Abstract
This study was performed to determine the effect of low-level exposure to a mixture of bisphenol A (BPA) and isobutylparaben (IBP) on male reproduction. Corn oil, BPA (0.05 mg/kg/day), IBP (2.5 mg/kg/day), and a BPA/IBP mixture (BPA 0.05 mg/kg/day and IBP 2.5 mg/kg/day) were administered once daily by oral gavage to female rats for 5 weeks from gestation day 6 to lactation day 21. Male pups were killed at postnatal day 70 and examined for developmental characteristics, body weight, testis and epididymis weight, steroid hormones, epididymal sperm count and motility, and histological changes in testis and epididymis. The BPA/IBP mixture produced a significant downregulation of epididymal sperm count and motility. BPA or IBP alone also reduced epididymal sperm count and motility compared to control. These results indicate that exposure to low-level BPA/IBP mixture, which showed no notable physiological response in early life stages, can decrease semen quality in adulthood.
Introduction
Increasing incidence of male endocrine-related disorders may be due to increase exposure to environmental estrogens, which has become a major concern. 1 –4 In particular, the early stages of development are especially vulnerable to perturbation by chemicals with estrogen hormone-like activity that cause permanent alterations, including male reproductive disorders. 5 –8 These findings support the concept of the “fetal origins of adult disease,” whereby exposure to environmental estrogens during critical periods of development can result in permanent changes that can manifest even in late adulthood. 9 The need to evaluate the potential health effects of exposure to mixtures of endocrine disruptors has increased as a result of humans being simultaneously exposed to larger numbers of diverse chemicals. 10 Indeed, in vitro 11,12 and in vivo 13 studies have shown considerable evidence of the combinatorial effects of exposure to low doses of environmental estrogens. These effects can occur even when individual components are present at levels below their observable effects. Thus, this study considered the effects of bisphenol A (BPA) and isobutylparaben (IBP) mixtures below their lowest observed adverse effect level (LOAEL).
BPA is widely used in the production of polycarbonate plastics and epoxy resins in food cans, food and beverage containers, and dental sealants 14 and shows 10,000–100,000 times lower estrogenic potency than 17β-estradiol in relative binding affinity serum-modified access assays. 15 The LOAEL of BPA has been identified to be 50 mg/kg/day, and a 1,000-fold lower dose (0.05 mg/kg/day) is defined as a reference dose by the Environmental Protection Agency. 16 BPA doses lower than the LOAEL have been shown to cause adverse effects in reproductive capacity including a decrease in epididymal sperm, 8 androgen receptor expression in the testis, 17 and steroidogenic enzyme gene expression in Leydig cells. 5 Parabens are used as preservatives in personal care products and cosmetics 18 and were once considered safe due to their low potential for toxicity 19 –21 and their relatively fast rate of metabolism. 22,23 However, there are now growing concerns about the estrogenic 19 and antiandrogenic properties 24,25 of parabens. The affinities of estrogen receptors for IBP are approximately 1,000-fold weaker than for 17β-estradiol. 26,27 The subcutaneous exposure of three daily doses of 1.2 mg IBP (approximately 72 mg/kg/day) increased the wet uterine weight in immature mice. 26 The oral administration of IBP 62.5 mg/kg/day in juvenile female rats induced histological abnormalities in the ovaries and uterus. 27 An adequate LOAEL of IBP has not yet been determined, 28 and the estrogenic effects of IBP have only been studied in female rodents. 26,27 Some parabens such as butylparaben have shown adverse effects on the male reproductive system. 6,24,25,29,30 Thus, the IBP concentration (2.5 mg/kg/day) in this study was determined based on the lowest dose of butylparaben (2 mg/kg/day). 30 Finally, individual concentrations of BPA (0.05 mg/kg/day) and IBP (2.5 mg/kg/day) were selected to examine their effects on the male reproductive system after exposure at an early life stage.
