Abstract
Background:
In this study, we investigated the alterations of matrix metalloproteinase (MMP) and tissue inhibitors of metalloproteinases (TIMPs), acute inflammation, and oxidative damage in the circulatory system and the intestine in response to mesenteric ischemia/reperfusion (I/R).
Methods:
Twenty-one rats were divided randomly into the following three groups (n = 7 in each group): a sham group (CG), an ischemic group (IG), and an I/R group (I/RG). MMP-9, TIMP-1, and myeloperoxidase (MPO) were measured using the enzyme-linked immunosorbent assay method, and lipid peroxidation (quantified as thiobarbituric acid reactive substances (TBARS) content), ischemia-modified albumin, the prooxidant–antioxidant balance (PAB), and ferric-reducing antioxidant power (FRAP) were measured spectrophotometrically. The degree of intestinal injury was evaluated according to the Chiu scoring system.
Results:
A significant difference between the mean serum TIMP-1 and MMP-9 levels and the alanine transaminase activity was found among the groups. Compared with the I/RG group a significant difference in the mean tissue MMP-9, MPO, and TBARS levels in addition to the PAB and FRAP was found between the CG and IG groups. The level of MMP-9 also demonstrated a strong, positive, and valid correlation with the TBA-RS levels. A significant morphological change was observed in both the IG and the I/RG groups. The degree of intestinal injury was more severe in the I/R group and was characterized by either villous denudation or villous loss.
Conclusions:
These results suggest that MMP-9, TIMP-1, MPO, and oxidative stress may be important in the intestinal injury development that is induced by acute mesenteric I/R in a rat model. MMP-9 overexpression may increase the extent of intestinal villous loss, particularly when MMP-9 is upregulated by the TBARS present in the intestinal injury.
Keywords
Introduction
Ischemia/reperfusion (I/R) injury is a fairly common problem in clinics. I/R syndrome has a basic influence on the pathophysiology of a considerable number of clinical surgery cases and may be a result of acute mesenteric arterial occlusion, intestinal intussusception, and hemodynamic shock. It is also observed that mesenteric I/R can cause lesions in small intestine transplants. 1
Oxidative damage is thought to be an essential factor in I/R injury initiation, even though the pathophysiological mechanisms that cause I/R injury are still difficult to define clearly. 2 It is widely demonstrated that when intestinal I/R occurs, neutrophils, cytokines—such as tumor necrosis factor α—free radicals, and chemokines are implicated in the large spectrum of inflammatory mediators that are included in the physiopathology of intestinal I/R-caused tissue injury. 3 The results of various preclinical studies regularly demonstrate that annihilating reactive oxygen species (ROS) and reactive nitrogen species, which limit oxidative damage, reduce the severity of I/R injury. 2 Myeloperoxidase (MPO) is one of the enzymes that functions in the oxygen-dependent killing of microorganisms and is released from the primary granules of neutrophils during acute inflammation. 4
Matrix metalloproteinases (MMPs), which are endopeptidases involved in extracellular matrix (ECM) degradation, are also implicated in several abnormal bioprocesses, such as tumor growth, invasion, and metastasis. For this reason, there is considerable interest surrounding the regulation of MMP activity as a possible therapeutic mechanism. Tissue inhibitors of metalloproteinases (TIMPs) mainly restrain MMPs and influence the maintenance of the balance between the destruction and the formation of the ECM. 5 MMPs appear to be crucial for both physiological and pathological healing in several organ systems.
In the present study, in an acute mesenteric I/R injury model, we investigated (1) the serum MMP-9 and TIMP-1 levels; (2) the presence of MPO, MMP-9, TIMP-1, lipid peroxidation (quantified as thiobarbituric acid reactive substances (TBARS) content), and ischemia-modified albumin (IMA) as well as the prooxidants–antioxidants balance (PAB), and ferric-reducing antioxidant power (FRAP) in intestinal tissue; and (3) the level of intestinal tissue injury using a grade scale.
