Abstract
Vigabatrin (VGB) is an antiepileptic drug thatincreases brain γ-aminobutyric acid (GABA) levels through irreversible inhibition of GABA transaminase. The aim of this study was to evaluate neurotoxicological effects of VGB measuring motor activity and genotoxic and mutagenic effects after a single and repeated administration. Male Wistar rats received saline, VGB 50, 100, or 250 mg/kg by gavage for acute and subchronic (14 days) treatments and evaluated in the rotarod task. Genotoxicity was evaluated using the alkaline version of the comet assay in samples of blood, liver, hippocampus, and brain cortex after both treatments. Mutagenicity was evaluated using the micronucleus test in bone marrow of the same animals that received subchronic treatment. The groups treated with VGB showed similar performance in rotarod compared with the saline group. Regarding the acute treatment, it was observed that only higher VGB doses induced DNA damage in blood and hippocampus. After the subchronic treatment, VGB did not show genotoxic or mutagenic effects. In brief, VGB did not impair motor activities in rats after acute and subchronic treatments. It showed a repairable genotoxic potential in the central nervous system since genotoxicity was observed in the acute treatment group.
Introduction
Vigabatrin (VGB) is an antiepileptic drug (AED) helpful in infantile spasms and refractory epilepsy. This drug increases brain γ-aminobutyric acid (GABA) level through irreversible inhibition of the GABA catabolic enzyme GABA transaminase. 1,2 Since VGB is a structural analog of GABA, the enzyme accepts it as a substrate. VGB is metabolized into a reactive intermediate that binds to the enzyme, inhibiting its activity. 3 VGB also seems to increase GABA concentrations, by inhibiting glial uptake or stimulating its release. 1 It is known that GABA is the major inhibitory neurotransmitter in mammals and that it has an important role in epileptogenesis, anxiety, and depression. 2 Therefore, drugs that act according to this neurotransmission system have become useful in therapy.
Different pharmacological and toxicological properties have been attributed to VGB using behavioral animal models. Low doses of VGB (50 mg/kg/day) for 28 days produced an anxiolytic-like effect on the elevated plus maze test. 3 Furthermore, Levav et al. 4 showed that VGB was able to induce behavioral changes in newborn mice, developmental delay, and impaired sensory motor function measured in the rotarod test after treatment with (50 mg/kg, subcutaneously) for 1 and 2 weeks.
VGB use has been restricted due to some adverse effects, like defects on the peripheral visual field and risk of retinopathy. 1,5 Currently, the benefits of VGB seem to outweigh the risks, under some conditions, but studies showed that VGB may be neurotoxic and should be used with caution. 6
Long-term oral administration of VGB at dosages of 50 mg/kg/day has been shown to induce intramyelinic edema in brain of adult rodents. 7 VGB-induced intramyelinic edema was histopathologically characterized by microvacuolation of specific regions of the brain caused by fluid accumulation and separation of the outer layers of myelin associated with reactive astrocytosis and microglial activation. 8,9 Several studies have shown the adverse effect of VGB on the developing postnatal rat brain 7,10 Young rats treated with VGB 50 mg/kg/day showed similar vacuolar brain lesions in the white and gray matter, compared with adult rats, suggesting injuries in brain development. 7
In order to expand knowledge of VGB toxicity, the aim of this study was to evaluate its effect on motor coordination after a single or repeated administration in rats using the rotarod task and to evaluate any possible DNA damage in brain (cortex and hippocampus), blood, and liver tissues using the comet assay. In addition, mutagenic activity was assessed using the micronucleus test in bone marrow. Thus, toxicity was evaluated in brain as target tissue and, systemically, using other tissues.
Materials and methods
Animals
Seventy-five male Wistar albino rats weighing between 200 and 250 g (2 months old) were obtained from the Lutheran University of Brazil (ULBRA). Rats were housed in plastic cages, with ad libitum access to water and food, under a 12-h light/12-h dark cycle and at a constant temperature of 23 ± 2°C. All experimental procedures were performed in accordance with the NIH guidelines for the Care and Use of Laboratory Animals and with the consent of the Ethics Committee of ULBRA (No. 2013-24P).
