Abstract
2,4-Dichlorophenol (2,4-DCP) is formed in drinking water as a result of its chlorination, and it is created in the environment during transformation of various xenobiotics such as triclosan or herbicide 2,4-dichlorophenoxyacetic acid (2,4-D). The molecular mechanism depicting the action of phenolic compounds on nucleated blood cells has been insufficiently studied, and therefore, we have assessed the effect of 2,4-DCP on the structure and viability of human peripheral blood mononuclear cells (PBMCs). We have evaluated necrotic, apoptotic, and morphological changes (alterations in the size and granulation) in PBMCs incubated with 2,4-DCP in the concentration ranging from 10 to 500 µg mL−1 for 4 h at 37°C. Moreover, we have estimated changes in reactive oxygen species (ROS) formation, lipid peroxidation, and protein carbonylation in the incubated cells. We have noted that 2,4-DCP increased ROS formation and lipid peroxidation (from 10 µg mL−1) and oxidized proteins (from 50 µg mL−1) in PBMCs. The compound studied also provoked apoptotic (from 50 µg mL−1), necrotic (from 100 µg mL−1) and alterations in the size and granulation (from 50 µg mL−1) in the incubated cells. The analysis of quinolinium 4-[(3-methyl-2(3H)-benzoxazolylidene)methyl]-1-[3-(trimethyl-ammonio)-propyl]-diiodide/propidium iodide staining revealed that 2,4-DCP (50–250 µg mL−1) more strongly increased the number of apoptotic than necrotic cells, which suggests that this cell death type is mainly provoked by this compound in PBMCs. The observed changes were caused by relatively high concentrations of 2,4-DCP, which cannot influence human organism during environmental exposure and thus may only occur as a result of acute or subacute poisoning with this compound.
Keywords
Introduction
2,4-Dichlorophenol (2,4-DCP) is formed in drinking water as a result of its chlorination. 1 The 2,4-DCP may also be formed in the environment during transformation of various pesticides, mainly phenoxyherbicides, such as 2,4-dichlorophenoxyacetic acid (2,4-D), 1,2 as well as the antibacterial and antifungal agent triclosan. 3 Chlorophenols are also biosynthesized by anaerobic microorganisms as a result of transformation of biogenic chlorinated hydroquinone metabolite. 4 2,4-DCP is also formed as a major metabolite of 2,4-D in animals. Moreover, 2,4-DCP is used as a precursor for the synthesis of 2,4-D, and it is also the main contaminant in the preparation of this herbicide. 5
Due to widespread occurrence of 2,4-DCP in the environment, and thus in human organism, the mechanism of action of this substance is still being investigated in vitro and in vivo. 6 –8 Up to now, it has been shown that phenolic compounds including 2,4-DCP may reveal genotoxic, mutagenic, and carcinogenic activities. 9,10
The presence of chlorophenols in blood and urine of the general population has been proven in some studies. For instance, in the population of the United States, chlorophenols have been detected in the concentrations ranging from 3 to 65 µg L−1 of blood serum. 11 Other studies have shown that workers permanently exposed to chlorophenols had high urinary levels of these substances. For instance, workers employed in sawmills, where chlorophenols were used to prevent fungal growth of lumber had high concentrations of these compounds (from 196 to 2320 µg L−1) in blood and urine. 12 In another study, it was observed that workers permanently exposed to pentachlorophenol (PCP) had enormous PCP amounts up to 84.9 mg L−1 in blood serum. 13
Peripheral blood mononuclear cells (PBMCs, mainly lymphocytes) are very important cells of the immune system because they are responsible for the organism’s defense against pathogens as well as maintaining homeostasis in the body, having the so-called function immunological supervision. 14 Moreover, PBMCs are potentially exposed to numerous toxicants entering human organism. 10,15 Nutrients, drugs, and toxic compounds are transported by blood in living organisms, which leads to numerous interactions between various xenobiotics and PBMCs, and thus changes in structure and functionality of these cells. Therefore, PMBCs have been repeatedly used as model cells in the investigation of xenobiotics toxicity on nucleated cells. 16 –18
Up to now, insufficient information has been obtained concerning the mechanism of phenolic compounds’ action on nucleated blood cells, and no study has been conducted to analyze the effect of 2,4-DCP on PBMCs. Therefore, the purpose of this study was to assess the effect of 2,4-DCP on necrotic, apoptotic, and morphological (size and granulation) alterations in PMBCs. Moreover, the effect of 2,4-DCP on lipid and protein damage as well as free radical formation in the cells studied was assessed. The 2,4-DCP was used in the concentrations that can enter human organism as a result of occupational exposure or acute (accidental) poisoning with this compound.
