Abstract
Biomedical application of silver nanoparticles (AgNPs) has been rapidly increasing. Owing to their strong antimicrobial activity, AgNPs are used in dermatology in the treatment of wounds and burns. However, recent evidence for their cytotoxicity gives rise to safety concerns. This study was undertaken as a part of an ongoing programme in our laboratory to develop a topical agent for wound healing. Here, we investigated the potential toxicity of AgNPs using normal human dermal fibroblasts (NHDF) and normal human epidermal keratinocytes (NHEK) with the aim of comparing the effects of AgNPs and ionic silver (Ag-I). Besides the effect of AgNPs and Ag-I on cell viability, the inflammatory response and DNA damage in AgNPs and Ag-I–treated cells were examined. The results showed that Ag-I were significantly more toxic than AgNPs both on NHDF and NHEK. Non-cytotoxic concentrations of AgNPs and Ag-I did not induce DNA strand breaks and did not affect inflammatory markers, except for a transient increase in interleukin 6 levels in Ag-I–treated NHDF. The results showed that AgNPs are more suitable for the intended application as a topical agent for wound healing up to the concentration 25 µg/mL.
Keywords
Introduction
Nanotechnology is a highly promising field for the development of new applications in a variety of areas including electronics, environmental remediation, and medical healthcare. 1 Nanomaterial is defined as a substance (natural, incidental or manufactured) containing particles in an unbound state, as an aggregate or as an agglomerate and where, for 50% or more of the particles in the number size distribution, one or more external dimensions is in a size ranging from 1 to 100 nm. 2 Nanoparticles (NPs) can take many different forms such as tubes, rods, wires or spheres, with more elaborate structures devised such as nano-onions and nano-pea pods. 3,4 Due to the large surface area and high reactivity of NPs compared with macro-/micro-particles, NPs exhibit excellent physical, chemical and biological properties. This makes nanomaterial attractive for exploitation in diverse products, and therefore, the field of nanotechnology is expanding at a rapid rate. 5,6
In terms of interest and utilization, silver NPs (AgNPs) seem to be the most popular. Ag compounds and ions have been used for both hygienic and healing purposes, due to their strong bactericidal effects as well as a broad spectrum of antimicrobial activity. 7,8 Nevertheless, topical application of Ag compounds (silver nitrate (AgNO3) and silver sulfadiazine) may cause cosmetic abnormality, for example, argyria (blue grey colouration) upon prolonged use or could arrest the healing process owing to fibroblast and epithelial cell toxicity. 9 The use of the Ag compounds and ions was suppressed with the advent of antibiotics and other disinfectants. 10,11 However, interest of Ag in medicine has re-emerged with the increased bacterial resistance to antibiotics and the production Ag in the form of NPs. Owing to their strong antimicrobial activity, AgNPs are used in surgical instruments and bone prostheses coated or embedded with AgNPs. 12 Further application of AgNPs is in dermatology in the treatment of wounds and burns. 13,14 With the increasing interest in the use of AgNPs in medicine, concerns about the potential toxicity of AgNPs also increase. Hence toxic effects on human health have been studied in vitro 15 –18 and in vivo. 15,19,20 Many studies have identified some degree of NPs toxicity.
