Abstract
Chemoprevention opens new window in the prevention of all types of cancers including colon cancer. Aloin, an anthracycline in plant pigment, can be utilized as a protective agent in cancer induction. In the present study, we have evaluated the chemopreventive efficacy of aloin against 1,2-dimethylhydrazine (DMH)-induced preneoplastic lesions in the colon of Wistar rats. DMH-induced aberrant crypt foci (ACF) and mucin-depleted foci (MDF) have been used as biomarkers of colon cancer. Efficacy of aloin against the colon toxicity was evaluated in terms of biochemical estimation of antioxidant enzyme activities, lipid peroxidation, ACF, MDF, histopathological changes, and expression levels of molecular markers of inflammation and tumor promotion. Aloin pretreatment ameliorates the damaging effects induced by DMH through a protective mechanism that involved reduction in increased oxidative stress enzymes (p < 0.001), ACF, MDF, cyclooxygenase-2, inducible nitric oxide synthase, interleukin-6, proliferating cell nuclear antigen protein expression, and tumor necrosis factor-α (p < 0.001) release. From the results, it could be concluded that aloin clearly protects against chemically induced colon toxicity and acts reasonably by inducing antioxidant level, anti-inflammatory and antiproliferative markers.
Introduction
Colon cancer results from both genetic and environmental factors and their interaction. Genetic disposition is the most important risk factor for some individuals; conversely, environmental factors and dietary habits such as smoking and obesity are the confounding factors in the induction of colon cancer. 1
The world burden of cancer continues to increase largely because of growth of an increasing adoption of cancer-causing behaviors worldwide, particularly smoking. Based on the data, about 12.7 million cancer cases and 7.6 million cancer deaths are estimated to have occurred in 2008; Out of those, 56% of the cases and 64% of the deaths occurred in the economically developing countries. 2 A total of 1,596,670 new cancer cases and 571,950 deaths from cancer are projected to be occurred in the United States in 2011. Colorectal cancer is the third most common cancer in men (663,000 cases, 10.0% of the total cancers) and the second in women (570,000 cases, 9.4% of the total cases) worldwide. 3 The age of the people between 55 and 64 years are at high risk of colon cancer incidence and mortality, accounting for over 600,000 deaths in 2008. 2,4
1,2-Dimethylhydrazine (DMH) is an effective colon carcinogen, inducing colorectal tumors in experimental animals 5,6 and is an extensively used model of chemically induced colon carcinogenesis to explore the signaling cascade involved in colon carcinogenesis. For the first time, Fiala reported that active metabolite of DMH is excreted through bile and is responsible for its carcinogenic effect on colon while passing through the digestive tract, consequential in the production of electrophilic diazonium ions, which are known to exert oxidative stress to induce DNA damage and cancer. 7 For over two decades, while its first discovery, azoxymethane (AOM)-induced rodent aberrant crypt foci (ACF) have served as substitute biomarkers in the screening of various anticarcinogens and carcinogens.
Several natural compounds like tannic acid and farnesol have been shown to possess chemopreventive properties. 8–10 Aloin is an anthraquinone glycoside, that is, anthraquinone skeleton has been modified by the addition of a sugar molecule. Anthraquinones are a common family of naturally occurring yellow, orange, and red pigments of which many have cathartic properties, attributes shared by aloin and have antitumor properties. 11,12 In the previously published reports, aloin has several biological effects, namely, inhibition of cell proliferation, adhesion, and invasion abilities on melanoma cells under noncytotoxic concentrations. 12,13 In pharmaceutical industries, aloin is used as a drastic purgative in veterinary medicine. 14
In a recent study, aloin has been shown to act as a growth inhibitor of gastric cancer cell. 15 Recent reports pointed out a potential cytotoxic effect of aloin through the induction of S-phase cell cycle arrest and apoptosis in human epithelial type breast and ovarian tumor cell lines. 16
Development of DMH-induced colon cancer in rodents is a multistep process involving a series of pathological changes, ranging from discrete microscopic mucosal lesions as ACF, mucin-depleted foci (MDF), and malignant tumors. ACF, which are morphologically irregular crypts in the colonic mucosa, are reliable intermediate biomarkers and early preneoplastic events of colorectal cancer. 17–19 ACF are putative preneoplastic lesions that are induced in colon of carcinogen-treated rodents and present in humans with increased risk of colon cancer. Femia et al. first described crypt foci with missing mucous production MDF in colon tissues induced by AOM in rat model. 20 MDF are simple to quantify in the complete unsectioned colon and show clear characteristics of dysplasia in histological section. These MDF have been suspected to recognize ACF with an increased probability of progressing to cancer. 18,21
Inflammatory cytokines tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) play an important role in the initiation and perpetuation of colon cancer. 22–24 TNF-α and IL-6 are the proinflammatory cytokines and several evidences have shown that the expression of TNF-α and IL-6 are dependent on the activation of nuclear factor-κB (NF-κB), which contributes to inflammation. 25 Therefore, targeting TNF-α and IL-6 is an effective approach for the treatment of colon cancer. 26 The release of an enormous amount of nitric oxide (NO) and prostaglandins is also associated with inflammatory conditions through inducible NO synthase (iNOS) and cyclooxygenase-2 (COX-2) as their overexpression is associated with colon cancer. 22
The present study was designed to examine the effects of aloin against DMH-induced ACF and MDF in the colon of Wistar rats. Furthermore, we explored the effects of aloin against DMH-induced oxidative stress and expression of cell proliferation and inflammatory biomarkers such as proliferating cell nuclear antigen (PCNA), COX-2, iNOS, IL-6, and TNF-α in colonic mucosa.