Experimental
Chemicals
2,4-DCP, Histopaque, Roswell Park Memorial Institute (RPMI) medium with
Human PBMCs isolation
Human PBMCs were obtained from leukocyte–platelet buffy coat taken from healthy donors in the Blood Bank of Łódź, Poland. The cells were isolated using Histopaque gradient by centrifugation at 1500 r min−1 for 30 min at 20°C. The cells were suspended in RPMI medium with
Analytical methods
Cell viability
Cell viability was quantified by measuring acetomethyl derivative of calcein (calcein-AM; evaluation of viable cells) and PI (evaluation of necrotic cells) entering PBMCs. 19,20
Calcein-AM penetrates the cells that maintain plasma membrane integrity and accumulates in the cytoplasm, while PI penetrates cells in which membrane lost its integrity and binds to DNA.
After incubation, the samples were centrifuged at 1000 r min−1 for 5 min at 4°C, the supernatant was discarded, and the cells were supplemented with RPMI. Then, the markers were added, and the samples were incubated for 15 min at 37°C in total darkness. The final concentration of calcein-AM and PI was 1 µM and 0.1 µM, respectively. The flow cytometry (FMC) gate on PBMCs has been established for data acquisition, and the fluorescence was measured with excitation/emission of 494/517 nm for calcein and 535/617 nm for PI, respectively.
Measurement of cell morphology
Cell morphology was analyzed using a flow cytometer (LSR II; Becton Dickinson, East Rutherford, New Jersey, USA). The FMC gate on PBMCs has been established for data acquisition, and cell size and granularity were evaluated with simultaneous separate detection of low-angle (forward scatter pulse area (FSC-A)) and right-angle (side scatter pulse area (SSC-A)) light scattering. The data obtained were displayed in the form of a diagram of cell number versus light scatter and were analyzed using the standard computer program WinMDI2.8. The light scattered near the forward direction (low angle) is expected to be proportional to particle size (volume), whereas scattering at the right angle depends on cell granulation (internal properties of the scattered particles). 21
Analysis of apoptosis
Apoptosis was quantified using the fluorescence probes YO-PRO-1 (quantification of apoptotic cells) and PI (quantification of necrotic cells). 22 Living cells exhibited a low level of green fluorescence, apoptotic cells exhibited incrementally higher level of green fluorescence, and necrotic cells revealed both red and green fluorescence. Percentage of apoptotic cells was defined as the percent of YO-PRO-1 stained cells within the cell population of interest. The positive control contained the cells in which the apoptosis was induced with 20 µM of camptothecin. The samples were incubated for 4 h at 37°C in total darkness. Then, the mixture of YO-PRO-1 and PI (0.1 µM each) was added to the samples. After incubation for 20 min on ice in total darkness, the samples were analyzed by flow cytometry (LSRII; Becton Dickinson) with excitation at 488 nm to visualize the YO-PRO-1 green fluorescence (520/30 bandpass filter) and PI red fluorescence (610/20 bandpass filter).
Hoechst 33342/PI staining
Morphological changes in PBMCs treated with 2,4-DCP were assessed by double staining with Hoechst 33342 and PI. The cells were treated with 2,4--DCP and incubated for 4 h at 37°C in total darkness. After incubation, the cells were centrifuged at 200g for 3 min at 4°C, and then the supernatant was discarded. The cells were suspended in 0.5 mL PBS, and then the mixture of 1 µL of Hoechst 33342 and 1 µL of PI (1 mg mL−1 each) was added to cells suspension. After 1 min incubation at 37°C in total darkness, the cells were analyzed by fluorescence microscopy (Olympus IX70, Japan) at 400× magnification. The cells were classified on the basis of their morphological staining characteristics: viable (blue fluorescence), early apoptotic (intensive bright blue fluorescence), late apoptotic (blue–violet fluorescence), and necrotic (red fluorescence). 23
The oxidation of H2DCFDA
The rate of H2DCFDA oxidation was measured by flow cytometry. This fluorescence probe is widely used for the detection of intracellular oxidants production. 24 Bartosz suggested that 2′,7′-dichlorodihydrofluorescein was originally introduced to detect “ultramicro quantities” of hydroperoxides, that is, hydrogen peroxide. The samples were incubated for 4 h at 37°C in total darkness. Then, fluorescence probe was added to the samples, which were incubated for 15 min at 37°C in total darkness. The final concentration of the probe was 10 µM. FMC gate on the PBMCs has been established for data acquisition, and fluorescence was measured by flow cytometer (LSR II, Becton Dickinson) with the excitation and emission maxima of 488 and 530 nm, respectively.