The additive or synergistic effects of BPA/IBP mixtures have been reported in GH3 rat pituitary cells by quantifying the expression of calbindin-D9K 31,32 and in algae by assessing the inhibition of spore release. 33 These in vitro studies have suggested potential adverse effects of BPA/IBP mixtures, however, they could not assess the development of adult onset disease. Animal studies are needed to better understand the biological activities of these compounds in humans.
Therefore, this study was performed to investigate reproductive changes in adult rats after exposure to a low-dose mixture of BPA and IBP in the early stages of life.
Materials and methods
Chemicals
BPA (purity ≥ 99%) and corn oil were purchased from Sigma Chemical Company (St. Louis, Missouri, USA), and IBP (purity ≥ 99%) was obtained from Tokyo Kasei Kogyo Co. LTD (Tokyo, Japan). The dosing solutions were prepared by dissolving BPA and IBP in corn oil.
Animals
This study was performed in accordance with the Good Laboratory Practice guidelines for Animal Experiments from the Korea Testing and Research Institute. Eight-week-old specific pathogen-free Sprague-Dawley rats were purchased from the Charles River Breeding Laboratory (Gyeonggi-do, Korea). These rats were maintained in a controlled environment (22 ± 2°C; lights on from 06:00 to 20:00 h) and had free access to pellet laboratory chow (Purina Korea Inc., Gyeonggi-do, Korea) and water purified by ultraviolet and reverse osmosis. The concentration of phytoestrogens in the diet was not evaluated, however, because food intake was equivalent for the control and experimental rats, all animals were exposed to the same levels of food-borne phytoestrogens. To minimize additional exposures to xenoestrogens, rats were housed in stainless steel cages with wood bedding, and purified water was supplied ad libitum in glass bottles with rubber stoppers surrounded by a steel ring.
Experimental design
A total of nine pregnant rats were randomly divided into four groups (n = 3/group), consisting of vehicle (corn oil), BPA (0.05 mg/kg/day), IBP (2.5 mg/kg/day), and a mixture of BPA and IBP (0.05 and 2.5 mg/kg/day, respectively). Corn oil, BPA, IBP, and the BPA/IBP mixture were administered once daily by oral gavage to the dam from gestation day (GD) 6 to postnatal day (PND) 21. BPA and IBP can enter the human body via ingestion, inhalation, and dermal absorption due to its wide use in a variety of products. 14,18 Although subcutaneous injection could induce more severe effects in laboratory animals than the same doses administered orally, this study selected the oral route because humans are mainly exposed to these chemicals by consuming foods. The day of birth was considered PND 0, and offspring were weaned on PND 21. After weaning, male pups received a regular chow diet.
After delivery, we recorded the number of pups, sex ratio, and birth weight in each litter. The number of male pups in each group was 12 (control), 13(BPA-only), 12 (IBP-only), and 11 (the BPA/IBP mixture) on PND 0. It was adjusted as eight male pups per group on PND 4 to reduce the litter size-related variability (n = 8/group). Male pups were killed using isophorone on PND 70. To minimize the effects of diurnal variation on hormone levels, male pups were selected one order per each group from 9:00 am to 12:00 pm (noon). Blood was taken from the aorta for hormone assays, and organs (the testis and epididymis) were removed and weighed. The caudal part of the epididymis was dissected for analysis of sperm count and motility. The other parts of testis and epididymis were preserved in Bouin’s solution for histological observations.
Evaluation of external physical signs and body weight
Each male pup was weighed and underwent a detailed physical examination on PND 1, 4, 7, 14, and 21, and then weekly thereafter. Clinical signs including weight-adjusted anogenital distance (wAGD, AGD/body weight (BW)(1/3) in mm/g(1/3)), pinna detachment, incisor eruption, thoracic nipple retention, eye opening, testis descent, and preputial separation were measured as previously described. 34
Histological analysis of the testis and epididymis
The testis and epididymis (caudal region) were preserved in Bouin’s solution for approximately 24 h, and later washed several times with ethanol (70%) before being embedded in paraffin. Embedded tissues were sectioned at 5 μm and tissue sections were stained with hematoxylin–eosin (H&E) for light microscopy (Olympus, Tokyo, Japan). The size and shape of the seminiferous tubules and the progression of spermatogenesis were observed in the cross sections of the testis. The thickness of the epithelium and spermatozoa in the epididymal tubule lumen were also examined in the epididymis (caudal region). The photographs in the present study were taken using a Fuji Digital Camera HC 300 Z/CL (Olympus, Tokyo, Japan).