Materials and methods
Chemicals
Reduced glutathione (GSH), nicotinamide adenine dinucleotide phosphate reduced (NADPH), ethylenediaminetetraacetic acid (EDTA), nicotinamide adenine dinucleotide reduced, thiobarbituric acid (TBA), trichloroacetic acid, bovine serum albumin (BSA), 1,2-dithio-bis-nitrobenzoic acid (DTNB), aloin, 1-chloro-2,4-dinitrobenzene (CDNB), DMH, and GSH reductase were obtained from Sigma Chemical Co. (St Louis, Missouri, USA). Ferric nitrate, ammonium thiocyanate, hydrogen peroxide (H2O2), magnesium chloride, disodium hydrogen phosphate, sodium dihydrogen phosphate, and sodium hydroxide (NaOH) were purchased from E Merck Limited (Mumbai, India). Primary antibodies were rabbit anti-COX-2 (dilution 1:200, Matrix Technology, Santa Cruz, California, USA), rabbit anti-iNOS (1:500, Jackson ImmunoResearch Laboratories, Inc., West Baltimore Pike, PA, USA), IL-6 (dilution 1:300, Matrix Technology, Santa Cruz, California, USA), TNF-α Kit (eBioscience, Inc., San Diego, California, USA), and PCNA (dilution 1:300, Matrix Technology, Santa Cruz, USA).
Animals and treatment schedule
The experiments were carried out using male albino Wistar rats, weighing 120–150 g, obtained from the Central Animal House of Hamdard University, New Delhi, India. A total of 40 animals were randomly divided into 5 groups with 8 animals in each group. The rats were kept in polypropylene cages and were kept in a room maintained at 25 ± 2°C with a 12-h light/dark cycle. They were given free access to diet and water. Figure 1 shows the treatment protocol followed; the rats of group I (control group) received distal water orally. Group II received DMH subcutaneously at a dose of 20 mg kg−1 body weight (b.w.) once a week for five consecutive weeks from second week till sixth week. Group III received aloin orally at a dose of 50 mg kg−1 b.w. on alternate days consecutively for 14 weeks. Groups IV and V (dose 2 of aloin) received aloin at a dose of 100 mg kg−1 b.w. on alternate days consecutively for 14 weeks from the first day till the end of the study. The rats of groups II, III, and IV were given DMH injection five times (dissolved in 1 mM EDTA solution with pH adjusted to 6.5 with 1 mM NaOH) at a dose of 20 mg kg−1 b.w. subcutaneously in the groin starting after one week of aloin treatment. The rats of groups I and V also received subcutaneous injection of 1 mM EDTA solution (pH adjusted to 6.5 with 1 mM NaOH), which was used as the vehicle for DMH. All the rats were anesthetized with mild anesthesia and killed by cervical dislocation after 14 weeks. The doses of aloin were selected on the basis of available preliminary studies performed by Zhou et al. 27 Our study was approved by the Institutional Animal Ethics Committee (IAEC) of the university with registration no. 599.

Treatment protocol for different groups.
PMS preparation and estimation of different parameters
Colons were removed quickly, cleaned free of irrelevant material, and immediately perfused with ice-cold saline (0.85% sodium chloride (NaCl)). The colons (10% w/v) were homogenized in chilled phosphate buffer (0.1 M, pH 7.4) using a Potter–Elvehjem homogenizer (Olympus, Tokyo, Japan). The homogenate was filtered through muslin cloth and were centrifuged at 3000 r min−1 for 10 min at 4°C by a refrigerated centrifuge (model RC 4100 D; Eltek Group, Goregaon(E), Mumbai, India) to separate the nuclear debris. The aliquot so obtained was centrifuged at 12,000 r min−1 for 20 min at 4°C to obtain postmitochondrial supernatant (PMS), which was used as a source for various enzymes. 28,29
Measurement of reduced GSH level
The GSH content in colon was determined by the method of Jollow et al. 30 in which 1.0 ml of PMS fraction (10%) was mixed with 1.0 ml of sulfosalicylic acid (4%). The samples were incubated at 4°C for at least 1 h and then subjected to centrifugation at 1200g for 15 min at 4°C. The assay mixture contained 0.4 ml filtered aliquot, 2.2 ml phosphate buffer (0.1 M, pH 7.4), and 0.4 ml DTNB (10 mM) in a total volume of 3.0 ml. The yellow color developed was read immediately at 412 nm on a spectrophotometer (Milton Roy Model-21 D). The GSH content was calculated as nanomoles of DTNB conjugate formed per gram of tissue using molar extinction coefficient of 13.6 × 103 M−1 cm−1.