Drugs and pharmacological procedures
VGB (Sabril®, Sanofi Aventis, Brazil) was dissolved in 0.9% saline solution. All solutions were prepared immediately prior to administration.
Behavioral (rotarod test) and genotoxic and mutagenic parameters (comet and micronucleus assays) were evaluated after a single (acute treatment) or repetitive administration (subchronic treatment, 14 days). Animals were divided in four groups that received saline (sodium chloride (NaCl) 0.9%) or VGB (50, 100, or 250 mg/kg). All drugs were orally administered by gavage as a volume of 10 mL/kg of weight. The same visual observer participated in all behavioral tests to reduce errors. After 3 h from single dose, the animals were killed and samples of cortex, hippocampus, blood, and liver were collected for the comet assay. In the subchronic treatment, animals were killed 24 h after the last administration (day 14).Then, samples used in the comet assay and bone marrow from femurs used in the micronucleus test were collected.
Rotarod performance
The method described by Leal et al. 11 was used. For evaluation of acute effects, animals were initially trained to remain on the rotarod apparatus (18 r/min for 120 s; UgoBasile), and those that did not remain on the bar for at least two out of three consecutive trials were excluded. VGB or saline was administered 24 h after this initial training, and latency to fall from the rotarod (one trial of maximum 60 s) was determined 30, 60, 90, and 120 min after administration of acute treatment. For the subchronic evaluation, animals received one dose/day for 14 days (same groups described before). Twenty-four hours after the last administration, animals were tested in the rotarod apparatus, and the latency to fall was determined (one trial of maximum 60 s).
Comet assay
The alkaline comet assay was performed according to Tice et al. 12 with minor modifications. Samples of blood, liver, cortex, and hippocampus were collected.
Liver and brain samples were placed in 0.5 mL cold phosphate-buffered saline and finely minced in order to obtain a cellular suspension, and blood samples were placed in 15 µL anticoagulant (heparin sodium 25,000 IU- Liquemine). These suspension cells from forebrain, liver, and peripheral blood (5.0 µL) were embedded in 95 µL 0.75% low melting point agarose (Gibco BRL, Gaithersburg, Maryland, USA). The mixture (cell/agarose) was spread on a fully frosted microscope slide coated with a layer of 300 µL normal melting agarose (1%) (Gibco BRL). After solidification, slides were placed in lysis buffer (2.5 M NaCl, 100 mM ethylenediaminetetraacetic acid (EDTA) and 10 mM Tris, freshly added 1% Triton X-100 (Sigma-Aldrich, St Louis, Missouri, USA) and 10% dimethyl sulfoxide, pH 10.0) for 48 h at 4°C. Subsequently, the slides were incubated in freshly prepared alkaline buffer (300 mM sodium hydroxide and 1 mM EDTA, pH > 13) for 20 min, at 4°C. An electric current of 300 mA and 25 V (0.90 V/cm) was applied for 15 min to induce DNA electrophoresis. The slides were then neutralized (0.4 M Tris, pH 7.5), stained with silver, and analyzed using an optical microscope. 13 Images of 100 randomly selected cells (50 cells from each of two replicate slides) were analyzed from each tissue per animal. Cells were also scored visually according to tail size into five classes, ranging from undamaged (0), to maximally damaged (4), resulting in a single DNA damage score for each animal, and consequently each group studied. The damage index (DI) can range from 0 (completely undamaged, 100 cells × 0) to 400 (with maximum damage, 100 × 4). Damage frequency (DF) was calculated based on number of cells with tail versus those with no tails. 14,15 Hydrogen peroxide (H2O2) of 0.25 mM concentration was used as the positive control.