Hormone assays
Collected blood was separated by centrifugation at 3000 r/min for 15 min at 4°C. Serum was distributed into sterile Cryo Vials (Greiner Labortechnik, Frickenhausen, Germany) in volumes of 500 µL and was immediately frozen at −80°C until analysis. Serum hormone levels were measured using a commercial IBL enzyme-linked immunosorbent assay (ELISA) kit (Germany). Serum luteinizing hormone (LH) levels and follicle-stimulating hormone (FSH) levels were measured using a rat LH ELISA kit (LH Detect®, INRA Tours, Nouzilly, France) and a rat FSH ELISA kit (Biocode-Hycel, Liege, Belgium), respectively. Serum testosterone and 17β-estradiol levels were measured using a rat testosterone and 17β-estradiol ELISA kit (Endocrine Technologies Inc., Newark, California, USA). All hormones were measured according to the manufacturer’s protocols. The absorbance was measured with a plate reader (Tecan Sunrise TW, Salzburg, Austria).
Sperm number and motility in epididymis
Five male rats per group were used to analyze the epididymal sperm number and motility. The caudal part of the epididymis was dissected (5 mm), and tubules were dispersed into 5 mL Hank’s balanced salt solution (Sigma, Yong-In, Korea) in a plastic petri dish. The petri dish was kept in the incubator at 37°C in 5% carbon dioxide for 10 min to allow the sperm to disperse. One milliliter aliquots of sperm suspension solution were mixed in 2 mL of Hank’s balanced salt solution, collected and loaded in an 80-μm glass chamber, and analyzed using an IVOS automated semen analyzer (Hamilton Thorne Biosciences, Beverly, Massachusetts, USA). The sperm count was expressed as the per gram weight of caudal epididymis tissue. The motility (%) was calculated from the number of motile sperms divided by the total number of sperm.
Data analysis
Data analysis was performed using STATA (version 10.0, StataCorp LP, College Station, Texas, USA). Statistical differences in the serum hormones, sperm count, and sperm motility between the control and exposed groups were evaluated by the Wilcoxon rank sum test. p Values less than 0.05 were considered to show a significant difference. The generalized extreme studentized deviate test 35 was used to detect outliers in steroid hormone levels, sperm count, and sperm motility. One 17β-estradiol value in the IBP-only group and one sperm count value and sperm motility value in the BPA/IBP mixture group were identified as outliers and excluded from statistical analysis.
Results
The evaluation of external physical signs, body weights, and relative organ weights
The evaluation of body characteristics, including pinna detachment, eruption of the incisors, separation of the eyelids, nipple retention, descent of the testis, and separation of the prepuce in the group treated with the BPA/IBP mixture showed no marked differences as compared to the control, the BPA group, and the IBP group (Table 1). Average body weight (Figure 1(a)) and wAGD (AGD/BW(1/3) in mm/g(1/3); Figure 1(b)) in the BPA/IBP group were similar to those of the control. There was no significant difference in relative testis and epididymis weight (organ weight/body weight × 100) in the BPA/IBP group as compared to the control, BPA group, and IBP group on PND 70 (Figure 1(c)).
Developmental characteristics of male pups in control (n = 8) and treatment groups (BPA, IBP, and mixture; n = 8/group). Data are mean and standard errors.