Measurement of MDA
Lipid peroxidation as evidenced by the formation of TBA reactive substances was estimated by the method described by Ohkawa et al. 31 To a 0.2 ml of tissue homogenate, 0.2 ml of 8.1% sodium dodecylsulfate, 1.5 ml of 20% acetic acid, and 1.5 ml of 0.8% TBA were added. The mixture was made up to 4.0 ml with distilled water and then heated in a boiling water bath at 95°C for 60 min. After cooling, 1.0 ml of water and 5.0 ml of n-butanol/pyridine mixture were added and shaken vigorously. After centrifugation at 600g for 10 min, the pink-colored chromogen formed by the reaction of 2-TBA with the breakdown products of lipid peroxidation was read at 535 nm. The results were expressed as the nanomoles of malondialdehyde (MDA) formed per minute per gram of tissue using a molar extinction coefficient of 1.56 × 105 M−1 cm−1.
Measurement of GPx activity
The glutathione peroxidase (GPx) activity was calculated by the method of Mohandas et al. 32 A total of 2 ml volume consisted of 0.1 ml EDTA (1 mM), 0.1 ml sodium azide (1 mM), 1.44 ml phosphate buffer (0.1 M, pH 7.4), 0.05 ml glutathione reductase (GR; 1 IU ml−1), 0.05 ml reduced GSH (1 mM), 0.1 ml NADPH (0.2 mM), 0.01 ml H2O2 (0.25 mM), and 0.1 ml 10% PMS. The depletion of NADPH at 340 nm was recorded at 25°C. The enzyme activity was calculated as nanomoles of NADPH oxidized per minute per milligram protein with the molar extinction coefficient of 6.22 × 103 M−1 cm−1.
Measurement of GR activity
The GR activity was measured by the method of Carlberg and Mannervik. 33 The assay system consisted of 1.65 ml phosphate buffer (0.1 M, pH 7.6), 0.1 ml EDTA (0.5 mM), 0.05 ml oxidized GSH (1.0 mM), 0.1 ml NADPH (0.1 mM), and 0.1 ml of 10% PMS in a total volume of 2.0. The enzyme activity was assessed at 25°C by measuring the disappearance of NADPH at 340 nm and was calculated as nanomoles of NADPH oxidized per minute per milligram of protein using the molar extinction coefficient of 6.22 × 103 M−1 cm−1.
Measurement of GST activity
The glutathione-S-transferase (GST) activity was measured by the method of Habig et al. 34 The reaction mixture consisted of 2.4 ml phosphate buffer (0.1 M, pH 6.5), 0.2 ml reduced GSH (1.0 mM), 0.2 ml CDNB (1.0 mM), and 0.2 ml of cytosolic fraction in a total volume of 3.0 ml. The changes in absorbance were recorded at 340 nm, and the enzyme activity was calculated as nanomoles of CDNB conjugate formed per minute per milligram protein using a molar extinction coefficient of 9.6 × 103 M−1 cm−1.
Measurement of catalase activity
The catalase activity was measured by the method of Claiborne. 35 In brief, the assay mixture consisted of 2.0 ml phosphate buffer (0.1 M, pH 7.4), 0.95 ml H2O2 (0.019 M), and 0.05 ml of PMS (10%) in a final volume of 3.0 ml. The changes in absorbance were recorded at 240 nm. The catalase activity was calculated in terms of nanomoles of H2O2 consumed per minute per milligram protein.
Measurement of SOD activity
The superoxide dismutase (SOD) activity was measured by the method of Marklund and Marklund. 36 The reaction mixture consisted of 2.875 ml tris(hydroxymethyl)aminomethane-hydrochloric acid (Tris–HCl) buffer (50 mM, pH 8.5), pyrogallol (24 mM in 10 mM HCl), and 100 μl PMS in a total volume of 3 ml. The enzyme activity was measured at 420 nm and was expressed as units per milligram of protein. One unit of enzyme is defined as the enzyme activity that inhibits auto-oxidation of pyrogallol by 50%.
Protein estimation
The protein concentration in all samples was determined by the method of Lowry et al. 37 using BSA as standard.
Assay for TNF-α level
TNF-α protein level was measured by enzyme-linked immunosorbent assay kit (eBioscience, Inc., San Diego, California, USA). Analysis was performed according to the manufacturer’s instruction.
ACF assay
Colons were excised from rats immediately, flushed with ice-cold saline (0.85% NaCl), opened longitudinally, and fixed flat between two sheets of filter paper in 10% formalin (Sigma chemical, St Quentin, France). Colons picked up in random order were stained for 6 min in a 0.05%-filtered solution of methylene blue. 38 The number of ACF per colon and number of crypts in each ACF were counted under light microscope (Olympus BX51, Olympus, Tokyo, Japan) at 40× magnification.
MDF assay
Colons, after being scored for ACF, were stained with high iron diamine–alcian blue (HID-AB) procedure to evaluate mucin production. 21 Briefly, colons were rinsed in distilled water and left for 18 h in freshly prepared HID solution (50 ml of distilled water with 120 mg N,N′-dimethyl-m-phenylene diamine, 20 mg N,N′-dimethyl-p-phenylene diamine, and 1.4 ml of 60% ferric chloride). After rinsing, colons were counterstained in 1% AB solution for 30 min. MDF number and number of crypts per MDF were counted under light microscope (Olympus BX51) at 40× magnification.