Due to the small size, NPs have higher mobility in the body compared to macro-/micro-particles. In vivo studies have shown the presence of AgNPs in various organs (e.g. brain, skin, lung, kidney, liver and testis). 19,21 Several in vitro studies further reported the ability of AgNPs to enter cells. 22 –25 However, the mode by which NPs enter cells has not been explained yet. Similarly, the mechanism of NPs, particularly AgNPs, and their toxic effects remain unclear. Several in vitro studies suggest that AgNPs cytotoxicity is connected with their effect on cellular energy metabolism. AgNPs induce mitochondrial dysfunction via interruption of mitochondrial membrane permeability and adenosine triphosphate synthesis. 22,26,27 AgNPs further stimulate generation of reactive oxygen species (ROS). 22,28,29 Excessive ROS generation results in oxidative stress and subsequent oxidative damage to biomolecules including lipids 30 and DNA as well as an inflammatory response. Recent studies have reported production of DNA single-strand breaks in AgNPs-exposed cells. 20,22,30,31 Carlson et al. 28 and Park et al. 16 have further shown increased levels of pro-inflammatory cytokines (interleukin-1β (IL-1β), IL-6 and tumour necrosis factor α (TNF-α)), Th1- and Th2-type cytokines (IL-4, IL-5, IL-10, IL-12 and interferon-γ) and transforming growth factor β after AgNPs treatment in vitro or in vivo. 16
The goal of the present work was to evaluate the potential toxicity of AgNPs and ionic silver (Ag-I) using normal human dermal fibroblasts (NHDF) and normal human epidermal keratinocytes (NHEK) and define a range of AgNPs and Ag-I concentration for the intended application as a topical agent for wound healing. Besides, influence of AgNPs and Ag-I on cell viability, the inflammatory response and DNA damage in AgNPs and Ag-I–treated cells were examined.
Materials and Methods
Chemicals
AgNPs and AgNO3 (Ag-I) were provided by NanoTrade s.r.o. (Czech Republic). Western blotting Luminol Reagent, cyclooxygenase 2 (COX-2) rabbit polyclonal antibody, IL-6 rabbit polyclonal antibody, actin (1–19) goat polyclonal antibody, horseradish peroxidase-conjugated goat anti-rabbit and rabbit anti-goat antibodies were received from Santa Cruz Biotechnology (Santa Cruz, California, USA). Protease inhibitor cocktail tablets (Complete™) were purchased from Roche (Mannheim, Germany). Keratinocyte basal medium-2 and KGM™-2 SingleQuots™ were purchased from East port Prague s.r.o. (Czech Republic). EpiLife® medium and human keratinocyte growth supplement kit were received from Life Technologies s.r.o. (Czech Republic). Dulbecco’s modified Eagle’s medium, Ham-F12 nutrient mixture, stabilized penicillin–streptomycin solution, amphotericin B, hydrocortisone, adenine, insulin, epidermal growth factor, 3,3′,5 triiod-
Synthesis and characterization of AgNPs
AgNPs were provided by NanoTrade s.r.o. The particle was synthesized by chemically reducing AgNO3 with sodium borohydride (NaBH4) as referred in Czech patent (CZ 304160 B6). 32 The prepared AgNPs were sonicated in a bath sonicator for 30 min prior to use. The size of prepared AgNPs was lower than 20 nm (Figure 1(a) and (b)). The ultraviolet–visible spectrum was collected on a ultraviolet–visible spectrophotometer (Helios Alpha, Thermo Electron Corporation, UK); Figure 1(c) and (d).

Characterization of AgNPs. Panel (a) represents typical TEM image of AgNPs captured with the TEM JEOL 2010. Scale bar represent 100 nm. Panel (b) shows the size distribution of AgNPs measured with Image J software. Panel (c) shows UV-Vis absorption spectra of AgNPs diluted in water and panel (d) shows UV-Vis absorption spectra of AgNPs diluted in serum-free medium. AgNPs: silver nanoparticle; TEM: transmission electron microscope; UV-Vis: ultraviolet–visible.
The primary particle size and morphology of AgNPs were examined using a transmission electron microscope (TEM; JEOL 2010, JEOL Ltd, Japan) at an accelerating voltage of 160 kV. An aqueous solution of AgNPs was sonicated (30 min) to reduce agglomeration, 5 µl of NPs suspension was then applied onto Formvar carbon-coated copper grid which was then dried. Image J software was used to measure the nanoparticle size. Particles were approximately spherical, and thereafter, the diameter measurements were performed at random. The mean and standard deviation (SD) of particle sizes were calculated from measuring over 700 individual NPs in random fields of view in addition to the images showing the general morphology of the NPs. 33 Zeta potential was also measured to determine the amount of aggregation of particles.
