends in the activation of iNOS, COX-2, TNF-α, and IL-1β via p65-NF-κB pathway. Enhanced levels of these mediators could damage function and transcription of the affected proteins and progression of epithelial cell transformation to invasive cancer in colonic mucosa through initiating the inflammation and progression of cancer. 43,44 Interestingly, UMB reduced considerably the expression of both iNOS and COX-2. It is an important essential constituent of a malignant cell autonomous network of inflammatory cytokines that includes IL-1β. This network then acts on the microenvironment, particularly affecting the infiltration and angiogenic actions of TNF-α, at least in part, its ability to cause differentiation of epithelial cells in the tumor microenviroment. 45,46 Hence, our study clearly indicates that the inhibition of p65-NF-κB expression by UMB regulates the inflammatory process through prevention of inflammatory cytokines in DMH-induced rats, which supports a previous study. 25 The possible mechanisms and action of UMB on DMH-induced rats are shown in Figure 8.
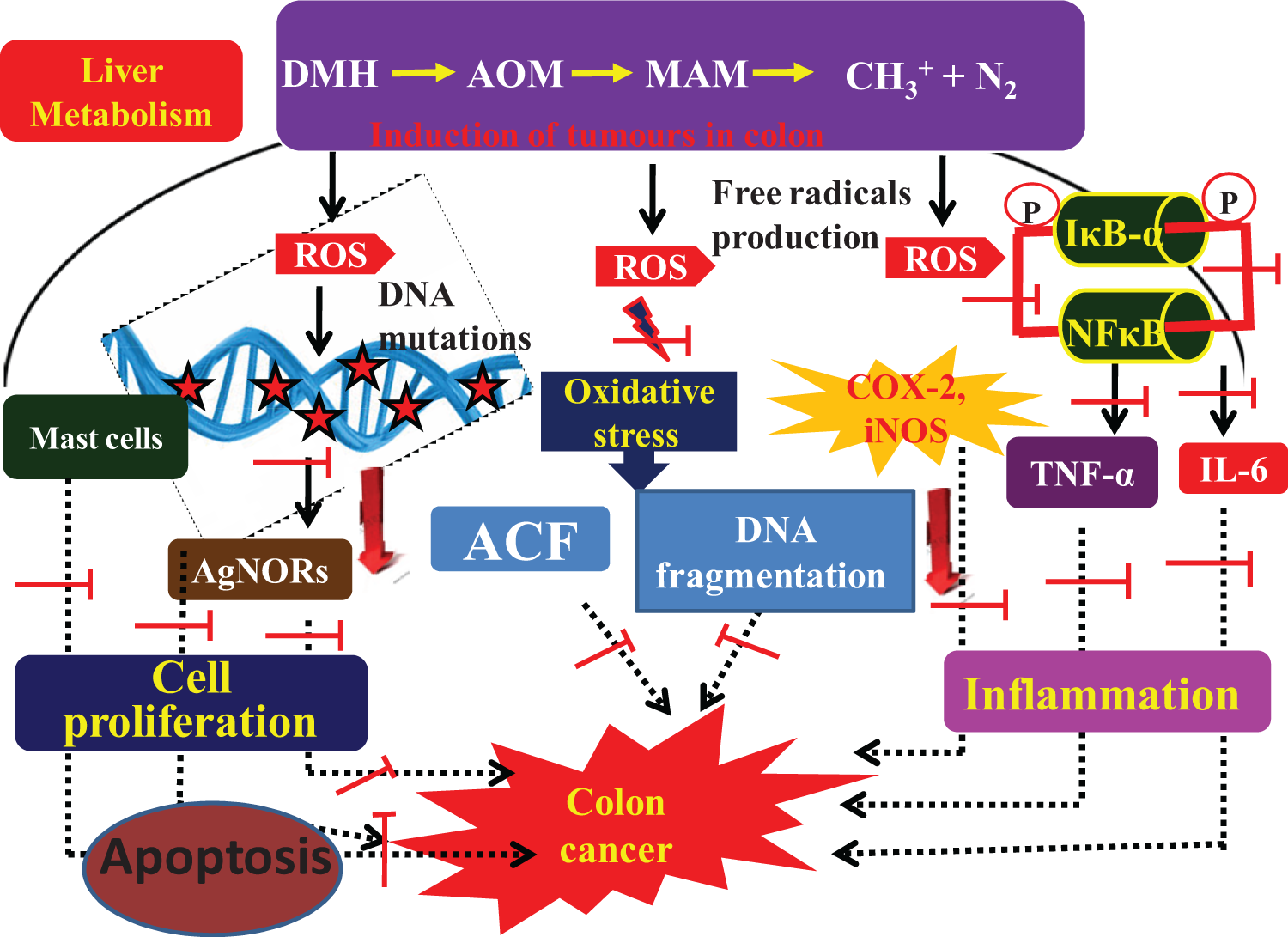
The mechanism of action of UMB in DMH-induced CRC. UMB: umbelliferone; DMH: 1,2-dimethylhydrazine; CRC: colorectal cancer.
To further confirm the apoptosis induced by UMB, the DNA fragmentation analysis was performed. We observed a DNA shear, characteristic of apoptotic cell death in DMH + UMB-treated rats. Also, in the cells undergoing apoptosis, DNA was degraded to fragments of low molecular weight and subsequently leaked out of the cells. These structural changes, such as plasma and nuclear membrane blebbings, chromatin condensation, protease activation, and DNA fragmentation, are considered to be hallmarks of the apoptotic process were also confirmed with TEM analysis and our observations are in concordance with previous reports. 47
Conclusion
Our results suggest that supplementation of UMB significantly inhibited the incidence of ACF, decreased the expression of inflammatory mediators such as p65-NF-κB, iNOS, COX-2, TNF-α and IL-1β in the DMH-induced CRC rats. Umbelliferone acts as an anti-inflammatory factor and reduced the mast cell recruitment and reduced the cell proliferation that was confirmed by AgNOR staining. On the other hand, umbelliferone can induce apoptosis by modulating the expressions of Bax, Bcl-2, caspase 3, and caspase 9. We also confirmed the apoptosis through DNA fragmentation and ultrastructural changes in the DMH-induced CRC rats. Further studies are in progress to assess the mechanism of action exhibited by umbelliferone, which might be developed as a successful chemotherapeutic agent to treat CRC.
Footnotes
Acknowledgement
The authors thank Dr Ashok K Pandurangan, Department of Pharmacology, University of Malaya, 50603 Kuala Lumpur, Malaysia, for his generous support and help in the restructuring of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