Histopathology
The colon were excised, flushed with saline, cut open longitudinally along the main axis, and then again washed with saline. These colonic sections were fixed in 10% buffered formalin for at least 24 h, and after fixation, the specimens were dehydrated in ascending grades of ethanol, cleared in benzene, and embedded in paraffin wax. Blocks were made and 5 µm thick sections were cut from the distal colon. The paraffin-embedded colonic tissue sections were deparaffinized using xylene and ethanol. The slides were washed with phosphate-buffered saline and permeabilized with permeabilization solution (0.1 M citrate, 0.1% Triton X-100). These sections stained with hematoxylin and eosin and were observed under light microscope at 10× and 40× magnifications to investigate the histoarchitecture of colonic mucosa.
Immunohistochemical staining for detection of iNOS, COX-2, and IL-6
Sections of 4 μm were cut from formalin-fixed, paraffin-embedded tissue blocks and mounted on poly-
Statistical analysis
The data from individual groups were presented as the mean ± standard error. Differences between the groups were analyzed using analysis of variance followed by Tukey–Kramer multiple comparison test, and the minimum criterion for statistical significance was set at p < 0.05 for all comparisons.
Results
Effect of aloin on the activities of GSH-dependent enzymes in colonic tissue
The activities of GPx, GR, and GST decreased significantly (p < 0.001) in group II (DMH 20 mg kg−1 b.w.) as compared to group I (control). Aloin pretreatment (group III) at the dose of (50 mg kg−1 b.w.) GPx and GR showed not much significant activities and also showed significant increase in the activities of GST (p < 0.05) in group III as compared to group II. Aloin (100 mg kg−1 b.w.) also showed significant increase in the activities of GPx (p < 0.001), GST (p < 0.001), and GR (p < 0.001) in group IV as compared to group II. The activities of these enzymes in group V did not change significantly as compared to group I. (Table 1)
Effects of aloin and DMH on activities of GSH, GPX, GST and GR.a

DMH: 1,2-dimethylhydrazine; GPx: glutathione peroxidase; GST: glutathione-S-transferase; GR: glutathione reductase; D1: dose 1; D2: dose 2; SEM: standard error of the mean; b.w.: body weight.
aResults represent mean ± SE of eight animals per group. D1 = 50 mg kg−1 b w.; D2 = 100 mg kg−1 b.w.
b p < 0.001: Results obtained are significantly different from control group.
c p < 0.01: Results obtained are significantly different from DMH-treated group.
d p < 0001: Results obtained are significantly different from DMH-treated group.
Effect of aloin on the activities of colonic antioxidant enzymes
DMH treatment (20 mg kg−1 b.w.) showed significant decrease in the activities of SOD (p < 0.01) and catalase (p < 0.001) in group II as compared to group I (control). Aloin pretreatment (50 mg kg−1 b.w.) significantly increased the activities of SOD (p < 0.05) and catalase (p < 0.05) in group III as compared to group II. Aloin (100 mg kg−1 b.w.) also showed significant increase in the activities of SOD (p < 0.01) and catalase (p < 0.01) in group IV as compared to group II. However, the activities of these enzymes in group V did not change significantly as compared to group I. (Figures 2 and 3)

Effect of aloin on the activities of colonic SOD enzyme. Values are expressed as mean ± SEM measured as unit per milligram per protein in tissue. SOD activity was significantly reduced in group II (DMH 20 mg kg−1 b.w.; ***p < 0.001) as compared to group I (control). Pretreatment with aloin significantly decreased SOD activity in group III (DMH as group II + aloin 50 mg kg−1 b.w.; ## p < 0.01) and group IV (DMH as group II + aloin 100 mg kg−1 b.w.; ### p < 0.001) as compared to group II (aloin 100 mg kg b.w.). SOD: superoxide dismutase; DMH: 1,2-dimethylhydrazine; b.w.: body weight.

Effect of aloin on the activities of colonic catalase enzyme. Values are expressed as mean ± SEM measured as nanomoles of H202 consumed per minute per milligram tissue. Catalase activity was significantly decreased in group II (DMH 20 mg kg−1 b.w.; ***p < 0.001) as compared to group I. Pretreatment with aloin significantly increased catalase activity in group III (DMH as group II + aloin 50 mg kg−1 b.w.; ## p < 0.05) and group IV (DMH as group II + aloin 100 mg kg−1 b.w.; ### p < 0.001) as compared to group II. H202: hydrogen peroxide; DMH: 1,2-dimethylhydrazine; b.w.: body weight.
Effect of aloin on the level of colonic GSH
The level of GSH was exhausted significantly (p < 0.001) in DMH-treated group (20 mg kg−1 b. w.; group II) as compared to control group (group I). Aloin pretreatment showed a significant increase in the level of GSH in group III (p < 0.001) and group IV (p < 0.001) when compared with group II. Group V exhibited no significant change in the level of GSH as compared to the control group. (Table 1)
Effect of aloin on the level of MDA content in colonic tissue
The levels of MDA content were significantly ( p < 0.001) enhanced in group II (DMH 20 mg kg−1 b. w.) as compared to group I (control). Aloin pretreatment (50 mg kg−1 b.w.) significantly (p < 0.001) decreased the level of MDA in group III as compared to group II. Higher dose of aloin (100 mg kg−1 b.w.) also significantly (p < 0.001) decreased the level of MDA content in group IV as compared to group II. No significant difference was found in the level of MDA between groups I and V (Figure 4).