Cell culture and characterization
NHDF and NHEK were cultivated from skin of healthy donors (see supplementary document). The tissue specimens were obtained from patients undergoing plastic surgery at the Department of Plastic and Aesthetic Surgery (University Hospital Olomouc). The tissue acquisition protocol adhered to the requirements of the local ethics committee. All patients had signed written informed consent.
The isolated primary cell cultures (NHDF and NHEK) were characterized by the morphology and immunocytochemically according to their phenotype at the Department of Histology and Embryology, Faculty of Medicine and Dentistry, Palacký University Olomouc (see supplementary document).
Treatment of cells
For evaluation of AgNPs and Ag-I effects on cell viability, inflammatory response, and DNA damage, NHDF and NHEK were treated with AgNPs and Ag-I in serum-free medium that was applied for 8 or 24 h. Then the media were collected and immediately frozen (−80°C) for a measurement of inflammatory markers particularly IL-6, IL-8, TNF-α, basic fibroblast growth factor (FGF basic) and granulocyte–macrophage colony-stimulating factor (GM-CSF). Cells were washed with phosphate-buffered saline (PBS), if necessary, further processed and then toxicity, DNA damage or expression of COX-2 and IL-6 were evaluated.
Cell viability
Reduction of tetrazolium salt, MTT, was used as a parameter for cytotoxicity assessment. MTT is reduced by intracellular dehydrogenases of viable living cells that lead to the formation of purple formazan crystals, insoluble in aqueous solutions. After dissolution in organic solvent, the absorbance is monitored. 34
The cells on 96-well microplates were treated with AgNPs (0–33 µg/mL) and Ag-I (0–33 µg/mL) in serum-free medium for 24 h. Control cells were treated with a serum-free medium without AgNPs or Ag-I. After the incubation period, the medium was removed, and serum-free medium supplemented with MTT (5 mg/mL) was applied to the cells for 2 h (37°C, dark). The solution was then removed and the crystals were dissolved in dimethyl sulfoxide with ammonia (1%, v/v). Afterwards, the solubilization solution (100 µl) from each well was transferred onto a new 96-well plate, and the absorbance was read from the bottom of the well to ensure that residual AgNPs or Ag-I on the bottom of the wells did not interfere with the absorbance. The absorbance was measured at a test wavelength 540 nm and a reference wavelength of 690 nm (Sunrise, Tecan, Switzerland). 35
The cytotoxicity of AgNPs/Ag-I can be evaluated using the following equation:
Genotoxicity
DNA strand breaks were used for genotoxicity evaluation. The alkaline version of single-cell gel electrophoresis (comet assay) was used. The NHDF on 24-well plates were treated with AgNPs (1, 10, and 25 µg/mL) and Ag-I (0.1, 0.5, and 1 µg/mL) in serum-free medium for 24 h. After the incubation period, cells were washed with PBS, harvested, and processed as described previously. 36
The extent of DNA damage was analysed after staining by ethidium bromide on an Olympus BX 70 fluorescence microscope (Tokyo, Japan). The lesions were assessed at least in 100 nuclei/slide area by visual scoring from 0 (undamaged, no discernible comet tail) to 4 (almost all DNA in tail, insignificant head) using the standard breakage scale (Figure 2). Total DNA damage was calculated: number of cells in class 0 × 0 + number of cells in class 1 × 1 + number of cells in class 2 × 2 + number of cells in class 3 × 3 + number of cells in class 4 × 4. The maximal damage was given a value of 400.

Comet examples for visual scoring of DNA damage.
Cytokine determination
Production of inflammatory markers particularly IL-6, IL-8, TNF-α, FGF basic and GM-CSF was measured in medium collected from NHDF treated with AgNPs (0.25 and 25 µg/mL) and Ag-I (0.025 and 0.25 µg/mL) after 8 and 24 h. For determination, the Bio-Plex Human Cytokines enzyme-linked immunosorbent assay (ELISA) kit and Bio-Plex 200 System (Bio-Rad, Hercules, California, USA) were used according to the manufacturer’s manual. The system allowed simultaneous measurement of all five cytokines in one sample.