Micronucleus test
The micronucleus assay was performed according to the US Environmental Protection Agency Gene-Tox Program. 16 The bone marrow was extracted from both femurs, suspended in fetal calf serum, and smeared on glass slides. After drying, the slides were fixed in methanol for 10 min and stained in 10% Giemsa for 7 min. Two thousand polychromatic erythrocytes (PCEs) were examined in each slide, and the incidence of micronuclei was observed. The ratio of PCE to normochromatic erythrocytes was scored in 2000 cells for each animal. All sides were coded for blind analysis.
Statistical analysis
Results of the rotarod test are expressed as mean ± SEM and were analyzed by means of analysis of variance (ANOVA), followed by a Duncan test. Comet and micronucleus data are expressed as means ± SD, and statistical significance was determined by one-way ANOVA complemented with the Dunnett’s test. In all comparisons, p < 0.05 indicated statistical significance.
Results
The behavioral pattern of the groups given saline or VGB (50, 100, or 250 mg/kg) was measured using the rotarod test after acute or subchronic treatment. No statistical difference was observed in the time animals remained in the apparatus, between the treated groups (VGB), in comparison with the saline group after both treatments (Figures 1 and 2).

Effect of VGB (50, 100, or 250 mg/kg) measured in the rotarod test at 30, 60, 90, and 120 min after a single administration. Latency (in seconds) expressed as mean ± SEM (N = 6–7). One-way ANOVA is used for statistical analysis. VGB: vigabatrin; SEM: standard error of mean; ANOVA: analysis of variance.

Effect of VBG (50, 100, or 250 mg/kg) measured in the rotarod test after a subchronic treatment. Latency (in seconds) expressed as mean ± SEM (N = 10).One-Way ANOVA is used for statistical analysis. VGB: vigabatrin; SEM: standard error of mean; ANOVA: analysis of variance.
As shown in Table 1, VGB 250 mg/kg increased DI and DF in blood tissue sampled 3 h after administration of VGB. The same was observed for DI, with VGB 100 mg/kg. In the hippocampus, only the highest VGB dose increased DF, though it did not induce DNA damage in liver and cortex. However, in the subchronic treatment, VGB was not able to increase DI and DF in any of the tissues and doses analyzed (Table 2).