Lipid peroxidation
Lipid peroxidation is associated with oxidative damage to lipids by xenobiotics and their transformation products, that is, organic radicals and reactive oxygen species (ROS). Evaluation of lipid peroxidation in PBMCs was conducted by measuring cis-parinaric acid (PnAC) fluorescence at excitation/emission of 320/432 nm. The method is based on initial incubation of the cells with PnAC, which leads to the incorporation of this substance into cellular membrane. Then, the cells are incubated with individual xenobiotic. Damage to lipids results in a decrease of PnAC fluorescence. The analysis was performed in 96-well plates using a microplate reader (Cary Eclipse, Varian, Palo Alto, California, USA). 25
Protein carbonylation
Protein carbonylation was determined spectrophotometrically by the method of Levine et al. 26 2,4-Dinitrophenylhydrazine was used to determine the concentration of carbonyl groups in PBMCs. As a result of the reaction, a yellow product, hydrazine, was formed. The content of carbonyl groups was calculated from the absorbance measurement of the sample at 370 nm, and the absorption coefficient for hydrazone was e = 21.01 mmol−1 cm−1.
2.4. Statistical analysis
Data are expressed as mean ± SD. Multiple comparisons among group mean differences were analyzed by one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test. Differences were considered to be statistically significant when p value was less than 0.05. All statistical analyses were done using STATISTICA software (StatSoft Inc., Tulusa, Oklahoma, USA). The mean value was calculated from three to five individual experiments, (3–5 blood donors), whereas for each individual an experimental point was a mean value of three replications.
Results
Cells viability and morphology
It was observed that 2,4-DCP in the concentrations of 250 µg mL−1 and 500 µg mL−1 induced statistically significant decrease in PBMCs viability (F5;77 = 72.80; p < 0.001; Figure 1). The 2,4-DCP at the highest concentration of 500 µg mL−1 decreased cell viability up to 58.32 ± 12.74%.
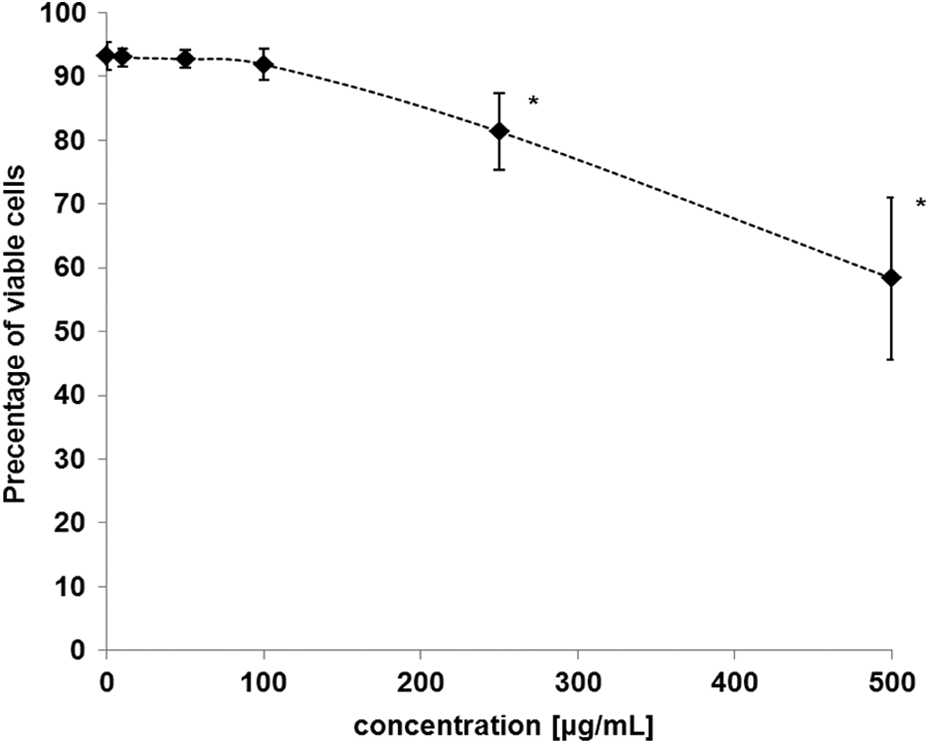
Changes in the viability of human blood mononuclear cells exposed to 2,4-DCP in the concentrations ranging from 10 to 500 µg mL−1. *Significant differences compared with controls (one-way ANOVA and a posteriori Tukey test). 2,4-DCP: 2,4-dichlorophenol; ANOVA: analysis of variance.