Negative control cells were treated with a serum-free medium without AgNPs and Ag-I. Positive control cells were treated with a LPS (10 µg/mL) in serum-free medium, as LPS is a well-known inflammatory agent. 37
Protein expression
The amount of COX-2 and IL-6 protein was evaluated using Western blot analysis. NHDF were treated with AgNPs (0.25 and 25 µg/mL) and Ag-I (0.025 and 0.25 µg/mL) in serum-free medium for 24 h. Fibroblasts were then washed properly with cold PBS and scraped into the ice-cold lysis buffer (20 mM Tris, 5 mM ethylene glycol tetra acetic acid, 150 mM sodium chloride, 20 mM glycerol phosphate, 1 mM sodium fluoride, 1% Triton X-100, 1 mM sodium orthovanadate, 0.1% Tween 20, and protease inhibitor cocktail tablet). After incubation (15 min, 4°C), the lysate was cleared by centrifugation (14,000 r/min, 10 min, 4°C). The supernatant protein concentration was determined by Bradford assay. Proteins were separated by 12.5% sodium dodecyl sulphate–polyacrylamide gel electrophoresis and transferred onto a PVDF membrane. Residual binding sites on the membrane were blocked using blocking buffer (5% non-fat dry milk (w/v) in 100 mM Tris-buffered saline (pH 7.5) with Tween 20 (0.05%, v/v) for 1 h at room temperature). The membrane was then incubated with a polyclonal primary antibody (rabbit anti-IL-6, goat anti-COX-2 or goat anti-actin (1–19), overnight at 4°C) and with a secondary horseradish peroxidase-conjugated antibody (goat anti-rabbit or rabbit anti-goat for 2 h at room temperature). COX-2, IL-6 and actin expression was detected by chemiluminiscence using Western Blotting Luminol Reagent and autoradiography using a Kodak BioMax light film. The quantification of proteins was performed by densitometric analysis using ElfoMan 2.6 software.
Statistical analysis
Data are expressed as means ± SD of four independent experiments performed in triplicate for each sample. The Student’s t-test was used for statistical analysis. Statistical significance was determined at p = 0.05. Western blot data are representatives of three independent experiments.
Results
Synthesis and characterization of AgNPs
AgNPs were synthesized chemically by reducing AgNO3 with NaBH4. The use of strong reducing agent like NaBH4 enables to produce AgNPs of size lower than 40 nm. The AgNPs were characterized before using them further. TEM analysis was used for the determination of accurate size, shape and state of dispersion. The TEM analysis revealed that AgNPs (Figure 1(a)) monodispersed with spherical morphology. The size distribution analysis showed that the 50% of the AgNPs occurring between 5 and 40 nm in size, and the overall average size matches the 10.43 ± 4.74 nm (Figure 1(b)). Zeta potential value was −22 mV (in water) and −14.6 mV (in serum-free medium). Spectrophotometrical analysis showed that the AgNPs diluted in water and serum-free medium have absorption maximum at 388 nm and 557 nm, respectively (Figure 1(c) and (d)).
Effect of AgNPs on cell viability
Toxicity is the most crucial problem for metal-based NPs in medical applications. Therefore, we evaluated the toxic effect of AgNPs and Ag-I on two cell cultures (NHDF and NHEK) after 24 h exposure. The MTT results revealed that the concentrations of Ag-I above 10 µg/mL were extremely toxic to both NHDF (Figure 3(a)) and NHEK (Figure 4(a)). The IC50 values (concentration yielding 50% growth inhibition) were 3.1 ± 0.3 µg/mL and 5.8 ± 0.8 µg/mL for the Ag-I on NHDF and NHEK, respectively. In contrast, AgNPs did not decrease cell viability in the chosen concentration range in NHDF (Figure 3(a)) and NHEK (Figure 4(a)). Similar results were obtained in morphologic observations (Figure 3(b) to (d) and Figure 4(b) to (d)).