Comet assay in the peripheral blood, liver, cortex, and hippocampus from rats treated once with VGB or saline solution.a
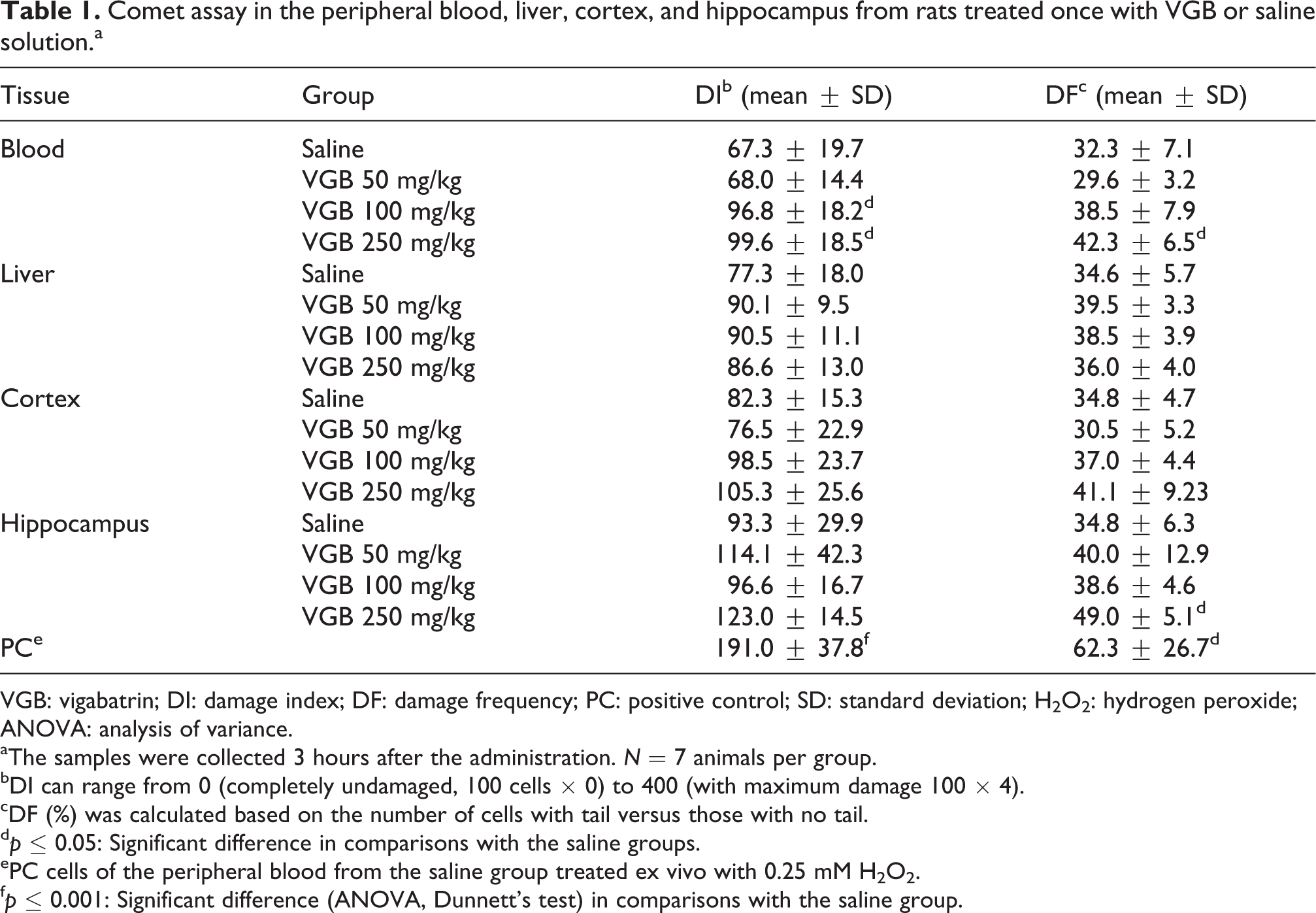
VGB: vigabatrin; DI: damage index; DF: damage frequency; PC: positive control; SD: standard deviation; H2O2: hydrogen peroxide; ANOVA: analysis of variance.
aThe samples were collected 3 hours after the administration. N = 7 animals per group.
bDI can range from 0 (completely undamaged, 100 cells × 0) to 400 (with maximum damage 100 × 4).
cDF (%) was calculated based on the number of cells with tail versus those with no tail.
dp ≤ 0.05: Significant difference in comparisons with the saline groups.
ePC cells of the peripheral blood from the saline group treated ex vivo with 0.25 mM H2O2.
fp ≤ 0.001: Significant difference (ANOVA, Dunnett’s test) in comparisons with the saline group.
Comet assay in the peripheral blood, liver, cortex, and hippocampus from rats treated with VGB or saline solution for 14 consecutive days.a

VGB: vigabatrin; DI: damage index; DF: damage frequency; PC: positive control; SD: standard deviation; H2O2: hydrogen peroxide; ANOVA: analysis of variance.
aN = 7 animals per group.
bDI can range from 0 (completely undamaged, 100 cells × 0) to 400 (with maximum damage 100 × 4).
cDF (%) was calculated based on the number of cells with tail versus those with no tail.
dPC cells of the peripheral blood from the saline group treated ex vivo with 0.25 mM H2O2.
ep ≤ 0.001: Significant difference (ANOVA, Dunnett’s test) in comparison with the saline group.
fp ≤ 0.01: Significant difference in comparison with the saline group.
As shown in Table 3, there was no increase in micronucleus frequency in treated groups in comparison with the saline group. The positive control group (cyclophosphamide) showed a significant increase in micronucleus frequency in PCE, as expected. The PCE/NCE ratio was similar across all groups, indicating no toxicity in bone marrow of the animals.