The analysis of FSC-A parameter allowed obtaining information about the size of PBMCs (Figure 2). It was observed that 2,4-DCP at 50 and 100 µg mL−1 caused statistically significant increase in PBMCs size, but in the highest concentration of 500 µg mL−1, it induced statistically significant decrease in FSC parameter (F(5, 98) = 98.10; p < 0.001).

Alterations (expressed as %) in the size (FSC-A) and granulation (SSC-A) of human blood mononuclear cells (exposed to 2,4-DCP at concentrations ranging from 10 to 500 µg mL−1. *Significant differences compared with controls (one-way ANOVA and a posteriori Tukey test). 2,4-DCP: 2,4-dichlorophenol; ANOVA: analysis of variance; FSC-A: forward scatter pulse area; SSC-A: side scatter pulse area.
The SSC-A parameter provided the information about granulation of PBMCs (Figure 2). A statistically significant increase in SSC parameter was noted after incubation of the cells with 2,4-DCP in the concentrations ranging from 50 to 500 µg mL−1 (F(5, 93) = 25.21; p < 0.001).
Apoptotic changes, photos of cells
The 2,4-DCP induced apoptosis of PBMCs, which was associated with changes in cell membrane permeability, and thus penetration of YO-PRO-1 probe into the cells (Figure 3(a)). The exemplary dot plots illustrating necrotic and apoptotic PBMCs measured by flow cytometry are shown in Figure 3(b). The 2,4-DCP in the concentration of 50 µg mL−1 provoked a strong increase in the number of apoptotic cells (F(4, 55) = 110; p < 0.001) but in the concentration of 100 µg mL−1 provoked statistically significant increase in the number of necrotic cells (F(4, 55) = 42.81; p < 0.001).

(a) Changes in the percentage of apoptotic and necrotic cells exposed to 2,4-DCP at concentrations ranging from 10 to 250 µg mL−1. *Significant differences compared with controls. (b) Quantification of viable (quadrant Q3), apoptotic (quadrant Q4), and necrotic (quadrant Q2) mononuclear blood cells after 2,4-DCP treatment. The analysis was performed by flow cytometry using YO-PRO-1 and PI stains. Control cells (1), cells treated with 2,4-DCP at 50 μg mL−1 (2), cells treated with 2,4-DCP at 250 μg mL−1 (3). 2,4-DCP: 2,4-dichlorophenol; YO-PRO-1: quinolinium 4-[(3-methyl-2(3H)-benzoxazolylidene)methyl]-1-[3-(trimethyl-ammonio)-propyl]-diiodide; PI: propidium iodide.
Figure 4 shows representative photomicrographs of Hoechst 33324/PI-stained PBMCs pretreated with PBS (control) and 2,4-DCP at 10, 50, and 250 µg mL−1. In control sample, mainly viable cells were observed. In samples treated with 2,4-DCP at 10 µg mL−1 mostly viable and a single apoptotic cells were noticed. In samples incubated with 2,4-DCP at 50 µg mL−1, several viable, a single necrotic and a few apoptotic cells appeared, while at 250 µg mL−1, mostly apoptotic and a single necrotic cell were observed.

The representative photomicrographs of Hoechst 33324/PI-stained PBMCs pretreated with PBS (control) and 2,4-DCP at 10, 50, and 250 µg mL−1. Viable (blue fluorescence), early apoptotic (intensive bright blue fluorescence), late apoptotic (blue-violet fluorescence) and necrotic (red fluorescence). PI: propidium iodide; PBMC: peripheral blood mononuclear cell; PBS: phosphate-buffered saline; 2,4-DCP: 2,4-dichlorophenol.
The level of ROS, lipid peroxidation and protein carbonylation
It was observed that 2,4-DCP in the concentrations ranging from 50 µg mL−1 to 500 µg mL−1 induced a statistically significant increase in H2DCFDA oxidation in the cells studied (F(5, 51) = 36.78; p < 0.001) (Figure 5(a)). The exemplary dot plots obtained by flow cytometry are shown in Figure 5(b).