Effect of aloin on the level of MDA content in the colonic tissue. Values are expressed as mean ± SEM measured as nanomoles of MDA formed per minute per gram tissue. MDA content was significantly (***p < 0.001) increased in DMH-treated group (group II) as compared to control group (group I). Pretreatment with aloin significantly (### p < 0.001, ### p < 0.001) attenuated the MDA content in groups III and IV, respectively, when compared with group II. MDA: malondialdehyde; DMH: 1,2-dimethylhydrazine.
Effect of aloin and DMH on the development of ACF in colonic tissue
In DMH-treated group (group II), the number of ACF/colon is 78.43 ± 11, while supplementation with aloin in group III (69.87 ± 13; p < 0.01) and group IV (47.97 ± 8; p < 0.001) significantly reduced the number of ACF. Original magnification is 10× (Figure 5(a) and (b)).
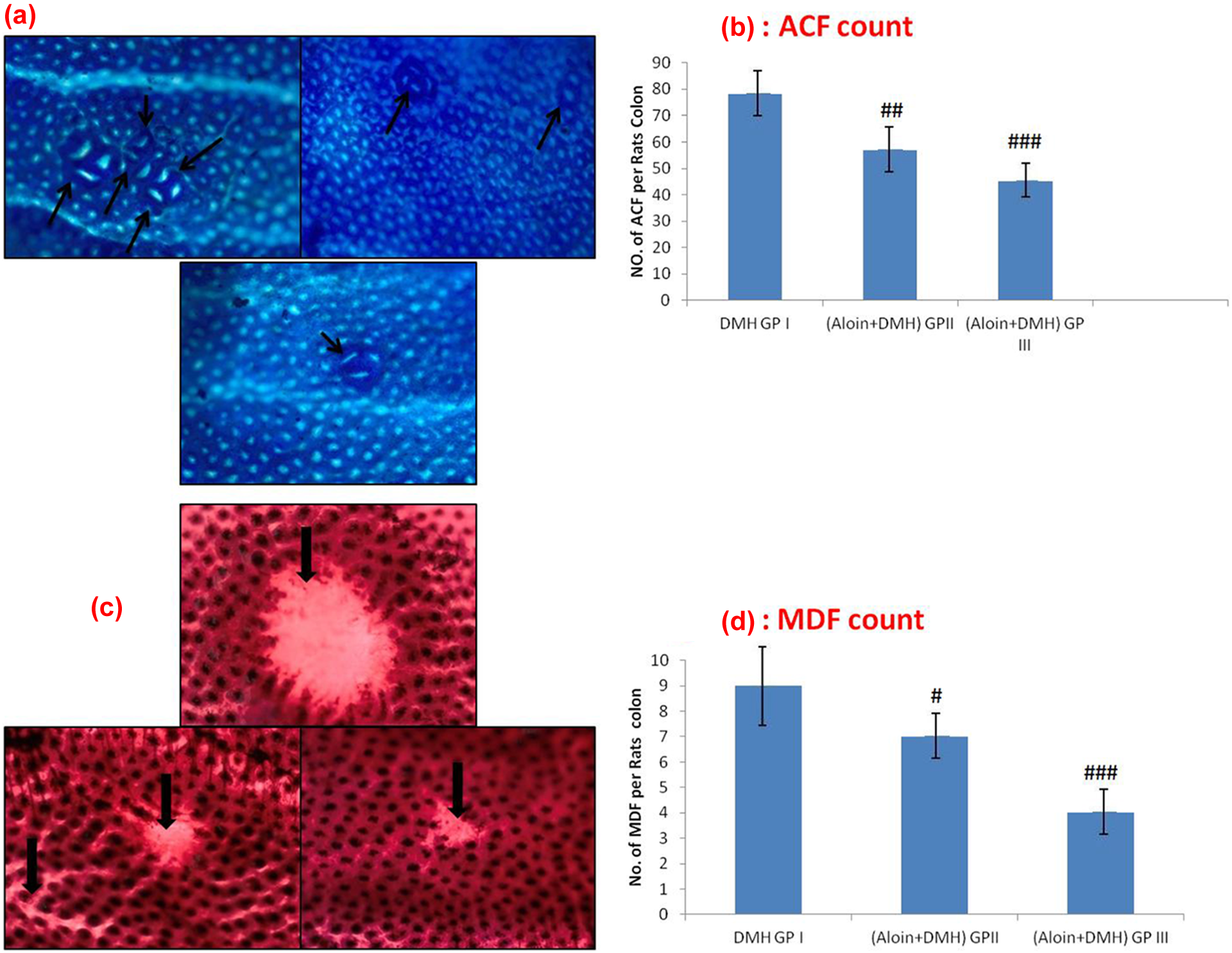
Effects of aloin and DMH on the development of ACF and MDF in colonic tissue. (a) ACF in the rat colon showing round and elongated ACF (groups II, III, and IV) with different crypt multiplicities. The colons were opened, stained with methylene blue, and observed on a glass slide. Original magnification: ×10. (b) Effects of DMH and aloin on incidence of ACF per rat colon. Values are expressed as mean ± SEM. Aloin significantly suppressed the development of ACF in groups III and IV (## p < 0.01, ### p < 0.001) as compared to DMH-treated group (group II). (c) MDF in the rat colon showing depleted mucin (groups II, III, and IV).The colons were opened, stained with HID and AB. Original magnification: ×10. (d) Effects of DMH and aloin on incidence of MDF per rat colon. Values are expressed as mean ± SEM. Aloin significantly suppressed the development of ACF in groups II and IV (# p < 0.05, ### p < 0.001) when compared with DMH-treated group (group II). ACF: aberrant crypt foci; MDF: mucin-depleted foci; DMH: 1,2-dimethylhydrazine; HID: high iron diamine; AB: alcian blue.