Mutagenic activity in bone marrow of rats treated with VGB or saline solution for 14 consecutive days.a
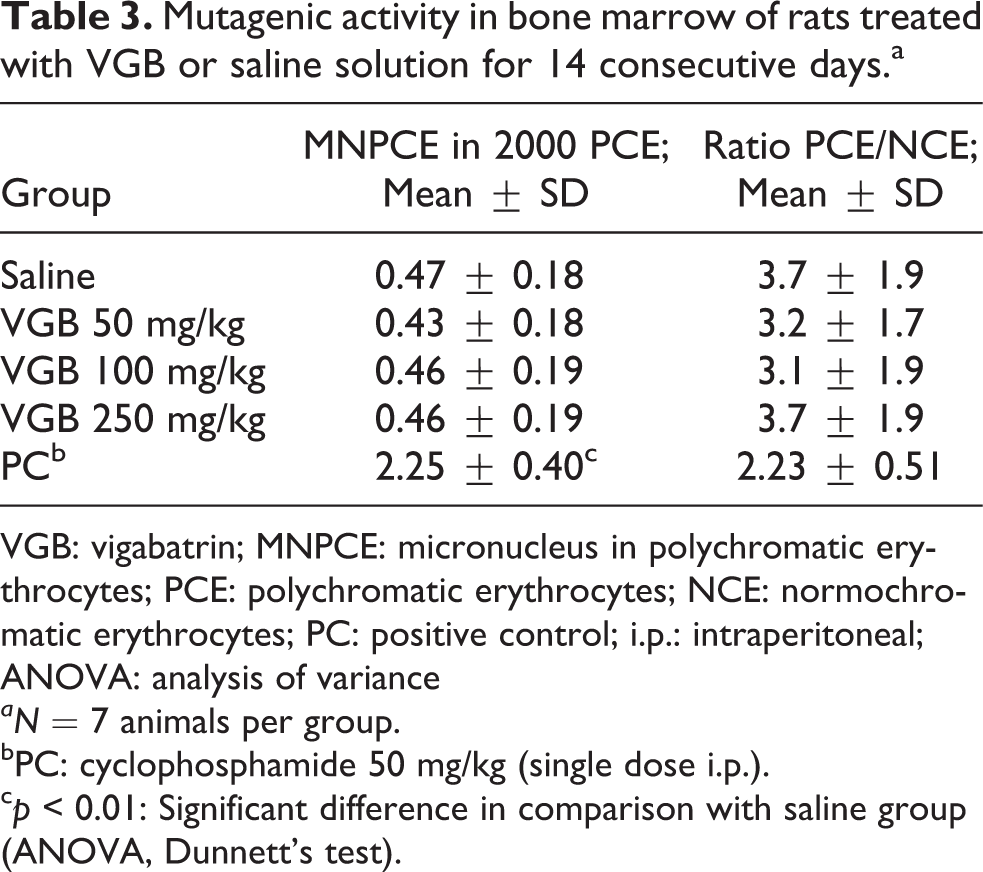
VGB: vigabatrin; MNPCE: micronucleus in polychromatic erythrocytes; PCE: polychromatic erythrocytes; NCE: normochromatic erythrocytes; PC: positive control; i.p.: intraperitoneal; ANOVA: analysis of variance
aN = 7 animals per group.
bPC: cyclophosphamide 50 mg/kg (single dose i.p.).
cp < 0.01: Significant difference in comparison with saline group (ANOVA, Dunnett’s test).
Discussion
Considering the lack of studies about toxicity of VGB, despite its clinical use in epilepsy patients, this study evaluated possible motor alterations and genotoxic effect induced by VGB, using the rotarod test, the comet assay, and the micronucleus test. VGB did not affect motor coordination, after acute and subchronic treatments, suggesting that the drug was not able to induce toxicological effects in the tested doses. The lack of systemic toxicity was later confirmed by similar PCE:NCE ratios of treated groups, in comparison with the saline group (Table 3).