Materials and methods
Animals
Male Sprague–Dawley rats (3 months old) weighing 250–300 g were maintained in standard animal cages where they could freely reach a standard pellet diet and water before surgery. The study was performed according to the animal care guidelines of the National Institutes of Health and with the approval of a local ethical committee in Turkey. The surgical procedures were carried out according to the criteria of the National Institutes of Health Guidelines on the use of laboratory animals.
Study groups and design
Twenty-one rats were divided randomly into the following three groups (n = 7 in each group): a sham operation group (CG: group 1), an ischemic group (IG: group 2), and an I/R group (I/RG: group 3). The animals were anesthetized using an intraperitoneal injection of 35 mg kg−1 ketamine hydrochloride (HCl; Ketalar, Parke Davis and Co. Inc., Pfizer Pharmaceuticals, Istanbul, Turkey) and 5 mg kg−1 xylazine HCl (Rompun, Bayer Healthcare Pharmaceuticals, Istanbul, Turkey). A midline laparotomy process followed.
Group 1 was not subjected to mesenteric ischemia or drug treatment. Following 120 min of anesthesia, the animals were killed after blood samples were taken from the vena cava inferior, and tissue samples were collected. Intestinal tissues were excised and fixed in a 10% buffered formalin solution for histological evaluation, flash which was frozen in liquid nitrogen and then stored at −80°C for the subsequent determination of the biochemical parameters.
After group 2 underwent laparotomy, the mesenteric artery was ligated; after confirmation was obtained by observing the pale color of the bowel and the loss of mesenteric pulsation, mesenteric ischemia was continued for the last 60 min of a total 120 min of anesthesia. Tissue samples were subsequently collected, and blood samples were then drawn from the rats. Intestinal tissues were immediately excised and fixed in a 10% buffered formalin solution for histological evaluation, flash which was frozen in liquid nitrogen and then stored at −80°C for the subsequent biochemical analyses.
After group 3 underwent laparotomy, the superior mesenteric artery was ligated, and after confirmation was obtained by observing the pale color of the bowel and the loss of mesenteric pulsation, mesenteric ischemia was continued for 60 min. The clamps were subsequently opened, and reperfusion was enabled for 60 min. Reperfusion was confirmed by the recovery of bowel color and pulsation in the artery. After ischemia and reperfusion lasting a total of 120 min, tissue and blood samples were drawn from the rats. Intestinal tissues were immediately excised and fixed in a 10% buffered formalin solution for histological evaluation, flash which was frozen in liquid nitrogen and then stored at −80°C for the subsequent biochemical analyses.
Preparation of samples
Blood samples were collected using tubes containing only a clot activator (9 mL, Greiner Bio-One, Kremsmünster, Austria, cat. no. 455092) and anticoagulant-free tubes. The samples were permitted to clot, subsequently centrifuged at 1000g for 10 min, and then stored at −80°C.
Tissue samples that were stored at −80°C were brought to room temperature, and after cleaning, the bowel contents were weighed (1058 ± 238 mg). All of the tissue samples were washed with a 0.1 M phosphate buffer (pH = 7.4), diluted 1:10, and subsequently homogenized using a Wiggen Hauser D130 handheld homogenizer (Selangor, Malaysia). The homogenate was sonicated using a Bandelin Sonopuls HD 2070 (Berlin, Germany) sonicator. The samples were centrifuged for 10 min at 1500g and 4°C. The supernatants were analyzed to determine the levels of MMP-9, TIMP-1, MPO, TBARS, and IMA in addition to the FRAP and PAB.
Histopathological evaluation
Intestinal samples were fixed in 10% buffered formalin, dehydrated with ethanol, and purified in xylene. The tissues were then embedded in paraffin and cut into 4–5 µm sections. After deparaffinization, the tissues were stained by hematoxylin and eosin and examined through a light microscope by researchers who were blinded to the conditions. Ten villi and crypts were randomly chosen and were evaluated in terms of the severity of intestinal tissue injury according to the criteria of the Chiu scoring system 6 as follows: grade 0 (normal mucosal villi), grade 1 (subepithelial space at the villus tip), grade 2 (extension of the subepithelial space with moderate lifting of the epithelial layer), grade 3 (massive epithelial lifting along the villus sides), grade 4 (denuded villi with exposed dilated capillaries), and grade 5 (digestion and disintegration of the lamina propria, hemorrhage, and ulceration).