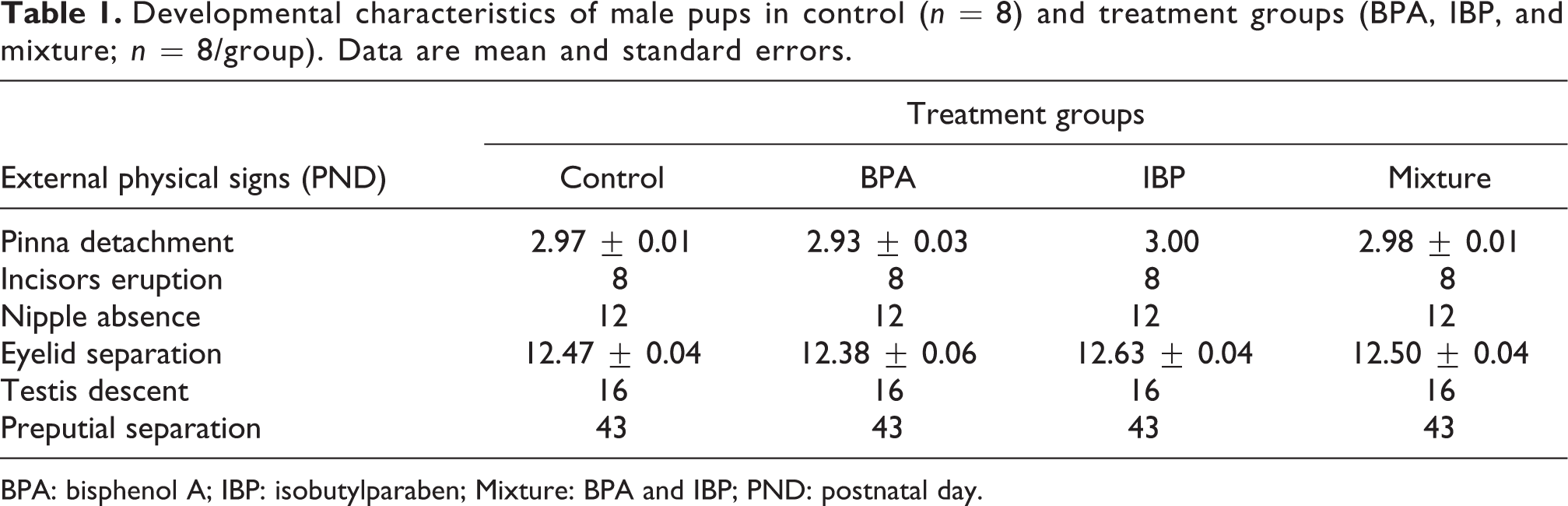
BPA: bisphenol A; IBP: isobutylparaben; Mixture: BPA and IBP; PND: postnatal day.

The changes of (a) body weights, (b) wAGD, and (c) relative testis and epididymis weight (organ weight/body weight × 100) in male rats between control (n = 8) and treatment groups (BPA, IBP, and mixture; n = 8/group). Body weight and anogenital distance (AGD) on PND 0 were observed in 12 (control), 13 (BPA-only), 12 (IBP-only), and 11 (the BPA/IBP mixture) male pups. Body weight, wAGD, and relative organ weights of testis and epididymis did not show statistical difference between control and treatment groups. Values are means and standard errors. BPA: bisphenol A; IBP: isobutylparaben; Mixture: BPA and IBP; wAGD: weight-adjusted anogenital distance; BW: body weight.
Hormone levels in serum
The rats’ exposure to the BPA/IBP mixture showed similar serum FSH, testosterone, and 17β-estradiol levels as compared to the control, BPA-only, and IBP-only groups (Table 2). Serum LH concentrations in the BPA/IBP group had high variability, hence reducing the possibility of detecting statistical significance. In the IBP group, one 17β-estradiol value was excluded because of its extremely high concentration (120.2 pg/mL). IBP treatment alone produced significantly lower 17β-estradiol levels (5.812 pg/mL, p = 0.0053) as compared to the control (7.874 pg/mL). BPA treatment did not result in significant changes in serum hormones as compared to the control.