Few studies evaluated VGB toxicity in vivo. Evidence shows that chronic use of VGB in animals causes lesions in some brain regions. Rats treated with a mean daily oral dose of VGB 312 mg/kg developed vacuoles in the white matter. 17 Also, rats treated with VGB 250 mg/kg/day developed lesions detected by magnetic resonance imaging in the cerebellar white matter in vivo. VGB-induced changes were detected, including reactive astrocytosis, microglial proliferation, and vacuolation in the rat brain. 18 Analysis of brain tissue in neonates and young rats showed that the vacuoles started as a development of the myelin sheath, a process consistent with intramyelinic edema. 7 Furthermore, VGB axonal degeneration was caused in the white matter, which proved to be irreversible in neonatal rats, after 5 days of treatment, using a mean daily dose of 40 mg/kg. 10
The rotarod test is an important behavioral model because it allows the screening of neurological difficulties produced by AED in terms of motor coordination and balance. This task is able to measure parameters that can be predictive of adverse effects, such as ataxia, in patients with epilepsy. 19 Cuadrado and Armijo 5 reported that VGB decreased the neurotoxicity of valproate evaluated in the rotarod test in mice. Results obtained in this study showed that VGB was not able to affect motor coordination measured after a single and subchronic treatment in the rotarod task, suggesting no neurotoxicity of the drug, even when it is administered at high doses.
Changes in the activity of the GABA-ergic system in the brain have been reported to affect the behavior of the animal, as shown by spontaneous motor activity and behavioral exploration. 20,21 The activity of GABA-transaminase has previously been found to be significantly reduced in the rat brain 24–48 h after the last injection of repeated treatment with a 100 mg/kg day of VGB for 11 days. 22 At this time point, the content of GABA in the brain has been found to rise to its maximal level. 23,24 In patients with refractory partial seizures treated with VGB 1.5 g twice daily, the drug was associated with a significant reduction in motor speed and overall score in a design learning test, in the first 20 weeks. 24 Although VGB is capable of producing a strong inhibition of GABA transaminase (half maximal inhibitory concentration (IC50) of 24 µM in neurons and 89 µM in astrocytes), 25 at the doses tested in this study, the performance of animals in the rotarod task was not altered by single or repeated administration of this drug.
The comet assay was used to detect recent DNA damage, such as single and double strand breaks, alkali-labile sites, DNA–DNA and DNA–protein cross-links. Biological samples were collected after acute and subchronic treatments in order to evaluate possible DNA repair, as recommended by Tice et al. 12 VGB was able to induce DNA damage in blood sample collected 3 h after a single dose at 100 and 250 mg/kg. However, this genotoxic activity was not observed after 14-day repeated administrations, suggesting that repair mechanisms are triggered during this period.
Plasmatic VGB concentration increases in a linear dose-dependent manner after single doses. 26 Pharmacokinetic studies have shown that peak VGB concentrations were achieved within 0.4 h in serum, after intraperitoneal administration of 500 and 1000 mg/kg doses, while the elimination half-life was 1.1–1.5 h, indicating rapid absorption and elimination. 27 In the present study, blood cells were challenged with the maximum concentration peak in the first hours after VGB oral administration, and the samples collected after 3 h showed an increase in DNA strain break in blood cells in the groups treated with 100 and 250 mg/kg, indicating also that the drug reached the systemic circulation and the target tissues, in the hippocampus, an increase in DF was observed (p < 0.05).