Effects of AgNPs and Ag-I on NHDF viability (a) determined by the MTT assay after 24 h treatment. The viability percentage was normalized to the untreated cells (without AgNPs or Ag-I). The values represent the mean ± SD of four sets of independent experiments. *p ≤ 0.05: significant difference from control. Morphological changes were observed after vehicle (b), AgNPs (10 µg/mL; (c)) and Ag-I (10 µg/mL; (d)). AgNPs: silver nanoparticles; Ag-I: ionic silver; NHDF: normal human dermal fibroblasts; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
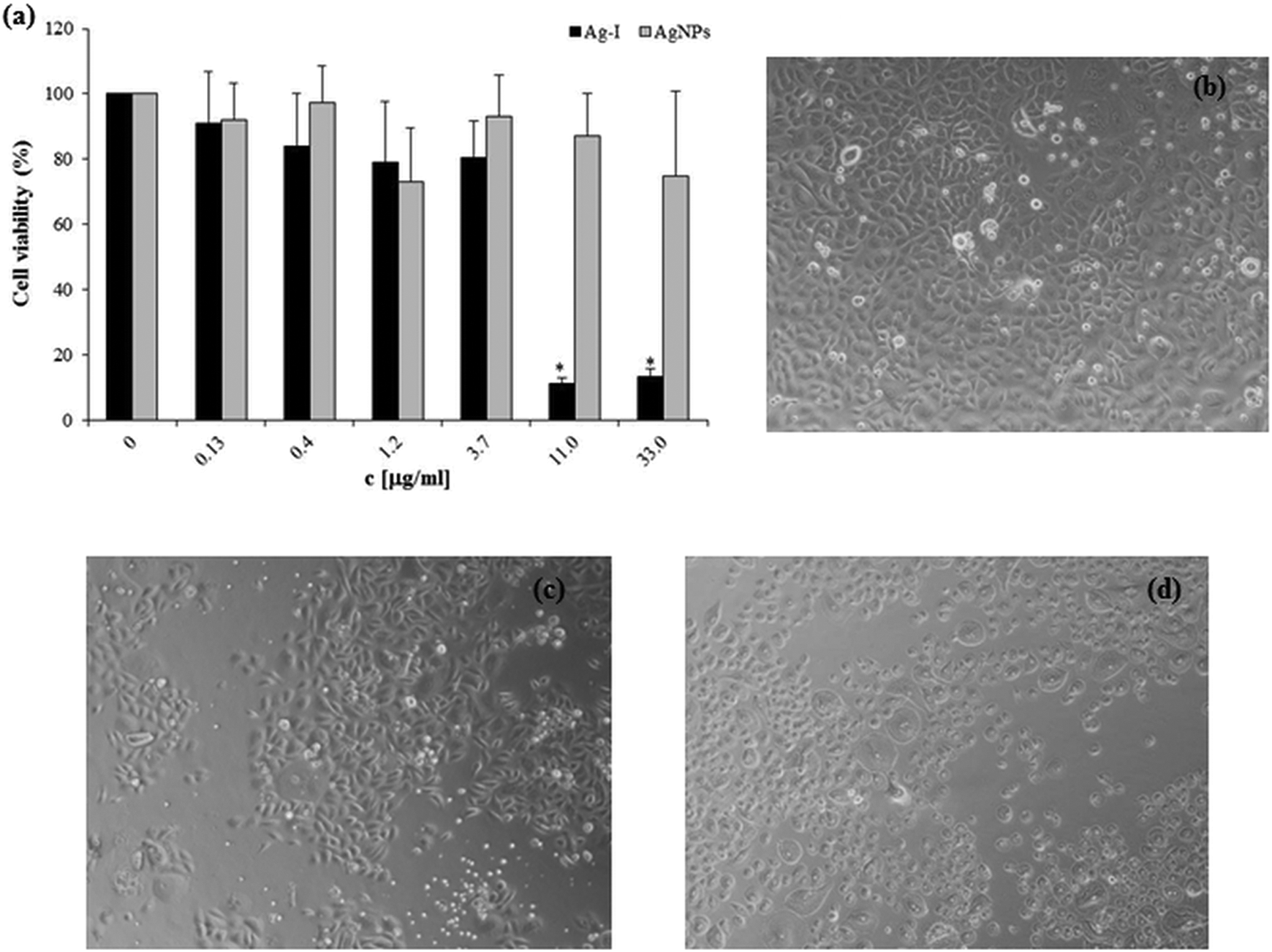
Effects of AgNPs and Ag-I on NHEK viability (a) determined by the MTT assay after 24 h treatment. The viability percentage was normalized to the untreated cells (without AgNPs or Ag-I). The values represent the mean ± SD of four sets of independent experiments. *p ≤ 0.05: significant difference from control. Morphological changes were observed after vehicle (b), AgNPs (10 µg/mL; (c)) and Ag-I (11 µg/mL; (d)). AgNPs: silver nanoparticles; Ag-I: ionic silver; NHEK: normal human epidermal keratinocytes; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; SD: standard deviation.