Comparison of serum hormones (FSH, LH, testosterone, and 17β-estradiol) in male rats between control (n = 8) and treatment groups (BPA, IBP, and mixture; n = 8/group). Hormones were measured using a commercial IBL ELISA kit from 9:00 am to 12:00 pm (noon). Data are mean and standard errors.

BPA: bisphenol A; IBP: isobutylparaben; Mixture: BPA and IBP; FSH: follicle-stimulating hormone; LH: luteinizing hormone.
*p < 0.05 compared with controls.
Sperm number and motility in epididymis
Epididymal sperm number and motility were significantly lower in the group treated with the BPA/IBP mixture relative to the control, BPA, and IBP groups (Figure 2). In particular, one offspring in the BPA/IBP mixture group showed extremely low sperm number (34.0 × 106/g of caudal epididymis) and motility (6.2%). These data were treated as an outlier and were excluded in the statistical analysis. Treatment with the BPA/IBP mixture produced a significant decrease in the average sperm number (192.4 × 106/g of caudal epididymis, p = 0.0209) and sperm motility (60.3%, p = 0.0209) compared to the control (529.8 × 106/g of caudal epididymis and 84.0%, respectively), and, the changes in sperm number and motility were not significantly different from those in the BPA-only and IBP-only groups. Treatment with BPA alone resulted in lower sperm number (227.2 × 106/g of caudal epididymis, p = 0.0209) and motility (68.4%, p = 0.0209) as compared to the control, while treatment with IBP alone also reduced sperm number (244.8 × 106/g of caudal epididymis, p = 0.0433) and motility (61.1%, p = 0.0209) as compared to the control. There was no significant difference in sperm number and motility between the BPA and IBP groups.

Comparison of epididymal (a) sperm count and (b) motility between control (n = 5) and treatment groups in male rats (n = 5/group). The number and motility of epididymal sperm in BPA-, IBP-, and mixture-treated male pups significantly decreased compared to control. Values are means and standard errors. BPA: bisphenol A; IBP: isobutylparaben; Mixture: BPA and IBP; *p < 0.05: compared with controls.
Histological evaluation of testis and epididymis
Histological examination of the testis and epididymis obtained from adult offspring that were exposed to the BPA/IBP mixture during early life stages showed findings similar to that of the control group. The progression of spermatogenesis in the testis was evaluated (Figure 3(a)). The BPA/IBP mixture did not induce adverse changes in the Sertoli cells of the seminiferous tubules and the Leydig cells in the testis as compared to the control group. The epididymal epithelium and spermatozoa in the epididymal tubule lumen were similar between the control and treatment groups (Figure 3(b)).

Histological findings in (a) testis and (b) epididymis (caudal region) in male rats between control (n = 8) and treatment groups (n = 8/group). The testis and epididymis were preserved in Bouin’s solution and then embedded in paraffin. Embedded tissue was sectioned and stained with H&E and examined under a light microscope (×400). No significant changes or gross abnormalities in BPA-, IBP-, and mixture-treated male pups were observed in micromorphology of testis and epididymis. BPA: bisphenol A; IBP: isobutylparaben; Mixture: BPA and IBP.
Discussion
This study was performed to evaluate whether additive or synergistic effects of low-dose BPA and IBP exposure during early life stages could induce reproductive changes in adults. Mixed exposure to low-dose BPA and IBP during early life stages decreased epididymal sperm number and motility in adult male offspring as compared to the control. BPA and IBP also individually decreased epididymal sperm number and motility in adult male offspring as compared to control. Nevertheless, combined exposure to the two chemicals had an even more pronounced effect.