Effect of aloin and DMH on the development of MDF in colonic tissue
In DMH-treated group (20 mg kg−1 b. w.; group II), the number of MDF/colon is 8.4 ± 1.44, while supplementation with aloin (50 mg kg−1 b. w.) in group III significantly reduced the number of MDF (7.43 ± 0.64; p < 0.05) level as compared to group II. In group IV, aloin (100 mg kg−1 b. w.), treatment with aloin significantly reduced the number of MDF (4.3 ± 0.57; p < 0.001) as compared to group II. Original magnification is 10× (Figure 5(c) and (d))
Effect of aloin on colon tissues of proinflammatory cytokine (TNF-α)
We have assessed the effect aloin on DMH-induced abnormal increase in TNF-α level (Figure 6). We found that there was a significant increase in the level of proinflammatory cytokines in DMH-treated group (20 mg kg−1 b. w.) compared with control group I (p < 0.001). Pretreatment with aloin (50 mg kg−1 b. w.) significantly inhibited abnormal increase in group III when compared with the only DMH-treated group II (p < 0.05). Pretreatment with aloin (100 mg kg−1 b. w.) significantly inhibited increase in TNF-α in group IV when compared with the only DMH-treated group II (p < 0.001) There was no significant difference in the levels of this proinflammatory cytokine between groups I and V.

Effect of aloin on colon tissues of proinflammatory cytokines (TNF-α). In DMH-treated group (group II), the level of TNF-α was increased significantly (***p < 0.001) as compared with the control group (group I). Treatment with aloin significantly attenuated the level of TNF-α in group III (## p < 0.01) and group IV (### p < 0.001) as compared with group II. There was no significant difference in the level of TNF-α in group V when compared with group I. DMH: 1,2-dimethylhydrazine; TNF-α: tumor necrosis factor-α.
Effects of aloin and DMH-induced overexpression of COX-2, IL6 and iNOS
In group II (DMH only), the COX-2 and iNOS proteins showed more positive staining compared with group I (control). In pretreatment group III aloin (50 mg kg−1 b.w.), COX-2 and iNOS expression showed considerable decrease compared to group II (DMH only). Group IV aloin (100 mg kg−1 b. w.) showed expression of COX-2 and iNOS protein as compared to group II (DMH only). No significant difference was observed in staining pattern in group V as compared to group I (control; Figures 7 to 9).

Effect of aloin on DMH induced colon expression of COX-2 in colon tissue. Representative photomicrographs with ×40 magnification. Brown color indicates COX-2 specific staining and blue color indicates hematoxylin staining. (a) Protein expression in control group almost shows no staining. (b) DMH-treated group (group II) shows more COX-2 immunopositive staining as compared to control group (group I). (c) Aloin pretreatment (group III) showed lesser COX-2 positive immunostaining as compared with group II. (d) Group IV aloin pretreatment shows reduced COX-2 expression as compared to group II. (e) However, there was no significant difference in the COX-2 immunostaining in group V as compared to group I. Black arrows indicate region of positive immunostaining reactivity. DMH: 1,2-dimethylhydrazine; COX-2: cyclooxygenase-2.

Effect of aloin on DMH-induced colon expression of iNOS in colon tissue. Representative photomicrographs with ×40 magnification. Brown color indicates iNOS-specific staining and blue color indicates hematoxylin staining. (a) Protein expression in control group almost shows no staining. (b) DMH-treated group (group II) shows more intense iNOS immunopositive staining as compared to control group (group I). (c, d) Aloin pretreatment groups III and IV reduce iNOS expression as compared to group II. (e) However, there was no significant difference in iNOS immunostaining in group V as compared to group I. Group IV pretreatment groups show aloin to be more a potent inhibitor of iNOS as compared to group III pretreatment groups. Black arrows indicate region of positive immunostaining reactivity. iNOS: inducible nitric oxide synthase; DMH: 1,2-dimethylhydrazine.

Effect of aloin on DMH-induced colon expression of IL-6 in colon tissue. Representative photomicrographs with ×40 magnification. Brown color indicates IL-6-specific staining and blue color indicates hematoxylin staining. (a) Protein expression in control group almost shows no staining. (b) DMH-treated group (group II) shows more IL-6 immunopositive staining as compared to control group (group I). (c) Group III aloin pretreatment showed lesser IL-6 positive immunostaining as compared to group II. (d) Group IV aloin pretreatment shows reduced IL-6 expression as compared to group II. (e) However, there was no significant difference in the IL-6 immunostaining in group V as compared to group I. Black arrows indicate region of positive immunostaining reactivity. IL-6: interleukin-6; DMH: 1,2-dimethylhydrazine.
Effect of aloin on DMH-induced increase in PCNA expression on colon tissue
In group II (DMH only), the PCNA showed increased number of positive nuclei staining compared with group I (control). In pretreatment group III aloin (50 mg kg−1 b.w.), the PCNA-positive staining appeared slightly decreased in colon tissue when compared with group II (DMH only). Pretreatment group IV, higher dose of aloin (100 mg kg−1 b.w.), showed substantial decrease in the number of PCNA-positive nuclei in colonic tissue as compared to group II (DMH only), but the decrease was more effective in pretreatment group IV than in pretreatment group III. There was no significant difference in staining pattern in group V as compared to group I (control; Figure 10).