Based on these results, we selected non-toxic concentration of AgNPs (0.25–25 µg/mL) and Ag-I (0.025–1 µg/mL) for further biochemical studies. Although these concentrations did not affect cell viability, we would like to verify whether they cause (or not) modification of DNA and cytokines level.
Effect of AgNPs on DNA strand breaks formation
Earlier studies have shown that AgNPs induce DNA damage either directly through physical attack, indirectly by triggering the production of ROS or by a combination of both factors. 18,22,38 To test whether non-toxic concentrations of AgNPs and Ag-I might affect DNA, NHDF were treated with AgNPs (1, 10 and 25 µg/mL) and Ag-I (0.1, 0.5 and 1 µg/mL) and the strand breaks of DNA were evaluated. Our results showed that neither AgNPs nor Ag-I induced DNA strand breaks formation in all tested concentrations after 24 h (Table 1).
Effect of AgNPs and Ag-I on DNA strand breaks formation in NHDF.a

AgNPs: silver nanoparticles; Ag-I: ionic silver; NHDF: normal human dermal fibroblasts; SD: standard deviation.
aToxicity scale (number of damaged nuclei in individual comet class) represents the mean of four sets of independent experiments. The values of total damage represent the mean ± SD of four sets of independent experiments. No significant difference was found in total DNA damage between the controls (0 µg/mL) and AgNPs (1–25 µg/mL) and Ag-I (0.1–1 µg/mL), respectively.
Effect of AgNPs on inflammatory markers
Media collected from NHDF exposed to AgNPs (0.25 and 25 µg/mL) and Ag-I (0.025 and 0.25 µg/mL) for 8 and 24 h were used for cytokine level determination by ELISA assay. The results showed that of the five chosen cytokines, only IL-6 level was changed after AgNPs and Ag-I treatment. Non-toxic concentrations of AgNPs (25 µg/mL) and Ag-I (0.025 and 0.25 µg/mL) increased the production of IL-6 (Figure 5(a)) after 8 h treatment. The level was normalized within 24 h. IL-8, TNF-α, FGF basic and GM-CSF levels were not affected by either AgNPs or Ag-I (Figure 5(b) to (e)) after 8 and 24 h.

Effects of AgNPs and Ag-I on production of inflammatory markers in NHDF. Cells were treated with AgNPs (0.25 and 25 µg/mL) and Ag-I (0.025 and 0.25 µg/mL) for 8 and 24 h. As the positive control, cells were treated with LPS (10 µg/mL) for 8 and 24 h. As negative control, cells were treated with medium without AgNPs or Ag-I for 8 and 24 h. The level of IL-6 (a), IL-8 (b), TNF-α (c), FGF basic (d) and GM-CSF (e) in supernatants were determined by ELISA analysis. *p ≤ 0.05: significant difference from control. AgNPs: silver nanoparticles; Ag-I: ionic silver; NHDF: normal human dermal fibroblasts; LPS: lipopolysaccharide; IL: interleukin; TNF: tumour necrosis factor; FGF: fibroblast growth factor; ELISA: enzyme-linked immunosorbent assay.