Determination of plasma and tissue MMP-9 concentrations
The concentration of MMP-9 in the samples was determined using a quantitative sandwich enzyme immunoassay kit (Rat Total MMP-9 Immunoassay, Quantikine® enzyme-linked immunosorbent assay (ELISA), R&D Systems, Inc., Minneapolis, Minnesota, USA) according to the manufacturer’s instructions.
Determination of plasma and tissue TIMP-1 concentrations
The concentration of TIMP-1 in the samples was determined using a quantitative sandwich enzyme immunoassay kit (Rat TIMP-1 Immunoassay, Quantikine® ELISA, R&D Systems, Inc.) according to the manufacturer’s instructions.
Determination of tissue MPO concentrations
The concentration of MPO in the samples was determined using a competitive ELISA kit (Hycult Biotech, Uden, the Netherlands) according to the manufacturer’s instructions.
Determination of tissue TBARS levels
The extent of lipid peroxidation was determined according to the procedure of Buege and Aust. 7 The coefficients of the intra- and inter-assay variations for the TBARS assay were 3.2% (n = 10) and 4.7% (n = 10), respectively.
Determination of tissue FRAP concentrations
The FRAP assay was fulfilled according to the protocol described by Benzie and Strain 8 with insignificant changes. The coefficients of the intra- and inter-assay variations were 4.9% (n = 10) and 6.3% (n = 10), respectively.
Determination of tissue PAB concentrations
The PAB assay was fulfilled according to the method of Alamdari et al. 9 with insignificant changes. The coefficients of the intra- and inter-assay variation were 5.9% (n = 10) and 6.1% (n = 10), respectively.
Determination of tissue IMA concentrations
IMA concentrations were assessed using a modification of the method of Bar-Or et al. 10 The coefficients of the intra- and inter-assay variations were 5.9% (n = 10) and 6.7% (n = 10), respectively.
The levels of serum alanine transaminase (ALT), aspartate transaminase (AST), lactate dehydrogenase (LDH), and total protein were measured using commercially available kits (Siemens Healthcare Diagnostic Inc., West Sacramento, California, USA).
Statistical analysis
Parameters used for biostatistical analysis of the study data included mean, standard deviation, frequency, and percentage. For the comparison of frequency and percentage between the groups, a χ2 test was used. For the comparison of more than two group means, a one-way analysis of variance (ANOVA) model was used. If a significant difference was found with the ANOVA model, the subgroup differences were analyzed with a post hoc “Tukey’s honestly significant difference” or “Tamhane” or “Dunnett” test for the pairwise comparisons. Spearman correlation analyses (rs) were used to determine and document the mathematical correlations between the variables. The significance level was set at p = 0.05. An SPSS version 11.5 software package was used for the statistical analyses.
Results
Histopathological alterations
The experimental groups were divided into subgroups according to the histopathological grades—which were designated as minimal–moderate change for groups 1, 2, and 3 and severe change for groups 4 and 5—and were then reevaluated. (Figure 1(b) to (d)). Morphologically, the damage to the intestinal mucosa was more severe in the I/R group than in the IG group, and the I/R group demonstrated villous denudation and/or loss. Therefore, the total thickness of the intestinal wall was reduced (Figure 1(d)). The intensity of neutrophil leukocyte infiltration corresponded to the mucosal damage.

(a) Microscopic view of normal intestinal mucosa (group 1, H&E). Villous structures seen as finger-like projections into the lumen. Both columnar absorptive cells and goblet cells (with apical clear vacuoles) cover the villous surfaces, which are supported by a thin layer of lamina propria. (b) Villous structures look more coarse and broader than their normal shape. Arrows show subepithelial edema at villous tips with capillary congestion (group 2, H&E). (c) Prominent epithelial lift along villous sides (group 2, H&E). (d) Loss of villous tissue. Because the upper half of the mucosal tissue disintegrates, the finger-like appearance of villous structures disappear (group 3, H&E). H&E: hematoxylin and eosin.