Effects of aloin on DMH expression of PCNA in colon cancer. Representative photomicrographs with ×40 magnification. Brown color indicates PCNA-specific staining and blue color indicates hematoxylin staining. (a) Protein expression in control group almost shows no staining. (b) DMH-treated group (group II) shows more PCNA positive cells as compared to control group (group I). (c, d) Aloin pretreatment groups III and IV inhibit PCNA expression as compared to group II. (e) However, there was no significant difference in PCNA immunostaining in group V as compared to group I. Group IV aloin pretreatment (d) shows aloin to be a more potent inhibitor of cellular proliferation PCNA as compared to group III aloin pretreatment (c). Black arrows represent PCNA positive cell. PCNA: proliferating cell nuclear antigen.
Histopathological findings
Histological sections of control group (group I) exhibited normal histoarchitecture of the colon. In DMH-treated group (group II), irregular glandular structure, regional destruction of the mucosa with severe inflammatory cells infiltration in mucosal and submucosal layers, as well as submucosal edema were examined. The mid-colon of a prolonged reactivation rat showed large lymphoid follicles invading the lamina propria, causing loss of mucosal architecture. In group III (pretreatment) and group IV (pretreatment), histological sections showed that aloin protected the mucosa from damage, and there was notable decrease in the inflammatory cells permeation and submucosal edema but the protection was not as effective as in the pretreatment group IV in comparison with group III. Group V (aloin only) histological sections exhibited typical histoarchitecture of the colon as similar to group I (control group; Figure 11).

(a) Group I (control group) shows the normal histoarchitecture of the colon with normal mucosal glandular structure with mild inflammatory cells infiltration. (b) Group II shows massive inflammatory cells infiltration (white small arrows) with crypt abscess formation (white star) and distorted mucosal glandular structure (black head arrows). (c) Group III shows crypt ablation(arrowhead) with less inflammatory cells infiltration as compared to group IV (d), which shows crypt abscess formation with mild inflammatory cells infiltration. (e) Group V shows normal histoarchitecture of the colon with normal mucosal glandular structure with mild inflammatory cells infiltration similar to group I.
Discussion
Chemoprevention opens new window in the avoidance of all types of cancer including colon cancer. Although great efforts have been made to avoid the development of various types of cancer, there is no complete and promising therapeutic agent. Cancer chemopreventive agents are gaining much consideration because natural products like fruits, vegetables, medicinal plants, and herbs have many pharmacological properties and have the potential to scrap against frequent human diseases associated with oxidative stress. 39 Several natural compounds of dietary sources have been reported to hamper various diseases including cancer 39,40 and aloin is one of them, which belongs to the category of anthracycline. Aloin, a natural anthracycline, found in aloe plants is a hydroxyanthraquinone derivative shown to have antitumor properties. 12 Aloin has the effect of inhibiting cancer induction by several mechanisms. 13 In the present study, we have showed that pretreatment with aloin resulted in the protection against DMH-induced colonic damage by ameliorating oxidative stress, hyperproliferation, and inflammation. In our laboratory, we have previously confirmed the role of oxidative stress in colonic damage induced by DMH. 41 DMH is a colon-specific carcinogen, which undergoes metabolism in the liver, leading to the generation of methyldiazonium ion, which is known to elicit oxidative stress that may contribute to initiation of lipid peroxidation via MDA formation. 42
MDA formation is one of the important markers of oxidative damage, and high level of MDA formed has been observed after treatment with DMH. 43,44 The highly reactive hydroxyl radical attacks unsaturated lipid biomembrane and leads to the generation of the lipid peroxyl radical, which has an unpaired electron and further forms chain reaction in peroxidation of the lipid molecules. 45 Consistent with earlier reports, our results also showed remarkable increase in the level of MDA after DMH treatment. Treatment with aloin at the dose of 50 and 100 mg kg−1 b.w. significantly attenuated the levels of MDA.
Endogenous nonenzymatic (GSH) and enzymatic (SOD, GPx, GR, catalase, etc.) antioxidants act as a major defense system against free radicals generated by DMH. 46 GSH and its oxidized counterpart represent a chief redox buffer system of the cell. GSH can act either as a nonenzymatic antioxidant by direct interaction of –SH group with reactive oxygen species (ROS), or it can be concerned in the enzymatic detoxification reaction against ROS as a cofactor or coenzyme.