Western blot analysis also revealed that AgNPs (0.25 and 25 µg/mL) and Ag-I (0.025 and 0.25 µg/mL) induced increase in IL-6 protein expression after 8 and 24 h (Figure 6(a) and (c)). No effect was found on COX-2 expression in AgNPs and Ag-I-treated cells after 8 and 24 h as shown in Figure 6(b) and (d).

Effects of AgNPs and Ag-I on COX-2 and IL-6 protein expression in NHDF. Cells were treated with AgNPs (0.25 and 25 µg/mL) and Ag-I (0.025 and 0.25 µg/mL) for 8 and 24 h. As the positive control, cells were treated with LPS (10 µg/mL) for 8 and 24 h. As negative control, cells were treated with medium without AgNPs or Ag-I for 8 and 24 h. Panel (a) and (b) show effects on the proteins expression. Panel (c) and (d) represent the quantification of proteins performed by densitometric analysis using ElfoMan 2.6 software. Data are representatives of three independent experiments. *p ≤ 0.05: significant difference from negative control . AgNPs: silver nanoparticles; Ag-I: ionic silver; COX-2: cyclooxygenase-2; IL: interleukin; NHDF: normal human dermal fibroblasts; LPS: lipopolysaccharide.
Discussion
AgNPs are toxic to a wide range of microorganisms and for this reason they are a promising antimicrobial agent. At the present time, many new products, utilized for the treatment of burns and chronic wound, containing AgNPs are developed. However, some studies also demonstrated that AgNPs may induce significant cytotoxicity in vitro and in vivo. The present study aimed to investigate the potential toxicity of AgNPs and Ag-I using primary skin cells (NHDF and NHEK) and define a range of AgNPs and Ag-I concentration for the intended application as a topical agent for wound healing.
In our study, the cytotoxicity of AgNPs was compared to that of various corresponding concentrations of Ag-I on two types of skin cells (Figure 3 and 4). We found a dramatic difference in cytotoxicity of AgNPs and Ag-I. While AgNPs were non-toxic in the tested range, Ag-I induced significant decrease in NHDF and NHEK viability with the IC50 values 3.1 µg/mL and 5.8 µg/mL, respectively. Comparably, Sur et al. did not found toxic effect of AgNPs on NHDF up to a concentration of 50 µg/mL. 31 In other study, Hidalgo and Domínguez showed cytotoxicity of Ag-I on NHDF lower than 8.2 µmol/L. 39 Samberg et al. compared the toxicity of unwashed, washed and carbon-coated AgNPs (20, 50 and 80 nm) on NHEK. They found that 20 nm and 50 nm unwashed AgNPs decreased viability at 1.7 µg/mL and 80 nm AgNPs at 0.34 µg/mL. Washed (20, 50 and 80 nm) and carbon-coated (20 and 35 nm) AgNPs did not affect NHEK viability. 40 Burd et al. tested the cytotoxicity of five commercially available Ag-based dressings that contained different amounts of AgNPs (Acticoat™ (934 µg/cm2), Aquacel® Ag (21 µg/cm2), Contreet® Foam (12 µg/cm2), PolyMem® Ag (139 µg/cm2) and Urgotul®SSD (85 µg/cm2)) in NHDF and NHEK. They observed that Acticoat™, Aquacel® Ag, and Contreet® Foam which release 12, 13 and 14 µg/mL of Ag in the culture medium were much more toxic than PolyMem® Ag and Urgotul®SSD, which release 4 and 3 µg/mL of Ag in the culture medium. 41 Studies using non-skin cells such as HepG2 23 and RAW 264.7 16 described approximately the same sensitivity of cells to AgNPs and Ag-I. Furthermore, some studies suggest that the cytotoxic effects of AgNPs compared to Ag-I may relate to the size of the NPs. Liu et al. reported that 5 nm AgNPs were more toxic than Ag-I, but 20 nm and 50 nm were less toxic than Ag-I. 42 Similarly, Piao et al. noted that 5–10 nm AgNPs showed two times higher toxicity than Ag-I. 30 Here, we showed that 10 nm AgNPs were less toxic than Ag-I. The discrepancy may be related to the fact that we used a mixture of AgNPs in a range 0–40 nm (Figure 1). These findings suggest that the toxicity of AgNPs and Ag-I depends on the cell type and also on NPs size.