Table 1 summarizes the histopathological degree of intestinal injury in the experimental groups. Figure 1(a) shows normal intestinal mucosa in the control group. As shown in Table 1, a highly significant difference was found using Pearson’s χ2 test for the distribution of pathologic grade (frequency and percentage) between the CG, IG, and I/RG groups (p = 0.003). A significant difference was found using Pearson χ2 test for the distribution of pathological grades (frequency and percentage) between the IG and I/RG groups (p = 0.031).
Histopathological degree of intestinal tissue injury of the experimental groups.a

CG: control group; IG: ischemic group; I/RG: ischemia–reperfusion group.
aGroup 1:CG; group 2: IG; group 3: I/RG. Percentage values given in parentheses are within each group.
bp = 0.003: Significant difference was found using χ2test for the distribution of histopathological grade between the CG, IG, and I/RG groups.
cp = 0.031: Significant difference was found using the Pearson chi-square test for the distribution of histopathological grade between the “IG and I/RG” groups.
The groups were divided into subgroups according to histopathological grade, groups 1, 2, and 3 were categorized as undergoing minimal–moderate change, and groups 4 and 5 were categorized as undergoing severe change. We then reevaluated that a more significant difference was found with Fisher’s exact test for the distribution of pathological grades (frequency and percentage) between the IG and I/RG groups (p = 0.005; Figure 1(b) to (d)). Microscopically, the morphological damage in the intestinal mucosa was more severe and observed as villous denudation and/or loss in the I/R group. Therefore, the total thickness of the intestinal wall was lower (Figure 1(d)). The intensity of neutrophil infiltration was parallel to mucosal damage.
Biochemical alterations
For the serum biochemical indices in the overall study group (n = 21), a strong (rs = 0.671), positive, significant (p = 0.001) correlation between TIMP-1 and MMP-9 levels was detected. The level of TIMP-1 demonstrated a strong (rs = 0.643), positive, and significant (p < 0.002) correlation with the level of AST and a strong (rs = 0.763), positive, and significant (p < 0.0001) correlation with the level of ALT. The level of TIMP-1 demonstrated a weak (rs = 0.461), positive, and significant (p = 0.041) correlation with the level of LDH. The level of MMP-9 demonstrated a strong (rs = 0.741), positive, and significant (p < 0.0001) correlation with the level of AST and a strong (rs = 0.697), positive, and significant (p < 0.001) correlation with the level of ALT. The level of AST demonstrated a very strong (rs = 0.945), positive, and significant (p < 0.0001) correlation with the level of ALT and a weak (rs = 0.468), positive, and significant (p = 0.038) correlation with the level of LDH (Table 2).
Biochemical circulating markers of the experimental groups.a

CG: control group; IG: ischemic group; I/RG: ischemia–reperfusion group; TIMP-1: tissue inhibitor of metalloproteinase-1; MMP-9: matrix metalloproteinase-9; AST: aspartate transaminase; ALT: alanine transaminase; LDH: lactate dehydrogenase; ANOVA: analysis of variance.
aGroup 1:CG; group 2: IG; group 3: I/RG. Percentage values given in parentheses are within each group.
bp value was calculated using one-way ANOVA (mean ± SD).
cp < 0.033: versus group 3.
dp < 0.005: versus group 1.
ep < 0.01: versus group 3.
fp < 0.001: versus group 1.
gp < 0.011: versus group 3.
hp < 0.003: versus group 1.
ip < 0.0001: versus group 3.
jp < 0.0001: versus group 1.
A significant difference in the mean tissue MPO levels in addition to the PAB and FRAP was found between the CG and IG groups compared with the I/RG group. Pairwise comparisons and their levels of significance are summarized in Table 3.