Perinatal exposure to the BPA/IBP mixture at levels below their LOAELs reduced epididymal sperm numbers and motility in male offspring in an additive manner (Figure 2). Furthermore, animals treated with BPA and IBP alone also exhibited significantly decreased epididymal sperm numbers and motility as compared to the control animals in adulthood. Oral ingestion of 1.2 µg/kg/day8 and 2.4 μg/kg/day 5 of BPA during the critical period of development at environmentally relevant doses resulted in the disruption of spermatogenesis and steroidogenesis enzyme gene expression in adulthood. However, exposure to BPA (2, 20, or 200 µg/kg/day) during pregnancy (GD 11–17) 36 and exposure to BPA (20, 100 μg/kg/day) during pregnancy (GD 6–21) 37 did not produce significant adverse effects on reproductive organs. Although BPA had inconsistent results on male reproduction at lower doses, perinatal exposure to BPA at 50 μg/kg/day caused adverse effects on the male reproductive system in adulthood. In addition, perinatal exposure to butylparaben 0.001%, 0.01%, 0.10%, and 1.0% by diet, 24 as well as 200 and 400 mg/kg/day by oral gavage 6,29 decreased sperm count and motility in the epididymis. Similar to these findings, perinatal exposure to bisphenol A diglycidyl ether at 375 mg/kg/day, which has an estrogenic potency 100 times lower then BPA, 38 induced the loss of spermatids in the adult testis and decreased testosterone levels in the blood. 39 The impairment of spermatogenesis might be induced by the disruption of endogenous hormone levels 5,8,40 and results from the changes in the expression of steroidogenic enzymes in the testis after exposure to environmental estrogens. 5,8 Immotile and immature spermatozoa that are released from the testis undergo maturation in the epididymis to become competent for fertilization. 41 Although the epididymis is an androgen-dependent tissue, 42 estrogens and their receptors can regulate the transport of fluid and sperm concentration in the epididymis. 43 This suggests that disruptions of estrogen receptor expression in the epididymis after exposure to environmental estrogens could affect the maturation of spermatozoa through impaired epithelial development and inhibited fluid reabsorption. 43,44 Adult male rats orally exposed to BPA 20 μg/kg/day showed remarkable degenerative histological changes in the epithelium and decreased sperm number in the epididymis. 45 Adult male pups perinatally exposed to butylparaben 400 and 1,000 mg/kg/day showed a reduction in seminiferous tubule layers and significant expansion of the seminiferous tubule lumen. 6 However, morphological changes in the testis and epididymis of adult rats were not seen after exposure to a low dose of BPA/IBP mixture (Figure 3). Similar to our results, subcutaneous exposure to BPA (0.002–97 mg/kg/day) during the neonatal period, 46 and perinatal exposure to BPA 1.2 and 2.4 μg/kg/day did not produce any significant changes in the adult male reproductive system. 8 Although degenerative histological changes were not observed in this study, the disruption in spermatozoa maturation might have resulted from changes in the endogenous hormones. Thus, serum hormones in adult male rats were measured to investigate the effects of low-dose BPA and IBP on sperm maturation.
The levels of endogenous hormones in the BPA/IBP mixture group did not show significant changes as compared to the control, however, treatment with IBP alone decreased serum 17β-estradiol levels relative to the control and the BPA/IBP mixture (Table 2). 17β-estradiol is derived from testosterone following the activity of the aromatase enzyme. The synthesis and secretion of steroid hormones are regulated by gonadotropins (FSH and LH) in the pituitary gland, which are the primary tropic hormones. This constant feedback of hormones might be an important factor in fetal development. Although this study did not show a significant difference in testosterone levels between the control and the BPA/IBP group, serum LH tended to increase in response to exposure to the BPA/IBP mixture. However, due to the small sample size and relatively large variation within groups, we were unable to determine whether the changes in the internal LH levels were related to abnormal function of the epididymis. Previously, the oral administration of BPA 2.4 μg/kg/day during the developmental period was shown to decrease serum LH and testosterone levels and steroidogenesis in Leydig cells. 5 BPA 2 μg/kg/day treatment for 14 days decreased serum testosterone and FSH, and disrupted spermatogenesis in adult rats. 40 Perinatal exposure to butylparaben 400 and 1000 mg/kg/day resulted in an increase in serum LH and a reduction in the epididymal sperm count in adult offspring. 6 Although hormonal changes due to BPA and IBP exposure were not observed in adult male rats, a disruption in the biosynthesis of any of these hormones in early life stages may alter gonadal development and induce permanent impairments, including reduced sperm number and motility.