(a) Oxidation of H2DCFDA in control human blood mononuclear cells exposed to 2,4-DCP in the concentrations ranging from 2 to 500 µg mL−1. *Significant differences compared with controls. (b) Histogram of changes in the intracellular ROS levels in human blood mononuclear cells after 2,4-DCP treatment; control and treated cells were stained with H2DCFDA; the fluorescence of the stain intensified in chlorophenol-treated cells. Count (relative cells number)/fluorescence of DCF. H2DCFDA: 6-carboxy-2′,7′-dichlorodihydrofluorescein diacetate; 2,4-DCP: 2,4-dichlorophenol; ROS: reactive oxygen species; 2,4-DCP: 2,4-dichlorophenol; H2DCFDA: 6-carboxy-2’,7’-dichlorodihydrofluorescein diacetate; DCF: 2′,7′-dichlorofluorescein.
We observed that 2,4-DCP at 10 µg mL−1 strongly induced lipid peroxidation (F(5, 121) = 224.87; p < 0.001) in the incubated cells (Table 1).
Lipid peroxidation and protein carbonylation in control cells and cells exposed to 2,4-DCP in the concentrations ranging from 10 to 500 µg mL−1.

2,4-DCP: 2,4-dichlorophenol; ANOVA: analysis of variance; PnAC: cis-parinaric acid.
aSignificant differences compared with controls (one-way ANOVA and a posteriori Tukey test).
The measurement of carbonyl group content proved that 2,4-DCP from 50 µg mL−1 caused statistically significant damage to proteins in human PBMCs (2,4-DCP; F(4, 61) = 9.01; p < 0.001; Table 1).
Discussion
Phenolic compounds have been proven to provoke oxidative changes in living cells. 27 –32 In this study, fluorescent probe H2DCFDA was used to detect oxidative potential of 2,4-DCP in human PBMCs. It was shown that 2,4-DCP at 10 µg mL−1 enhanced oxidation of the probe (Table 1). Those findings are in agreement with our previous results obtained on human erythrocytes, which showed that 2,4-DCP significantly increased ROS level in this cell type. 7
It is known that various ROS (hydroxyl radical, hydrogen peroxide, etc.) may induce lipid peroxidation. Lipid peroxidation can be defined as the oxidative deterioration of lipids containing any number of carbon–carbon double bonds. Destruction of membrane lipids induces alteration of its integrity, fluidity, and permeability as well as causes functional loss of biomembranes and modification of low-density lipoprotein (LDL) to proatherogenic and proinflammatory forms. 33
We found that 2,4-DCP at 10 µg mL−1 strongly induced lipid peroxidation (Table 1). The effect of 2,4-DCP on lipid peroxidation in mammalian model was observed by Clerhata et al. 34 who administrated this compound to guinea pigs. They observed that 2,4-DCP significantly increased lipid peroxidation in the liver of the animals studied. Similarly, Luo et al. 35 noticed that 2,4-DCP increased the content of malondialdehyde (the marker of cellular lipid damage) in liver of Carassius auratus. Moreover, Duchnowicz et al., observed that chlorinated aromatics induced lipid peroxidation in human erythrocytes. They suggested that it may be an additive effect of primary processes, that is, direct generation of free radicals by these xenobiotics and secondary processes like generation of ROS by oxyhemoglobin released during hemolysis of the erythrocytes. 31
Protein damage is associated with oxidation of some amino acids residues (e.g. lysine, arginine, and proline) and oxidative cleavage of the peptide backbone by way of the α-amidation pathway, which results in carbonyl derivative formation. 36,37 Our research shows much stronger oxidation of lipids than proteins by 2,4-DCP (Table 1). 2,4-DCP at 50 µg mL−1 decreased fluorescence of PnAC by 19%, while statistically significant changes in the oxidation of proteins occurred from the concentration of 50 µg mL−1 of this compound. 2,4-DCP at 250 µg mL−1 increased lipid peroxidation in PBMCs by 76%, while it depleted carbonyl groups level only by 37%. The oxidation of proteins is likely to be a secondary effect of 2,4-DCP action, which is a consequence of lipid peroxidation and increased ROS level induced by this chlorophenol.
Cellular membranes are efficient barriers between the intracellular and extracellular environment; thus, they have to be overcome by toxic compounds entering the cell. As the consequence, morphological changes in permeability and fluidity of those compartments occur. 38
Changes in membrane structure and organization can be analyzed by flow cytometry that is cell-by-cell qualitative and quantitative measurement technique, in which free cells in suspension are assessed for cytosolic or surface constituents. 39
In this study, human PBMCs morphology was determined based on cell size and granulation as measured by FSC and SSC light scattering characteristics.