Tissue levels of the evaluated biochemical markers of the experimental groups.a
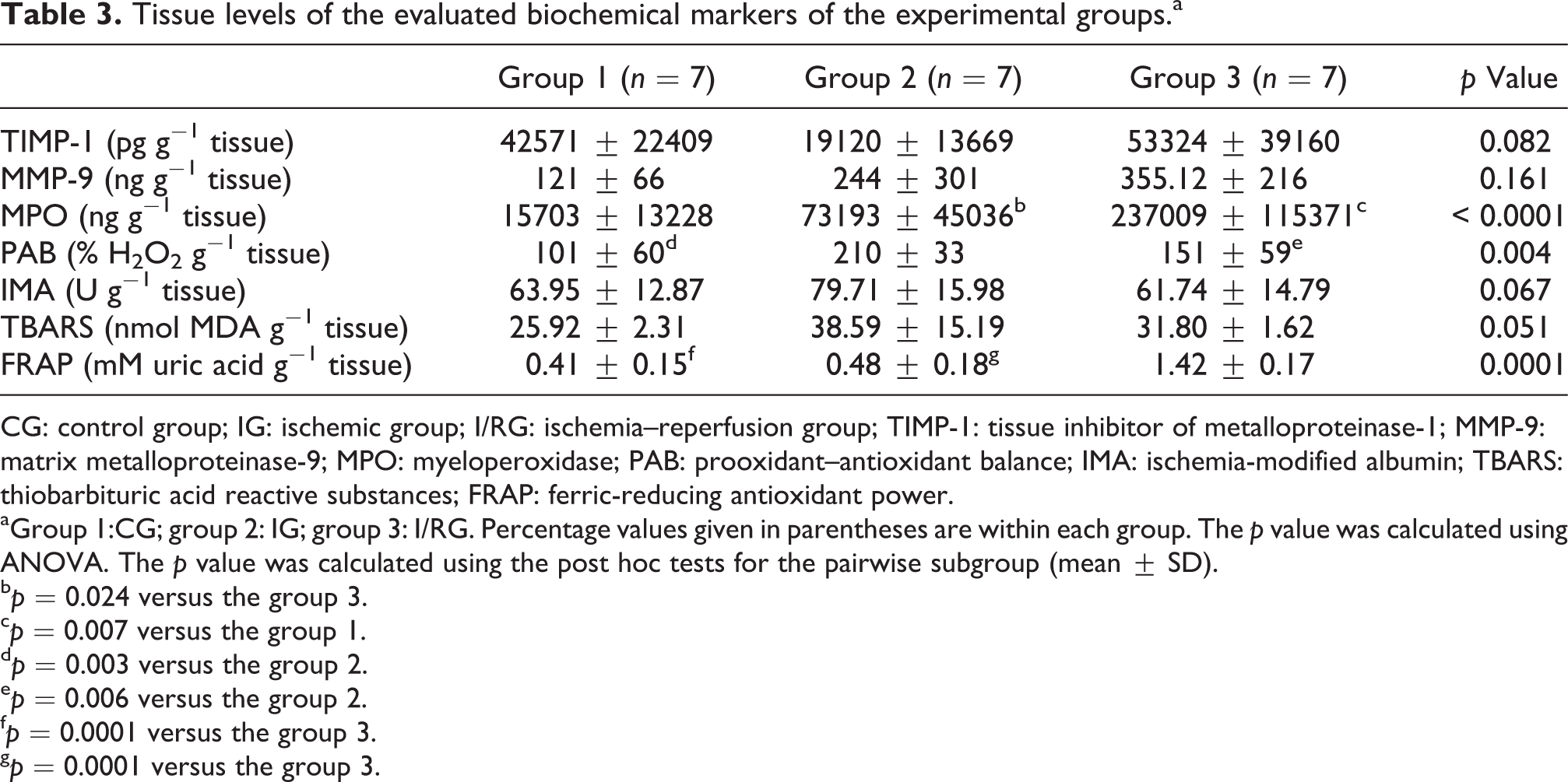
CG: control group; IG: ischemic group; I/RG: ischemia–reperfusion group; TIMP-1: tissue inhibitor of metalloproteinase-1; MMP-9: matrix metalloproteinase-9; MPO: myeloperoxidase; PAB: prooxidant–antioxidant balance; IMA: ischemia-modified albumin; TBARS: thiobarbituric acid reactive substances; FRAP: ferric-reducing antioxidant power.