Physiological changes including body weight and developmental parameters were similar between the control, BPA, IBP, and BPA/IBP groups (Figure 1(a) and 1(b), and Table 1). Changes in body weight and development parameters provide general information about the systemic impact of environmental estrogens beyond reproductive toxicity. Indeed, changes in body weight caused by environmental chemical exposure might be a result of estrogenic effects, as estrogens are associated with food consumption and energy expenditure. Nevertheless, changes in body weight tend to receive less attention than weight loss because developmental delays in the fetus and in neonates might be due to a reduction in body weight. A decrease in expected body weight gain and shortened AGD on PND 1 and PND 21 were observed following perinatal exposure to butylparaben 400 and 1,000 mg/kg/day in male offspring. 6 In addition, exposure to BPA 1.2 μg/kg/day8 and BPA 100 μg/kg/day 47 during the perinatal period increased body weight as compared to control treatment. Thus, changes in body weight may represent the impact of these chemicals on normal development after exposure to environmental estrogens. However, previous studies reported no dose-related body weight changes following a 3-week exposure to IBP 62.5, 250, and 1,000 mg/kg/day in juvenile female rats 27 and an 8-week exposure to butylparaben 0.001%, 0.01%, 0.10%, and 1.0% in the diet. 24 Although that particular study did not observe changes in body weight after exposure to environmental estrogens, significant effects on the reproductive system were identified, suggesting that low-dose BPA and IBP may affect the development of hormone-dependent organs more than systemic development in fetuses and neonates.
Experimental approaches assessing mixed treatment are performed to evaluate combinatorial effects including additivity, synergy, and antagonism. 48 Specifically, the independent actions of different treatments are considered “additive,” whereas synergistic effects are larger than expected and antagonistic effects are smaller than expected. 48 Although BPA and IBP have lower estrogenic potency than 17β-estradiol, 15,26,27 it is possible that simultaneous exposure might result in additive or synergistic effects because these chemicals could cause estrogenic activity by binding to hormone receptors at even extremely low doses. In this study, perinatal exposure to mixture of BPA and IBP at concentrations lower than their individual LOAELs may induce additive effects on reproduction in adult male offspring. A mixture of 23.84 mg/L of BPA and 15.65 mg/L of IBP, which are the median sporulation inhibition concentrations of these chemicals, were shown to produce greater effects on sporulation in Ulva pertusa as compared with individual treatments; however, mixtures at concentrations lower than the median effective concentration of BPA (0.05 mg/L) and IBP (2.5 and 5 mg/L) did not significantly inhibit sporulation as compared to controls. 33 Furthermore, co-treatment with BPA and IBP induced a dose-dependent increase in estrogenic effects in GH3 cells, 31,32 and the mixture of BPA (10-5M) and IBP (10-7M and 10-6M) significantly increased luciferase activity as compared to individual chemical treatments in the hormone-based reporter assays. 31 These studies showed dose-dependent effects in both single treatment and combination treatment groups; thus, only one dose of the BPA/IBP mixture was tested to evaluate for enhanced or diminished effects on male reproduction.
In conclusion, perinatal exposure to a BPA/IBP mixture, even below the LOAELs of the individual chemicals, contributed to the additive perturbation of sperm maturation in adults, which was similar to the changes induced by treatments with either BPA or IBP alone. This reduction in sperm number and motility may increase the risk of infertility. Thus, it will be important to further evaluate different combinations of BPA and IBP to better understand their effects on development. The impairment of male reproduction after exposure in the maternal generation may be explained by the direct influence of these chemical in utero. Further studies are needed to examine the effects of BPA and IBP on steroid-responsive organs in ensuing generation that have had no direct exposure to estrogenic chemicals.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