Previous studies showed that VGB was not able to protect against glutamate-induced toxicity in hippocampal neurons in vitro and that, conversely it induced toxicity, which was attributed to an increase in GABA concentration acting on GABAA receptors. 28 However, in vivo, VGB distribution in the brain has been shown to be region specific, with cortex concentrations being two to three times greater than VGB concentrations achieved in the hippocampus. Frontal cortex and hippocampal extracellular fluids showed concentrations VGB peaking within 0.7–1.0 h, somewhat later than in serum (0.4 h), and the concentrations between serum and brain extracellular fluid were equilibrated only 7–8 h after VGB administration. 27 Here, DNA damage in blood was more pronounced than in brain tissue, which probably reflects the different distribution of VGB between these tissues, with concentration in blood being higher than in brain in the first hours after administration.
It is known that GABA concentrations begin to increase approximately 3 h after VGB administration. 27 A single VGB 200 mg/kg dose significantly decreased brain GABA-transaminase activity after 4 h. 29 In the present study, it is possible that 3 h was not enough for VGB to increase GABA concentrations in brain via inhibition of GABA transaminase. Furthermore, VGB administration (1000 mg/kg) has been associated with a significant increase in extracellular fluid GABA concentrations in the frontal cortex, but not in the hippocampus. 27 Hence, a direct effect is expected, in which VGB induces DNA damage in brain without association with an increase in GABA, in the acute treatment, differently to the subchronic treatment, when the GABA concentrations may be increased by VGB. In a study using the wing somatic mutation and recombination test in Drosophila melanogaster, phenytoin and gabapentin showed genotoxic effects, while pregabalin displayed lower genotoxicity, when compared with the other two antiepileptics, all structurally related to VGB. 30
VGB did not induce DNA damage in liver after acute and subchronic treatments, suggesting that the drug is not hepatotoxic. In fact, VGB is not metabolized in liver, but, rather, it is exclusively eliminated renally. 7 Similarly, no genotoxic activity was detected in blood and brain tissues in subchronic treatment. The lack of genotoxicity after subchronic treatment could be explained in light of the rapid absorption and elimination of VGB. Since the samples were collected 24 h after the last administration, the genotoxic effects in blood reached a peak in the first hours after each dose administration and decreased by repair and/or elimination of the drug, leading to negative results for genotoxicity. Plasma accumulation should not be expected because of the short half-life of VGB and the large interval of administration (24 h) used in this study.
A marked inhibition in brain GABA transaminase is more pronounced after VGB multiple doses are administered, in comparison with a single dose. 23 In fact, repeated VGB administration leads to an overall elevation in GABA concentration by inhibition of GABA transaminase, affecting several GABA receptors. 26,31,32 The enhanced GABA function may reduce neuronal degeneration by inhibiting glutaminergic neurons and, consequently, inhibiting the release of cytotoxic levels of glutamate. 33 In this sense, VGB could promote protective effects on brain tissue via action mechanisms that affect GABA transaminase. This could explain the lack of genotoxic effects on brain tissues in the subchronic treatment.
However, DNA repair can be free of errors, or subject to these events, when it may lead to mutations. In order to evaluate possible induction of mutagenicity, the micronucleus test was performed at the end of the experimental procedures to detect clastogenic/aneugenic activities, which lead to increasing micronucleus frequency, and suggests mutagenic effects at chromosomal level. The micronucleus test is internationally recommended to evaluate the safety of drugs. 16 As shown in Table 3, VGB was not able to increase the micronucleus frequency in bone marrow, suggesting that this drug does not present mutagenic effects. Pregabalin, a structural analog of VGB, also has not shown mutagenic activities evaluated in Salmonella/microsome assay and in micronucleus test in bone marrow from rodents given single oral doses of up to the maximum dose of 2000 mg/kg. 34 To our knowledge, this study is the first showing the lack of mutagenicity in VGB after repeated administration.
In conclusion, our results indicate that VGB was able to induce a repairable genotoxic potential in the central nervous system since DNA damage was observed only in the acute treatment group. However, further studies should be conducted to evaluate other adverse effects on the neural tissue, and the effects of the combination of VGB with other drugs, since in other investigations VGB proved toxic in rodents.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Council of Technological and Scientific Development (CNPq, no. 307064/2013-1).