aGroup 1:CG; group 2: IG; group 3: I/RG. Percentage values given in parentheses are within each group. The p value was calculated using ANOVA. The p value was calculated using the post hoc tests for the pairwise subgroup (mean ± SD).
bp = 0.024 versus the group 3.
cp = 0.007 versus the group 1.
dp = 0.003 versus the group 2.
ep = 0.006 versus the group 2.
fp = 0.0001 versus the group 3.
gp = 0.0001 versus the group 3.
For the tissue biochemical indices in the overall study group (n = 21), the level of MMP-9 demonstrated a weak (rs = 0.544), positive, and significant (p = 0.011) correlation with the level of MPO. The level of MPO demonstrated a strong (rs = 0.684), positive, and significant (p = 0.001) correlation with the level of TBARS and a strong (rs = 0.574), positive, and significant (p = 0.007) correlation with the FRAP. The level of TBARS demonstrated a strong (rs = 0.744), positive, and significant (p = 0.0001) correlation with the level of PAB and a weak (rs = 0.519), positive, and significant (p = 0.016) correlation with the level of IMA.
Discussion
Studies using rat models have shown that cranial mesenteric vein occlusion stimulates a smaller reduction in energy metabolism than cranial mesenteric artery occlusion. Conversely, hemorrhage and tissue injury resulting from venous occlusion causes irremediable tissue loss in the intestines of the animals. 1 I/R injury is a fairly common clinical problem. Several studies have indicated that hepatic and pulmonary dysfunction in addition to renal and intestinal injury may occur following mesenteric I/R. The main reason for choosing this rat model is to simulate the real clinical condition. Despite these findings, the effect of mesenteric I/R on the MMP-9, TIMP-1, MPO, and oxidative stress levels of intestinal tissue has not been investigated. The formation of ROS following reperfusion may lead to the oxidative damage of lipids, proteins, and nucleic acids. Our results illustrated that the increase of TBARS levels in the intestinal tissue following mesenteric I/R was significant, indicating lipid peroxidation. The level of MMP-9 also demonstrated a strong, positive, and significant correlation with the TBARS levels. MMP-9 activity is elevated in the intestine during acute I/R, which stimulates inflammation and tissue injury. Yurdakan et al. 11 revealed that the level of TBARS, which functions as a sign of lipid peroxidation, is increased considerably in the renal tissue following intestinal I/R. Lipid peroxidation plays an important role in the formation of cellular damage caused by I/R injury and results in the increased permeability of the plasma, mitochondrial, and lysosomal membranes. 11 Rosário et al. 12 demonstrated that an increase in the levels of MMP-9 in the wall of the small intestine is observed while the intestinal I/R occurs. It was discovered that the MMP-9 is largely associated with neutrophils. After the intestinal mucosal barrier opens during I/R, the pancreatic trypsin penetrates into the intestinal wall and supports the neutrophils and MMP-9 accumulation in the intestine. Additionally, trypsin functions as a strong activator of the MMP-9 pro-form that is released from neutrophils.
Most members of the MMP group—including MMP-9—are inhibited by TIMP-1. MMPs and TIMPs are crucial to maintain the wholeness and well-being of healthy tissues by creating a balance in the breakdown and deposition of the ECM. A disproportion in the expression of these proteins causes diseases linked to undue ECM degradation. We discovered a significant positive correlation between serum levels of MMP-9 and TIMP-1 among all study groups in this study. This result is consistent with the fact that a disproportion between MMP-9 and TIMP-1 tissue levels is an important factor in the development of I/R injury. We observed that the level of tissue MMP-9 better correlated with the levels of MPO and TBARS in addition to the PAB and FRAP than with the level of TIMP-1, which appeared to indicate that MMP-9 might be a more suitable biological marker than TIMP-1 for intestinal tissue injury following acute mesenteric I/R.