The results showed that 2,4-DCP changed PBMCs morphology. It was observed that 2,4-DCP at lower concentrations (up to 50 µg mL−1) increased SSC and FSC values, while at its highest concentration of 500 µg mL−1 it decreased cell size and increased cell granulation. The observed changes in cell morphology might have been associated with significant cytotoxic effect of 2,4-DCP on PBMCs (Figure 1). It may be supposed that at the beginning, the cell was swelling, which resulted in an increase of its size, followed by the occurrence of cell shrinking, which led to a decrease of the PBMCs size. Simultaneously, gradual increase in PBMCs granulation was observed.
Bukowska et al. 40 noticed that chlorophenols were capable of interacting with proteins and lipids near the surface of the cellular membrane and between the lipid chains of membrane hydrophobic core. They also observed that chlorophenols induced changes in the content of erythrocyte membrane proteins including spectrin, band 3 and 6 proteins as well as low–molecular-weight proteins. In another work, Marczak et al. proved that each change in the content of spectrin altered cell shape, thereby decreased cellular deformability and cell survival. 41
It must also be remembered that hydrophobicity and ionization of each substance, which is expressed by their log P and pK a values, determine its behavior in the cellular environment. 42,43 It was proven that the increase in log P value and a decrease in pK a value enhance toxicity of weak acids like phenolic compounds. 2,4-DCP has relatively high log P o/w value of 3.23 and moderate pK a value of 7.81.
Changes in cell morphology induced by chlorophenols were also observed by other scientists. Biberstein and Braunbeck observed that 3,5-dichlorophenol (3,5-DCP) caused alterations in shape of fibrocyte-like fish cell line (RTG-2) from rainbow trout. 44 They noticed that 3,5-DCP provoked the appearance of surface blebs as well as progressive retraction of extensions of the cells studied. In another study, Bukowska observed that 2,4,5-trichlorophenol (2,4,5-TCP) caused significant morphological changes in human erythrocytes. 45
Changes in cell size are often associated with apoptotic alterations, in which cytoplasmic condensation occurs with simultaneous maintenance of cell membrane integrity. Moreover, alterations in cell granulation (observed in our work) that are associated with degradation of nucleus and other organelles may be characteristic feature of apoptotic cell death. Apoptosis is an essential event in the organism development and homeostasis. Apoptosis is characterized by various biochemical changes such as increase in cell membrane permeability and the occurrence of number of corrugations on the cell surface. In order to analyze apoptosis, we used YO-PRO-1 dye, which enters the cells with disturbed membrane permeability characteristic for this process. It was found that 2,4-DCP increased the number of apoptotic PBMCs (from 50 µg mL−1) (Figure 4(b)).
The studies of Bironaite et al. showed that phenol was capable of inducing apoptosis in HL-60 cells and human bone marrow endothelial cells. 8 Other study, conducted by Jiang et al., revealed apoptotic alterations in fibroblast L929 and human liver cancer HepG2 cells incubated with chlorophenols. 46 Recently, Li et al. observed that 2,4-DCP induced apoptosis in primary hepatocytes of grass carp by mitochondrial pathway. 47
Based on the obtained data, we can suggest the following order of toxicity of 2,4-DCP in PBMCs: increase in ROS level and lipid peroxidation (10 µg mL−1), protein carbonylation, apoptotic alterations and decrease in cell size, an increase in cell granulation (50 µg mL−1), and finally an increase of necrotic changes (100 µg mL−1). In summary, our results indicate that 2,4-DCP toxicity in PBMCs is associated with significant oxidative properties of this compound, which are responsible for cell death mostly through the induction of apoptosis.
The observed changes were induced by relatively high concentrations of 2,4-DCP, which cannot influence human organism during environmental exposure. Nevertheless, similar chlorophenols concentration has been determined in the blood serum of workers poisoned with these substances, 48 which may suggest that some toxic effects such as ROS formation or damage to lipids (observed in our study) may occur during acute or subacute exposure to 2,4-DCP.
Footnotes
Acknowledgement
We are thankful to Professor Aneta Koceva-Chyła (Department of Termobiology) for allowing us to work on the fluorescence microscope (Olympus IX70, Japan).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was granted from the Research 505/387/R admitted for Department of Biophysics of Environmental Pollution University of Lodz.