Besides cytotoxicity, AgNPs were reported to cause DNA single-strand breaks. Asharani et al. studied the genotoxicity of AgNPs (6–20 nm) on normal human lung fibroblasts and human glioblastoma (U251) cells after 48 h treatment. They observed an increase in DNA damage with increasing AgNPs concentration up to 400 µg/mL in cancer U251 cells. In the lung fibroblasts, DNA damage increased up to 100 µg/mL and at higher concentrations of AgNPs the damage remained same. 22 Ghosh et al. assessed the DNA damage caused by AgNPs (70–125 nm) on human lymphocytes. The greatest damage was found at a concentration of 25 µg/mL after 3 h, whilst at higher concentrations of AgNPs (50–150 µg/mL) DNA damage decreased. 20 Piao et al. also found the formation of DNA strand breaks in human liver cells by AgNPs (5–10 nm; 4 µg/mL; 3 h). 30 Although our experimental conditions were nearly comparable to those of the above-mentioned studies, we found no DNA strand break production either in AgNPs or in Ag-I–treated cells.
Several studies have also demonstrated that AgNPs induce the release of a number of pro-inflammatory markers in various cell types. 16 –18,29 Asharani et al. reported a threefold increase in IL-6 level, eightfold increase in IL-8 level and moderate increase in GM-CSF in normal human lung fibroblasts. 18 Increased levels of IL-6 and IL-10 were also observed using THP-1 and RAW 264.7 macrophages. 16,17 Moreover, AgNPs of various sizes (15, 30 and 50 nm) stimulated the secretion of cytokines in alveolar macrophages, particularly TNF-α, IL-1β and IL 10. 16,28 In our study, we found moderate increase in IL-6 level in NHDF exposed to AgNPs and Ag-I (Figure 5(a)). However, we observed no changes in the levels of other markers, IL-8, TNF-α, FGF basic or GM-CSF (Figure 5(b) to (e)). Even though the size of the AgNPs was nearly the same, Asharani et al. used a 16-fold higher concentration of AgNPs (400 µg/mL) compared to our study. 18 On the other hand, some studies have described the anti-inflammatory effects of AgNPs. 43 –45 The discrepancy between our results and the results of the above studies could be explained by the AgNPs concentrations used. Moreover, it should be noted that the size, type and source of the AgNPs, manufacturers’ synthesis method, type of stabilizer used during the manufacturing process, residues within the AgNPs and also the cell type may influence the response.
Conclusion
Our study showed that Ag-Is were significantly more toxic than AgNPs on NHDF and NHEK. In addition, non-cytotoxic concentrations of AgNPs and Ag-I did not induce DNA strand breaks and did not affect inflammatory markers, except for transient increase in IL-6 levels in AgNPs and Ag-I–treated NHDF. AgNPs seem to be more suitable for the intended application as a topical agent for wound healing up to the concentration 25 µg/mL though further investigation is necessary to exclude any possible adverse effect such as evaluation of safety after topical application and skin absorption of AgNPs using human skin explants.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant from Palacký University (IGA_LF_2014_014; NA, AG), by the National Programme for Sustainability I (LO1304; AG, JU), by the National Programme for Sustainability LO1305; MS) and by Operational Program Education for Competitiveness – European Social Fund (CZ.1.07/2.3.00/30.0004; VP). We thank the Mgr. Kateřina Čížková (Faculty of Medicine and Dentistry, Palacký University) for the characterization of primary cell cultures (NHDF and NHEK).