In humans, an increase of MPO activity in plasma or other body fluids is considered a marker of neutrophil activation in several disease conditions, including I/R injury, respiratory distress syndrome, glomerulonephritis, arthritis, peptic ulcer formation, gastric cancer, and cardiovascular disease. 13 –16 The level of inflammation seen in intestinal tissues during I/R is measured by the MPO activity. 17 An important difference in the mean tissue level of MPO was found between the CG and IG groups compared with the I/R group. The highest level of MPO was found in the I/R group. The level of MMP-9 also correlated with the level of MPO in this study. The role that MMP-9, TIMP-1, and MPO play may be important in the physiological turnover of the ECM through degradation and remodeling during inflammation. Dong et al. 18 discovered that, compared with the CG, there was an obvious increase in the levels of expression in the case of MPO and TBARS in the I/R group.
A decrease was observed in the MPO activity and TBARS levels with treatment using resveratrol. Oxidative stress is crucial in the development of intestinal I/R injury. Okudan et al. 19 demonstrated that there was a lower increase of MPO activity in intestinal I/R than in other studies that demonstrated a higher increase of MPO activity in comparison with the CG. The duration of ischemia and/or reperfusion is important to this difference because a longer period of ischemia and/or reperfusion may prompt more neutrophil infiltration. A previous study found that lipid peroxidation products are increased by I/R injury as reflected by TBARS levels, and the direct measurement of ROS using chemiluminescence yielded a similar result of increased levels in the I/R group compared with the CG. Our findings are consistent with previous reports. 19 –21 Intestinal I/R trigger a cascade of events, including an increase in vascular permeability; an alteration in the absorption, activation, and adhesion of polymorphonuclear neutrophils; the release of pro-inflammatory substances; and bacterial translocation. Mediators released from mast cells and macrophages—which are normally located in close adjacency to postcapillary venules—aggravate the inflammatory response that is induced by I/R. 22
The mechanisms of development of I/R injury are not completely understood. Hypoxia occurs during ischemia, but reperfusion injury occurs after the reconstitution of blood flow. 20 According to the recent studies on biochemical markers in acute mesenteric ischemia, time is vital, and an appropriately sensitive and specific marker that allows for early diagnosis to increase survival is not available. 23 Serum IMA concentrations have emerged as a new, sensitive biomarker of ischemia and oxidative stress. I/R brings about changes in the albumin-binding capacity for cobalt caused by acidosis, decreased oxygen tension, and free radical production. 24 Many recent studies demonstrate an increase in IMA levels in different cases of acute ischemia, including cerebral, myocardial, pulmonary, and mesenteric infarction, suggesting the idea of its employment as a diagnostic marker. 10,24 –34 However, high IMA concentrations are not the only result of purely myocardial involvement. There are other organs that may also be responsible for the increase in IMA concentrations. IMA concentrations may increase when I/R—which may affect any organ—occurs. They cannot be taken into consideration specifically in relation to oxidative stress. 34 Dundar et al. 35 performed a study that only compared the serum IMA levels with other indicators in acute mesenteric ischemia. On the other hand, in our study, using a rat model, the relationships among IMA levels, lactate, and malondialdehyde—which are markers of ischemia—were assessed. We also discovered that the differences in tissue IMA concentrations among the studied subjects were not significant. Although plasma IMA concentrations are commonly used to reflect oxidative status and ischemia, the tissue IMA concentrations may not be considered a specific prognostic marker for acute mesenteric ischemia. These introductory findings reveal that intestinal tissue IMA concentrations may not necessarily indicate an important parameter in the early diagnosis of acute mesenteric ischemia and that further studies are needed to determine the role of serum IMA levels.