DMH treatment leads to the depletion of GSH, 9,47 and our findings showed that in DMH-treated rats, the colonic GSH level has been decreased significantly as compared to control group. Treatment with aloin restored a normal level of GSH indicating protective efficacy of aloin against DMH-induced oxidative damage. 48
DMH treatment generates free radicals in the colonic tissue and their level is controlled by GSH and other enzymatic antioxidants like SOD, catalase, GPx, GR, and so on. These antioxidants play an important role in protection of the colonic tissue by scavenging the free radicals. Thus, treatment with DMH results in depletion in the level of antioxidants. 44,47 The protective efficacy of aloin against carcinogen-induced oxidative stress can be recognized as its ability to induce antioxidant enzymes (Catalase, SOD, GR, and GPx) and phase II detoxifying enzymes (GST) in rats as reported previously. 49
Oxidative stress and inflammation play an important role in carcinogenesis and are documented to manipulate tumor initiation and promotion. 50 COX-2, an important inflammatory marker, is induced during inflammation, and it is reported to be overexpressed in colonic adenomas and tumors 51,52 COX-2 has been shown to play a key role in polyp formation and its inhibition has proven to be an important strategy for chemopreventive treatment of colon-related disorders. 53 We observed that DMH-treated rats showed enhanced expression of COX-2, while pretreatment with aloin at the dose of 100 mg kg−1 b.w. efficiently inhibited the expression of COX-2 and pretreatment with aloin at the dose of 50 mg kg−1 b.w. also inhibited expression of COX-2 but not as effectively as in the case of pretreatment with higher dose (100 mg kg−1 b.w.). iNOS, another inflammatory marker, has been shown to be upregulated in DMH-induced colon tumorogenesis 54,55 and its inhibition is an additional important approach to prevent colon cancer. 56 We also observed increased pattern of iNOS expression in DMH-treated group and pretreatment with aloin at the dose of 50 and 100 mg kg−1 b.w. attenuated the expression of iNOS.
Inflammation is one of the complex and major event during tumor promotion and metastasis. The key molecules besides cytokines and chemokines are the NO-dependent iNOS activation and upregulation of COX-2. The increase in the NO levels upregulates the gene and protein expression for the iNOS, which through further activation of prostaglandins leads to the enhanced COX activities. 57 Although the signaling involving these molecules is also dependent on various other molecular targets, our results show evidence that iNOS and COX-2 play a major role in inflammation-dependent pathology in the colon. 58
In previous studies, aloin efficiently reduced the expressions of TNF-α and IL-6 in DMH-induced colonic tumors and thus exhibited its anti-inflammatory activity. 59
TNF-α and IL-6 are the proinflammatory cytokines, and several evidences have shown that the expression of TNF-α and IL-6 are dependent on the activation of NF-κB, which is one of the key inflammatory mediators. 25 Therefore, targeting TNF-α and IL-6 is an effective approach for the treatment of colon cancer. 26 We observed that DMH-treated rats showed enhanced expression of IL-6, while a higher dose of aloin (100 mg kg−1 b.w.) in group IV markedly ameliorates the expression of IL-6 and a lower dose of aloin (50 mg kg−1 b.w.) in group III also ameliorated the expression of IL-6 but not as effectively as compared to higher dose of aloin (100 mg kg−1 b.w.) in group IV. Our data suggest that aloin effectively suppresses the production of these cytokines, which implicates its possible role in chemoprevention of colon cancer.
PCNA is a nonhistone acidic protein expressed in the nuclei of proliferating cells throughout G1 and S phase of cell cycle. 60–62 Overexpression of PCNA protein was directly related to increased proliferation, because it plays an important role in nucleic acid metabolism as a part of the replication. 63 It is previously reported to be an important biomarker in gastrointestinal cancer. 63,64 In this study, DMH-treated group showed enhanced expression of PCNA in the colonic tissue as compared to the control group, while treatment with aloin reduced the expression of PCNA at both the doses.
The above findings of antioxidative, anti-inflammatory, and antitumor-promoting potential of aloin were supported by histopathological findings, which confirmed that aloin concealed the inflammatory responses in the colon by decreasing the massive infiltration of the inflammatory cells in the mucosal and submucosal layers induced by DMH. It also decreased the severity of submucosal edema and regional destruction of mucosal layer induced by DMH in the colon of Wistar rats. These histological findings exhibited the protective efficacy of aloin against damaging effects of DMH in the colon of Wistar rats.
ACF and MDF are microscopic lesions that are probable precursors of adenomas in the colon and can be used as markers to neoplasia and precancerous stage. 64 MDF possess dysplastic features similar to those encountered in colon tumors, and their number and multiplicity are correlated with carcinogenesis. ACF have been described as early lesions in carcinogen-treated rodents and humans at risk, 3,65,66 and their formation is regarded as a necessary step in the development of colon cancer. In this study, it was observed that aloin supplementation effectively suppresses the development of ACF and MDF as compared to DMH-treated group.

Graphical representation of the mechanism of action of aloin on DMH induced rat colon inflammation and early cancer markers.
In conclusion, the findings of the present study showed that aloin supplementation effectively suppressed the initial phases of colon carcinogenesis possibly by attenuating the oxidative damage, inflammatory mediators (COX-2, iNOS, TNF-α, and IL-6), hyperproliferation (PCNA), and early neoplastic transitions (ACF and MDF) induced by DMH in the colon of Wistar rats. Therefore, aloin may prevent the tumor-promoting effects of DMH through its antioxidant and anti-inflammatory properties and might be clinically useful after further chemopreventive studies.
Highlights
Aloin helps to maintain antioxidant armory in DMH-induced colon toxicity.
Aloin treatment resulted in decline in expression of COX-2 and iNOS induced by DMH.
Aloin treatment helps to normalize DMH-induced increase in PCNA.
Aloin increase expression of IL-6 and TNF-α level on DMH treatment.
Footnotes
Funding
The author OOH has been provided with a fellowship to carry out the research from the University Grants Commission, New Delhi, India, and Ministry of Higher Education, Republic of Iraq.