An additional marker that was assessed in the present study was the PAB as an oxidative stress marker. To our knowledge, no modified PAB methods have been utilized to determine the oxidant–antioxidant status in I/R injury. Oxidative stress could represent a considerable risk indicator in the atherosclerotic process, and the evaluation of the PAB, together with the other risk factors, may be beneficial in risk prediction for cardiovascular events. Through the proper interventions, an adequate and effectual primary prevention may reduce the progress of cardiovascular disease. The PAB values were found to be higher in the IG group than in the I/R group. To date, various methods have been developed that individually determine the total prooxidant and antioxidant capacities and are therefore time consuming, difficult, costly, and uncertain. For our evaluation of the PAB, we used a simple, fast, and reasonable modified PAB assay method, which correlated with the levels of both MMP-9 and MPO. We suggest that this PAB assay can be favorable as an ischemia risk predictor in various cases of acute ischemic conditions—including cerebral, myocardial, pulmonary, and mesenteric infarction—and may aid in the identification of patients with higher oxidative stress for early intervention to prevent vascular and ischemic disease.
Many studies have evaluated the capacity of antioxidative defense systems—such as oxygen radical absorbance capacity, FRAP, and antioxidant enzymatic activities—in human and animals model of I/R. In our research, we have performed the FRAP assay in accordance with Benzie and Strain, 8 with minor modifications. The FRAP assay is favored not only for the measurement of a single compound’s antioxidant capacity but also for its ability to measure the total antioxidant capacity (TAC) of all antioxidants in serum, foodstuff, and biological tissues. In this study, the increase in TAC levels that was measured by the total antioxidant status was significant in I/R comparing to that of the other groups. It appears that the other antioxidants did not contribute to the phenomenon of conditioning in the intestinal tissue, and the increase in the tissue TAC levels was not sufficient to prevent oxidative damage. Moreover, it is not clear which antioxidant components are responsible for the increased TAC. If there is no activation of the oxidation systems, there is no increase in TAC. The TAC decreases if oxidative stress is seen as a result of physiological stress. Serum or plasma TAC has not consistently reflected increased oxidative stress. 36 Zou et al. 37 demonstrated a decrease in the activities of superoxide dismutase, glutathione, catalase, and the TAC in the kidneys of the I/R injury group compared with the sham-operated group. Pretreatment performed by the use of pioglitazone also brought about an increase in the TAC of the kidney that was subjected to ischemia. The enzymatic activities and TAC signify a whole range of antioxidant activities against different kinds of reactive oxygen/nitrogen radicals. Bostankolu et al. 38 demonstrated a decrease in the serum TBARS levels in comparison with the basal values at 5 and 20 min after tourniquet release (ATR) and that, compared to basal values, the TAC was lower at 1 min before and at 5 min ATR, reaching the basal level at 20 min ATR. Yet these results were similar to those obtained from kidneys that were not treated with dexmedetomidine. The scavenging capacity of antioxidant enzymes and ROS is usually in balance. As a result of this balance, the determined TAC is a sensitive marker of the general protective power of the antioxidants. Although a decrease in TAC levels is predicted, a higher TAC was found in the I/R group in our study. It is likely that the high levels of other antioxidants may have contributed to this unexpected finding. However, this could be confirmed by further studies.
Histological damage that occurs after intestinal I/R is caused by necrosis, loss of villus epithelium, the shortening of the villus length, and invasion of inflammatory cells. 35,39 –41 In this study, significant morphological changes were observed in both the IG and the I/RG groups. In the ischemic group, the degree of mucosal injury was minimal to moderate, and this was observed in a morphological spectrum from subepithelial edema at the villous tips to extensive edema and congestion. The most severe injury was observed in the reperfusion group as extensive villous denudation or loss. The intensity of neutrophil infiltration additionally corresponded to the mucosal damage. This observation may indicate a successful model of I/R injury in the small intestine in our study.
In conclusion, mesenteric I/R may generate several inflammatory mediators, including MMP-9, TIMP-1, MPO, reactive oxygen metabolites such as TBARS, and IMA in addition to oxidative stress as assessed by the PAB and FRAP in intestinal tissue injury, which was histopathologically confirmed. These results suggest that a multifactorial drug that possesses antioxidant, anti-inflammatory, and antineutrophil infiltration properties may be a therapeutic option to prevent intestinal injury after acute mesenteric I/R. Further work is necessary to better understand these parameters in wider patient populations with acute mesenteric I/R.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Siemens Healthcare Diagnostic Inc., West Sacramento, California, USA.
